Submitted:
09 April 2024
Posted:
10 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methodology
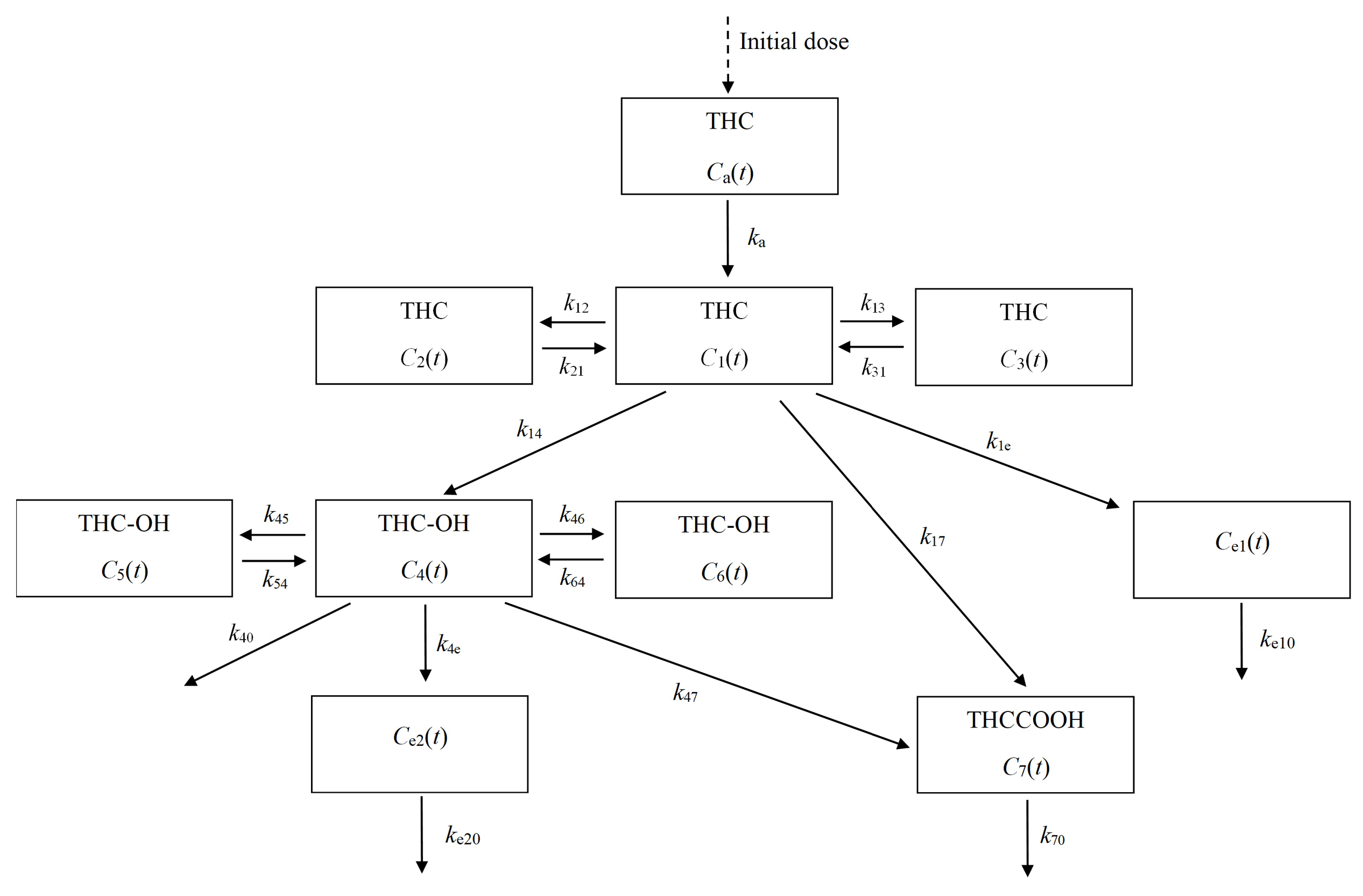
2.1. Compartmental Modeling
