Submitted:
11 March 2024
Posted:
12 March 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Patients and Methods
Study Design
Endocrinological Examination
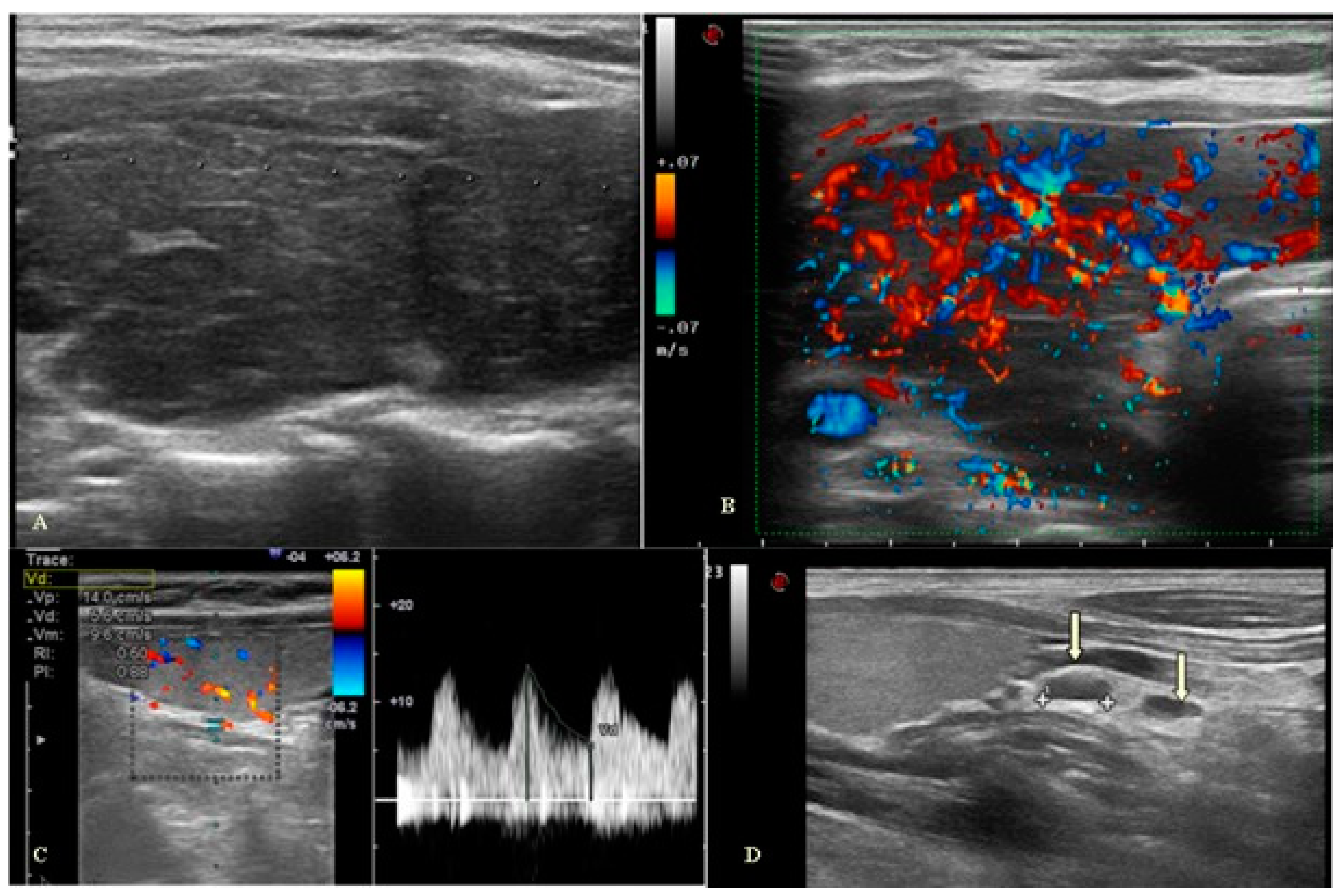
Ultrasound Evaluation
Dermatological Examination
Statistical Analysis
Results
Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Ethics
Data Availability Statement
Acknowledgments
Conflicts of interest
References
- Skevaki, C.; Wesemann, D.R. Antibody repertoire and autoimmunity. J Allergy Clin Immunol 2023, 151, 898–900. [Google Scholar] [CrossRef]
- Huffaker, M.F.; Sanda, S.; Chandran, S.; Chung, S.A.; St Clair, E.W.; Nepom, G.T.; Smilek, D.E. Approaches to establishing tolerance in immune mediated diseases. Front Immunol 2021, 12, 744804. [Google Scholar] [CrossRef]
- Hu, Y.; Zhang, L.; Chen, H.; Liu, X.; Zheng, X.; Shi, H.; Jiang, L.; Cui, D. Analysis of regulatory T cell subsets and their expression of helios and PD-1 in patients with Hashimoto thyroiditis. Int J Endocrinol 2019, 2019, 5368473. [Google Scholar] [CrossRef]
- Caturegli, P.; De Remigis, A.; Rose, N.R. Hashimoto thyroiditis: Clinical and diagnostic criteria. Autoimmun Rev 2014, 13, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Ragusa, F.; Fallahi, P.; Elia, G.; Gonnella, D.; Paparo, S.R.; Giusti, C.; Churilov, L.P.; Ferrari, S.M.; Antonelli, A. Hashimotos' thyroiditis: Epidemiology, pathogenesis, clinic and therapy. Best Pract Res Clin Endocrinol Metab 2019, 33, 101367. [Google Scholar] [CrossRef] [PubMed]
- Braverman LECooper DSWerner SCIngbar, S.H. Werner & Ingbar's the Thyroid: A Fundamental and Clinical Text. 10th ed. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins Health; 2013.
- Weetman, A.P. An update on the pathogenesis of Hashimoto's thyroiditis. J Endocrinol Invest 2021, 44, 883–890. [Google Scholar] [CrossRef]
- Mincer, D.L.; Jialal, I. Hashimoto Thyroiditis. [Updated 2023 Jul 29]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459262/.
- Virmani, V.; Hammond, I. Sonographic patterns of benign thyroid nodules: Verification at our institution. AJR Am J Roentgenol 2011, 196, 891–895. [Google Scholar] [CrossRef]
