Submitted:
26 March 2024
Posted:
26 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
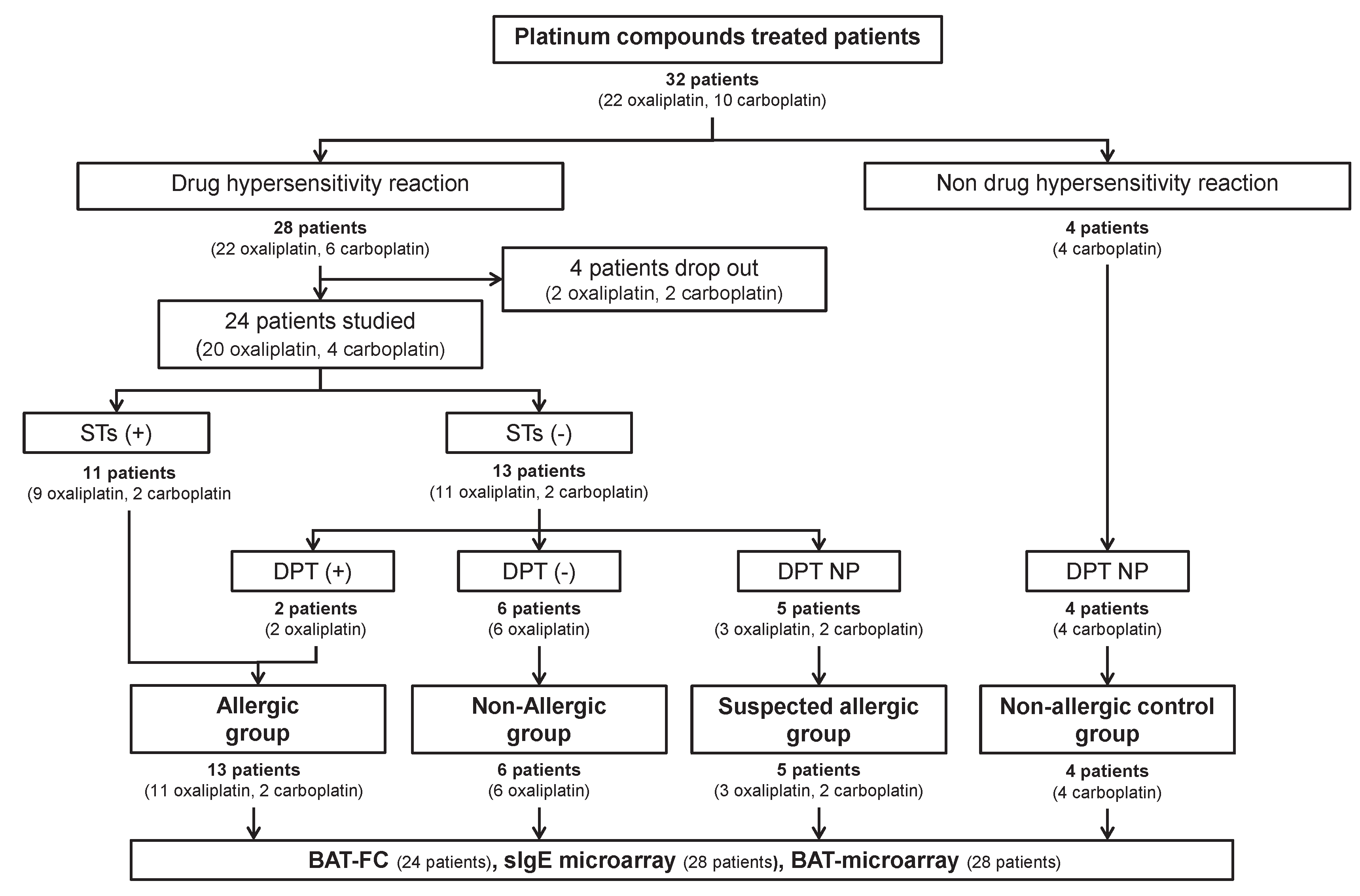
2.1. Characteristics of the Patients
2.2. Allergological Work-Up Outcomes
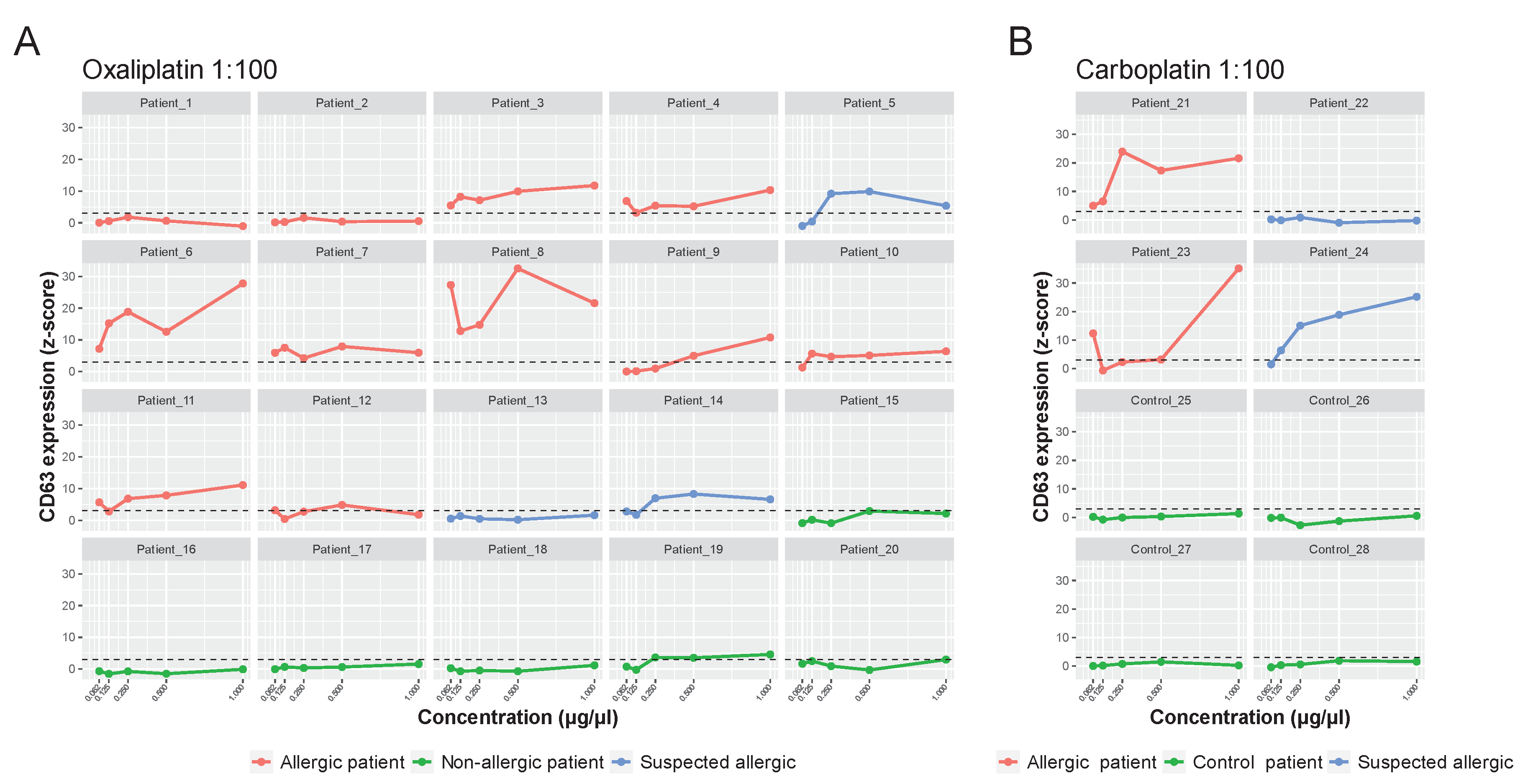
2.3. Basophil Activation Test by Flow Cytometry (BAT-FC)
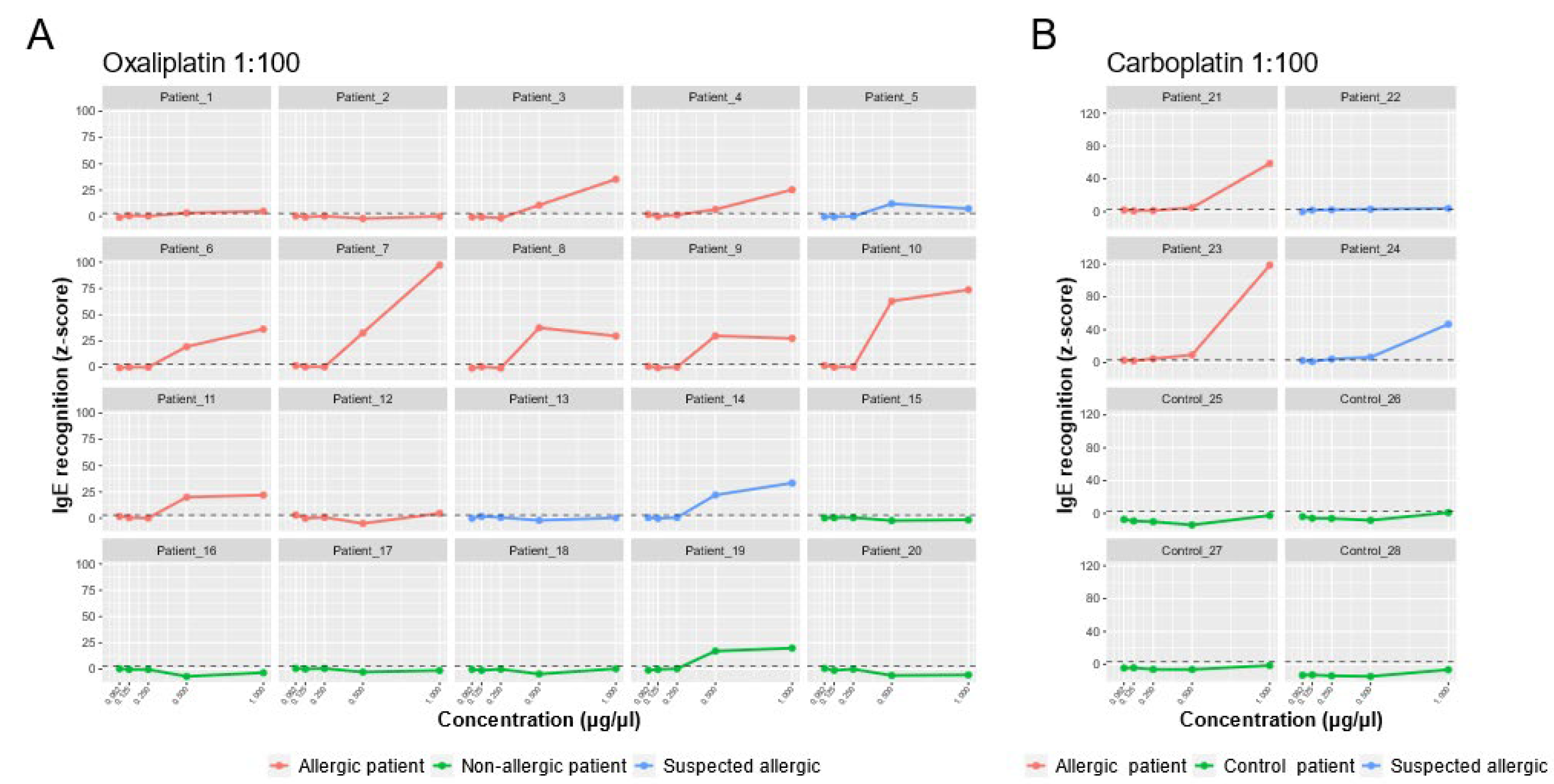
2.4. sIgE Microarrays Immunoassay
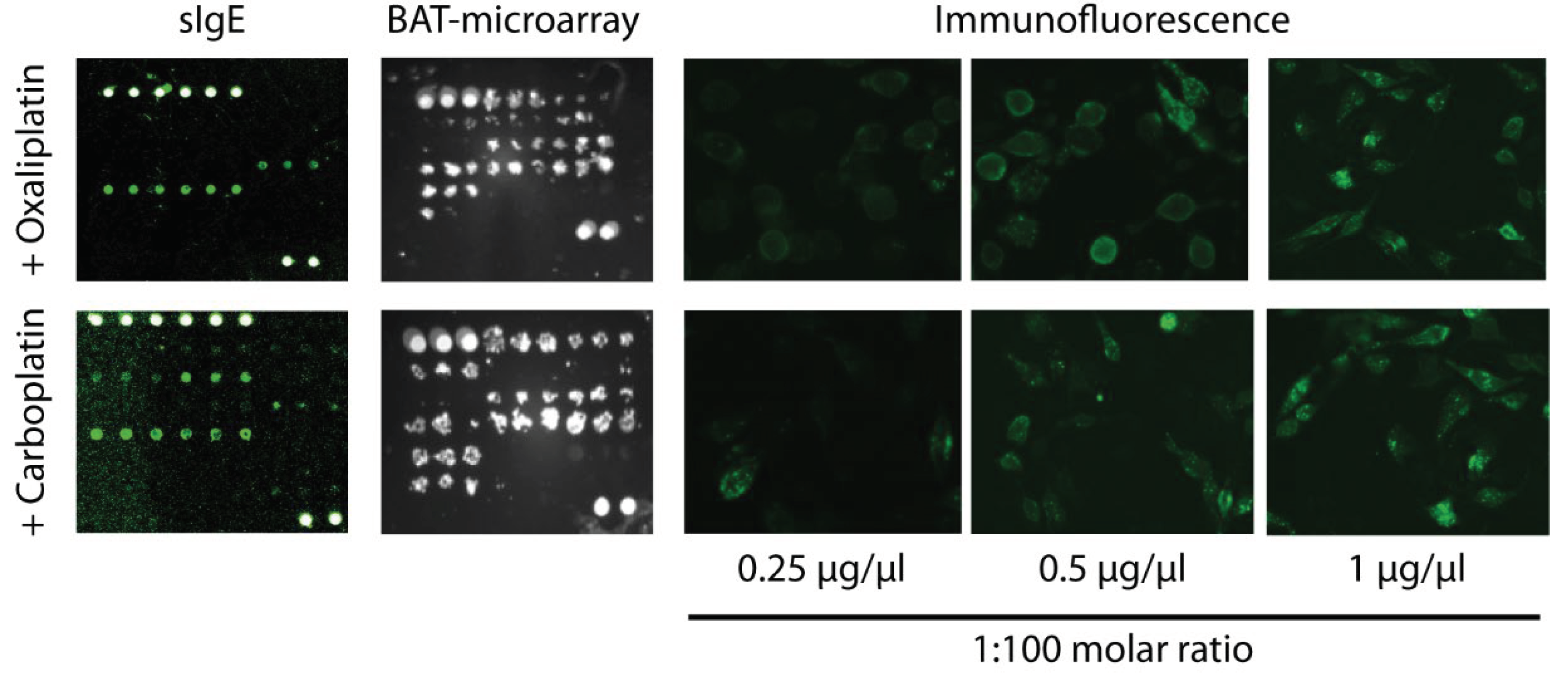
2.5. Basophil Activation Test on Microarray Support (BAT-Microarray)
3. Discussion
4. Materials and Methods
4.1. Patient Population and Study Design
4.2. Basophil Activation Test by Flow Cytometry (BAT-FC)
4.3. Microarray Printing
4.5. Basophil Activation Test on Microarray Support (BAT-Microarray)
4.6. Data Analysis.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Silver, J.; Garcia-Neuer, M.; Lynch, D. M.; Pasaoglu, G.; Sloane, D. E.; Castells, M., Endophenotyping Oxaliplatin Hypersensitivity: Personalizing Desensitization to the Atypical Platin. The journal of allergy and clinical immunology. In practice 2020, 8, (5), 1668-1680 e2. [CrossRef]
