Submitted:
13 March 2024
Posted:
13 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Oxidized LDL and Atherosclerosis
3. The Protective Role of HDL
4. Transport of Lipophilic Substances Other Than Plasma Lipids
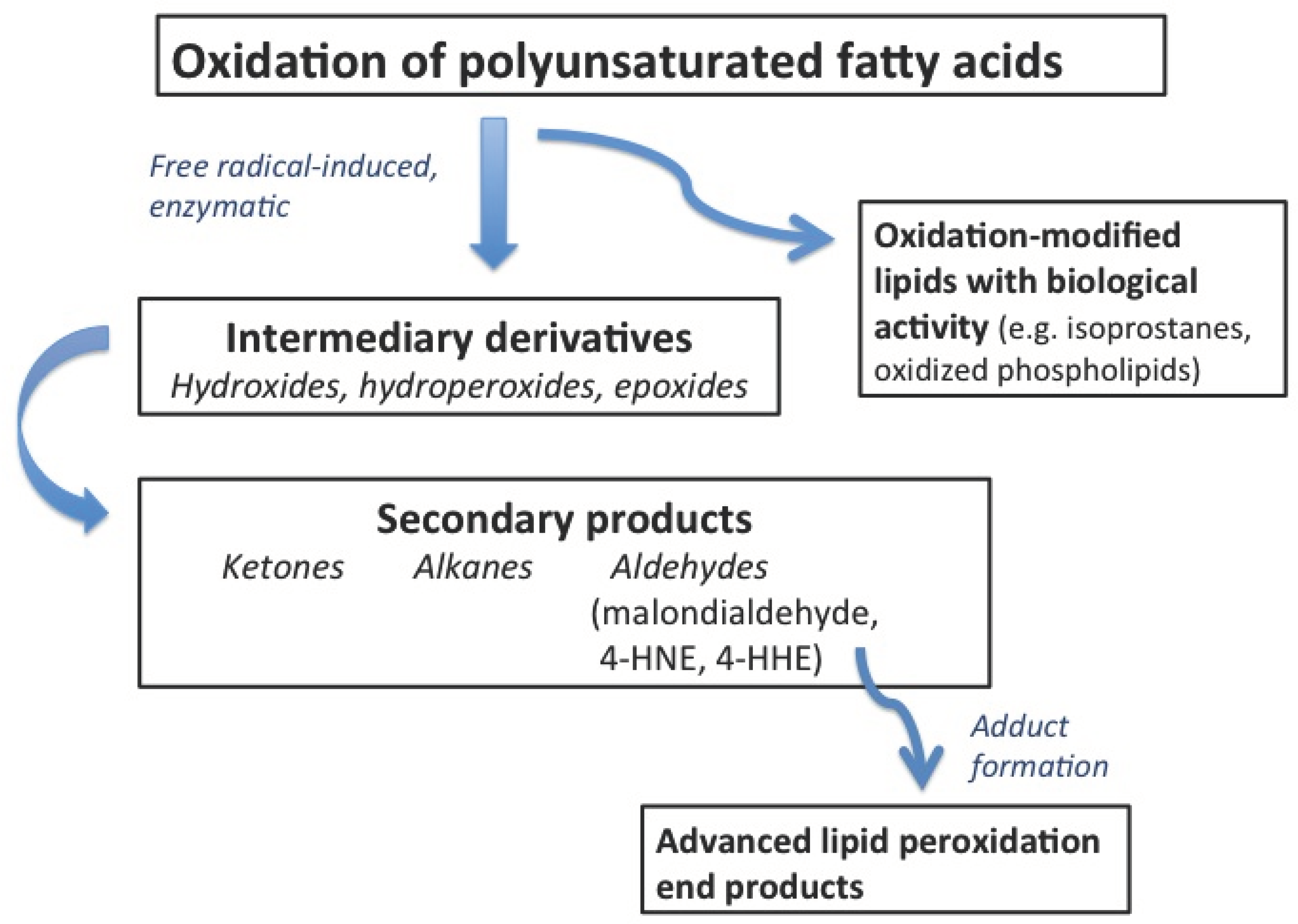
5. Lipid Oxidation Products in Human Plasma
6. Lipoprotein-Specific Transport of LOP
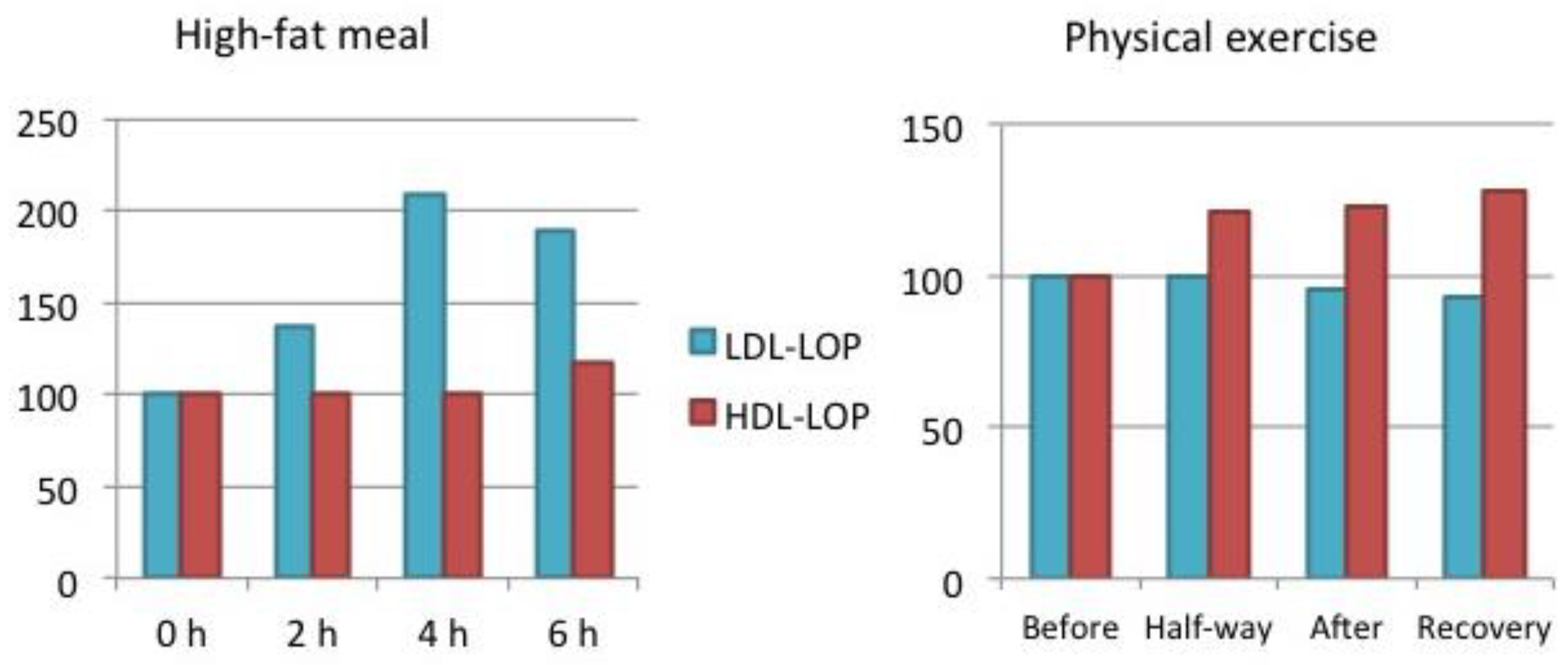
6.1. Studies on Physiological Oxidative Stress in Human Volunteers
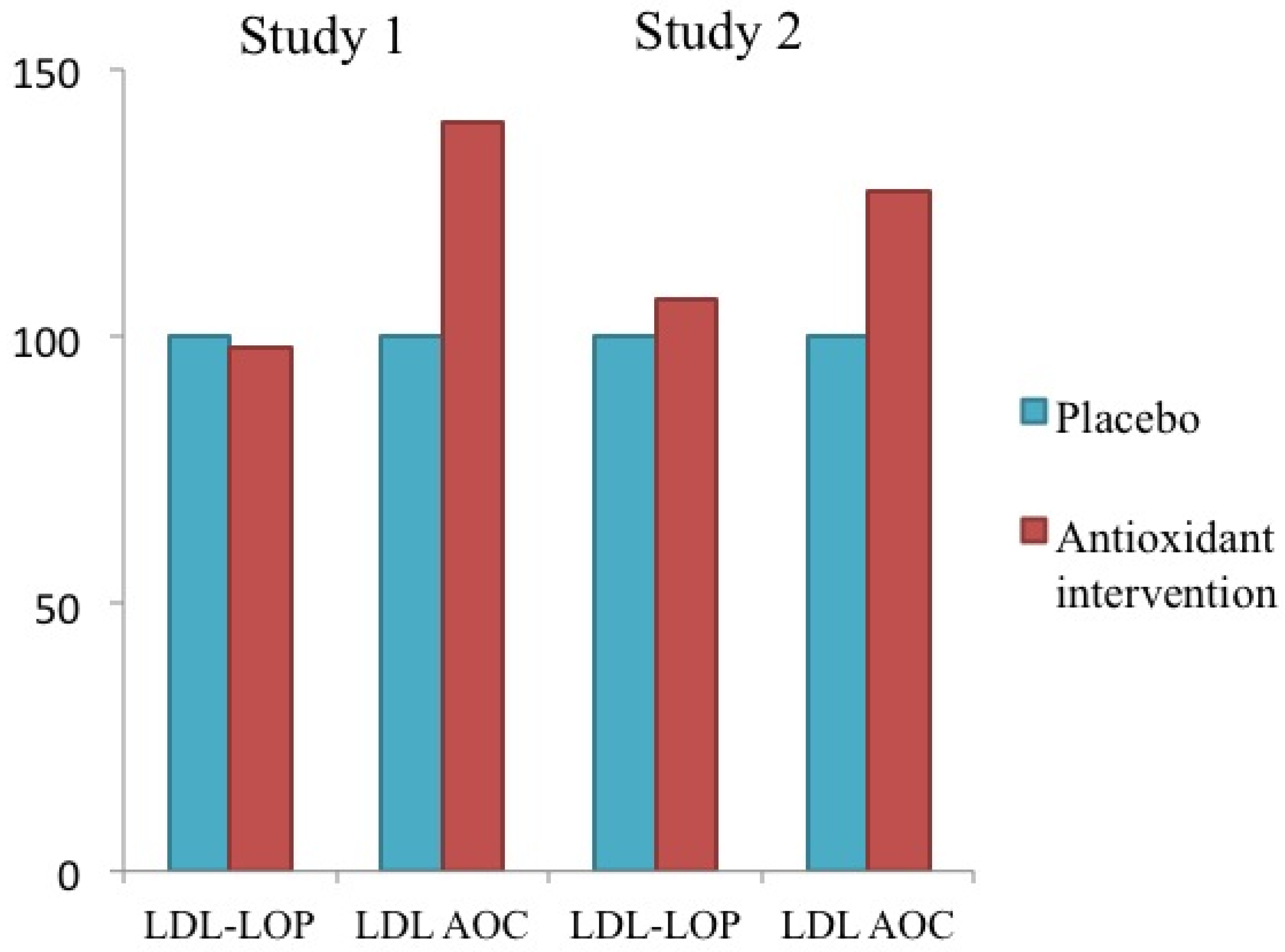
6.2. Experimental Evidence of the Role of HDL
7. Potential Significance of Lipoprotein LOP Transport for Oxidative Stress
8. Measurement of Lipoprotein LOP Transport
9. Lipoprotein LOP and the Risk of Atherosclerosis
9.1. Common Risk Factors
9.2. Physical Activity
9.3. Obesity and Weight Reduction
9.4. Dietary Factors
9.5. Metabolic Disorders Related to Atherosclerosis
9.6. Subclinical Atherosclerosis
9.7. Clinically Verified Atherosclerosis
9.8. Mortality
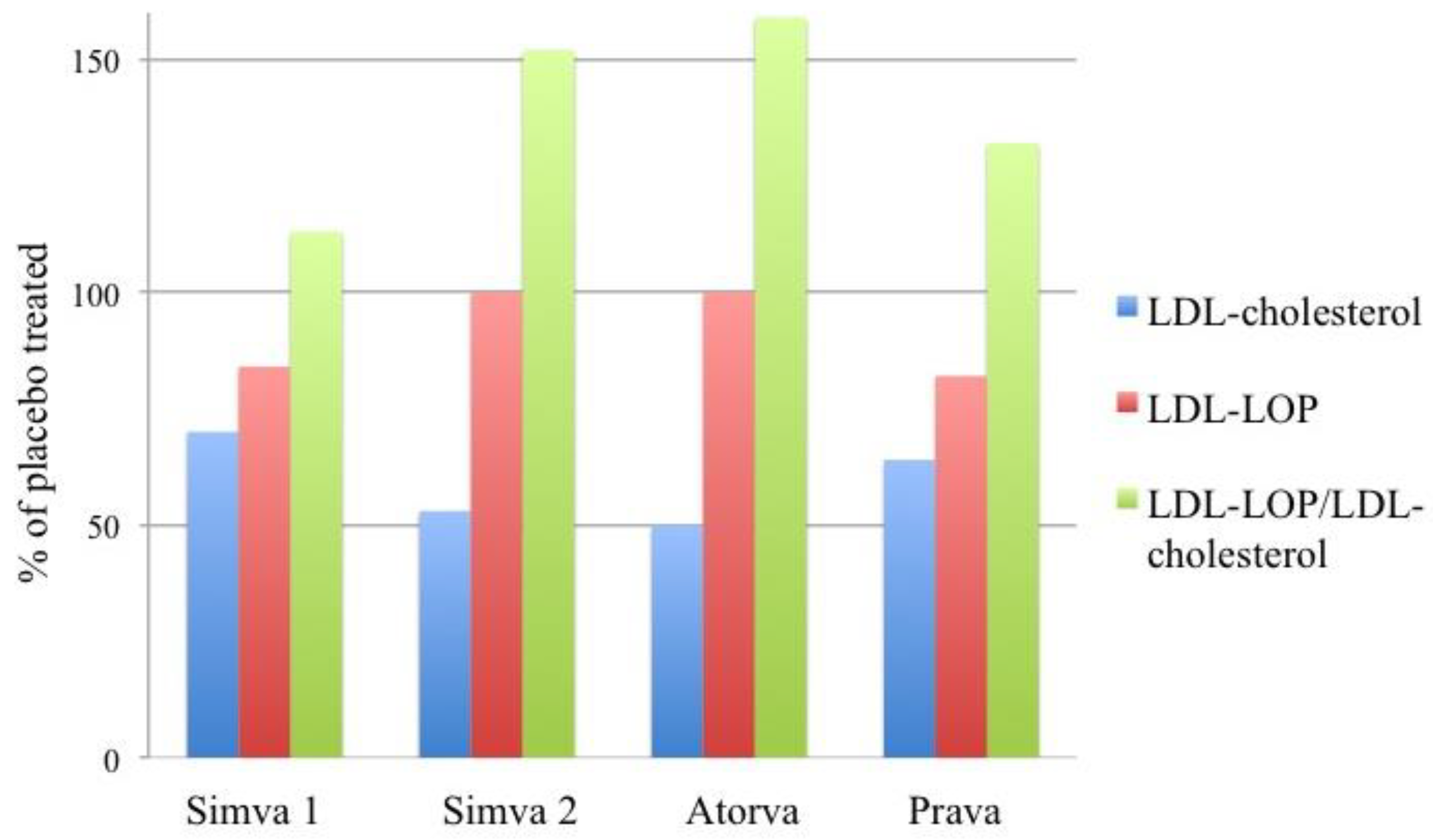
9.9. Statin Treatment
10. LOP and Cholesterol: Commonalities and Disparities
11. Lipoprotein LOP as a Potential Risk Factor
11.1. LOP as Dietary Risk Factor
11.2. HDL and the Clearance of LOP
12. Significance for Other Oxidative Stress-Dependent Diseases
13. Perspective Regarding Prevention and Treatment
14. Consistence with Present Theories
15. Open Questions and Future Directions
16. Conclusions
Funding
Conflicts of Interest
References
- Feingold, K. R. Introduction to lipids and lipoproteins. In Endotext; Feingold, K. R., Anawalt, B., Blackman, M. R., Boyce, A., Chrousos, G., Corpas, E., de Herder, W. W., Dhatariya, K., Dungan, K., Hofland, J., Kalra, S., Kaltsas, G., Kapoor, N., Koch, C., Kopp, P., Korbonits, M., Kovacs, C. S., Kuohung, W., Laferrère, B., Levy, M., McGee, E. A., McLachlan, R., New, M., Purnell, J., Sahay, R., Shah, A. S., Singer, F., Sperling, M. A., Stratakis, C. A., Trence, D. L., Wilson, D. P., Eds.; MDText.com, Inc.: South Dartmouth (MA), 2000. [Google Scholar]
- Steinberg, D.; Parthasarathy, S.; Carew, T. E.; Khoo, J. C.; Witztum, J. L. Beyond cholesterol. Modifications of low-density lipoprotein that increase its atherogenicity. N Engl J Med. [CrossRef]
- Bowry, V. W.; Stanley, K. K.; Stocker, R. High density lipoprotein is the major carrier of lipid lydroperoxides in human blood plasma from fasting donors. Proc. Natl. Acad. Sci. U.S.A. 1992, 89(21), 10316–10320. [Google Scholar] [CrossRef] [PubMed]
- Esterbauer, H.; Gebicki, J.; Puhl, H.; Jürgens, G. The role of lipid peroxidation and antioxidants in oxidative modification of LDL. Free Radical Biology and Medicine 1992, 13(4), 341–390. [Google Scholar] [CrossRef] [PubMed]
- Staprans, I.; Rapp, J. H.; Pan, X. M.; Feingold, K. R. The effect of oxidized lipids in the diet on serum lipoprotein peroxides in control and diabetic rats. J. Clin. Invest. 1993, 92(2), 638–643. [Google Scholar] [CrossRef] [PubMed]
- Staprãns, I.; Rapp, J. H.; Pan, X. M.; Kim, K. Y.; Feingold, K. R. Oxidized lipids in the diet are a source of oxidized lipid in chylomicrons of human serum. Arterioscler Thromb 1994, 14(12), 1900–1905. [Google Scholar] [CrossRef] [PubMed]
- Staprans, I.; Rapp, J. H.; Pan, X. M.; Feingold, K. R. Oxidized lipids in the diet are incorporated by the liver into very low density lipoprotein in rats. J Lipid Res 1996, 37(2), 420–430. [Google Scholar] [CrossRef] [PubMed]
- Ahotupa, M. Oxidized lipoprotein lipids and atherosclerosis. Free Radical Research 2017, 51(4), 439–447. [Google Scholar] [CrossRef] [PubMed]
- Shao, B.; Heinecke, J. W. HDL, lipid peroxidation, and atherosclerosis. Journal of Lipid Research 2009, 50(4), 599–601. [Google Scholar] [CrossRef]