- Khati, N.; Adamson, T.; Johnson, K.S.; Hill, M.C. Ultrasound of the thyroid and parathyroid glands. Ultrasound Q 2003, 19, 162–176. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.; Middleton, W.D.; Teefey, S.A.; Reading, C.C.; Langer, J.E.; Desser, T.; Szabunio, M.M.; Hildebolt, C.F.; Mandel, S.J.; Cronan, J.J. Hashimoto thyroiditis: Part 1, sonographic analysis of the nodular form of Hashimoto thyroiditis. AJR Am J Roentgenol 2010, 195, 208–215. [Google Scholar] [CrossRef]
- Bogazzi, F.; Bartalena, L.; Brogioni, S.; Burelli, A.; Manetti, L.; Tanda, M.L.; Gasperi, M.; Martino, E. Thyroid vascularity and blood flow are not dependent on serum thyroid hormone levels: Studies in vivo by color flow doppler sonography. Eur J Endocrinol 1999, 140, 452–456. [Google Scholar] [CrossRef]
- Lagalla, R.; Caruso, G.; Novara, V.; Cardinale, A.E. Analisi flussimetrica nelle malattie tiroidee: Ipotesi di integrazione con lo studio qualitativo con color-Doppler [Flowmetric analysis of thyroid diseases: Hypothesis on integration with qualitative color-Doppler study]. Radiol Med 1993, 85, 606–610. (In Italian) [Google Scholar]
- Ceylan, I.; Yener, S.; Bayraktar, F.; Secil, M. Roles of ultrasound and power Doppler ultrasound for diagnosis of Hashimoto thyroiditis in anti-thyroid marker-positive euthyroid subjects. Quant Imaging Med Surg 2014, 4, 232–238. [Google Scholar] [CrossRef]
- Sarikaya, B.; Demirbilek, H.; Akata, D.; Kandemir, N. The role of the resistive index in Hashimoto's thyroiditis: A sonographic pilot study in children. Clinics (Sao Paulo) 2012, 67, 1253–1257. [Google Scholar] [CrossRef]
- Jones, M.R.; Mohamed, H.; Catlin, J.; April, D.; Al-Qurayshi, Z.; Kandil, E. The presentation of lymph nodes in Hashimoto's thyroiditis on ultrasound. Gland Surg 2015, 4, 301–306. [Google Scholar] [CrossRef]
- Campanati, A.; Martina, E.; Offidani, A. The challenge arising from new knowledge about immune and inflammatory skin diseases: Where we are today and where we are going. Biomedicines 2022, 10, 950. [Google Scholar] [CrossRef]
- Cianfarani F, Baldini E, Cavalli A, Marchioni E, Lembo L, Teson M, Persechino S, Zambruno G, Ulisse S, Odorisio T, D'Armiento M. TSH receptor and thyroid-specific gene expression in human skin. J Invest Dermatol 2010, 130, 93–101. [Google Scholar] [CrossRef]