- Pagani, M.; Bavbek, S.; Alvarez-Cuesta, E.; Berna Dursun, A.; Bonadonna, P.; Castells, M.; Cernadas, J.; Chiriac, A.; Sahar, H.; Madrigal-Burgaleta, R.; Sanchez Sanchez, S., Hypersensitivity reactions to chemotherapy: an EAACI Position Paper. Allergy 2022, 77, (2), 388-403. [CrossRef]
- Madrigal-Burgaleta, R.; Bernal-Rubio, L.; Berges-Gimeno, M. P.; Carpio-Escalona, L. V.; Gehlhaar, P.; Alvarez-Cuesta, E., A Large Single-Hospital Experience Using Drug Provocation Testing and Rapid Drug Desensitization in Hypersensitivity to Antineoplastic and Biological Agents. The journal of allergy and clinical immunology. In practice 2019, 7, (2), 618-632. [CrossRef]
- Caiado, J.; Picard, M., Diagnostic tools for hypersensitivity to platinum drugs and taxanes: skin testing, specific IgE, and mast cell/basophil mediators. Current allergy and asthma reports 2014, 14, (8), 451. [CrossRef]
- Alpan, O.; Wasserman, R. L.; Kim, T.; Darter, A.; Shah, A.; Jones, D.; McNeil, D.; Li, H.; Ispas, L.; Rathkopf, M.; Perez, E.; Siri, D.; O'Connor, M.; Plassmeyer, M.; Romito, K.; Pettibone, C.; O'Reilly, S.; Sonder, S. U.; Marti, G., Towards an FDA-cleared basophil activation test. Front Allergy 2022, 3, 1009437. [CrossRef]