2.2. NSFD Schemes
2.3. Pharmacokinetic Parameters
2.4. Actual Data
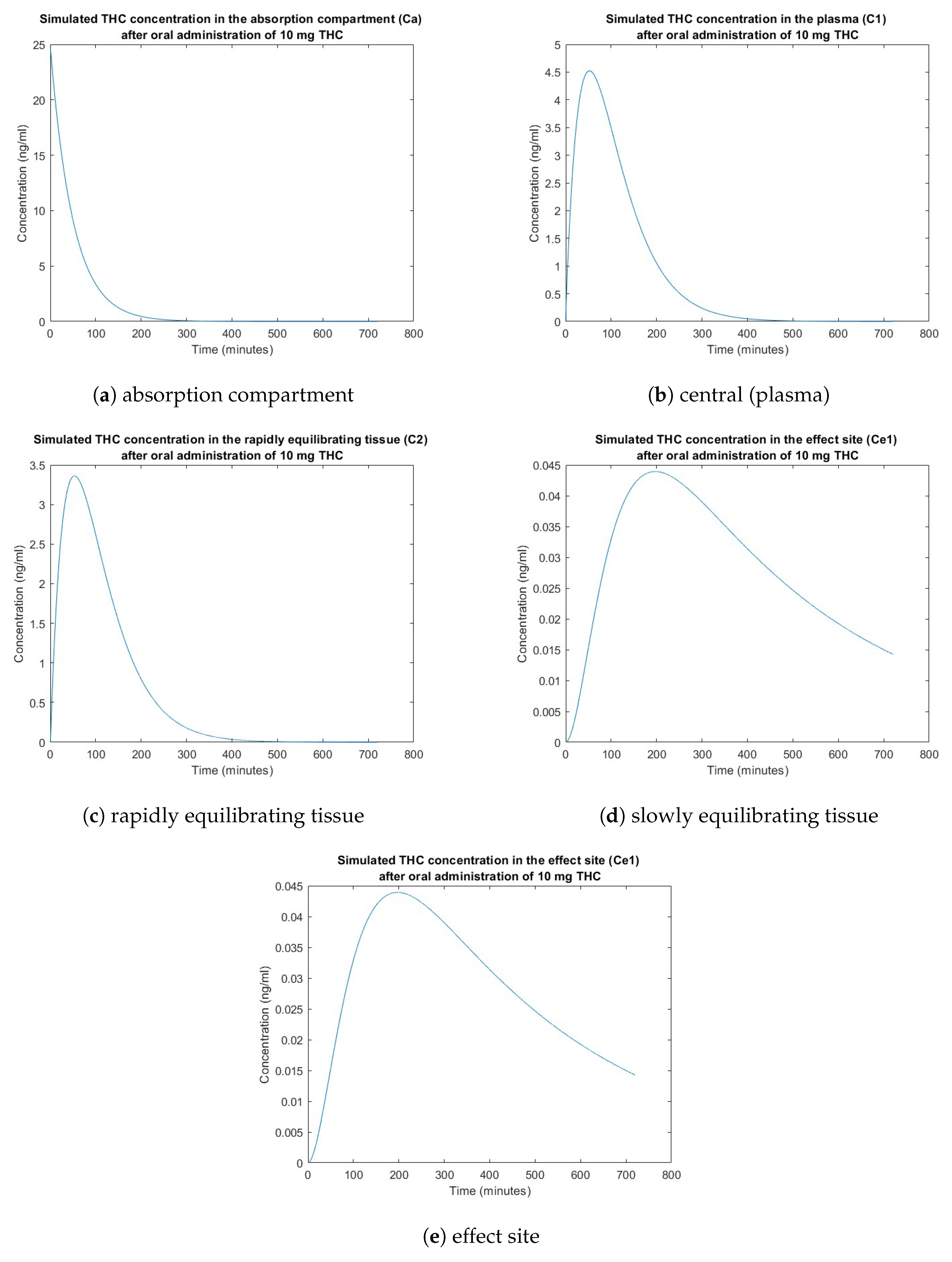
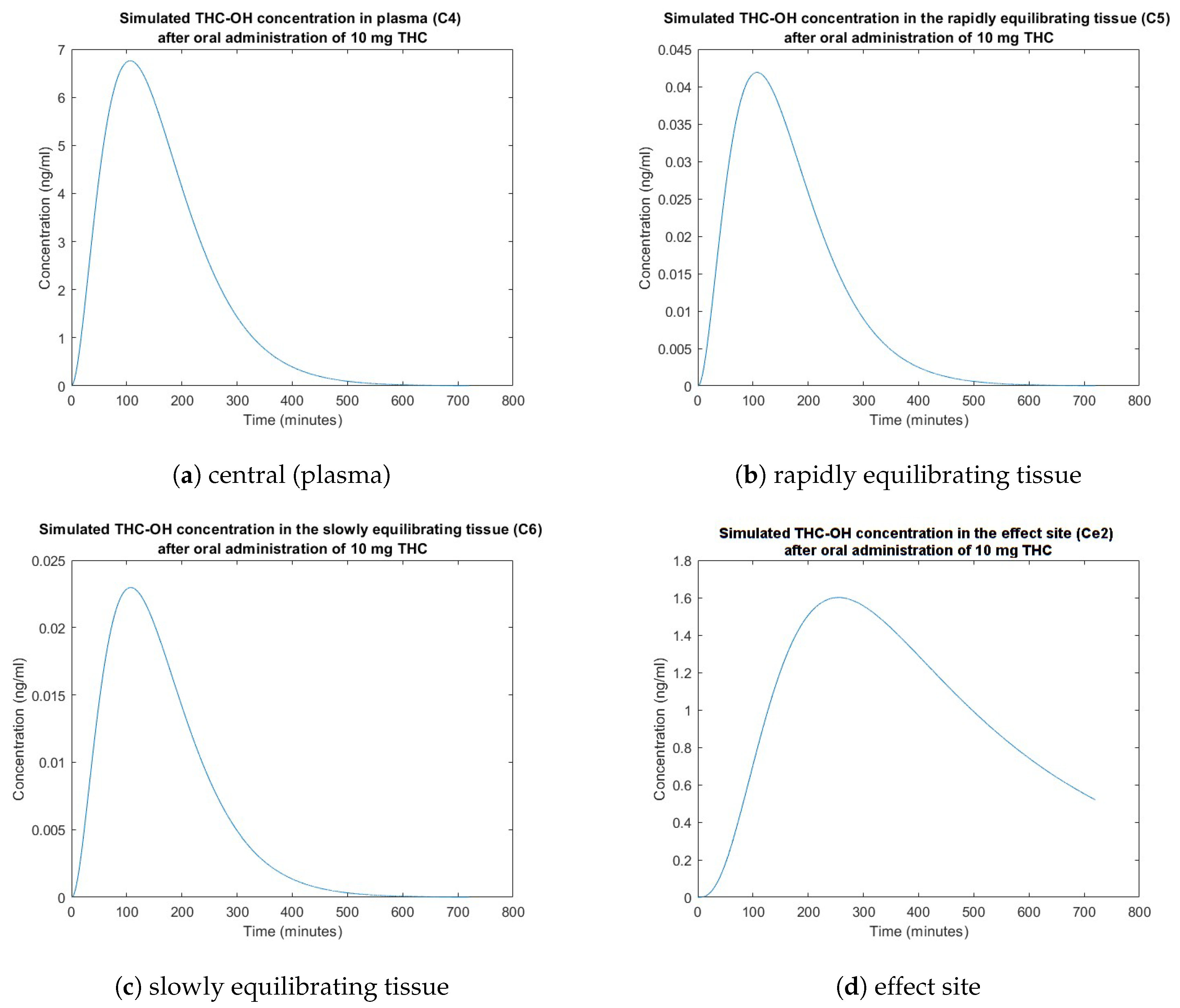