- Brankov, N.; Conic, R.Z.; Atanaskova-Mesinkovska, N.; Piliang, M.; Bergfeld, W.F. Comorbid conditions in lichen planopilaris: A retrospective data analysis of 334 patients. Int J Womens Dermatol 2018, 4, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Kasumagic-Halilovic, E.; Begovic, B. Thyroid autoimmunity in patients with skin disorders. In: Thyroid Hormone. Agrawal NK. (Eds). InTech Open. (2012). [CrossRef]
- Aithal, S.; Ganguly, S.; Kuruvila, S. Coexistence of mucous membrane pemphigoid and vitiligo. Indian Dermatol Online J 2014, 5, 485–487. [Google Scholar] [CrossRef] [PubMed]
- Antonelli, A.; Fallahi, P.; Mosca, M.; Ferrari, S.M.; Ruffilli, I.; Corti, A.; Panicucci, E.; Neri, R.; Bombardieri, S. Prevalence of thyroid dysfunctions in systemic lupus erythematosus. Metabolism 2010, 59, 896–900. [Google Scholar] [CrossRef] [PubMed]
- Bagnato, G.L.; Roberts, W.N.; Fiorenza, A.; Arcuri, C.; Certo, R.; Trimarchi, F.; Ruggeri, R.M.; Bagnato, G.F. Skin fibrosis correlates with circulating thyrotropin levels in systemic sclerosis: Translational association with Hashimoto's thyroiditis. Endocrine 2016, 51, 291–297. [Google Scholar] [CrossRef]
- Fallahi, P.; Ruffilli, I.; Giuggioli, D.; Colaci, M.; Ferrari, S.M.; Antonelli, A.; Ferri, C. Associations between systemic sclerosis and thyroid diseases. Front Endocrinol (Lausanne) 2017, 8, 266. [Google Scholar] [CrossRef]
- Baldini, E.; Odorisio, T.; Tuccilli, C.; Persechino, S.; Sorrenti, S.; Catania, A.; Pironi, D.; Carbotta, G.; Giacomelli, L.; Arcieri, S.; Vergine, M.; Monti, M.; Ulisse, S. Thyroid diseases and skin autoimmunity. Rev Endocr Metab Disord 2018, 19, 311–323. [Google Scholar] [CrossRef]
- Kunzler, E.; Hynan, L.S.; Chong, B.F. Autoimmune diseases in patients with cutaneous lupus erythematosus. JAMA Dermatol 2018, 154, 712–716. [Google Scholar] [CrossRef]
- Mahajan, V.K.; Vashist, S.; Chauhan, P.S.; Mehta, K.I.S.; Sharma, V.; Sharma, A. Clinico-Epidemiological Profile of Patients with Vitiligo: A Retrospective Study from a Tertiary Care Center of North India. Indian Dermatol Online J 2019, 10, 38–44. [Google Scholar] [CrossRef]