- Iwamoto, T.; Yuta, A.; Tabata, T.; Sugimoto, H.; Gabazza, E. C.; Hirai, H.; Kojima, S.; Okuda, M., Evaluation of basophil CD203c as a predictor of carboplatin-related hypersensitivity reaction in patients with gynecologic cancer. Biol Pharm Bull 2012, 35, (9), 1487-95. [CrossRef]
- Giavina-Bianchi, P.; Galvao, V. R.; Picard, M.; Caiado, J.; Castells, M. C., Basophil Activation Test is a Relevant Biomarker of the Outcome of Rapid Desensitization in Platinum Compounds-Allergy. The journal of allergy and clinical immunology. In practice 2017, 5, (3), 728-736. [CrossRef]
- Ornelas, C.; Caiado, J.; Campos Melo, A.; Pereira Barbosa, M.; Castells, M. C.; Pereira Dos Santos, M. C., The Contribution of the Basophil Activation Test to the Diagnosis of Hypersensitivity Reactions to Oxaliplatin. Int Arch Allergy Immunol 2018, 177, (3), 274-280. [CrossRef]
- Caiado, J.; Venemalm, L.; Pereira-Santos, M. C.; Costa, L.; Barbosa, M. P.; Castells, M., Carboplatin-, oxaliplatin-, and cisplatin-specific IgE: cross-reactivity and value in the diagnosis of carboplatin and oxaliplatin allergy. The journal of allergy and clinical immunology. In practice 2013, 1, (5), 494-500. [CrossRef]
