Submitted:
19 April 2024
Posted:
23 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Ethical Aspects
2.2. Participants
2.3. Questionnaire Development
2.4. Statistical Analysis
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Union of Innovative Pharmaceutical Companies INFARMA, the Polish Association of Clinical Research Organizations POLCRO. Report Industry Clinical Trials in Poland. Possibilities to increase number and scope of trials in Poland, 2021; pp. 16-17, 22-23.
- Union of Innovative Pharmaceutical Companies INFARMA, Association for Good Clinical Practice in Poland GCPpl, the Polish Association of Clinical Research Organizations POLCRO: Report Clinical Trials in Poland, 2015; pp.5.
- The Office for Registration of Medicinal Products, Medical Devices and Biocidal Products: Bulletin of products human medicinal products, 2016; pp. 2.
- The Office for Registration of Medicinal Products, Medical Devices and Biocidal Products: Bulletin of products human medicinal products, 2019; pp. 1.
- The Office for Registration of Medicinal Products, Medical Devices and Biocidal Products: Bulletin of products human medicinal products, 2022; pp. 6.
- The Office for Registration of Medicinal Products, Medical Devices and Biocidal Products: Annual Report, 2022; pp.51.
- Brodniewicz Teresa: Clinical Trials. CeDeWu 2016; pp. 48-49.
- Lara, Primo & Paterniti, Debora & Chiechi, Christine & Turrell, Corinne & Morain, Claudia & Horan, Nora & Montell, Lisa & Gonzalez, Jose & Davis, Sharon & Umutyan, Ari & Martel, Cynthia & Gandara, David & Beckett, Laurel & Chen, Moon. (2006). Evaluation of Factors Affecting Awareness of and Willingness to Participate in Cancer Clinical Trials. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 23. 9282-9. [CrossRef]
- MD, Jean & PhD, MPH & MHS, Gabriel & PhD, MHS & MD, MPH & MD, MPH & MD, MPH & PhD, MPH & BA, Teerath & MS, Renee & MBA, Neil & MD, MPH & Ford, Jean & Lai, Gabriel & Gary-Webb, Tiffany & Bolen, Shari & Gibbons, Michael & Tilburt, Jon & Baffi, Charles & Bass, Eric. (2008). Barriers to Recruiting Underrepresented Populations to Cancer Clinical Trials: A Systematic Review. Cancer. 112. 228 - 242. [CrossRef]
- Quality Certificate of Ariadna Company. Available online: https://www.pkjpa.pl/certyfikat-jakosci/ (accessed on 25 August 2023).
- Ariadna Company. Available online: https://panelariadna.pl/wiedza/badania-cawi (accessed on 25 August 2023).
- Sridharan, Kannan & Mehta, Mansi & Sivaramakrishnan, Gowri. (2016). Awareness and attitude of general public about clinical trials in a developing country. American Journal of Experimental and Clinical Research. 33. 146-148.
- Abu Farha, Rana & Alzoubi, Karem & Khabour, Omar & Mukattash, Tareq. (2020). Factors Influencing Public Knowledge and Willingness to Participate in Biomedical Research in Jordan: A National Survey. Patient Preference and Adherence. Volume 14. 1373-1379. [CrossRef]
- Sridharan, Kannan & Mehta, Mansi & Sivaramakrishnan, Gowri. (2016). Awareness and attitude of general public about clinical trials in a developing country. American Journal of Experimental and Clinical Research. 33. 146-148.
