Submitted:
26 March 2024
Posted:
28 March 2024
Read the latest preprint version here
Abstract
Keywords:
Undeniable Evidence in Brief
 . The facts are well known in the fields of Biology, Chemistry and Physics as following:
. The facts are well known in the fields of Biology, Chemistry and Physics as following:- Osmosis: two types of fluids can be separated by a semipermeable membrane like cellophane, frog’s skin, sheep bladder or fish bladder. The separated fluids can have different vapour pressures.
- Vapour Pressure and Raoult's Law: the vapour pressure of different liquids are different based on the solute molecules concentration at the same temperature.
- Pressure Gradient: Pressure gradient including vapour pressure gradient is a known source of energy.

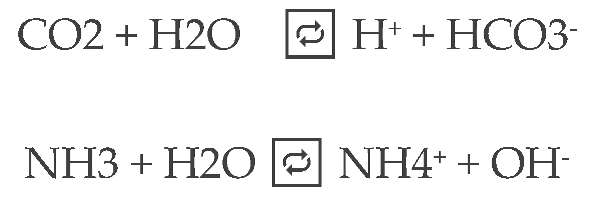
Extended Gibbs Donnan Equilibrium
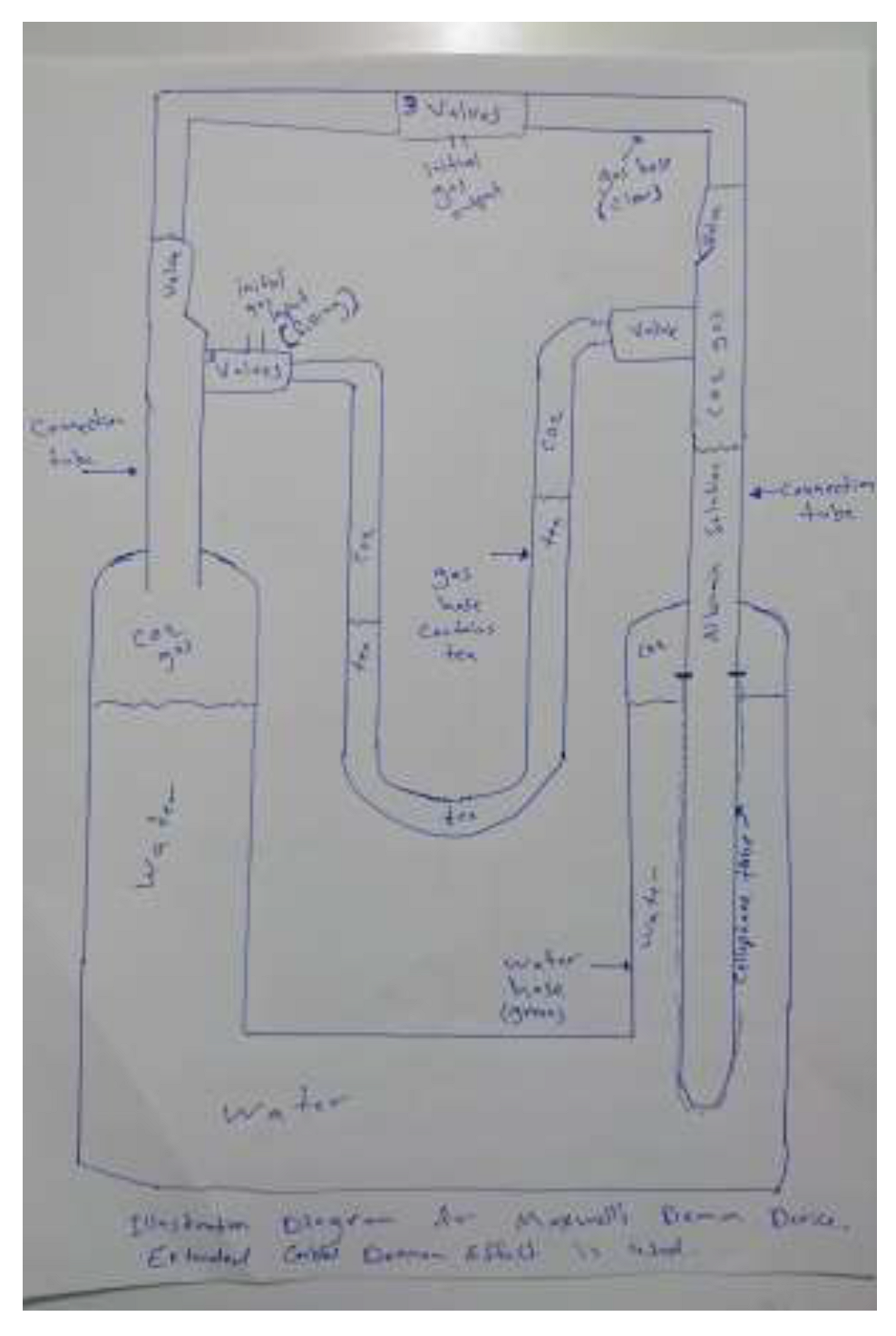
Experimental Evidence
- Cellophane Dialysis Tube: Cellophane is commonly used by laboratories for demonstration of osmosis.