- Tomer, Y. Genetic susceptibility to autoimmune thyroid disease: Past, present, and future. Thyroid 2010, 20, 715–725. [Google Scholar] [CrossRef]
- Furue, K.; Ito, T.; Tsuji, G.; Kadono, T.; Nakahara, T.; Furue, M. Autoimmunity and autoimmune co-morbidities in psoriasis. Immunology 2018, 154, 21–27. [Google Scholar] [CrossRef]
- Li, D.; Liang, G.; Calderone, R.; Bellanti, J.A. Vitiligo and Hashimoto's thyroiditis: Autoimmune diseases linked by clinical presentation, biochemical commonality, and autoimmune/oxidative stress-mediated toxicity pathogenesis. Med Hypotheses 2019, 128, 69–75. [Google Scholar] [CrossRef]
- Diotallevi, F.; Campanati, A.; Martina, E.; Radi, G.; Paolinelli, M.; Marani, A.; Molinelli, E.; Candelora, M.; Taus, M.; Galeazzi, T.; Nicolai, A.; Offidani, A. The role of nutrition in immune-mediated, inflammatory skin disease: A narrative review. Nutrients 2022, 14, 591. [Google Scholar] [CrossRef] [PubMed]
- McInnes, I.B.; Gravallese, E.M. Immune-mediated inflammatory disease therapeutics: Past, present and future. Nat Rev Immunol 2012, 21, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Baldini, E.; Odorisio, T.; Sorrenti, S.; Catania, A.; Tartaglia, F.; Carbotta, G.; Pironi, D.; Rendina, R.; D'Armiento, E.; Persechino, S.; Ulisse, S. Vitiligo and Autoimmune Thyroid Disorders. Front Endocrinol (Lausanne) 2017, 8, 290. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.M.; Lee, J.H.; Yun, J.S.; Han, B.; Han, T.Y. Vitiligo and overt thyroid diseases: A nationwide population-based study in Korea. J Am Acad Dermatol. 2017, 76, 871–878. [Google Scholar] [CrossRef]
- Liu, C.W.; Huang, Y.C. Vitiligo and autoantibodies: A systematic review and meta-analysis. J Dtsch Dermatol Ges 2018, 16, 845–851. [Google Scholar] [CrossRef]
- Vrijman, C.; Kroon, M.W.; Limpens, J.; Leeflang, M.M.; Luiten, R.M.; van der Veen, J.P.; Wolkerstorfer, A.; Spuls, P.I. The prevalence of thyroid disease in patients with vitiligo: A systematic review. Br J Dermatol 2012, 167, 1224–1235. [Google Scholar] [CrossRef]
