Submitted:
05 March 2024
Posted:
06 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Yeast Strains and Isolation of Old C. neoformans Cells
2.2. Macrophage Cell Line Culture
2.3. Urease Activity
2.4. Analysis of the Golgi Apparatus
2.5. Polysaccharide Capsule Analysis
2.6. DNA Staining and Cell Cycle Analysis
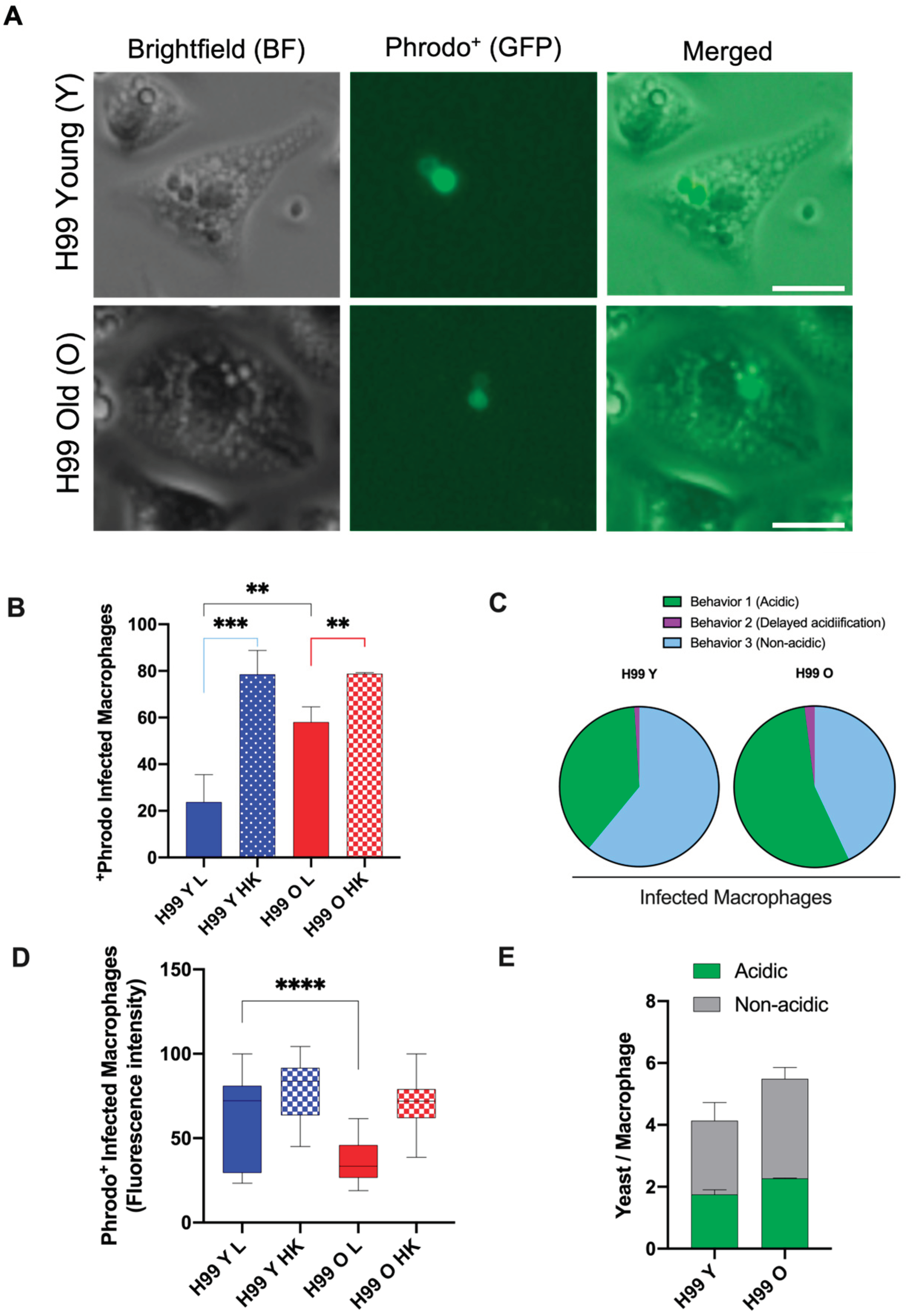