- Ahotupa, M.; Suomela, J.-P.; Vuorimaa, T.; Vasankari, T. Lipoprotein-specific transport of circulating lipid peroxides. Annals of Medicine 2010, 42(7), 521–529. [Google Scholar] [CrossRef]
- Soran, H.; Schofield, J. D.; Durrington, P. N. Antioxidant properties of HDL. Front. Pharmacol. 2015, 6. [Google Scholar] [CrossRef]
- Stocker, R.; Keaney, J. F. Role of oxidative modifications in atherosclerosis. Physiological Reviews 2004, 84(4), 1381–1478. [Google Scholar] [CrossRef]
- Steinbrecher, U. Receptors for oxidized low density lipoprotein. Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids. [CrossRef]
- Miller, Y. I.; Shyy, J. Y.-J. Context-dependent role of oxidized lipids and lipoproteins in inflammation. Trends in Endocrinology & Metabolism. [CrossRef]
- Von Eckardstein, A.; Nordestgaard, B. G.; Remaley, A. T.; Catapano, A. L. High-density lipoprotein revisited: biological functions and clinical relevance. European Heart Journal 2023, 44(16), 1394–1407. [Google Scholar] [CrossRef] [PubMed]
- Asztalos, B. F.; Tani, M.; Schaefer, E. J. Metabolic and functional relevance of HDL subspecies. Current Opinion in Lipidology 2011, 22(3), 176–185. [Google Scholar] [CrossRef] [PubMed]
- Ben-Aicha, S.; Badimon, L.; Vilahur, G. Advances in HDL: much more than lipid transporters. IJMS 2020, 21(3), 732. [Google Scholar] [CrossRef] [PubMed]
- Khera, A. V.; Cuchel, M.; De La Llera-Moya, M.; Rodrigues, A.; Burke, M. F.; Jafri, K.; French, B. C.; Phillips, J. A.; Mucksavage, M. L.; Wilensky, R. L.; Mohler, E. R.; Rothblat, G. H.; Rader, D. J. Cholesterol efflux capacity, high-density lipoprotein function, and atherosclerosis. N Engl J Med 2011, 364(2), 127–135. [Google Scholar] [CrossRef] [PubMed]
- Spindler-Vomachka, M.; Vodicnik, M. J.; Lech, J. J. Transport of 2,4,5,2′,4′,5′-Hexachlorobiphenyl by lipoproteins in vivo. Toxicology and Applied Pharmacology 1984, 74(1), 70–77. [Google Scholar] [CrossRef] [PubMed]
- Borlakoglu, J. T.; Welch, V. A.; Wilkins, J. P. G.; Dils, R. R. Transport and cellular uptake of polychlorinated biphenyls (PCBs)—I. Biochemical Pharmacology 1990, 40(2), 265–272. [Google Scholar] [CrossRef] [PubMed]
- Lamon-Fava, S.; Sadowski, J.; Davidson, K.; O’Brien, M.; McNamara, J.; Schaefer, E. Plasma lipoproteins as carriers of phylloquinone (vitamin K1) in humans. The American Journal of Clinical Nutrition 1998, 67(6), 1226–1231. [Google Scholar] [CrossRef]
- Meng, Q.-H.; Wähälä, K.; Adlercreutz, H.; Tikkanen, M. J. Antiproliferative efficacy of lipophilic soy isoflavone phytoestrogens delivered by low density lipoprotein particles into cultured U937 cells. Life Sciences 1999, 65(16), 1695–1705. [Google Scholar] [CrossRef]
- Gershkovich, P.; Hoffman, A. Effect of a high-fat meal on absorption and disposition of lipophilic compounds: the importance of degree of association with triglyceride-rich lipoproteins. European Journal of Pharmaceutical Sciences 2007, 32(1), 24–32. [Google Scholar] [CrossRef]
- Li, Y.; Wongsiriroj, N.; Blaner, W. S. The multifaceted nature of retinoid transport and metabolism. Hepatobiliary Surg Nutr 2014, 3(3), 126–139. [Google Scholar] [CrossRef]
- Yamamoto, H.; Takada, T.; Yamanashi, Y.; Ogura, M.; Masuo, Y.; Harada-Shiba, M.; Suzuki, H. VLDL/LDL acts as a drug carrier and regulates the transport and metabolism of drugs in the body. Sci Rep 2017, 7(1), 633. [Google Scholar] [CrossRef] [PubMed]
- Saito, Y.; Noguchi, N. Oxidized lipoprotein as a major vessel cell proliferator in oxidized human serum. PLoS ONE 2016, 11(8), e0160530. [Google Scholar] [CrossRef] [PubMed]