- Yuan, J.; Sun, C.; Jiang, S.; Lu, Y.; Zhang, Y.; Gao, X.H.; Wu, Y.; Chen, H.D. The prevalence of thyroid disorders in patients with vitiligo: A systematic review and meta-analysis. Front Endocrinol (Lausanne) 2019, 9, 803. [Google Scholar] [CrossRef] [PubMed]
- Kheradmand, M.; Afshari, M.; Damiani, G.; Abediankenari, S.; Moosazadeh, M. Melasma and thyroid disorders: A systematic review and meta-analysis. Int J Dermatol 2019, 58, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Kiguradze, T.; Bruins, F.M.; Guido, N.; Bhattacharya, T.; Rademaker, A.; Florek, A.G.; Posligua, A.; Amin, S.; Laumann, A.E.; West, D.P.; Nardone, B. Evidence for the association of Hashimoto's thyroiditis with psoriasis: A cross-sectional retrospective study. Int J Dermatol 2017, 56, 553–556. [Google Scholar] [CrossRef]
- Ghazanfar, M.N.; Kibsgaard, L.; Thomsen, S.F.; Vestergaard, C. Risk of comorbidities in patients diagnosed with chronic urticaria: A nationwide registry-study. World Allergy Organ J 2020, 13, 100097. [Google Scholar] [CrossRef]
- Najafipour, M.; Zareizadeh, M.; Najafipour, F. Relationship between chronic urticaria and autoimmune thyroid disease. J Adv Pharm Technol Res 2018, 9, 158–161. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Diaz, S.N.; Sanchez-Borges, M.; Rangel-Gonzalez, D.M.; Guzman-Avilan, R.I.; Canseco-Villarreal, J.I.; Arias-Cruz, A. Chronic urticaria and thyroid pathology. World Allergy Organ J 2020, 13, 100101. [Google Scholar] [CrossRef]
- Gill, L.; Zarbo, A.; Isedeh, P.; Jacobsen, G.; Lim, H.W.; Hamzavi, I. Comorbid autoimmune diseases in patients with vitiligo: A cross-sectional study. J Am Acad Dermatol 2016, 74, 295–302. [Google Scholar] [CrossRef]
- Han, T.Y.; Lee, J.H.; Noh, T.K.; Choi, M.W.; Yun, J.S.; Lee, K.H.; Bae, J.M. Alopecia areata and overt thyroid diseases: A nationwide population-based study. J Dermatol 2018, 45, 1411–1417. [Google Scholar] [CrossRef]
- Lee, S.; Lee, Y.B.; Kim, B.J.; Lee, W.S. Screening of thyroid function and autoantibodies in patients with alopecia areata: A systematic review and meta-analysis. J Am Acad Dermatol 2019, 80, 1410–1413.e4. [Google Scholar] [CrossRef]