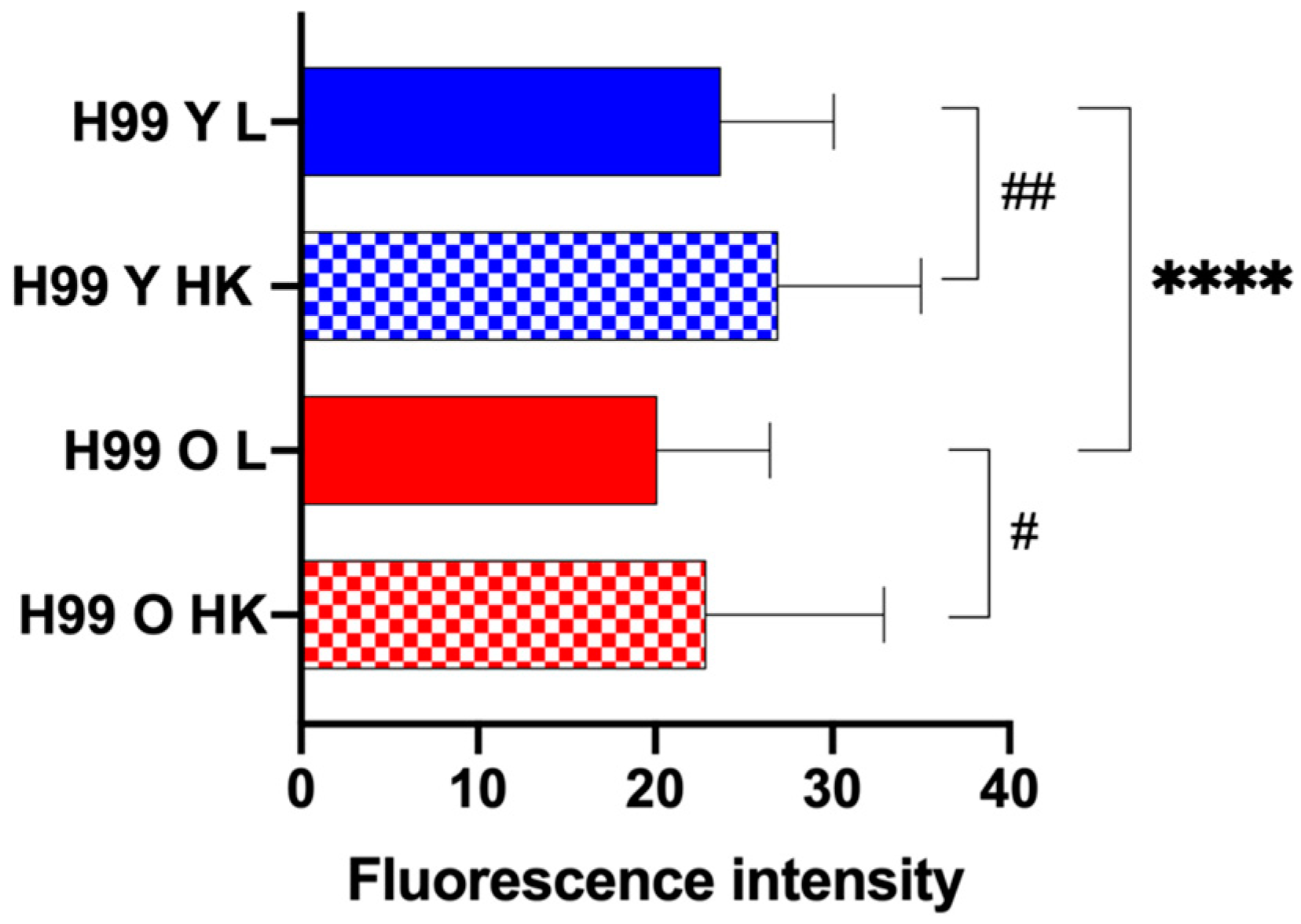
2.7. Phagocytosis Assay, Phagosomal Acidification, and Phagosomal Permeability
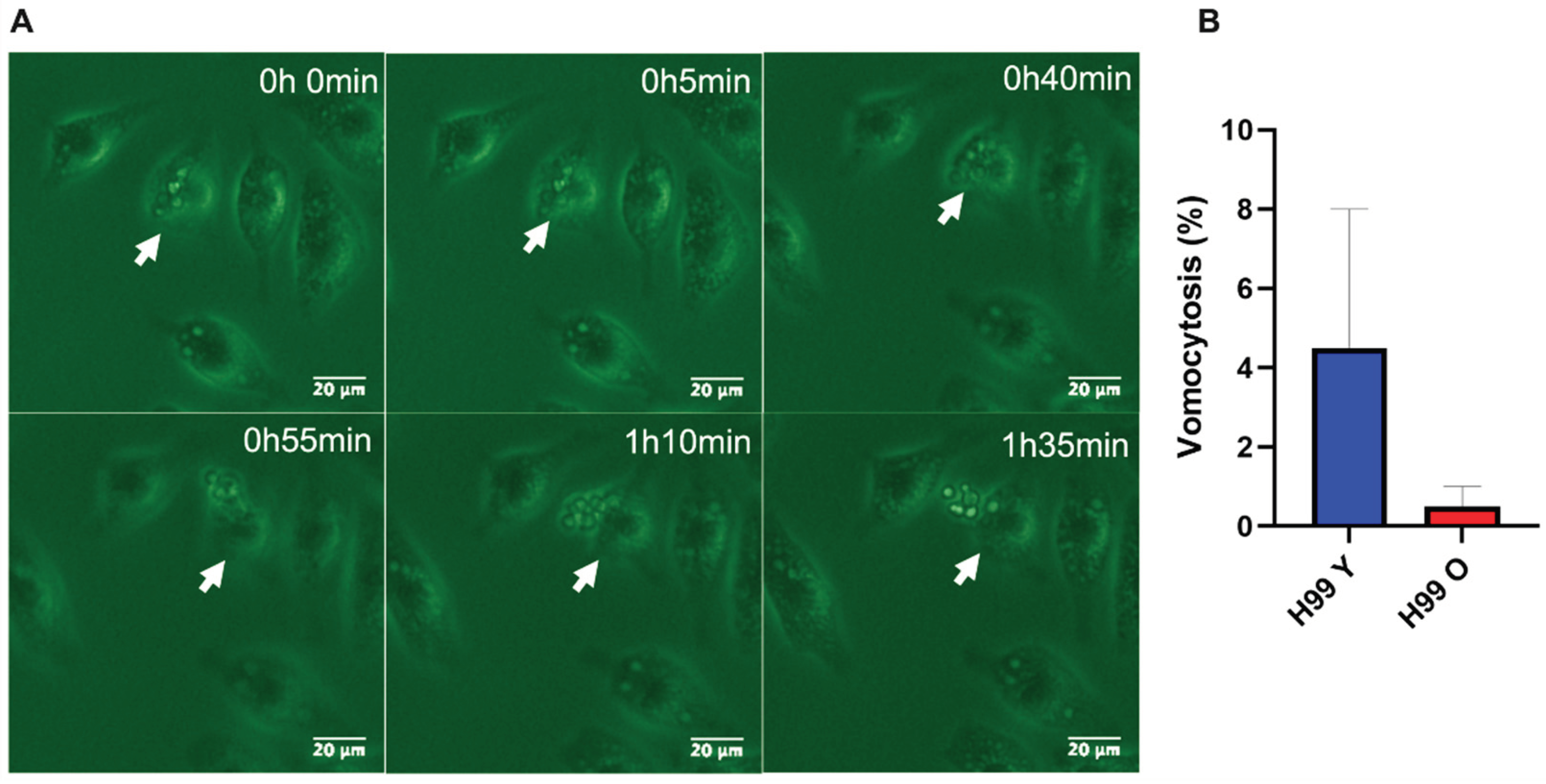