- Zheng, A.; Dubuis, G.; Georgieva, M.; Mendes Ferreira, C. S.; Serulla, M.; Del Carmen Conde Rubio, M.; Trofimenko, E.; Mercier, T.; Decosterd, L.; Widmann, C. HDLs extract lipophilic drugs from cells. Journal of Cell Science 2022, 135(5), jcs258644. [Google Scholar] [CrossRef] [PubMed]
- Negre-Salvayre, A.; Coatrieux, C.; Ingueneau, C.; Salvayre, R. Advanced lipid peroxidation end products in oxidative damage to proteins. Potential role in diseases and therapeutic prospects for the inhibitors. Potential role in diseases and therapeutic prospects for the inhibitors. British J Pharmacology 2008, 153(1), 6–20. [Google Scholar] [CrossRef]
- Vistoli, G.; De Maddis, D.; Cipak, A.; Zarkovic, N.; Carini, M.; Aldini, G. Advanced glycoxidation and lipoxidation end products (AGEs and ALEs): An overview of their mechanisms of formation. Free Radical Research 2013, 47 (sup1), 3–27. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J. M. C. Free Radicals in Biology and Medicine, 3rd ed.; Oxford science publications; Clarendon Press ; Oxford University Press: Oxford : New York, 1999. [Google Scholar]
- Fisher-Wellman, K.; Bloomer, R. J. Acute exercise and oxidative stress: A 30 year history. Dyn Med 2009, 8(1), 1. [Google Scholar] [CrossRef] [PubMed]
- Vasankari, T.; Kujala, U.; Heinonen, O.; Kapanen, J.; Ahotupa, M. Measurement of serum lipid peroxidation during exercise using three different methods: diene conjugation, thiobarbituric acid reactive material and fluorescent chromolipids. Clin Chim Acta. [CrossRef]
- Suomela, J.; Ahotupa, M.; Kallio, H. Triacylglycerol oxidation in pig lipoproteins after a diet rich in oxidized sunflower seed oil. Lipids 2005, 40(5), 437–444. [Google Scholar] [CrossRef] [PubMed]
- Wolff, S. P.; Nourooz-Zadeh, J. Hypothesis: UK consumption of dietary lipid hydroperoxides — a possible contributory factor to atherosclerosis. Atherosclerosis 1996, 119(2), 261–263. [Google Scholar] [CrossRef]
- Tarvainen, M.; Phuphusit, A.; Suomela, J.-P.; Kuksis, A.; Kallio, H. Effects of antioxidants on rapeseed oil oxidation in an artificial digestion model analyzed by UHPLC-ESI-MS. J Agric Food Chem 2012, 60(14), 3564–3579. [Google Scholar] [CrossRef]
- Nuora, A.; Chiang, V. S.-C.; Milan, A. M.; Tarvainen, M.; Pundir, S.; Quek, S.-Y.; Smith, G. C.; Markworth, J. F.; Ahotupa, M.; Cameron-Smith, D.; Linderborg, K. M. The impact of beef steak thermal processing on lipid oxidation and postprandial inflammation related responses. Food Chemistry 2015, 184, 57–64. [Google Scholar] [CrossRef]
- Tiainen, S.; Ahotupa, M.; Ylinen, P.; Vasankari, T. High density lipoprotein level is negatively associated with the increase of oxidized low density lipoprotein lipids after a fatty meal. Lipids 2014, 49(12), 1225–1232. [Google Scholar] [CrossRef]
- Cortés, B.; Núñez, I.; Cofán, M.; Gilabert, R.; Pérez-Heras, A.; Casals, E.; Deulofeu, R.; Ros, E. Acute effects of high-fat meals enriched with walnuts or olive oil on postprandial endothelial function. Journal of the American College of Cardiology 2006, 48(8), 1666–1671. [Google Scholar] [CrossRef] [PubMed]
- Välimäki, I.; Vuorimaa, T.; Ahotupa, M.; Vasankari, T. Effect of continuous and intermittent exercises on oxidised HDL and LDL lipids in runners. Int J Sports Med 2016, 37(14), 1103–1109. [Google Scholar] [CrossRef] [PubMed]
- Välimäki, I. A.; Vuorimaa, T.; Ahotupa, M.; Vasankari, T. J. Strenuous physical exercise accelerates the lipid peroxide clearing transport by HDL. Eur J Appl Physiol 2016, 116(9), 1683–1691. [Google Scholar] [CrossRef] [PubMed]
- Van Lenten, B. J.; Wagner, A. C.; Anantharamaiah, G. M.; Navab, M.; Reddy, S. T.; Buga, G. M.; Fogelman, A. M. Apolipoprotein A-I mimetic peptides. Curr Atheroscler Rep 2009, 11(1), 52–57. [Google Scholar] [CrossRef] [PubMed]