- Huang, H. J.; Campana, R.; Akinfenwa, O.; Curin, M.; Sarzsinszky, E.; Karsonova, A.; Riabova, K.; Karaulov, A.; Niespodziana, K.; Elisyutina, O.; Fedenko, E.; Litovkina, A.; Smolnikov, E.; Khaitov, M.; Vrtala, S.; Schlederer, T.; Valenta, R., Microarray-Based Allergy Diagnosis: Quo Vadis? Frontiers in immunology 2020, 11, 594978. [CrossRef]
- Sanchez-Ruano, L.; de la Hoz, B.; Martinez-Botas, J., Clinical utility of microarray B-cell epitope mapping in food allergies: A systematic review. Pediatr Allergy Immunol 2020, 31, (2), 175-185. [CrossRef]
- Falcone, F. H.; Alcocer, M. J.; Okamoto-Uchida, Y.; Nakamura, R., Use of humanized rat basophilic leukemia reporter cell lines as a diagnostic tool for detection of allergen-specific IgE in allergic patients: time for a reappraisal? Current allergy and asthma reports 2015, 15, (11), 67. [CrossRef]
- Blanc, F.; Adel-Patient, K.; Drumare, M. F.; Paty, E.; Wal, J. M.; Bernard, H., Capacity of purified peanut allergens to induce degranulation in a functional in vitro assay: Ara h 2 and Ara h 6 are the most efficient elicitors. Clin Exp Allergy 2009, 39, (8), 1277-85. [CrossRef]
