Submitted:
26 March 2024
Posted:
01 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Patients
Plasma
Bone Marrow Mesenchymal Stromal Cells Isolation
Cell Culture
Determination of Glypicans and Notum in Plasma and Cell Culture Supernatants
mRNA Analysis
Statistical Analysis
3. Results
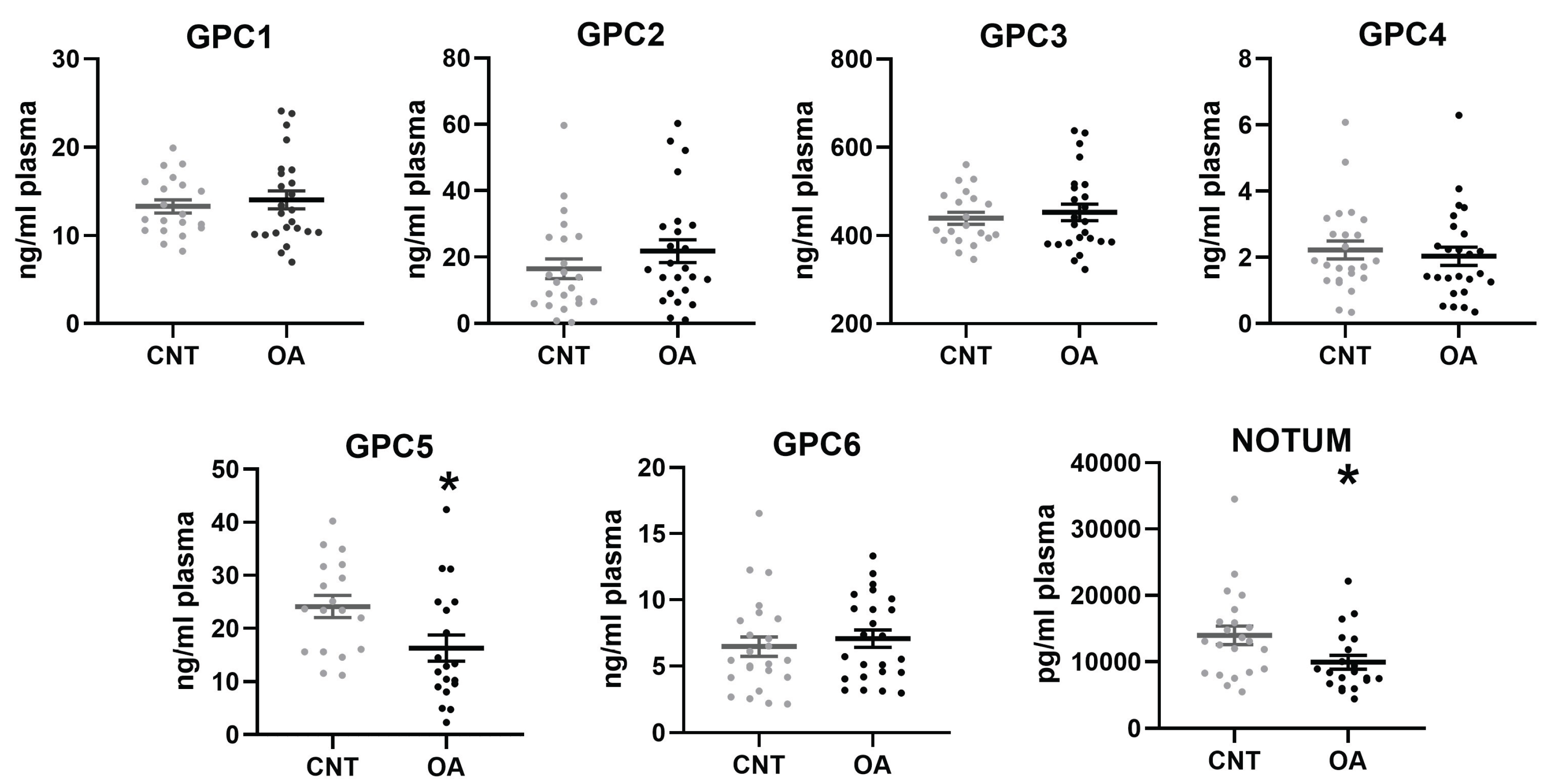
3.1. Circulating Levels of Glypicans and Notum
3.1.1. Demographics Characteristics of the Study Population
3.1.2. Plasma Levels in Controls and Patients with Osteoarthritis
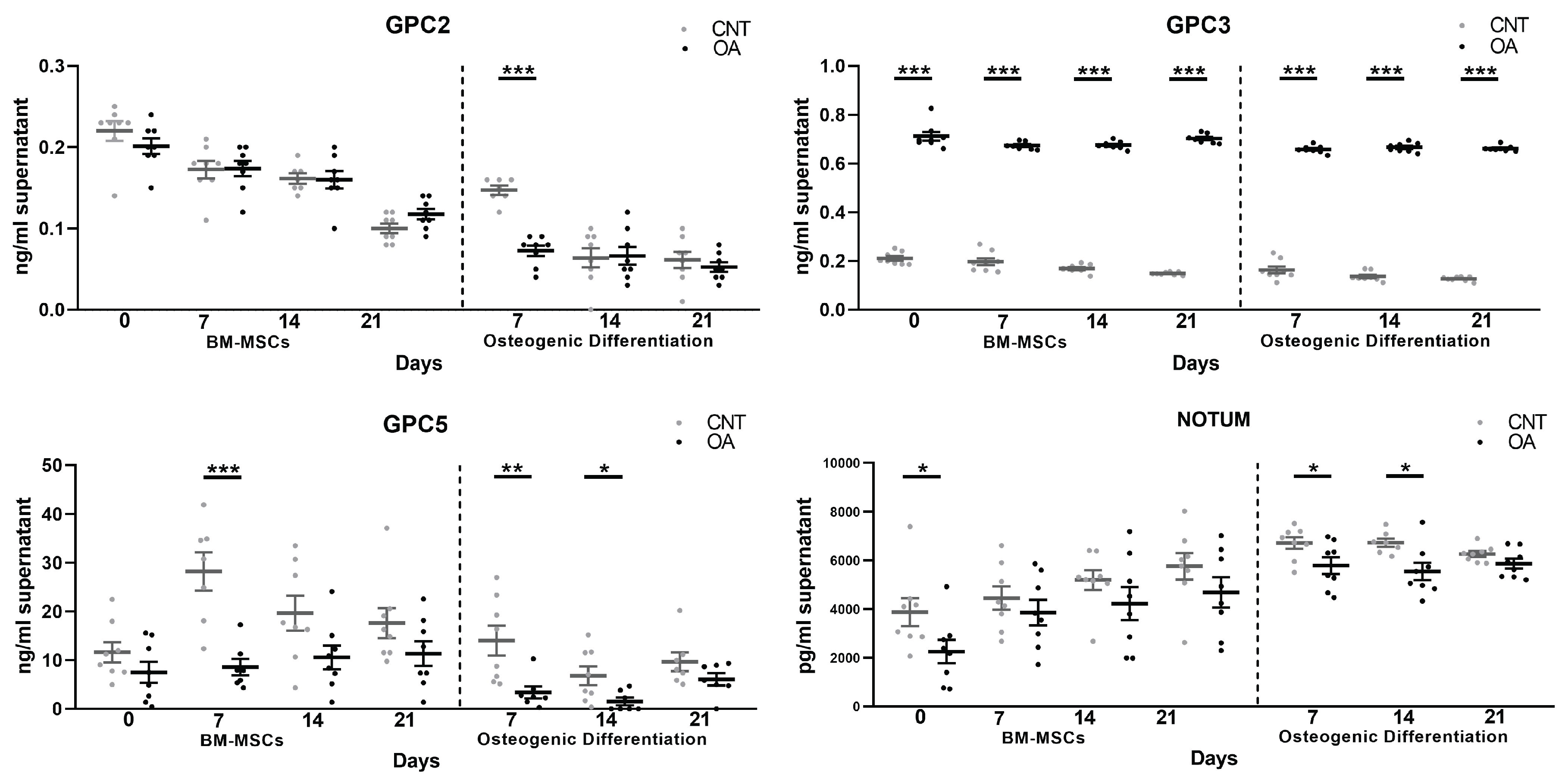
3.2. BM-MSCs and Osteoblasts
3.2.1. Expression Gene MSCs and during Osteogenic Differentiation
3.2.2. Protein Levels in Supernatant
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Palazzo, C.; Nguyen, C.; Lefevre-Colau, M.M.; Rannou, F.; Poiraudeau, S. Risk factors and burden of osteoarthritis. Ann Phys Rehabil Med. 2016, 59, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Martel-Pelletier, J.; Barr, A.J.; Cicuttini, F.M.; et al. Osteoarthritis. Nat Rev Dis Prim. 2016, 2, 16072. [Google Scholar] [CrossRef] [PubMed]
- Hunter, D.J.; McDougall, J.J.; Keefe, F.J. The Symptoms of Osteoarthritis and the Genesis of Pain. Rheum Dis Clin North Am. 2008, 34, 623–643. [Google Scholar] [CrossRef] [PubMed]
- Hunter, D.J.; Felson, D.T. Osteoarthritis. BMJ 2006, 332, 639–642. [Google Scholar] [CrossRef] [PubMed]
- Uccelli, A.; Moretta, L.; Pistoia, V. Mesenchymal stem cells in health and disease. Nat Rev Immunol. 2008, 8, 726–736. [Google Scholar] [CrossRef] [PubMed]
- Murphy, J.M.; Dixon, K.; Beck, S.; Fabian, D.; Feldman, A.; Barry, F. Reduced chondrogenic and adipogenic activity of mesenchymal stem cells from patients with advanced osteoarthritis. Arthritis Rheum. 2002, 46, 704–713. [Google Scholar] [CrossRef] [PubMed]
- Kwan Tat, S.; Lajeunesse, D.; Pelletier, J.P.; Martel-Pelletier, J. Targeting subchondral bone for treating osteoarthritis: what is the evidence? Best Pract Res Clin Rheumatol. 2010, 24, 51–70. [Google Scholar] [CrossRef] [PubMed]