- Imaizumi, S.; Grijalva, V.; Navab, M. ; J. Van Lenten, B.; C. Wagner, A.; Anantharamaiah, G. M.; M. Fogelman, A.; T. Reddy, S. L-4F differentially alters plasma levels of oxidized fatty acids resulting in more anti-inflammatory HDL in mice. DML. [CrossRef]
- Morgantini, C.; Imaizumi, S.; Grijalva, V.; Navab, M.; Fogelman, A. M.; Reddy, S. T. Apolipoprotein A-I mimetic peptides prevent atherosclerosis development and reduce plaque inflammation in a murine model of diabetes. Diabetes 2010, 59(12), 3223–3228. [Google Scholar] [CrossRef] [PubMed]
- Elsøe, S.; Ahnström, J.; Christoffersen, C.; Hoofnagle, A. N.; Plomgaard, P.; Heinecke, J. W.; Binder, C. J.; Björkbacka, H.; Dahlbäck, B.; Nielsen, L. B. Apolipoprotein M binds oxidized phospholipids and increases the antioxidant effect of HDL. Atherosclerosis 2012, 221(1), 91–97. [Google Scholar] [CrossRef] [PubMed]
- Christison, J.; Karjalainen, A.; Brauman, J.; Bygrave, F.; Stocker, R. Rapid reduction and removal of HDL- but not LDL-associated cholesteryl ester hydroperoxides by rat liver perfused in situ. Biochemical Journal 1996, 314(3), 739–742. [Google Scholar] [CrossRef]
- Fluiter, K.; Vietsch, H.; Biessen, E. A.; Kostner, G. M.; van Berkel, T. J.; Sattler, W. Increased selective uptake in vivo and in vitro of oxidized cholesteryl esters from high-density lipoprotein by rat liver parenchymal cells. Biochem J 1996, 319 ( Pt 2) (Pt 2), 471–476. [Google Scholar] [CrossRef]
- Nourooz-Zadeh, J. Measures of oxidative stress in heterozygous familial hypercholesterolaemia. Atherosclerosis 2001, 156(2), 435–441. [Google Scholar] [CrossRef]
- Mollazadeh, H.; Carbone, F.; Montecucco, F.; Pirro, M.; Sahebkar, A. Oxidative burden in familial hypercholesterolemia. Journal Cellular Physiology 2018, 233(8), 5716–5725. [Google Scholar] [CrossRef] [PubMed]
- Shahsavari, G.; Raoufi, A.; Toolabi, A.; Hosseninejadmir, N.; Ahmadvand, H.; Safariebrahimsarabie, M. The Effect of atorvastatin treatment duration on oxidative stress markers and lipid profile in patients with coronary artery diseases: a case series study. ARYA Atheroscler 2017, 13(6), 282–287. [Google Scholar] [PubMed]
- Sørensen, A. L.; Hasselbalch, H. C.; Nielsen, C. H.; Poulsen, H. E.; Ellervik, C. Statin treatment, oxidative stress and inflammation in a danish population. Redox Biology 2019, 21, 101088. [Google Scholar] [CrossRef] [PubMed]
- Rosenson, R. S.; Brewer, H. B.; Ansell, B.; Barter, P.; Chapman, M. J.; Heinecke, J. W.; Kontush, A.; Tall, A. R.; Webb, N. R. Translation of high-density lipoprotein function into clinical practice: current prospects and future challenges. Circulation 2013, 128(11), 1256–1267. [Google Scholar] [CrossRef]
- Ahotupa, M.; Marniemi, J.; Lehtimäki, T.; Talvinen, K.; Raitakari, O. T.; Vasankari, T.; Viikari, J.; Luoma, J.; Ylä-Herttuala, S. Baseline diene conjugation in LDL lipids as a direct measure of in vivo LDL oxidation. Clinical Biochemistry 1998, 31(4), 257–261. [Google Scholar] [CrossRef] [PubMed]
- Kresanov, P.; Ahotupa, M.; Vasankari, T.; Kaikkonen, J.; Kähönen, M.; Lehtimäki, T.; Viikari, J.; Raitakari, O. T. The associations of oxidized high-density lipoprotein lipids with risk factors for atherosclerosis: The Cardiovascular Risk in Young Finns Study. Free Radical Biology and Medicine 2013, 65, 1284–1290. [Google Scholar] [CrossRef] [PubMed]
- Sniderman, A. D.; Islam, S.; McQueen, M.; Pencina, M.; Furberg, C. D.; Thanassoulis, G.; Yusuf, S. Age and cardiovascular risk attributable to apolipoprotein B, low-density lipoprotein cholesterol or non-high-density lipoprotein cholesterol. J Am Heart Assoc 2016, 5(10), e003665. [Google Scholar] [CrossRef] [PubMed]