- Staniszewska, Anna & Lubiejewska, Adriana & Czerw, Aleksandra & Dąbrowska-Bender, Marta & Duda-Zalewska, Aneta & Olejniczak, Dominik & Juszczyk, Grzegorz & Bujalska-Zadrozny, Magdalena. (2018). Awareness and attitudes towards clinical trials among Polish oncological patients who had never participated in a clinical trial. Advances in clinical and experimental medicine: official organ Wroclaw Medical University. 27. [CrossRef]
- Leiter, Amanda & Diefenbach, Michael & Doucette, John & Oh, William & Galsky, Matt. (2015). Clinical trial awareness: Changes over time and sociodemographic disparities. Clinical trials (London, England). 12. [CrossRef]
- Figer, BrinalH & Lamture, SwastikaS & Gandhi, Tanmay & Chauhan, Ashnik & Gvalani, Anchal & Gogtay, NithyaJaideep & Thatte, UrmilaMukund. (2020). A survey of knowledge and variables influencing perceptions about clinical research: A cross-sectional study from Mumbai. Perspectives in Clinical Research. 12. 10.4103/picr.PICR_97_19.
- Burt, Tal & Dhillon, Savita & Sharma, Pooja & Khan, Danish & Mv, Deepa & Alam, Sazid & Jain, Sarika & Alapati, Bhavana & Mittal, Sanjay & Singh, Padam. (2013). PARTAKE Survey of Public Knowledge and Perceptions of Clinical Research in India. PloS one. 8. e68666. 10.1371/journal.pone.0068666.
- The Need for Awareness of Clinical Research, National Institutes of Health 2016.
- Wp.pl. Available online: https://finanse.wp.pl/mozesz-dorobic-3-tys-zl-musisz-byc-zdrowy-i-pelnoletni-6114350320687233a?ticaid=118dc2 (accessed on 25 August 2023).
- Rp.pl. Available online: https://www.rp.pl/ochrona-zdrowia/art16792241-polowanie-na-kroliki-doswiadczalne (accessed on 25 August 2023).
- Vanity Fair, 2014.
- Mackenzie, Isla & Wei, li & Rutherford, Daniel & Findlay, Evelyn & Saywood, Wendy & Campbell, Marion & Macdonald, Thomas. (2010). Promoting public awareness of randomised clinical trials using the media: The 'Get Randomised' campaign. British journal of clinical pharmacology. 69. 128-35. [CrossRef]
- Williams, Courtney & Everson, Nicole & Shelburne, Nonniekaye & Norton, Wynne. (2021). Demographic and Health Behavior Factors Associated WithClinical Trial Invitation and Participation in the United States. JAMA Network Open. 4. e2127792. [CrossRef]
- Patel EU, Zhu X, Quinn TC, Tobian AAR. Public Knowledge and Attitudes Toward Clinical Trials in the COVID-19 Era. Am J Prev Med. 2022 Mar;62(3):469-471. [CrossRef]
- Research! America Public Perception of Clinical Trials, 2017 America Speaks: Poll Data Summary.
- Morris MC, Besner D, Vazquez H, Nelson RM, Fischbach RL. Parental opinions about clinical research. J Pediatr. 2007 Nov;151(5):532-7, 537.e1-5. [CrossRef]
- Schupmann, Will & Li, Xiaobai & Wendler, David. (2021). Do the Potential Medical Benefits of Phase 1 Pediatric Oncology Trials Justify the Risks? Views of the US Public. The Journal of Pediatrics. 238. [CrossRef]
- Andrade, C. The Limitations of Online Surveys. Indian J. Psychol. Med. 2020, 42, 575–576. [Google Scholar] [CrossRef] [PubMed]
- Kevin, B. A: Researching Internet-Based Populations: Advantages and Disadvantages of Online Survey Research, Online Questionnaire Authoring Software Packages, and Web Survey Services, Journal of Computer-Mediated Communication, Volume 10, Issue 3, 1 April 2005.
- The Centre for Social Opinion Research in Poland: Internet usage in 2023, June 2023; pp. 1.