- Guarneri, F.; Giuffrida, R.; Di Bari, F.; Cannavò, S.P.; Benvenga, S. Thyroid autoimmunity and Lichen. Front Endocrinol (Lausanne) 2017, 8, 146. [Google Scholar] [CrossRef]
- Robledo-Sierra, J.; Landin-Wilhelmsen, K.; Filipsson Nyström, H.; Eggertsen, R.; Larsson, L.; Dafar, A.; Warfvinge, G.; Mattsson, U.; Jontell, M. A mechanistic linkage between oral lichen planus and autoimmune thyroid disease. Oral Dis 2018, 24, 1001–1011. [Google Scholar] [CrossRef]
- Serni, L.; Barbato, L.; Nieri, M.; Mallardi, M.; Noce, D.; Cairo, F. Association between oral lichen planus and Hashimoto thyroiditis: A systematic review. Oral Dis 2023, Epub ahead of print. [CrossRef]
- Triggianese, P.; Conigliaro, P.; Chimenti, M.S.; Barbato, C.; Greco, E.; Kroegler, B.; De Carolis, C.; Perricone, R. Systemic sclerosis: Exploring the potential interplay between thyroid disorders and pregnancy outcome in an Italian Cohort. Isr Med Assoc J 2017, 19, 473–477. [Google Scholar]
- Niepomniszcze, H.; Huaier Amad, R. Skin disorders and thyroid diseases. J Endocrinol Invest 2001, 24, 628–638. [Google Scholar] [CrossRef]
- Mysliwiec, J.; Oklota, M.; Nikolajuk, A.; Waligorski, D.; Gorska, M. Serum CD40/CD40L system in Graves' disease and Hashimoto's thyroiditis related to soluble Fas, FasL and humoral markers of autoimmune response. Immunol Invest 2007, 36, 247–257. [Google Scholar] [CrossRef]
- Tandon N, Zhang L, Weetman AP. HLA associations with Hashimoto's thyroiditis. Clin Endocrinol (Oxf) 1991, 34, 383–386. [Google Scholar] [CrossRef]
- Chen, H.; Poon, A.; Yeung, C.; Helms, C.; Pons, J.; Bowcock, A.M.; Kwok, P.Y.; Liao, W. A genetic risk score combining ten psoriasis risk loci improves disease prediction. PLoS ONE 2011, 6, e19454. [Google Scholar] [CrossRef]
- Vita, R.; Di Bari, F.; Perelli, S.; Capodicasa, G.; Benvenga, S. Thyroid vascularization is an important ultrasonographic parameter in untreated Graves' disease patients. J Clin Transl Endocrinol 2019, 15, 65–69. [Google Scholar] [CrossRef]
- Yuen, H.Y.; Wong, K.T.; Ahuja, A.T. Sonography of diffuse thyroid disease. Australas J Ultrasound Med 2016, 19, 13–29. [Google Scholar] [CrossRef]
- Medlock, D.; Chaljub, E.; Gavin, M.; Peiris, A.N. Shifting cervical lymphadenopathy in Hashimoto's disease. Proc (Bayl Univ Med Cent) 2019, 32, 235–236. [Google Scholar] [CrossRef]