- Kujala, U. M.; Ahotupa, M.; Vasankari, T.; Kaprio, J.; Tikkanen, M. J. Low LDL oxidation in veteran endurance athletes. Scandinavian Med Sci Sports 1996, 6(5), 303–308. [Google Scholar] [CrossRef]
- Kujala, U. M.; Ahotupa, M.; Vasankari, T. J.; Kaprio, J.; Tikkanen, M. J. Familial aggregation of LDL oxidation. Scandinavian Journal of Clinical and Laboratory Investigation 1997, 57(2), 141–146. [Google Scholar] [CrossRef]
- Vasankari, T.; Ahotupa, M.; Toikka, J.; Mikkola, J.; Irjala, K.; Pasanen, P.; Neuvonen, K.; Raitakari, O.; Viikari, J. Oxidized LDL and thickness of carotid intima-media are associated with coronary atherosclerosis in middle-aged men: lower levels of oxidized LDL with statin therapy. Atherosclerosis 2001, 155(2), 403–412. [Google Scholar] [CrossRef]
- Toikka, J. O.; Laine, H.; Ahotupa, M.; Haapanen, A.; Viikari, J. S. A.; Hartiala, J. J.; Raitakari, O. T. Increased arterial intima-media thickness and in vivo LDL oxidation in young men with borderline hypertension. Hypertension 2000, 36(6), 929–933. [Google Scholar] [CrossRef] [PubMed]
- Linna, M. S.; Ahotupa, M.; Irjala, K.; Pöllänen, P.; Huhtaniemi, I.; Mäkinen, J.; Perheentupa, A.; Vasankari, T. J. Smoking and low serum testosterone associates with high concentration of oxidized LDL. Annals of Medicine 2008, 40(8), 634–640. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Siegrist, J. Physical activity and risk of cardiovascular disease—a meta-analysis of prospective cohort studies. IJERPH 2012, 9(2), 391–407. [Google Scholar] [CrossRef] [PubMed]
- Martin, S. S.; Aday, A. W.; Almarzooq, Z. I.; Anderson, C. A. M.; Arora, P.; Avery, C. L.; Baker-Smith, C. M.; Barone Gibbs, B.; Beaton, A. Z.; Boehme, A. K.; Commodore-Mensah, Y.; Currie, M. E.; Elkind, M. S. V.; Evenson, K. R.; Generoso, G.; Heard, D. G.; Hiremath, S.; Johansen, M. C.; Kalani, R.; Kazi, D. S.; Ko, D.; Liu, J.; Magnani, J. W.; Michos, E. D.; Mussolino, M. E.; Navaneethan, S. D.; Parikh, N. I.; Perman, S. M.; Poudel, R.; Rezk-Hanna, M.; Roth, G. A.; Shah, N. S.; St-Onge, M.-P.; Thacker, E. L.; Tsao, C. W.; Urbut, S. M.; Van Spall, H. G. C.; Voeks, J. H.; Wang, N.-Y.; Wong, N. D.; Wong, S. S.; Yaffe, K.; Palaniappan, L. P. ; on behalf of the American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. 2024 Heart disease and stroke statistics: a report of US and global data from the American Heart Association. Circulation, 0000. [Google Scholar] [CrossRef]
- Vasankari, T.; Lehtonen-Veromaa, M.; Möttönen, T.; Ahotupa, M.; Irjala, K.; Heinonen, O.; Leino, A.; Viikari, J. Reduced mildly oxidized LDL in young female athletes. Atherosclerosis 2000, 151(2), 399–405. [Google Scholar] [CrossRef] [PubMed]
- Vasankari, T. J.; Kujala, U. M.; Vasankari, T. M.; Ahotupa, M. Reduced oxidized LDL levels after a 10-month exercise program: Medicine & Science in Sports & Exercise 1998, 30 (10), 1496–1501. [CrossRef]
- Välimäki, I. A.; Vuorimaa, T.; Ahotupa, M.; Kekkonen, R.; Korpela, R.; Vasankari, T. Decreased training volume and increased carbohydrate intake increases oxidized LDL levels. Int J Sports Med 2012, 33(4), 291–296. [Google Scholar] [CrossRef] [PubMed]
- Vuorimaa, T.; Ahotupa, M.; Irjala, K.; Vasankari, T. Acute prolonged exercise reduces moderately oxidized LDL in healthy men. Int J Sports Med 2005, 26(6), 420–425. [Google Scholar] [CrossRef]
- Tiainen, S.; Luoto, R.; Ahotupa, M.; Raitanen, J.; Vasankari, T. 6-mo aerobic exercise intervention enhances the lipid peroxide transport function of HDL. Free Radical Research 2016, 50(11), 1279–1285. [Google Scholar] [CrossRef]