2.8. Time-Lapse Microscopy and Vomocytosis Rate
2.9. Data Analysis
3. Results
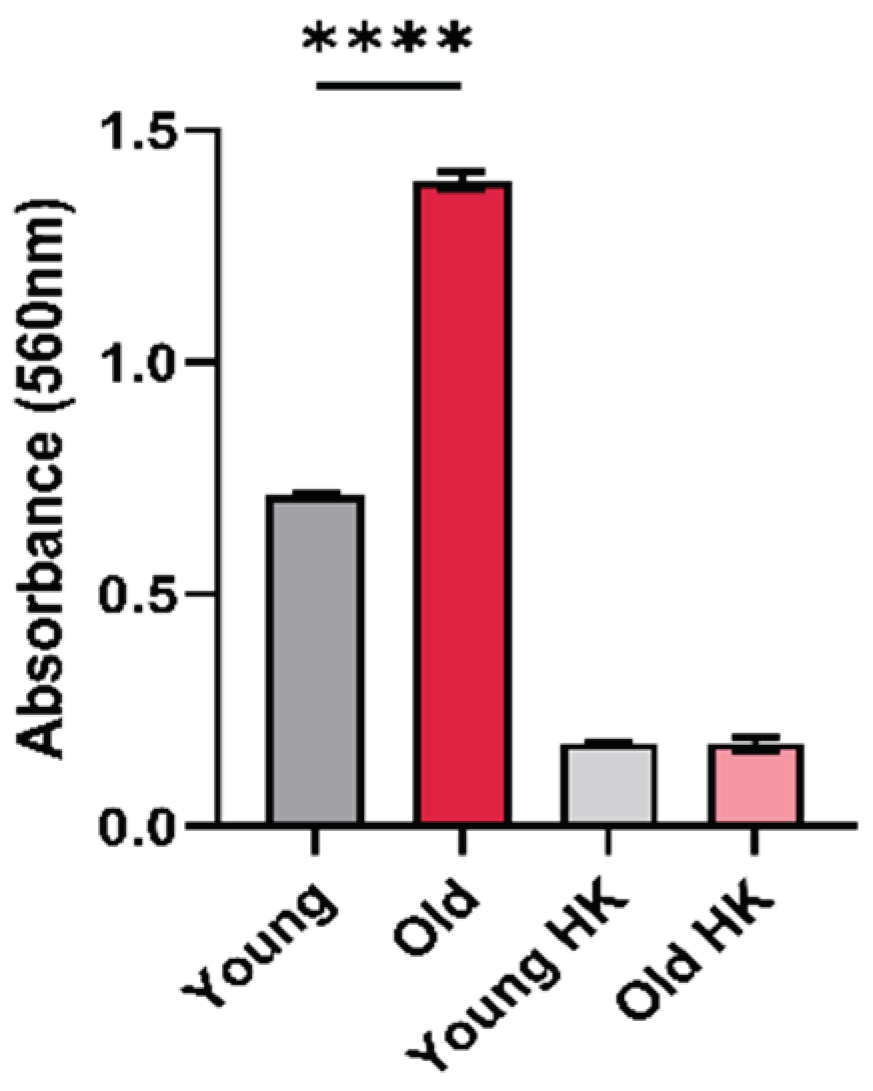
3.1. Effects of Aging on Urease Activity in C. neoformans