- Lin, J.; Renault, N.; Haas, H.; Schramm, G.; Vieths, S.; Vogel, L.; Falcone, F. H.; Alcocer, M. J., A novel tool for the detection of allergic sensitization combining protein microarrays with human basophils. Clin Exp Allergy 2007, 37, (12), 1854-62. [CrossRef]
- Wang, X.; Cato, P.; Lin, H. C.; Li, T.; Wan, D.; Alcocer, M. J.; Falcone, F. H., Optimisation and use of humanised RBL NF-AT-GFP and NF-AT-DsRed reporter cell lines suitable for high-throughput scale detection of allergic sensitisation in array format and identification of the ECM-integrin interaction as critical factor. Molecular biotechnology 2014, 56, (2), 136-46. [CrossRef]
- Demoly, P.; Adkinson, N. F.; Brockow, K.; Castells, M.; Chiriac, A. M.; Greenberger, P. A.; Khan, D. A.; Lang, D. M.; Park, H. S.; Pichler, W.; Sanchez-Borges, M.; Shiohara, T.; Thong, B. Y., International Consensus on drug allergy. Allergy 2014, 69, (4), 420-37. [CrossRef]
- Brown, S. G., Clinical features and severity grading of anaphylaxis. J Allergy Clin Immunol 2004, 114, (2), 371-6. [CrossRef]
- Brockow, K.; Romano, A.; Blanca, M.; Ring, J.; Pichler, W.; Demoly, P., General considerations for skin test procedures in the diagnosis of drug hypersensitivity. Allergy 2002, 57, (1), 45-51.