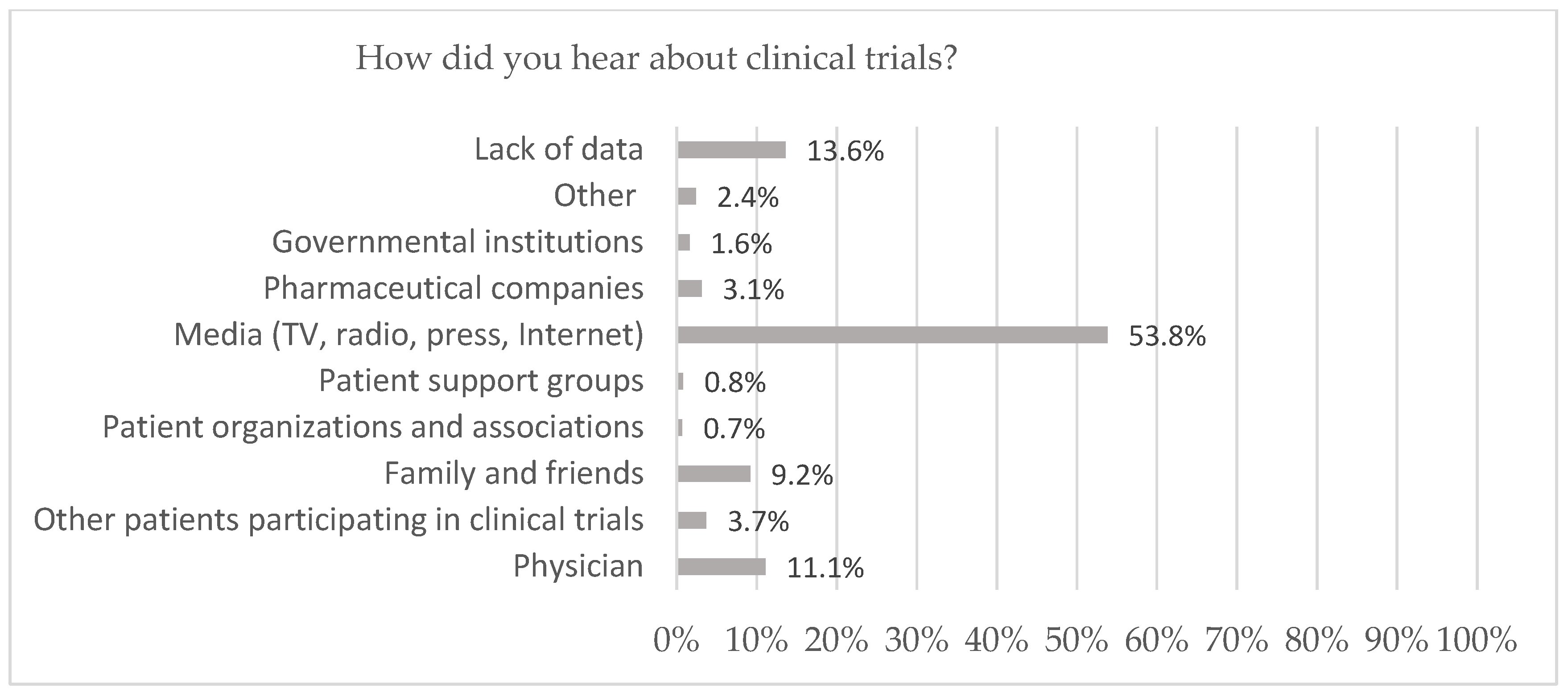
| N | (%) | ||
|---|---|---|---|
| Age (years), mean SD | 44.96 | ||
| 18-24 | 141 | 13.1 | |
| 25-34 | 216 | 20.0 | |
| 35-44 | 180 | 16.7 | |
| 45-54 | 187 | 17.3 | |
| 55 and more | 355 | 32.9 | |
| Sex | |||
| Female | 568 | 52.6 | |
| Male | 511 | 47.4 | |
| Region | |||
| Village | 404 | 37.4 | |
| Small town1 | 140 | 13.0 | |
| Medium town lower2 | 96 | 8.9 | |
| Medium town upper3 | 115 | 10.7 | |
| Large city lower4 | 87 | 8.1 | |
| Large city upper5 | 103 | 9.5 | |
| Big city6 | 134 | 12.4 | |
| Education | |||
| Primary or middle school | 40 | 3.7 | |
| Basic | 111 | 10.3 | |
| Secondary school | 338 | 31.3 | |
| Post secondary school | 111 | 10.3 | |
| Bachelor's degree | 96 | 8.9 | |
| Master's degree | 383 | 35.5 | |
| Voivodship | |||
| Dolnośląskie | 62 | 5.7 | |
| Kujawsko–pomorskie | 59 | 5.5 | |
| Łódzkie | 68 | 6.3 | |
| Lubelskie | 71 | 6.6 | |
| Lubuskie | 31 | 2.9 | |
| Małopolskie | 97 | 9.0 | |
| Mazowieckie | 155 | 14.4 | |
| Opolskie | 24 | 2.2 | |
| Podkarpackie | 63 | 5.8 | |
| Podlaskie | 48 | 4.4 | |
| Pomorskie | 55 | 5.1 | |
| Śląskie | 136 | 12.6 | |
| Świętokrzyskie | 34 | 3.2 | |
| Warminsko–mazurskie | 30 | 2.8 | |
| Wielkopolskie | 104 | 9.6 | |
| Zachodniopomorskie | 42 | 3.9 | |
| Family status | |||
| Single | 333 | 30.9 | |
| Married | 575 | 53.3 | |
| Divorced | 116 | 10.8 | |
| Widowed | 55 | 5.1 | |
| N | (%) | |
|---|---|---|
| Awareness of clinical trials | ||
| Yes | 933 | 86.5 |
| No | 146 | 13.5 |
| Self-reported knowledge about clinical | ||
| Very strong | 18 | 1.7 |
| Strong | 160 | 14.8 |
| A little | 466 | 43.2 |
| Nothing at all | 252 | 23.4 |