- Tubes with valves: for leak free connection of gases and liquids.
- Egg white Albumin: as a source of albumin and carbonic anhydrase enzyme.
- Carbon Dioxide: we produced CO2 from reaction of citric acid with sodium bicarbonate (baking soda) solution.
- Gas hose with tea as colouring agent : we used U shaped hose filled with tea simplest sensitive manometer in our experiment.

Energy Booster
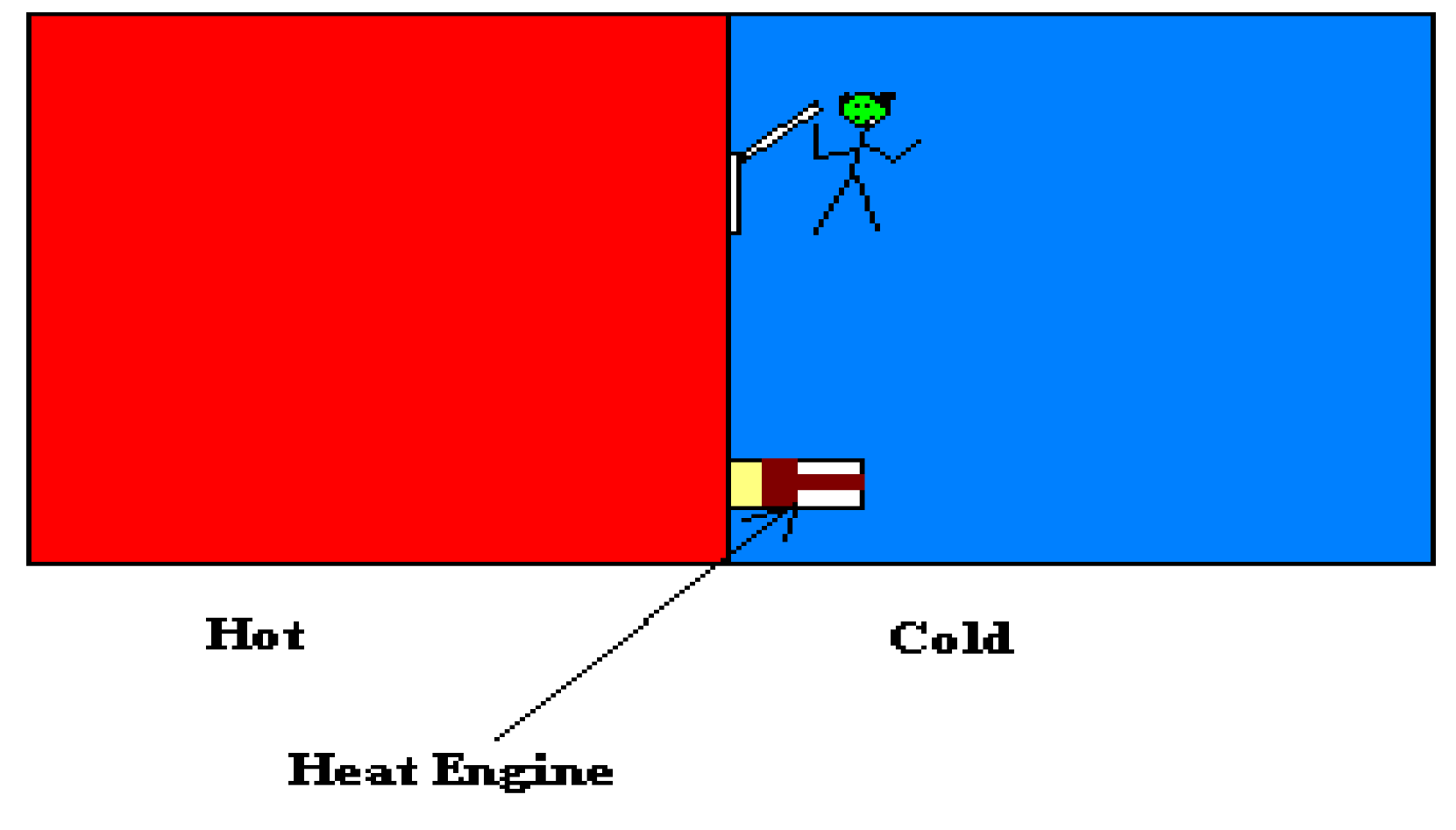
Maxwell’s Demon (Thought Experiment)
Three Versions of Maxwell’s Demon
- Selective demon: as shown above, high and low energy particles are separated by the demon.
- One way demon: this demon act like a valve only permits particles in one direction regardless of their energy state.