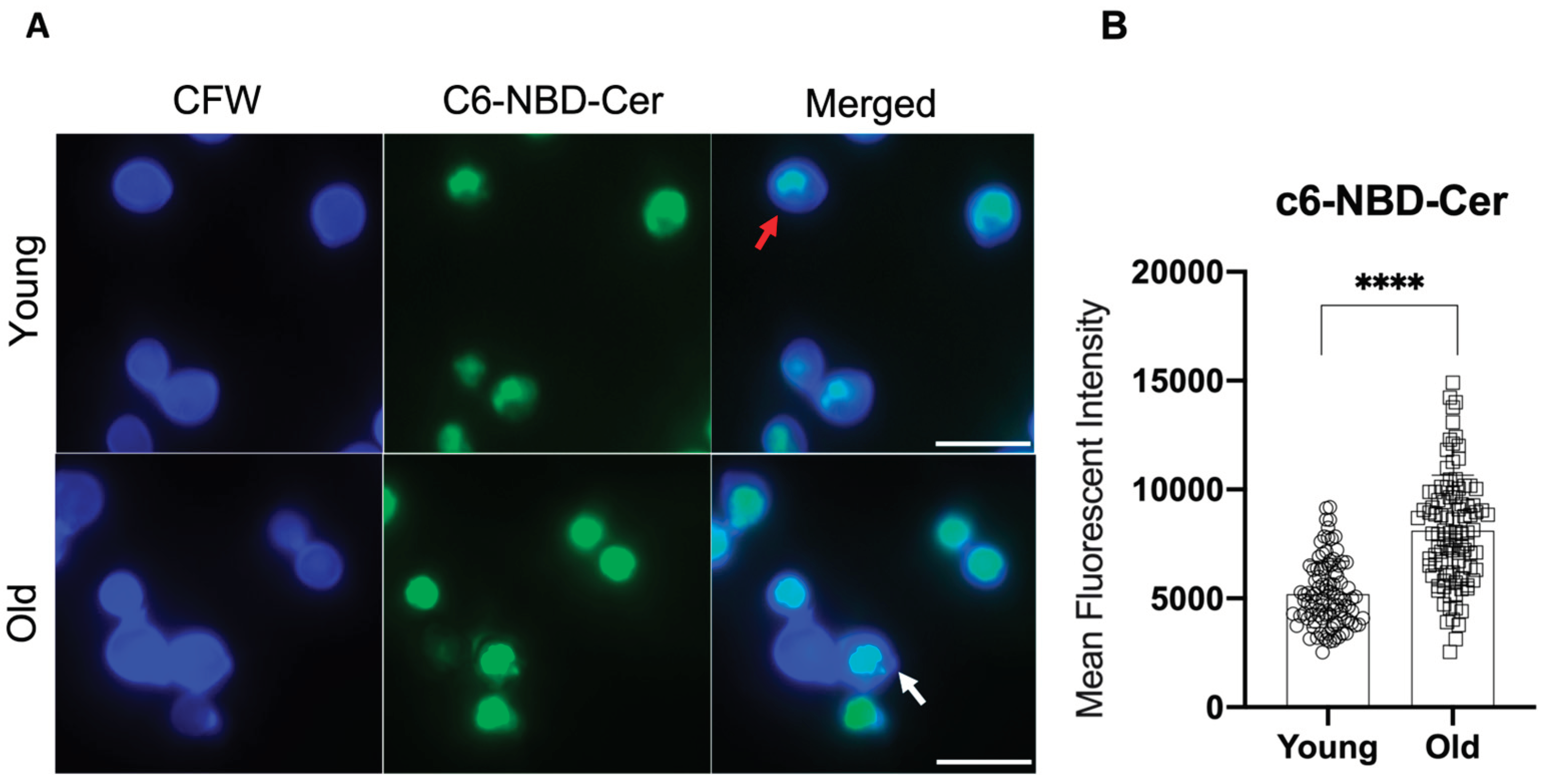
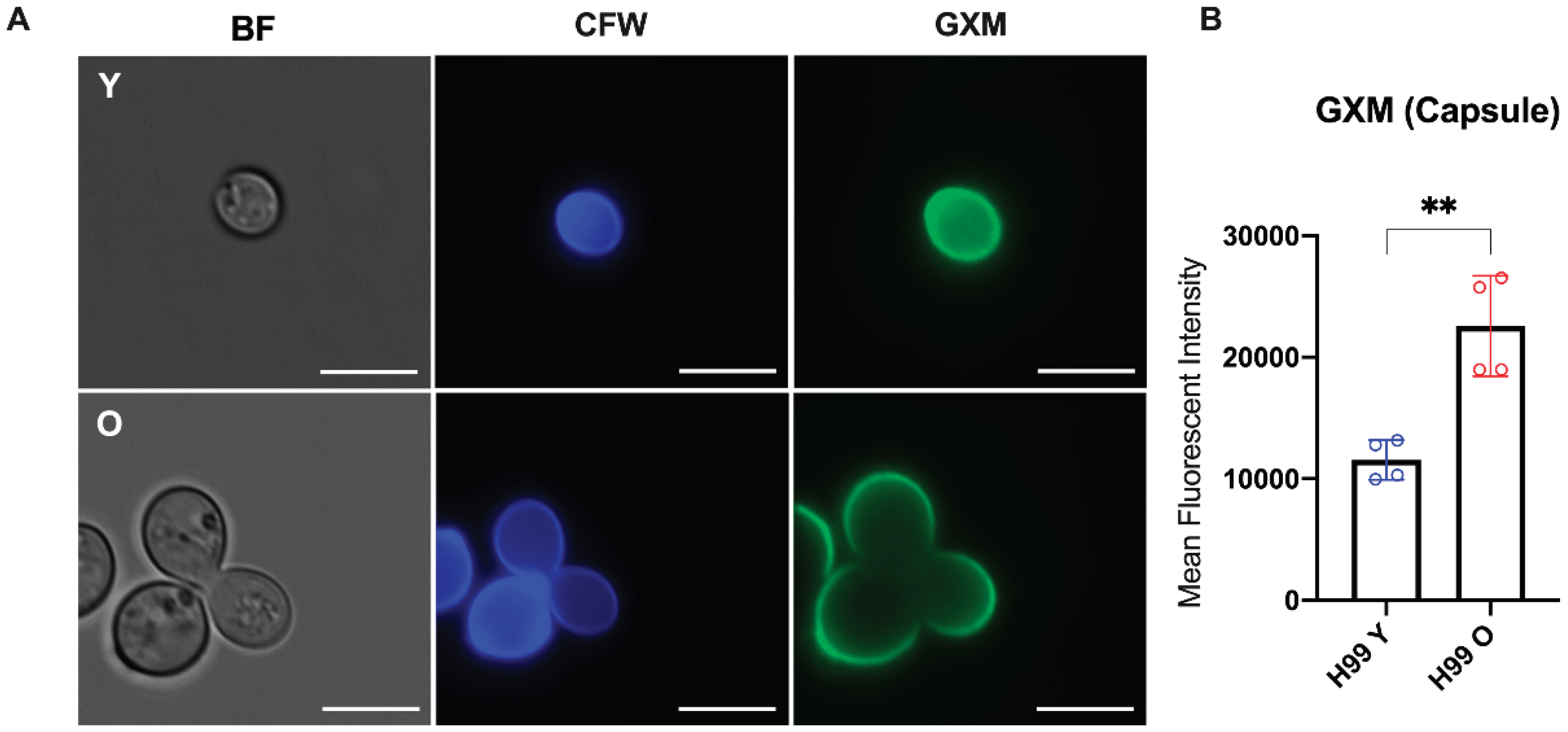
3.2. Longevity in C. neoformans Affects Golgi apparatus and Capsular GXM