- Duan, P.; Bonewald, L.F. The role of the wnt/β-catenin signaling pathway in formation and maintenance of bone and teeth. Int J Biochem Cell Biol. 2016, 77, 23–29. [Google Scholar] [CrossRef]
- Escobar-Gómez, F.; Jódar, E.; Hawkins, F. Receptor Wnt: fisiología, fisiopatología y potenciales nuevas dianas terapéuticas. Rev Española Enfermedades Metabólicas Óseas. 2009, 18, 39–44. [Google Scholar] [CrossRef]
- Corr, M. Wnt–β-catenin signaling in the pathogenesis of osteoarthritis. Nat Clin Pract Rheumatol. 2008, 4, 550–556. [Google Scholar] [CrossRef]
- Tornero-Esteban, P.; Peralta-Sastre, A.; Herranz, E.; et al. Altered Expression of Wnt Signaling Pathway Components in Osteogenesis of Mesenchymal Stem Cells in Osteoarthritis Patients. Shi XM, ed. PLoS One 2015, 10, e0137170. [Google Scholar] [CrossRef]
- Blom, A.B.; Brockbank, S.M.; van Lent, P.L.; et al. Involvement of the Wnt signaling pathway in experimental and human osteoarthritis: Prominent role of Wnt-induced signaling protein 1. Arthritis Rheum. 2009, 60, 501–512. [Google Scholar] [CrossRef]
- Held, A.; Glas, A.; Dietrich, L.; et al. Targeting β-catenin dependent Wnt signaling via peptidomimetic inhibitors in murine chondrocytes and OA cartilage. Osteoarthr Cartil. 2018, 26, 818–823. [Google Scholar] [CrossRef] [PubMed]
- Filmus, J.; Capurro, M.; Rast, J. Glypicans. Genome Biol. 2008, 9, 224. [Google Scholar] [CrossRef] [PubMed]
- Boudin, E.; Fijalkowski, I.; Piters, E.; Van Hul, W. The role of extracellular modulators of canonical Wnt signaling in bone metabolism and diseases. Semin Arthritis Rheum. 2013, 43, 220–240. [Google Scholar] [CrossRef]
- Kaur, S.P.; Verma, A.; Lee, H.K.; Barnett, L.M.; Somanath, P.R.; Cummings, B.S. Inhibition of glypican-1 expression induces an activated fibroblast phenotype in a human bone marrow-derived stromal cell-line. Sci Rep. 2021, 11, 9262. [Google Scholar] [CrossRef] [PubMed]
- Nie, J.; Yang, T.; Li, H.; et al. Frequently expressed glypican-3 as a promising novel therapeutic target for osteosarcomas. Cancer Sci. 2022, 113, 3618–3632. [Google Scholar] [CrossRef]
- Dwivedi, P.P.; Grose, R.H.; Filmus, J.; et al. Regulation of bone morphogenetic protein signalling and cranial osteogenesis by Gpc1 and Gpc3. Bone 2013, 55, 367–376. [Google Scholar] [CrossRef]