- Selectively One way demon: as we presented in our experiments above, this type of demon allow some particles to move more in one direction than the opposite direction. In our example the water vapour moves into the solution more than out of it as described by Raoult’s Law and the cycle maintained by the semipermeable membrane .
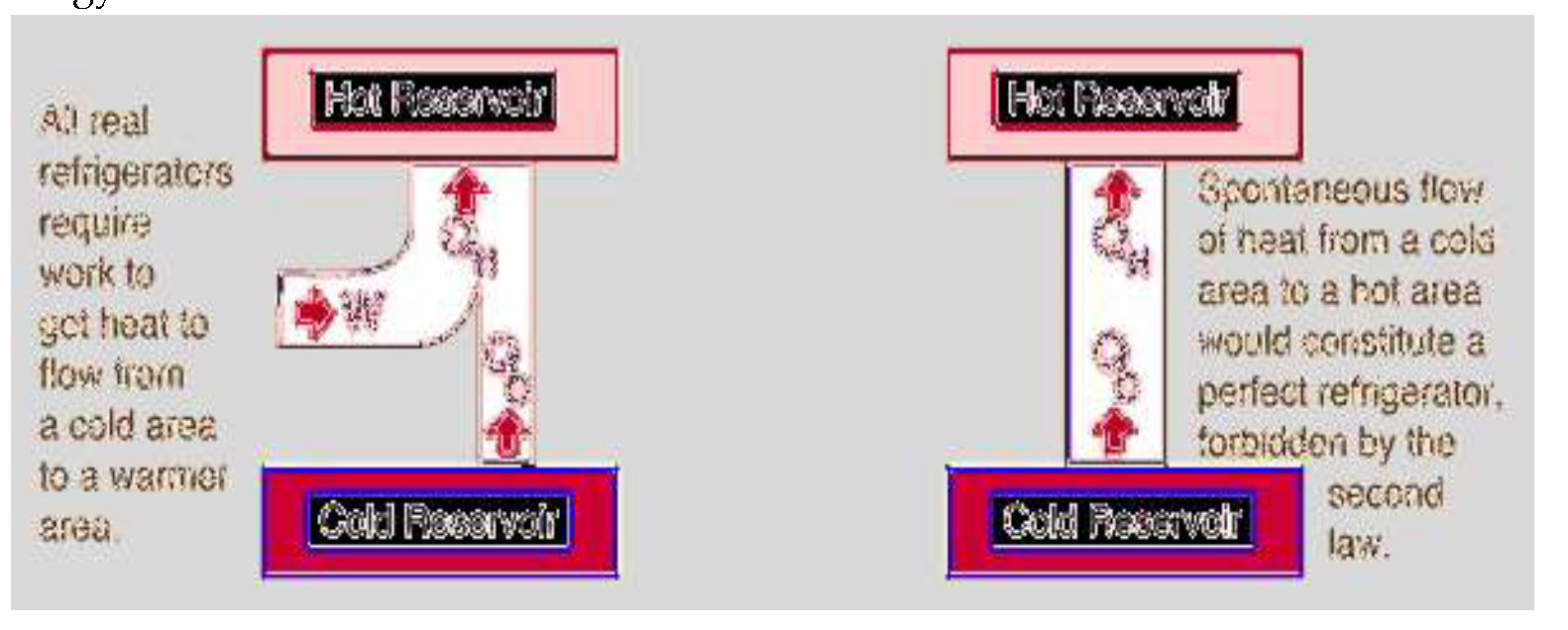