- Vogel, L.; Luttkopf, D.; Hatahet, L.; Haustein, D.; Vieths, S., Development of a functional in vitro assay as a novel tool for the standardization of allergen extracts in the human system. Allergy 2005, 60, (8), 1021-8. [CrossRef]
- Lin, J.; Bardina, L.; Shreffler, W. G.; Andreae, D. A.; Ge, Y.; Wang, J.; Bruni, F. M.; Fu, Z.; Han, Y.; Sampson, H. A., Development of a novel peptide microarray for large-scale epitope mapping of food allergens. J Allergy Clin Immunol 2009, 124, (2), 315-22, 322 e1-3. [CrossRef]
- Kolkhir, P.; Ali, H.; Babina, M.; Ebo, D.; Sabato, V.; Elst, J.; Frischbutter, S.; Pyatilova, P.; Maurer, M., MRGPRX2 in drug allergy: What we know and what we do not know. J Allergy Clin Immunol 2023, 151, (2), 410-412. [CrossRef]
- Hoffmann, H. J.; Santos, A. F.; Mayorga, C.; Nopp, A.; Eberlein, B.; Ferrer, M.; Rouzaire, P.; Ebo, D. G.; Sabato, V.; Sanz, M. L.; Pecaric-Petkovic, T.; Patil, S. U.; Hausmann, O. V.; Shreffler, W. G.; Korosec, P.; Knol, E. F., The clinical utility of basophil activation testing in diagnosis and monitoring of allergic disease. Allergy 2015, 70, (11), 1393-405. [CrossRef]
- Depince-Berger, A. E.; Sidi-Yahya, K.; Jeraiby, M.; Lambert, C., Basophil activation test: Implementation and standardization between systems and between instruments. Cytometry. Part A : the journal of the International Society for Analytical Cytology 2017, 91, (3), 261-269. [CrossRef]
- Santos, A. F.; Alpan, O.; Hoffmann, H. J., Basophil activation test: Mechanisms and considerations for use in clinical trials and clinical practice. Allergy 2021, 76, (8), 2420-2432. [CrossRef]
- Berges Gimeno, M. P.; Bernal Rubio, L.; Roldan, E.; Rita, C. G., Positive Basophil Activation Test Result in a Patient With Anaphylaxis to Cotrimoxazole. Journal of investigational allergology & clinical immunology 2022, 32, (5), 410-412. [CrossRef]
- Phillips-Angles, E.; Gonzalez-Munoz, M.; Dominguez-Ortega, J.; Cabanas, R.; Quirce, S., Usefulness of basophil activation test in diagnosis of hypersensitivity to etoricoxib. The journal of allergy and clinical immunology. In practice 2016, 4, (2), 350-1. [CrossRef]
- Puan, K. J.; Andiappan, A. K.; Lee, B.; Kumar, D.; Lai, T. S.; Yeo, G.; Bercin, D.; Starke, M.; Haase, D.; Lum, J.; Chew, F. T.; Connolly, J.; Wong, S. C.; Zolezzi, F.; Poidinger, M.; Wang, Y.; Rotzschke, O., Systematic characterization of basophil anergy. Allergy 2017, 72, (3), 373-384. [CrossRef]
- Falcone, F. H.; Gibbs, B. F., Purification of Basophils from Peripheral Human Blood. Methods Mol Biol 2020, 2163, 35-48.
- Ladics, G. S.; van Bilsen, J. H.; Brouwer, H. M.; Vogel, L.; Vieths, S.; Knippels, L. M., Assessment of three human FcepsilonRI-transfected RBL cell-lines for identifying IgE induced degranulation utilizing peanut-allergic patient sera and peanut protein extract. Regulatory toxicology and pharmacology : RTP 2008, 51, (3), 288-94.