| Hard to say | 183 | 17.0 |
| Willingness to learn more about clinical trials | ||
| Definitely yes | 238 | 22.7 |
| Rather yes | 567 | 52.5 |
| Probably not | 120 | 11.1 |
| Definitely not | 28 | 2.6 |
| Hard to say | 126 | 11.7 |
| Have you ever taken part in a clinical trial? | ||
| Yes | 124 | 11.5 |
| No | 955 | 88.5 |
| Definitely yes |
Rather yes |
Probably not | Definitely not | Hard to say |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | (%) | N | (%) | N | (%) | N | % | N | (%) | |
| Participation in a clinical trial is entirely voluntary | 392 | 36.3 | 487 | 45.1 | 39 | 3.6 | 9 | 0.8 | 152 | 14.1 |
| Hospitals participating in clinical trials provide better healthcare | 131 | 12.1 | 449 | 41.6 | 109 | 10.1 | 24 | 2.2 | 366 | 33.9 |
| The results of clinical trials are made available to the public | 52 | 4.8 | 321 | 29.7 | 313 | 29.0 | 89 | 8.2 | 304 | 28.2 |
| You can withdraw from a clinical trial at any time | 126 | 11.7 | 395 | 36.6 | 190 | 17.6 | 28 | 2.6 | 340 | 31.5 |
| The clinical trial patient is insured | 179 | 16.6 | 459 | 42.5 | 110 | 10.2 | 13 | 1.2 | 318 | 29.5 |
| All clinical trial patients receive an investigational medicinal product | 59 | 5.5 | 259 | 24.0 | 243 | 22.5 | 228 | 21.1 | 290 | 26.9 |
| Clinical trials should be conducted with children | 70 | 6.5 | 257 | 23.8 | 226 | 20.9 | 160 | 14.8 | 366 | 33.9 |
| I would allow my child to participate in a clinical trial as the only therapeutic option (there is no other treatment) | 237 | 22.0 | 459 | 42.5 | 74 | 6.9 | 66 | 6.1 | 243 | 22.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions, or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