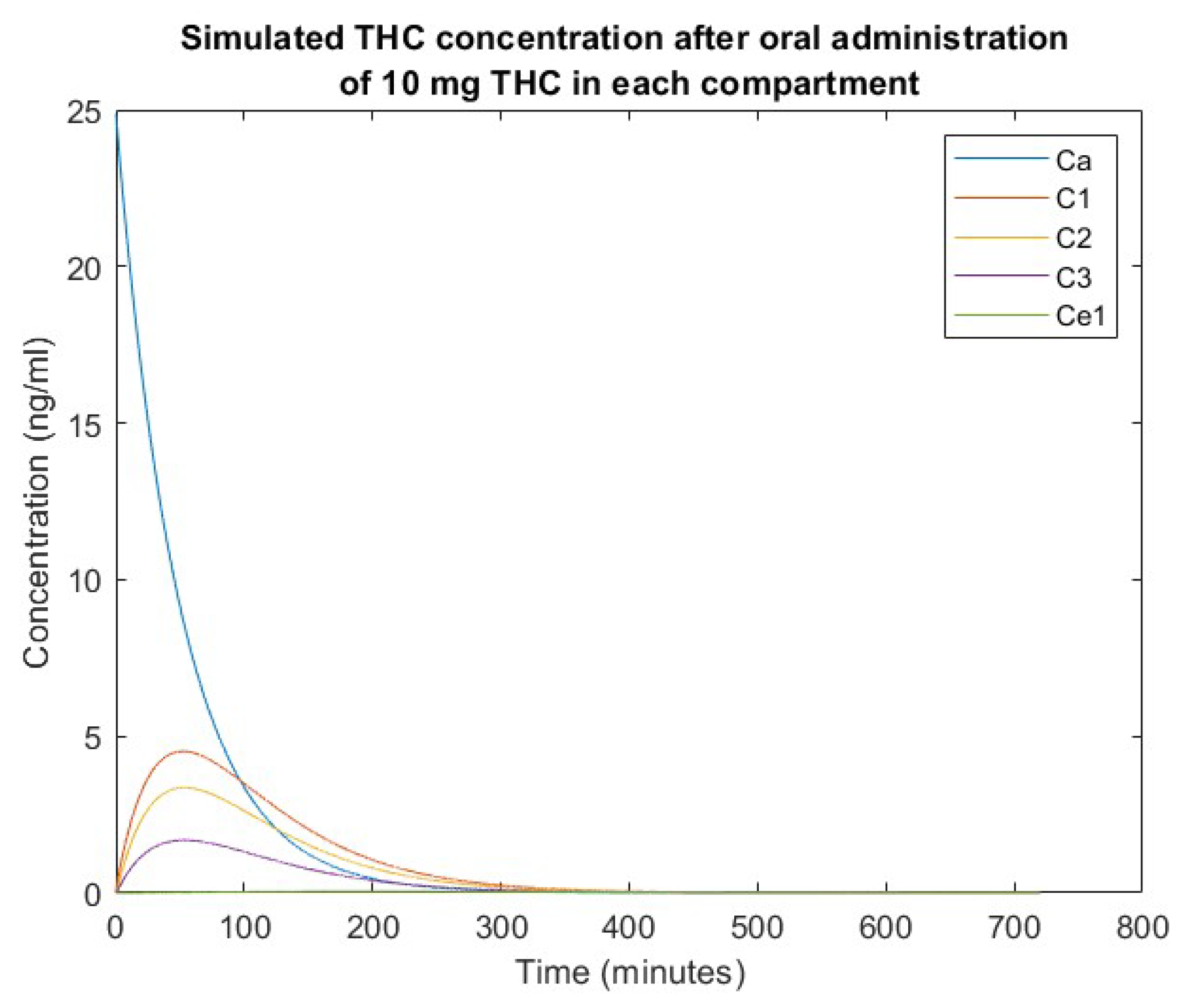
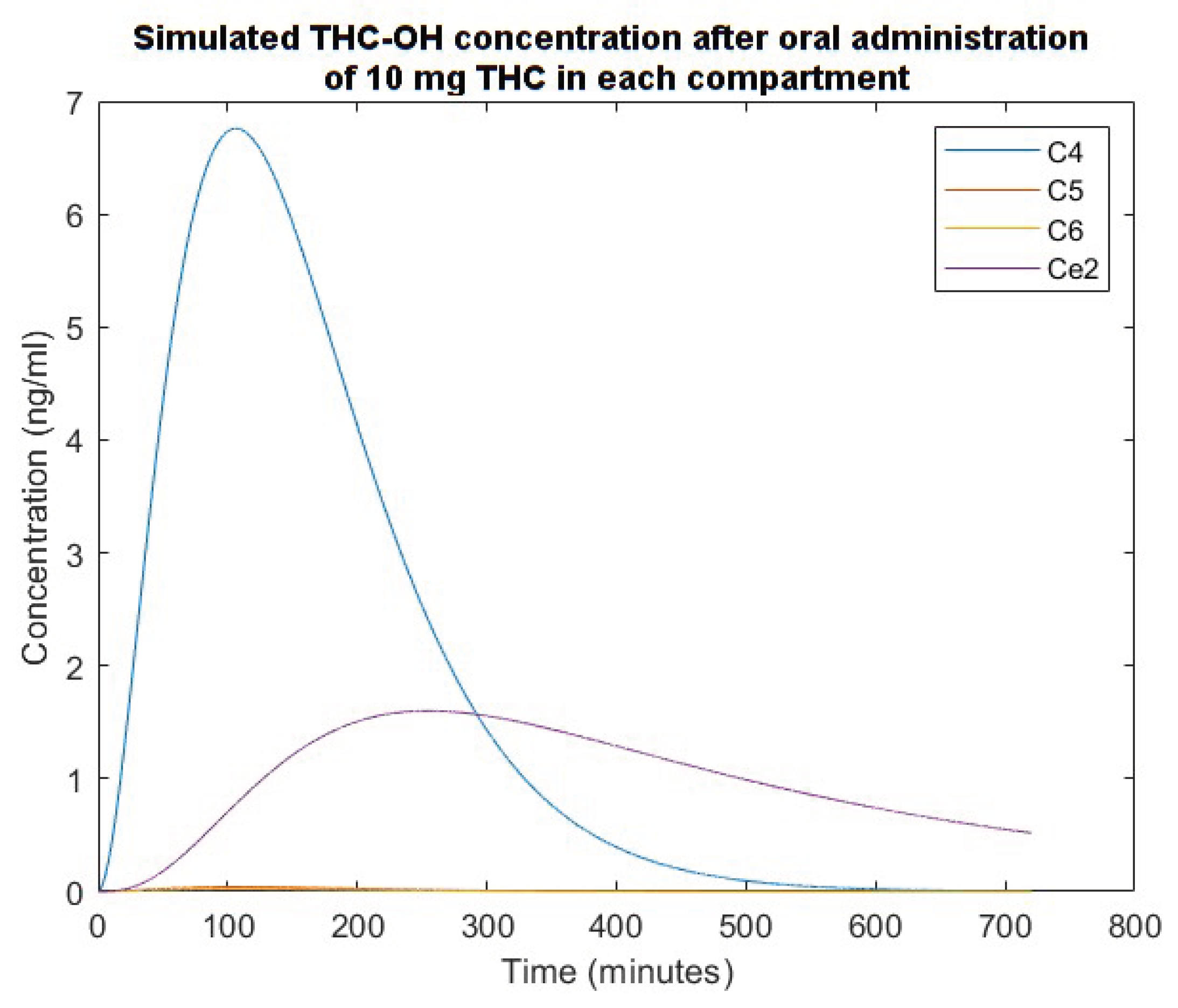
3. Simulation Results
4. Conclusion and Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| THC | Delta9-tetrahydrocannabinol |
| THC-OH | 11-hydroxy-delta9-tetrahydrocannabinol |
| THCCOOH | 11-nor-9-carboxy-delta9-tetrahydrocannabinol |
| NSFD | Nonstandard finite difference |
References
- Woodbridge, M. A Primer to Medicinal Cannabis: An introductory text to the therapeutic use of cannabis, Bedrocan International 2018, 1-57.