Breaking The Second Law of Thermodynamics
Brownian Motion
Brownian Ratchet or Feynman’s Ratchet (Though Experiment)
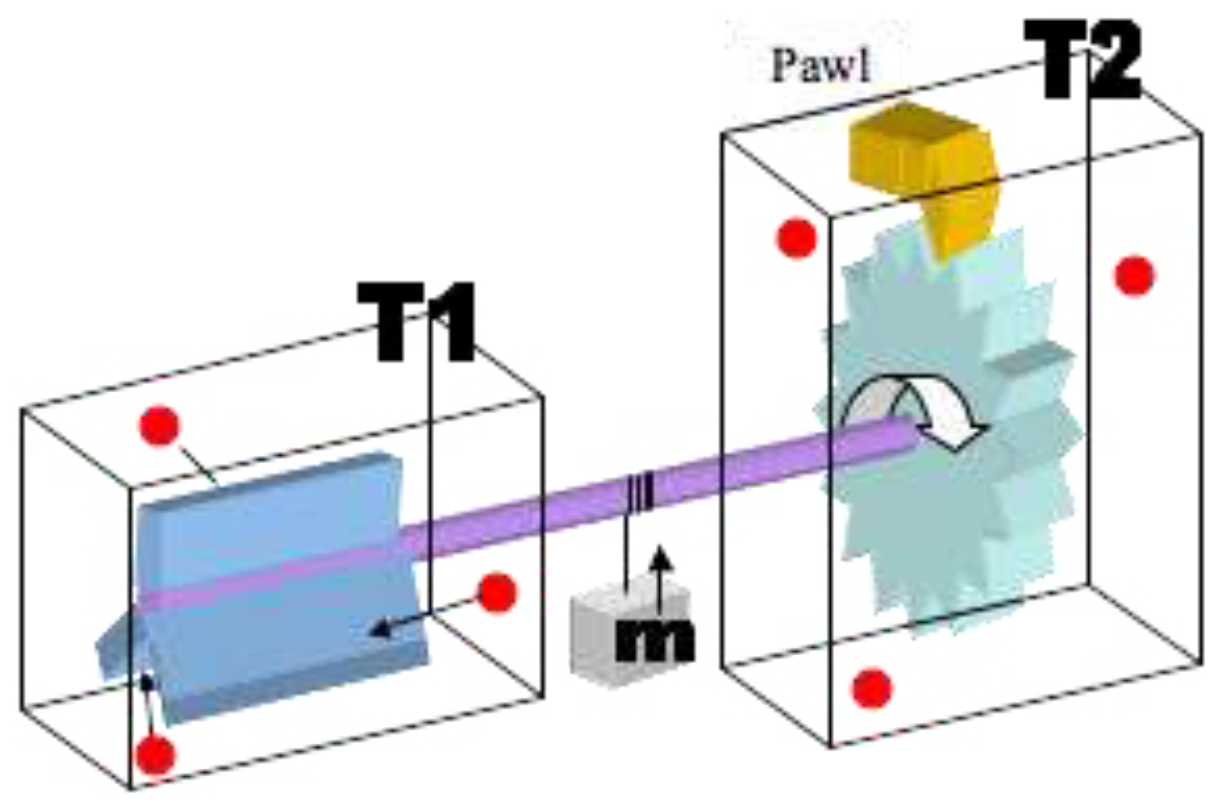
Ferrofluid Generator
Rectification Problem
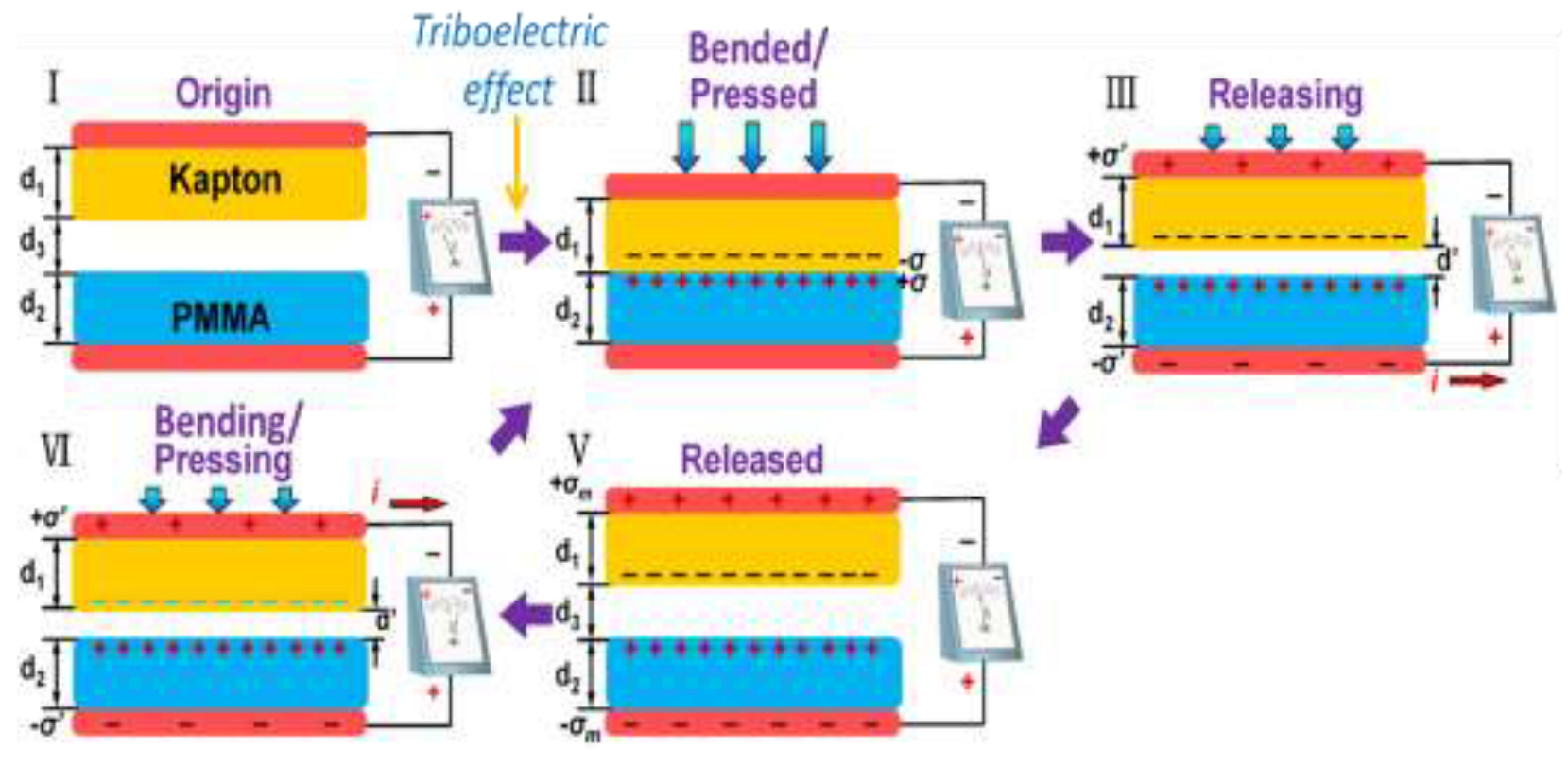
Triboelectric Nanogenerator (TENG)
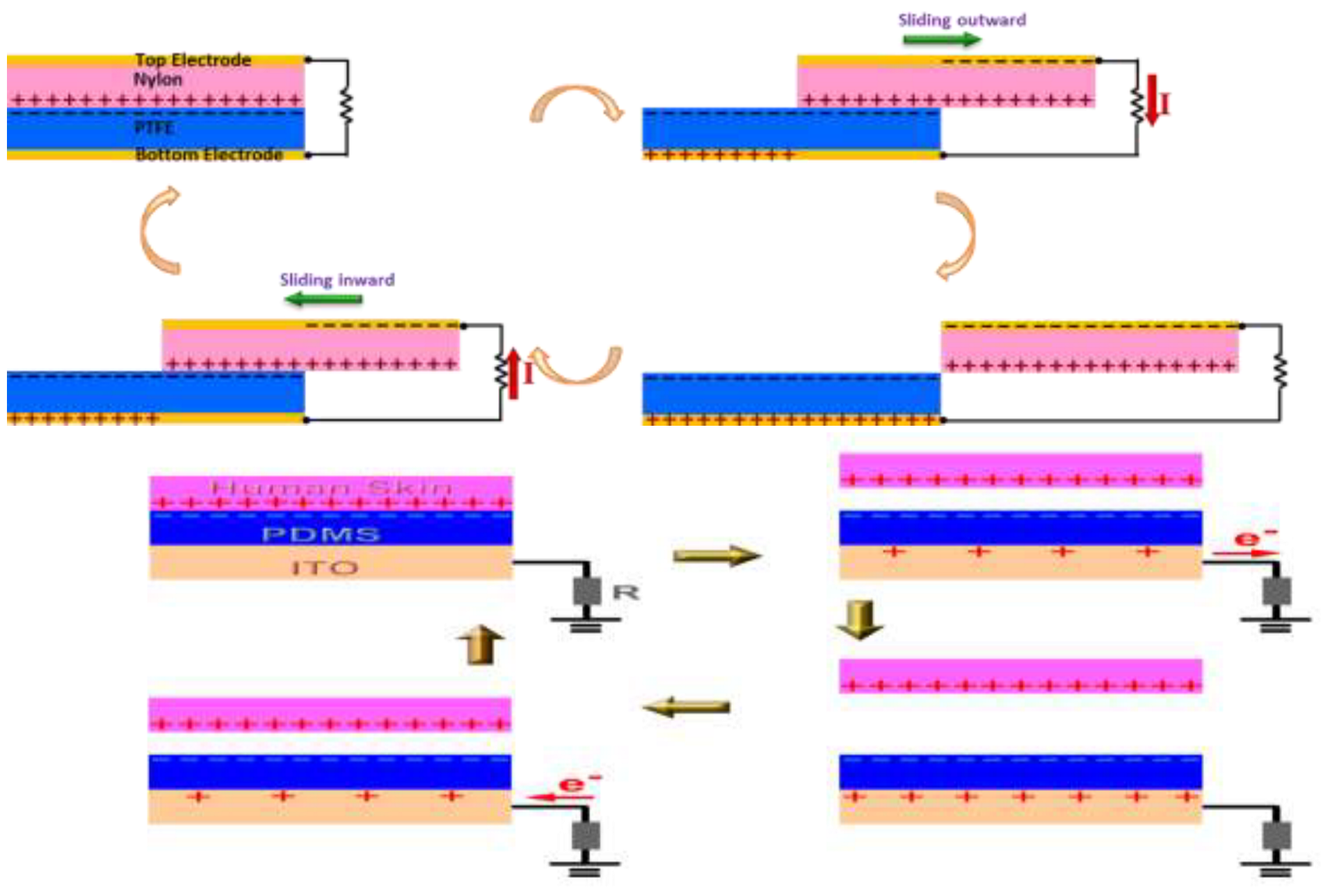
Lateral sliding mode TENG

The Water Evaporative Energy
- -
- The evaporation-driven engines can power common tasks like locomotion and electricity generation. These engines start and run autonomously when placed at air–water interfaces. They generate rotary and piston-like linear motion using specially designed, biologically based artificial muscles responsive to moisture fluctuations (Chen, 2015).