- Vasankari, T. J.; Kujala, U. M.; Vasankari, T. M.; Vuorimaa, T.; Ahotupa, M. Effects of acute prolonged exercise on serum and LDL oxidation and antioxidant defences. Free Radical Biology and Medicine 1997, 22(3), 509–513. [Google Scholar] [CrossRef]
- Vasankari, T.; Kujala, U.; Vasankari, T.; Vuorimaa, T.; Ahotupa, M. Increased serum and low-density lipoprotein antioxidant potential after antioxidant supplementation in endurance athletes. The American Journal of Clinical Nutrition 1997, 65(4), 1052–1056. [Google Scholar] [CrossRef]
- Hubert, H. B.; Feinleib, M.; McNamara, P. M.; Castelli, W. P. Obesity as an independent risk factor for cardiovascular disease: a 26-year follow-up of participants in the Framingham Heart Study. Circulation 1983, 67(5), 968–977. [Google Scholar] [CrossRef]
- Calle, E. E.; Thun, M. J.; Petrelli, J. M.; Rodriguez, C.; Heath, C. W. Body mass index and mortality in a prospective cohort of U.S. adults. S. adults. N Engl J Med 1999, 341(15), 1097–1105. [Google Scholar] [CrossRef] [PubMed]
- Sorokin, A. V.; Kotani, K.; Elnabawi, Y. A.; Dey, A. K.; Sajja, A. P.; Yamada, S.; Ueda, M.; Harrington, C. L.; Baumer, Y.; Rodante, J. A.; Gelfand, J. M.; Chen, M. Y.; Joshi, A. A.; Playford, M. P.; Remaley, A. T.; Mehta, N. N. Association between oxidation-modified lipoproteins and coronary plaque in psoriasis: an observational cohort study. Circ Res 2018, 123(11), 1244–1254. [Google Scholar] [CrossRef] [PubMed]
- Vasankari, T.; Fogelholm, M.; Kukkonen-Harjula, K.; Nenonen, A.; Kujala, U.; Oja, P.; Vuori, I.; Pasanen, P.; Neuvonen, K.; Ahotupa, M. Reduced oxidized low-density lipoprotein after weight reduction in obese premenopausal women. Int J Obes 2001, 25(2), 205–211. [Google Scholar] [CrossRef] [PubMed]
- Linna, M. S.; Borg, P.; Kukkonen-Harjula, K.; Fogelholm, M.; Nenonen, A.; Ahotupa, M.; Vasankari, T. J. Successful weight maintenance preserves lower levels of oxidized LDL achieved by weight reduction in obese men. Int J Obes 2007, 31(2), 245–253. [Google Scholar] [CrossRef] [PubMed]
- Raitakari, M.; Ilvonen, T.; Ahotupa, M.; Lehtimäki, T.; Harmoinen, A.; Suominen, P.; Elo, J.; Hartiala, J.; Raitakari, O. T. Weight reduction with very-low-caloric diet and endothelial function in overweight adults: role of plasma glucose. Arterioscler Thromb Vasc Biol 2004, 24(1), 124–128. [Google Scholar] [CrossRef] [PubMed]
- Lee, C. D.; Blair, S. N.; Jackson, A. S. Cardiorespiratory fitness, body composition, and all-cause and cardiovascular disease mortality in men. The American Journal of Clinical Nutrition 1999, 69(3), 373–380. [Google Scholar] [CrossRef] [PubMed]
- Kosola, J.; Ahotupa, M.; Kyröläinen, H.; Santtila, M.; Vasankari, T. Good aerobic or muscular fitness protects overweight men from elevated oxidized LDL. Medicine & Science in Sports & Exercise. [CrossRef]
- Kaikkonen, J. E.; Kresanov, P.; Ahotupa, M.; Jula, A.; Mikkilä, V.; Viikari, J. S. A.; Kähönen, M.; Lehtimäki, T.; Raitakari, O. T. High serum n6 fatty acid proportion is associated with lowered LDL oxidation and inflammation: The Cardiovascular Risk in Young Finns Study. Free Radical Research 2014, 48(4), 420–426. [Google Scholar] [CrossRef]
- Harris, W. S.; Mozaffarian, D.; Rimm, E.; Kris-Etherton, P.; Rudel, L. L.; Appel, L. J.; Engler, M. M.; Engler, M. B.; Sacks, F. Omega-6 fatty acids and risk for cardiovascular disease: a science advisory from the American Heart Association Nutrition Subcommittee of the Council on Nutrition, Physical Activity, and Metabolism; Council on Cardiovascular Nursing; and Council on Epidemiology and Prevention. Circulation 2009, 119(6), 902–907. [Google Scholar] [CrossRef]