- LeClair, E.E.; Mui, S.R.; Huang, A.; Topczewska, J.M.; Topczewski, J. Craniofacial skeletal defects of adult zebrafish Glypican 4 (knypek) mutants. Dev Dyn. 2009, 238, 2550–2563. [Google Scholar] [CrossRef]
- Amor, D.J.; Stephenson, S.E.M.; Mustapha, M.; et al. Pathogenic Variants in GPC4 Cause Keipert Syndrome. Am J Hum Genet. 2019, 104, 914–924. [Google Scholar] [CrossRef]
- Trajanoska, K.; Rivadeneira, F. The genetic architecture of osteoporosis and fracture risk. Bone 2019, 126, 2–10. [Google Scholar] [CrossRef]
- Kemp, J.P.; Morris, J.A.; Medina-Gomez, C.; et al. Identification of 153 new loci associated with heel bone mineral density and functional involvement of GPC6 in osteoporosis. Nat Genet. 2017, 49, 1468–1475. [Google Scholar] [CrossRef] [PubMed]
- Traister, A.; Shi, W.; Filmus, J. Mammalian Notum induces the release of glypicans and other GPI-anchored proteins from the cell surface. Biochem J. 2008, 410, 503–511. [Google Scholar] [CrossRef]
- Kakugawa, S.; Langton, P.F.; Zebisch, M.; et al. Notum deacylates Wnt proteins to suppress signalling activity. Nature 2015, 519, 187–192. [Google Scholar] [CrossRef]
- Lietman, C.; Wu, B.; Lechner, S.; et al. Inhibition of Wnt/β-catenin signaling ameliorates osteoarthritis in a murine model of experimental osteoarthritis. JCI Insight 2018, 3(3). [Google Scholar] [CrossRef]
- Deshmukh, V.; Hu, H.; Barroga, C.; et al. A small-molecule inhibitor of the Wnt pathway (SM04690) as a potential disease modifying agent for the treatment of osteoarthritis of the knee. Osteoarthr Cartil. 2018, 26, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Fan, X.; Xing, L.; Tian, F. Wnt signaling: a promising target for osteoarthritis therapy. Cell Commun Signal. 2019, 17, 97. [Google Scholar] [CrossRef] [PubMed]
- Song, H.H.; Shi, W.; Xiang, Y.Y.; Filmus, J. The Loss of Glypican-3 Induces Alterations in Wnt Signaling. J Biol Chem. 2005, 280, 2116–2125. [Google Scholar] [CrossRef]