- -
- In new work, nanotechnology researchers have shown that evaporation from the surface of a variety of nanostructured carbon materials can be used to generate electricity: the evaporation driven water flow in nanoporous carbon film converts ambient thermal energy into electricity via the water molecules' interaction with the carbon material (www.nanowerk.com, n.d.).
Acknowledgments
References
- Alazmi, S.; Xu, Y.; Daqaq, M.F. Harvesting energy from the sloshing motion of ferrofluids in an externally excited container: Analytical modeling and experimental validation. Phys. Fluids 2016, 28, 077101. [Google Scholar] [CrossRef]
- CChen, X.; Goodnight, D.; Gao, Z.; Cavusoglu, A.H.; Sabharwal, N.; DeLay, M.; Driks, A.; Sahin, O. Scaling up nanoscale water-driven energy conversion into evaporation-driven engines and generators. Nat. Commun. 2015, 6, 7346. [Google Scholar] [CrossRef] [PubMed]
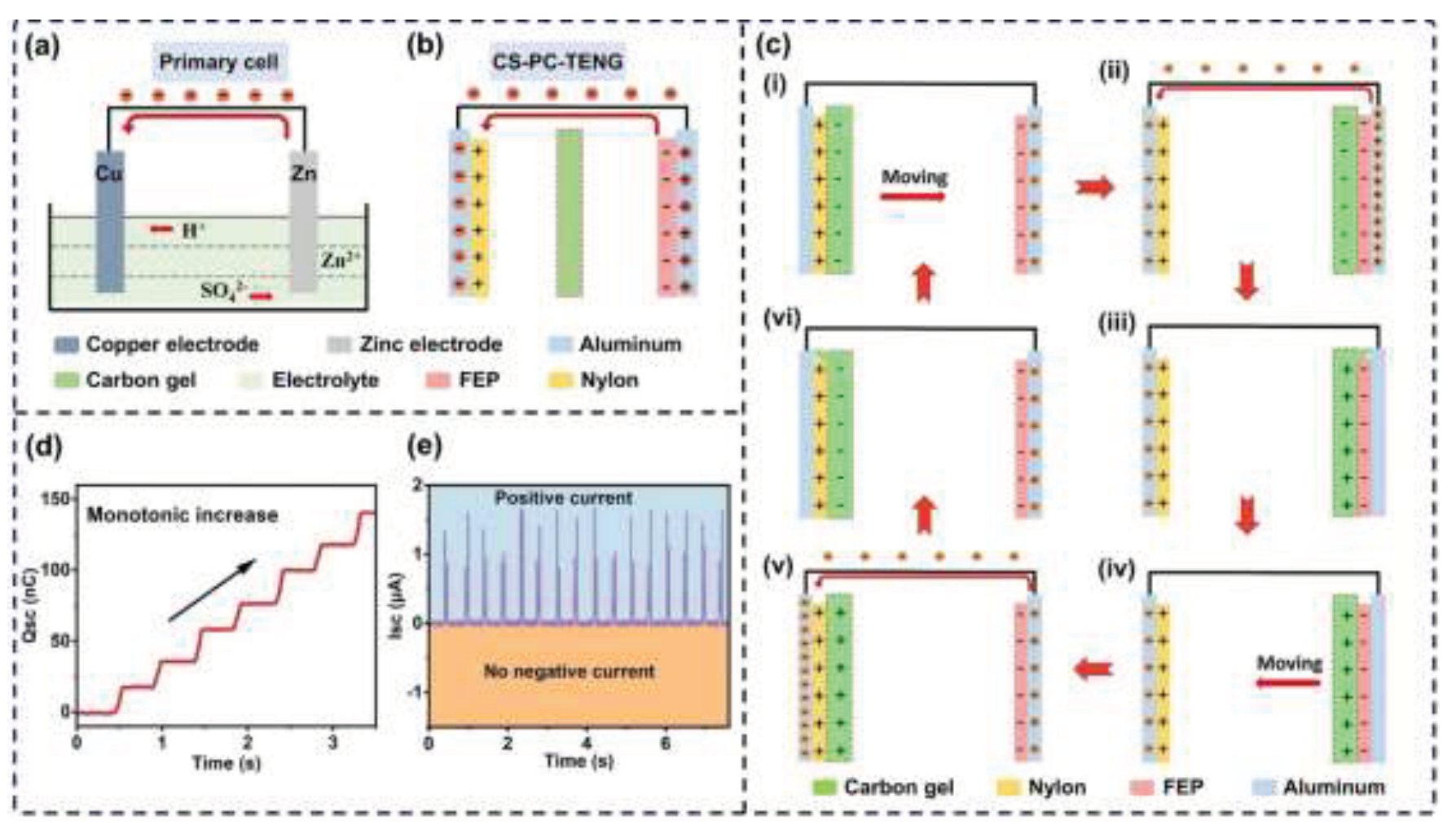
- Fu, S.; He, W.; Wu, H.; Shan, C.; Du, Y.; Li, G.; Wang, P.; Guo, H.; Chen, J.; Hu, C. High Output Performance and Ultra-Durable DC Output for Triboelectric Nanogenerator Inspired by Primary Cell. Nano-Micro Lett. 2022, 14, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Gsu.edu, 2019. Second Law of Thermodynamics. [Online] Available at: http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/seclaw.html[Accessed 1 Apr 2023].
- Helmenstine, A. M., 2019. An Introduction to Brownian Motion. [Online] Available at: https://www.thoughtco.com/brownian-motion-definition-and-explanation-4134272[Accessed 7 Feb 2023].