- Jula, A.; Marniemi, J.; Huupponen, R.; Virtanen, A.; Rastas, M.; Rönnemaa, T. Effects of diet and simvastatin on serum lipids, insulin, and antioxidants in hypercholesterolemic men: a randomized controlled trial. JAMA 2002, 287(5), 598. [Google Scholar] [CrossRef]
- Agarwal, S.; Rao, A. V. Tomato lycopene and low density lipoprotein oxidation: a human dietary intervention study. Lipids 1998, 33(10), 981–984. [Google Scholar] [CrossRef]
- Jenkins, D. J. A.; Kendall, C. W. C.; Marchie, A.; Parker, T. L.; Connelly, P. W.; Qian, W.; Haight, J. S.; Faulkner, D.; Vidgen, E.; Lapsley, K. G.; Spiller, G. A. Dose response of almonds on coronary heart disease risk factors: blood lipids, oxidized low-density lipoproteins, lipoprotein(a), homocysteine, and pulmonary nitric oxide: a randomized, controlled, crossover trial. Circulation 2002, 106(11), 1327–1332. [Google Scholar] [CrossRef] [PubMed]
- Marniemi, J.; Hakala, P.; Mäki, J.; Ahotupa, M. Partial resistance of low density lipoprotein to oxidation in vivo after increased intake of berries. Nutr Metab Cardiovasc Dis 2000, 10(6), 331–337. [Google Scholar] [PubMed]
- Suomela, J.-P.; Ahotupa, M.; Yang, B.; Vasankari, T.; Kallio, H. Absorption of flavonols derived from sea buckthorn ( Hippophaë Rhamnoides L.) and their effect on emerging risk factors for cardiovascular disease in humans. J. Agric. Food Chem. 7364. [Google Scholar] [CrossRef]
- Linna, M. S.; Ahotupa, M.; Kukkonen-Harjula, K.; Fogelholm, M.; Vasankari, T. J. Co-Existence of insulin resistance and high concentrations of circulating oxidized LDL lipids. Annals of Medicine 2015, 47(5), 394–398. [Google Scholar] [CrossRef] [PubMed]
- Kosola, J.; Vaara, J. P.; Ahotupa, M.; Kyröläinen, H.; Santtila, M.; Oksala, N.; Atalay, M.; Vasankari, T. Elevated concentration of oxidized LDL together with poor cardiorespiratory and abdominal muscle fitness predicts metabolic syndrome in young men. Metabolism 2013, 62(7), 992–999. [Google Scholar] [CrossRef] [PubMed]
- Targher, G.; Day, C. P.; Bonora, E. Risk of cardiovascular disease in patients with nonalcoholic fatty liver disease. N Engl J Med 2010, 363(14), 1341–1350. [Google Scholar] [CrossRef] [PubMed]
- Kaikkonen, J. E.; Kresanov, P.; Ahotupa, M.; Jula, A.; Mikkilä, V.; Viikari, J. S. A.; Juonala, M.; Hutri-Kähönen, N.; Kähönen, M.; Lehtimäki, T.; Kangas, A. J.; Soininen, P.; Ala-Korpela, M.; Raitakari, O. T. Longitudinal study of circulating oxidized LDL and HDL and fatty liver: The Cardiovascular Risk in Young Finns Study. Free Radical Research 2016, 50(4), 396–404. [Google Scholar] [CrossRef] [PubMed]
- Raitakari, O. T.; Toikka, J. O.; Laine, H.; Ahotupa, M.; Iida, H.; Viikari, J. S. A.; Hartiala, J.; Knuuti, J. Reduced myocardial flow reserve relates to increased carotid intima-media thickness in healthy young men. Atherosclerosis 2001, 156(2), 469–475. [Google Scholar] [CrossRef] [PubMed]
- Toikka, J. O.; Niemi, P.; Ahotupa, M.; Niinikoski, H.; Viikari, J. S. A.; Rönnemaa, T.; Hartiala, J. J.; Raitakari, O. T. Large-artery elastic properties in young men: relationships to serum lipoproteins and oxidized low-density lipoproteins. ATVB 1999, 19(2), 436–441. [Google Scholar] [CrossRef]
- Linna, M.; Ahotupa, M.; Lopponen, M. K.; Irjala, K.; Vasankari, T. Circulating oxidised LDL lipids, when proportioned to HDL-c, emerged as a risk factor of all-cause mortality in a population-based survival study. Age and Ageing 2013, 42(1), 110–113. [Google Scholar] [CrossRef]
- Randomised trial of cholesterol lowering in 4444 patients with coronary heart disease: The Scandinavian Simvastatin Survival Study (4S). Lancet 1994, 344(8934), 1383–1389.