- Ahmed, R.; Al-Shaikh, S.; Akhtar, M. Hashimoto thyroiditis: A century later. Adv Anat Pathol 2012, 19, 181–186. [Google Scholar] [CrossRef]
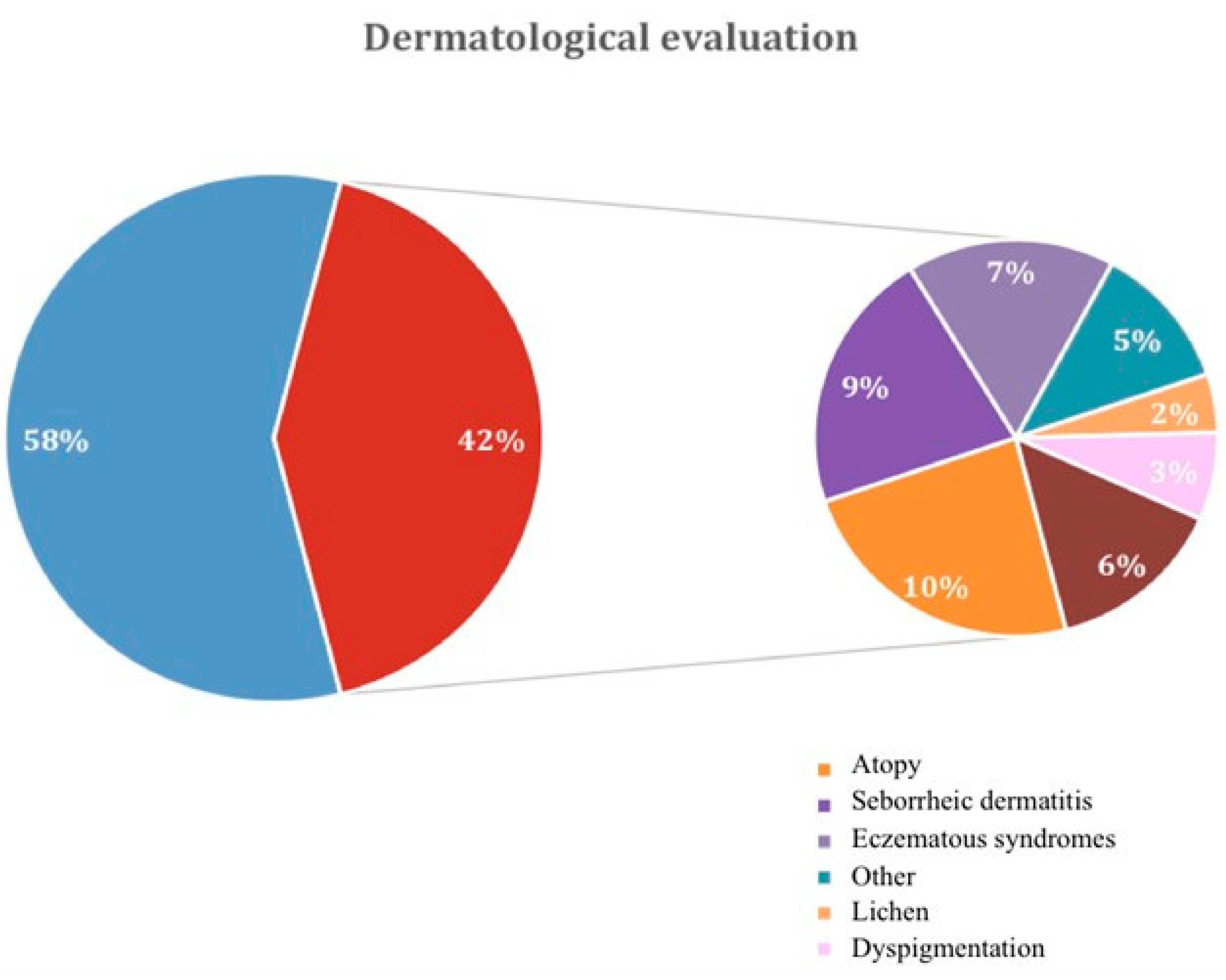
| Dermatological disease | p-value | ||
|---|---|---|---|
| No (n=58) | Yes (n=42) | ||
| Age (years), median (min–max) | 48 (19–72) | 44.5 (22–72) | 0.869† |
| BMI, median (min–max) | 23.7 (17.3–43.9) | 22.2 (17.6–35.7) | 0.262‡ |
| Sex, n (%): | |||
|
53 (91.4) | 39 (92.9) | 0.999§ |
|
5 (8.6) | 3 (7.1) | |
| Familiarity, n (%): | |||
|
45 (77.6) | 20 (47.6) | 0.002¶ |
|
13 (22.4) | 22 (52.4) | |
| Dermatological disease | Mann–Whitney test (p-value) | ||
|---|---|---|---|
| No (n=58), median (min–max) | Yes (n=42), median (min–max) | ||
| TSH: | 2.9 (0.1–22.2) | 2.8 (0.1–12.5) | 0.632 |
|
42 (72.4) | 33 (78.6) | 0.483† |
|
16 (27.6) | 9 (21.4) | |
| FT3 | 3 (0.9–12.7) | 3.1 (0.8–20.0) | 0.955 |
| FT4 | 1.2 (0.2–26.0) | 1.1 (0.6–12.0) | 0.834 |
| Ab-TPO | 155.5 (9.0–11,728.0) | 193.5 (0.7–13,000.0) | 0.617 |
| Ab-TG | 152.5 (0.3–3,854.0) | 97.0 (4.6–1,000.0) | 0.078 |
| Dermatological disease, median (min–max)/n (%) | Mann–Whitney test (p-value) | |||
|---|---|---|---|---|
| No (n=58) | Yes (n=42) | |||
| AP thyroid size (mm): | ||||
|
14.5 (1.54–26.0) | 15 (8.0–35.0) | 0.517 | |
|
15.0 (1.7–26.0) | 15.4 (9.0–49.0) | 0.230 | |
| Thyroid nodule size (mm) | 7.0 (2.0–27.0) | 7.0 (0.7–20.0) | 0.525 | |
| Regular margins: | ||||
|
27 (46.6) | 12 (29.3) | 0.083† | |
|
31 (53.4) | 29 (70.7) | ||
| Thyroid echogenicity: | ||||
|
50 (86.2) | 37 (90.2) | 0.648† | |
|
7 (12.1) | 4 (9.8) | ||
|
1 (1.7) | 0 (0.0) | ||
| Parenchymal vascularity: | ||||
|
13 (22.4) | 13 (31.7) | 0.301† | |
|
45 (77.6) | 28 (68.3) | ||
| Lymph node level: | ||||
|
7 (12.3) | 0 (0.0) | 0.039‡ | |
|
50 (87.7) | 41 (100.0) | ||
| Inferior thyroid RI and diameter: | ||||
|
0.6 (0.5–0.9) | 0.7 (0.5–0.8) | 0.237 | |
|
0.6 (0.5–0.8) | 0.7 (0.5–0.7) | 0.116 | |
|
1.3 (0.8–16.0) | 1.6 (0.8–16.0) | 0.023 | |
|
1.2 (0.7–3.0) | 1.5 (0.9–2.9) | 0.005 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