| Patient | Type of tumor | Sex | Age (years) |
Drug | Grade of initial reaction Brown/HRYC | Lifetime Exposure (previous cycles) |
Time to index reaction (minutes) | Symptoms | History of atopic disease/HR |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Colorectal | F | 68 | Oxaliplatin | 1.1 | 7 | 60 | Urticaria, erythema, pruritus | No/No |
| 2 | Colorectal | M | 69 | Oxaliplatin | 2.2 | 20 | 5 | Urticaria, erythema, pruritus, nausea | No/No |
| 3 | Colon | M | 76 | Oxaliplatin | 1.1 | 4 | 30 | Erythema, pruritus, warm sensation | No/No |
| 4 | Sigmoid | F | 63 | Oxaliplatin | 2.1-2 | 9 | 60 | Warm, diaphoresis, chills | No/No |
| 5 | Colon | M | 71 | Oxaliplatin | 3.3 | 10 | 120 | Erythema, flushing, pruritus, desaturation | No/No |
| 6 | Gastric | F | 53 | Oxaliplatin | 2.3 | 6 | 45 | Erythema, flushing, pruritus, chest tightness | No/No |
| 7 | Rectal | F | 64 | Oxaliplatin | 1.1 | 3 | 60 | Erythema, flushing | No/No |
| 8 | Sigmoid and rectal | M | 63 | Oxaliplatin | 3.3 | 9 | 70 | Pruritus, maculopapular rash, desaturation | Yes/Yes |
| 9 | Colon | M | 68 | Oxaliplatin | 3.3 | 3 | 60 | Cough, desaturation, throat tightness | No/No |
| 10 | Colorectal | F | 58 | Oxaliplatin | 3.4 | 3 | 60 | Erythema, flushing, dyspnea, desaturation, syncope, confusion, dizziness | No/Yes |
| 11 | Colon | M | 70 | Oxaliplatin | 3.3 | 5 | 20 | Erythema, flushing, pruritus, desaturation | Yes/No |
| 12 | Sigmoid | F | 71 | Oxaliplatin | 1.1 | 9 | 5 | Localized urticaria, pruritus | No/No |
| 13 | Sigmoid | M | 60 | Oxaliplatin | 2.2 | 1 | 120 | Dysesthesias, dyspnea, dysphonia, uvula edema | Yes/Yes |
| 14 | Sigmoid | F | 70 | Oxaliplatin | 1.1 | 3 | 60 | Localized urticaria, pruritus | No/No |
| 15 | Colon | M | 49 | Oxaliplatin | 1.1 | 8 | 70 | Erythema,Dysesthesias | Yes/No |
| 16 | Sigmoid | F | 68 | Oxaliplatin | 2.2 | 1 | 60 | Dyspnea,dysphonia, tachycardia | No/No |
| 17 | Sigmoid | F | 75 | Oxaliplatin | 2.3 | 1 | 120 | Dyspnea, wheeze | Yes/No |
| 18 | Rectal | M | 82 | Oxaliplatin | 1.1 | 22 | 480 | Flushing | Yes/No |
| 19 | Colon | F | 57 | Oxaliplatin | 2.3 | 1 | 60 | Dysesthesias, dyspnea, throat tightness | No/No |
| 20 | Colorectal | F | 58 | Oxaliplatin | 1.1 | 3 | 48 | Pruritus, dysesthesia | No/No |
| 21 | Endometrial | F | 57 | Carboplatin | 3.3 | 6 | 15 | Dyspnea, desaturation, hypertension, chest pain | No/Yes |
| 22 | Ovarian | F | 48 | Carboplatin | 2.2 | 15 | Not Known | Localized urticaria, dyspnea | No/Yes |
| 23 | Lung | M | 75 | Carboplatin | 3.3 | 7 | 60 | Pruritus, dyspnea, desaturation | No/No |
| 24 | Ovarian | F | 69 | Carboplatin | 1.1 | 9 | 15 | Urticaria, hands and foot pruritus | No/No |