- Shepherd, J.; Cobbe, S. M.; Ford, I.; Isles, C. G.; Lorimer, A. R.; Macfarlane, P. W.; McKillop, J. H.; Packard, C. J. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. N Engl J Med 1995, 333(20), 1301–1308. [Google Scholar] [CrossRef] [PubMed]
- Vasankari, T.; Ahotupa, M.; Viikari, J.; Nuotio, I.; Vuorenmaa, T.; Strandberg, T.; Vanhanen, H.; Tikkanen, M. J. Effects of statin therapy on circulating conjugated dienes, a measure of LDL Oxidation. Atherosclerosis 2005, 179(1), 207–209. [Google Scholar] [CrossRef] [PubMed]
- Janatuinen, T.; Knuuti, J.; Toikka, J. O.; Ahotupa, M.; Nuutila, P.; Rönnemaa, T.; Raitakari, O. T. Effect of pravastatin on low-density lipoprotein oxidation and myocardial perfusion in young adults with type 1 diabetes. ATVB 2004, 24(7), 1303–1308. [Google Scholar] [CrossRef] [PubMed]
- et al. Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90 056 participants in 14 randomised trials of statins. The Lancet 2005, 366(9493), 1267–1278. [Google Scholar] [CrossRef] [PubMed]
- Lawler, P. R.; Akinkuolie, A. O.; Chu, A. Y.; Shah, S. H.; Kraus, W. E.; Craig, D.; Padmanabhan, L.; Glynn, R. J.; Ridker, P. M.; Chasman, D. I.; Mora, S. Atherogenic lipoprotein determinants of cardiovascular disease and residual risk among individuals with low low-density lipoprotein cholesterol. JAHA 2017, 6(7), e005549. [Google Scholar] [CrossRef] [PubMed]
- Marschang, P.; Götsch, C.; Kirchmair, R.; Kaser, S.; Kähler, C. M.; Patsch, J. R. Postprandial, but not postabsorptive low-density lipoproteins increase the expression of intercellular adhesion molecule-1 in human aortic endothelial cells. Atherosclerosis 2006, 186(1), 101–106. [Google Scholar] [CrossRef] [PubMed]
- Sumida, Y.; Niki, E.; Naito, Y.; Yoshikawa, T. Involvement of free radicals and oxidative stress in NAFLD/NASH. Free Radical Research 2013, 47(11), 869–880. [Google Scholar] [CrossRef] [PubMed]
- Bigagli, E.; Lodovici, M. Circulating oxidative stress biomarkers in clinical studies on type 2 diabetes and its complications. Oxidative Medicine and Cellular Longevity 2019, 2019, 1–17. [Google Scholar] [CrossRef]
- Oberg, B. P.; McMenamin, E.; Lucas, F. L.; McMonagle, E.; Morrow, J.; Ikizler, T. A. L. P.; Himmelfarb, J. Increased prevalence of oxidant stress and inflammation in patients with moderate to severe chronic kidney disease. Kidney International 2004, 65(3), 1009–1016. [Google Scholar] [CrossRef]
- Tucker, P. S.; Scanlan, A. T.; Dalbo, V. J. Chronic kidney disease influences multiple systems: describing the relationship between oxidative stress, inflammation, kidney damage, and concomitant disease. Oxidative Medicine and Cellular Longevity 2015, 2015, 1–8. [Google Scholar] [CrossRef]
- Ho, H.-J.; Shirakawa, H. Oxidative stress and mitochondrial dysfunction in chronic kidney disease. Cells 2022, 12(1), 88. [Google Scholar] [CrossRef] [PubMed]
- Ruan, X. Z.; Varghese, Z.; Moorhead, J. F. An Update on the lipid nephrotoxicity hypothesis. Nat Rev Nephrol 2009, 5(12), 713–721. [Google Scholar] [CrossRef] [PubMed]
- De Ferranti, S. D.; De Boer, I. H.; Fonseca, V.; Fox, C. S.; Golden, S. H.; Lavie, C. J.; Magge, S. N.; Marx, N.; McGuire, D. K.; Orchard, T. J.; Zinman, B.; Eckel, R. H. Type 1 diabetes mellitus and cardiovascular disease: a scientific statement from the American Heart Association and American Diabetes Association. Circulation 2014, 130(13), 1110–1130. [Google Scholar] [CrossRef] [PubMed]
- Bae, J. C.; Han, J. M.; Kwon, S.; Jee, J. H.; Yu, T. Y.; Lee, M. K.; Kim, J. H. LDL-C/apoB and HDL-C/apoA-1 ratios predict incident chronic kidney disease in a large apparently healthy cohort. Atherosclerosis 2016, 251, 170–176. [Google Scholar] [CrossRef]
- Jialal, I.; Devaraj, S. Antioxidants and atherosclerosis: don’t throw out the baby with the bath water. Circulation 2003, 107(7), 926–928. [Google Scholar] [CrossRef] [PubMed]
- Ahotupa, M.; Vasankari, T. J. Baseline diene conjugation in LDL lipids: Free Radical Biology and Medicine 1999, 27 (11–12), 1141–1150. [CrossRef]
- Bacot, S.; Bernoud-Hubac, N.; Chantegrel, B.; Deshayes, C.; Doutheau, A.; Ponsin, G.; Lagarde, M.; Guichardant, M. Evidence for in situ ethanolamine phospholipid adducts with hydroxyalkenals. Journal of Lipid Research 2007, 48(4), 816–825. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J. L.; Brown, M. S. The LDL receptor. ATVB 2009, 29(4), 431–438. [Google Scholar] [CrossRef]
|
|
|
|
|
|
|
|
|
| Influence of / association with | Oxidized LDL lipids | Oxidized HDL lipids |
|---|---|---|
| Age | Increases with age | Decreases with age |
| Gender | Male > female | Female > male |
| Body mass index | Positive association | Negative association |
| Weight reduction | Substantial decrease | Not studied |
| Tobacco smoking | Elevated among smokers | Not studied |
| Statin treatment | Substantial decrease | |
| Acute physical exercise | No change / decrease | Increase |
| Physically active lifestyle | Substantial decrease | Slight increase |
| Fatty meal | Substantial increase within 2 hours | Slight increase after several hours |
| Saturated fat | Positive association | Not studied |
| Polyunsaturated fat | Negative association with n-6 PUFA | Not studied |
| Atherosclerosis | Positive association with brachial, carotid, and coronary atherosclerosis | Negative association with coronary atherosclerosis |
| Insulin resistance | Positive association | Not studied |
| Metabolic syndrome | Positive association | Not studied |
| Development of fatty liver | Positive association | Negative association |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