- Hazekamp, A.; Grotenhermen, F. Review on clinical studies with cannabis and cannabinoids 2005-2009. Cannabinoids; 2010; 5, pp. 1–21. [Google Scholar]
- Whiting, P.F.; Wolff, R.F.; Deshpande, S.; Di Nisio, M.; Duffy, S.; Hernandez, A.V.; Keurentjes, J.C.; Lang, S.; Misso, K.; Ryder, S.; Schmidlkofer, S.; Westwood, M.; Kleijnen, J. Cannabinoids for Medical Use: A Systematic Review and Meta-analysis. JAMA 2015, 313, 2456–2473. [Google Scholar] [CrossRef] [PubMed]
- Mahahong, T.; Saleewong, T. A Compartment Pharmacokinetics Model of THC and Its Metabolites after Smoking. Eng. Proc. 2023, 55, 4. [Google Scholar] [CrossRef]
- Wall, M.E.; Sadler, B.M.; Brine, D.; Taylor, H.; Perez-Reyes, M. Metabolism, disposition, and kinetics of delta-9-tetrahydrocannabinol in men and women. Clin. Pharmacol. Ther. 1983, 34(3), 352–363. [Google Scholar] [CrossRef] [PubMed]
- Abouchedid, R.; Ho, J.H.; Hudson, S.; Dines, A.; Archer, J.R.; Wood, D.M.; Dargan, P.I. Acute toxicity associated with use of 5F-derivations of synthetic cannabinoid receptor agonists with analytical confirmation. J. Med. Toxicol. 2016, 12(4), 396–401. [Google Scholar] [CrossRef] [PubMed]
- Huestis, M.A.; Henningfield, J.E.; Cone, E.J. Blood cannabinoids. I. Absorption of THC and formation of 11-OH-THC and THCCOOH during and after smoking marijuana. J. Anal. Toxicol. 1992, 16, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Heuberger, J.A.; Guan, Z.; Oyetayo, O.O.; Klumpers, L.; Morrison, P.D.; Beumer, T.L.; van Gerven, J.M.; Cohen, A.F.; Freijer, J. Population Pharmacokinetic Model of THC Integrates Oral, Intravenous, and Pulmonary Dosing and Characterizes Short- and Long-term Pharmacokinetics. Clinical Pharmacokinetics 2015, 54, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, R. Application of Modeling-based Approaches to Study the Pharmacokinetics and Pharmacodynamics of Delta-9-Tetrahydrocannabinol (THC) and Its Active Metabolite, The University of Iowa 2017.