| 25 | Lung | M | 73 | Carboplatin | NR‡ | 1 | NA† | NA† | No/No |
| 26 | Vulvar | F | 53 | Carboplatin | NR‡ | 2 | NA† | NA† | Yes/Yes |
| 27 | Ovarian | F | 43 | Carboplatin | NR‡ | 1 | NA† | NA† | No/Yes |
| 28 | Breast | F | 34 | Carboplatin | NR‡ | 4 | NA† | NA† | No/Yes |
| Patient | Drug | Skin tests | DPT | Tryptase (basal) | Tryptase (reaction) | Total IgE | BAT-FC CD63 | BAT-FC CD203c | sIgE-microarray | BAT-microarray |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Oxaliplatin | + (IDT 0.5 mg/ml) | NP† | 6.67 | NP† | 354 | - | NP† | 4.96 | ND‡ |
| 2 | Oxaliplatin | + (IDT 0.5 mg/ml | NP† | 7.57 | NP† | 71 | - | - | 0.02 | 0.55 |
| 3 | Oxaliplatin | - | + | <1 | NP† | 4 | - | - | 35.39 | 11.75 |
| 4 | Oxaliplatin | - | + | 13.8 | 13.70 | 6 | - | - | 25.40 | 10.33 |
| 5 | Oxaliplatin | - | NP† | <1 | NP† | 157 | - | - | 7.37 | 5.39 |
| 6 | Oxaliplatin | + (IDT 5 mg/ml) | NP† | 1.03 | NP† | 43 | + | + | 36.37 | 27.76 |
| 7 | Oxaliplatin | + (IDT 0.5 mg/ml) | NP† | 7.34 | NP† | 608 | - | - | 97.20 | 5.96 |
| 8 | Oxaliplatin | + (SPT 0.5-5 mg/ml) | NP† | 6.69 | NP† | 798 | - | - | 29.79 | 21.57 |
| 9 | Oxaliplatin | + (IDT 5 mg/ml) | NP† | 5.09 | NP† | 240 | - | - | 27.45 | 10.75 |
| 10 | Oxaliplatin | + (IDT 0.005 mg/ml) | NP† | 7.49 | NP† | 23 | - | - | 73.75 | 6.38 |
| 11 | Oxaliplatin | + (IDT 0.5 mg/ml) | NP† | 6.56 | NP† | 17 | - | - | 21.99 | 11.12 |
| 12 | Oxaliplatin | + (IDT 0.5mg/ml) | NP† | 6.06 | NP† | 29 | - | - | 4.57 | 4.84* |
| 13 | Oxaliplatin | - | NP† | 5.08 | NP† | 90 | - | - | 0.38 | 1.61 |
| 14 | Oxaliplatin | - | NP† | 5.81 | NP† | 71 | NP† | NP† | 33.45 | 6.60 |
| 15 | Oxaliplatin | - | - | 6.32 | NP† | 38 | - | - | ND‡ | 2.11 |
| 16 | Oxaliplatin | - | - | 3.6 | NP† | 32 | - | - | 0.32 | ND‡ |
| 17 | Oxaliplatin | - | - | 4.37 | NP† | 9 | NV§ | - | ND | 1.57 |
| 18 | Oxaliplatin | - | - | 3.02 | NP† | 633 | - | - | 0.36 | 1.16 |
| 19 | Oxaliplatin | - | - | 3.41 | NP† | 463 | NV§ | NP† | 19.91 | 4.60 |
| 20 | Oxaliplatin | - | - | 7.32 | NP† | 41 | - | NP† | ND‡ | 2.90 |
| 21 | Carboplatin | + (IDT 1 mg/ml | NP† | 2.14 | NP† | 743 | + | - | 58.76 | 21.62 |
| 22 | Carboplatin | - | NP† | 4.48 | NP† | 54 | - | - | 3.92 | ND‡ |
| 23 | Carboplatin | + (IDT 1mg/ml) | NP† | 7.41 | NP† | 49 | - | - | 118.72 | 35.11 |
| 24 | Carboplatin | - | NP† | 3.67 | NP† | 5 | - | - | 46.76 | 25.21 |
| 25 | Carboplatin | NP† | NP† | NP† | NP† | NP† | - | - | ND‡ | 1.33 |
| 26 | Carboplatin | NP† | NP† | NP† | NP† | NP† | - | - | 1.03 | 0.56 |
| 27 | Carboplatin | NP† | NP† | NP† | NP† | NP† | NP† | NP† | ND‡ | 0.26 |
| 28 | Carboplatin | NP† | NP† | NP† | NP† | NP† | NP† | NP† | ND‡ | 1.59 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