- Poláček, J.; Alexa, P. Brownian motion of magnetic nanoparticles as a source of energy? Int. J. Nanotechnol. 2013, 10, 1109. [Google Scholar] [CrossRef]
- Rattinacannou, J.-S. , 2014. A promising new energy source: The Brownian motion of nanoresonator arrays. EPJ Web of Conferences,.
- University of Arkansas News, n.d. Using the Natural Motion of 2D Materials to Create a New Source of Clean Energy. [Online] Available at: https://news.uark.edu/articles/40324/using-the-natural-motion-of-2d-materials-to-create-a-new-source-of-clean-energy[Accessed 2 Apr 2023].
- WWang, Z.L. Triboelectric Nanogenerators as New Energy Technology for Self-Powered Systems and as Active Mechanical and Chemical Sensors. ACS Nano 2013, 7, 9533–9557. [Google Scholar] [CrossRef] [PubMed]
- Waniewski, J.; Pietribiasi, M.; Pstras, L. Calculation of the Gibbs–Donnan factors for multi-ion solutions with non-permeating charge on both sides of a permselective membrane. Sci. Rep. 2021, 11, 22150. [Google Scholar] [CrossRef] [PubMed]
- webhome.auburn.edu, n.d. Maxwell’s Demon. [Online] Available at: https://webhome.auburn.edu/~smith01/notes/maxdem.htm[Accessed 21 Mar 2023].
- Wikipedia Contributors, 2019. Nanogenerator. [Online] Available at: https://en.wikipedia.org/wiki/Nanogenerator[Accessed 15 Feb 2023].
- Wikipedia, 2022. Brownian ratchet. [Online] Available at: https://en.wikipedia.org/wiki/Brownian_ratchet.
- www.nanowerk.com, n.d. A nanotechnology approach to generating electricity from water evaporation. [Online] Available at: https://www.nanowerk.com/spotlight/spotid=45918.php[Accessed 8 Mar 2023].
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