- Sempio, C.; Huestis, M.A.; Mikulich-Gilbertson, S.K.; Klawitter, J.; Christians, U.; Henthorn, T.K. Population pharmacokinetic modeling of plasma Delta9-tetrahydrocannabinol and an active and inactive metabolite following controlled smoked cannabis administration. Br. J. Clin. Pharmacol. 2020, 86(3), 611–619. [Google Scholar] [CrossRef] [PubMed]
- Mickens, R.E. Dynamic consistency: a fundamental principle for constructing nonstandard finite difference schemes for differential equations. Journal of Difference Equations and Applications 2005, 11, 645–653. [Google Scholar] [CrossRef]
- Mickens, R.E. Calculation of Denominator Functions for Nonstandard Finite Difference Schemes for Differential Equations Satisfying a Positivity Condition. Numerical Methods for Partial Differential Equations 2006, 23, 672–691. [Google Scholar] [CrossRef]
- Egbelowo, O.; Harley, C.; Jacobs, B. Nonstandard Finite Difference Method Applied to a Linear Pharmacokinetics Model. Bioengineering 2017, 4, 40. [Google Scholar] [CrossRef] [PubMed]
- Egbelowo, O. Nonlinear Elimination of Drug in One-Compartment Pharmacokinetic Models: Nonstandard Finite Difference Approach for Various Routes of Administration. Mathematical and Computational Applications 2018, 23, 27. [Google Scholar] [CrossRef]
- Egbelowo, O.F. Nonstandard finite difference approach for solving 3-compartment pharmacokinetic models. Int. J. Numer. Methods Biomed. Eng. 2018, 34(9), e3114. [Google Scholar] [CrossRef] [PubMed]
- Saadah, A.M.; Widodo, I. Drug Elimination in Two-Compartment Pharmacokinetic Models with Nonstandard Finite Difference Approach. IAENG International Journal of Applied Mathematics 2020, 50, 1–7. [Google Scholar]
- Guy, G.W.; Robson, P.J. A Phase I, Open Label, Four-Way Crossover Study to Compare the Pharmacokinetic Profiles of a Single Dose of 20 mg of a Cannabis Based Medicine Extract (CBME) Administered on 3 Different Areas of the Buccal Mucosa and to Investigate the Pharmacokinetics of CBME per Oral in Healthy Male and Female Volunteers. Journal of Cannabis 2003, 3, 79–120. [Google Scholar]
- Nadulski, T.; Sporkert, F.; Schnelle, M.; Stadelmann, A.M.; Roser, P.; Schefter, T.; Pragst, F. Simultaneous and sensitive analysis of THC, 11-OH-THC, THC-COOH, CBD, and CBN by GC-MS in plasma after oral application of small doses of THC and cannabis extract. J Anal. Toxicol. 2005, 29(8), 782–789. [Google Scholar] [CrossRef] [PubMed]
| Parameters | Value | Unit | Parameters | Value | Unit |
|---|---|---|---|---|---|
| 24.85 | ng/ml | 0.0001 | |||
| 0.02 | 0.0150 | ||||
| 0.7438 | 0.0062 | ||||
| 1.0000 | 1.0000 | ||||
| 0.3718 | 0.0034 | ||||
| 1.0000 | 1.0000 | ||||
| 0.0380 | 0.0015 | ||||
| 0.0001 | 0.0020 | ||||
| 0.0025 | 0.0030 |
| Time (min) | THC (ng/ml) | THC-OH (ng/ml) |
|---|---|---|
| 0 | 0.00 | 0.00 |
| 15 | 0.08 | 0.04 |
| 30 | 2.94 | 2.59 |
| 45 | 4.97 | 5.82 |
| 60 | 4.29 | 6.19 |
| 75 | 4.23 | 6.75 |
| 90 | 3.94 | 6.50 |
| 105 | 3.09 | 5.78 |
| 120 | 2.57 | 5.13 |
| 135 | 2.34 | 4.71 |
| 150 | 2.04 | 4.18 |
| 165 | 2.02 | 3.71 |
| 180 | 1.80 | 3.59 |
| 210 | 1.17 | 2.69 |
| 240 | 0.88 | 2.30 |
| 270 | 0.79 | 1.91 |
| 300 | 0.56 | 1.54 |
| 330 | 0.39 | 1.23 |
| 360 | 0.31 | 1.08 |
| 480 | 0.17 | 0.73 |
| 720 | 0.13 | 0.48 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