- Capurro, M.; Martin, T.; Shi, W.; Filmus, J. Glypican-3 binds to frizzled and plays a direct role in the stimulation of canonical Wnt signaling. J Cell Sci. Published online January 1. 2014. [Google Scholar] [CrossRef]
- Sun, Y.; Xu, K.; He, M.; Fan, G.; Lu, H. Overexpression of Glypican 5 (GPC5) Inhibits Prostate Cancer Cell Proliferation and Invasion via Suppressing Sp1-Mediated EMT and Activation of Wnt/β-Catenin Signaling. Oncol Res Featur Preclin Clin Cancer Ther. 2018, 26, 565–572. [Google Scholar] [CrossRef]
- Udomsinprasert, W.; McConachie, E.; Ngarmukos, S.; Theerawattanapong, N.; Tanavalee, A.; Honsawek, S. Plasma and Joint Fluid Glypican-3 Are Inversely Correlated with the Severity of Knee Osteoarthritis. Cartilage 2021, 12, 505–511. [Google Scholar] [CrossRef]
- Torisu, Y.; Watanabe, A.; Nonaka, A.; et al. Human homolog of NOTUM, overexpressed in hepatocellular carcinoma, is regulated transcriptionally by β-catenin/TCF. Cancer Sci. 2008, 99, 1139–1146. [Google Scholar] [CrossRef] [PubMed]
- Movérare-Skrtic, S.; Nilsson, K.H.; Henning, P.; et al. Osteoblast-derived NOTUM reduces cortical bone mass in mice and the NOTUM locus is associated with bone mineral density in humans. FASEB J. 2019, 33, 11163–11179. [Google Scholar] [CrossRef] [PubMed]
- Capurro, M.; Martin, T.; Shi, W.; Filmus, J. Glypican-3 binds to frizzled and plays a direct role in the stimulation of canonical Wnt signaling. J Cell Sci. Published online January 1. 2014. [Google Scholar] [CrossRef] [PubMed]
- Hill, T.P.; Später, D.; Taketo, M.M.; Birchmeier, W.; Hartmann, C. Canonical Wnt/β-Catenin Signaling Prevents Osteoblasts from Differentiating into Chondrocytes. Dev Cell. 2005, 8, 727–738. [Google Scholar] [CrossRef] [PubMed]
- De Robertis, M.; Arigoni, M.; Loiacono, L.; et al. Novel insights into Notum and glypicans regulation in colorectal cancer. Oncotarget 2015, 6, 41237–41257. [Google Scholar] [CrossRef]
- Yang, P.; Li, C.; Kou, Y.; et al. Notum suppresses the osteogenic differentiation of periodontal ligament stem cells through the Wnt/Beta catenin signaling pathway. Arch Oral Biol. 2021, 130, 105211. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