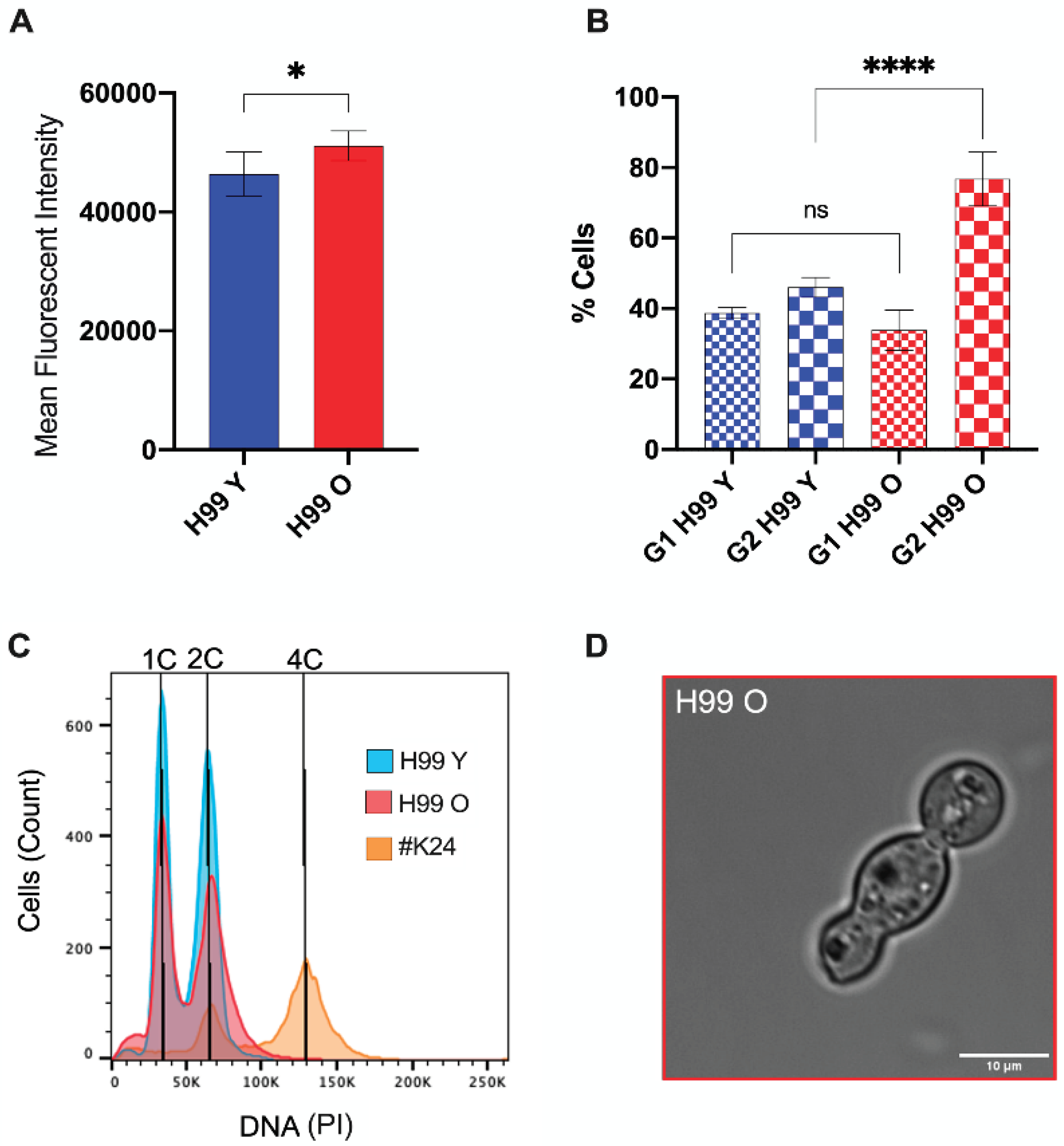
3.3. Old C. neoformans Presented an Increase in DNA Content and G2 Arrest
3.4. Vomocytosis Phenomenon during Replicative Aging in C. neoformans
3.5. Old C. neoformans Prefer to Reside in Acidified Phagosomes
3.2. Generational age in C. neoformans Influences Lysosomal Permeabilization
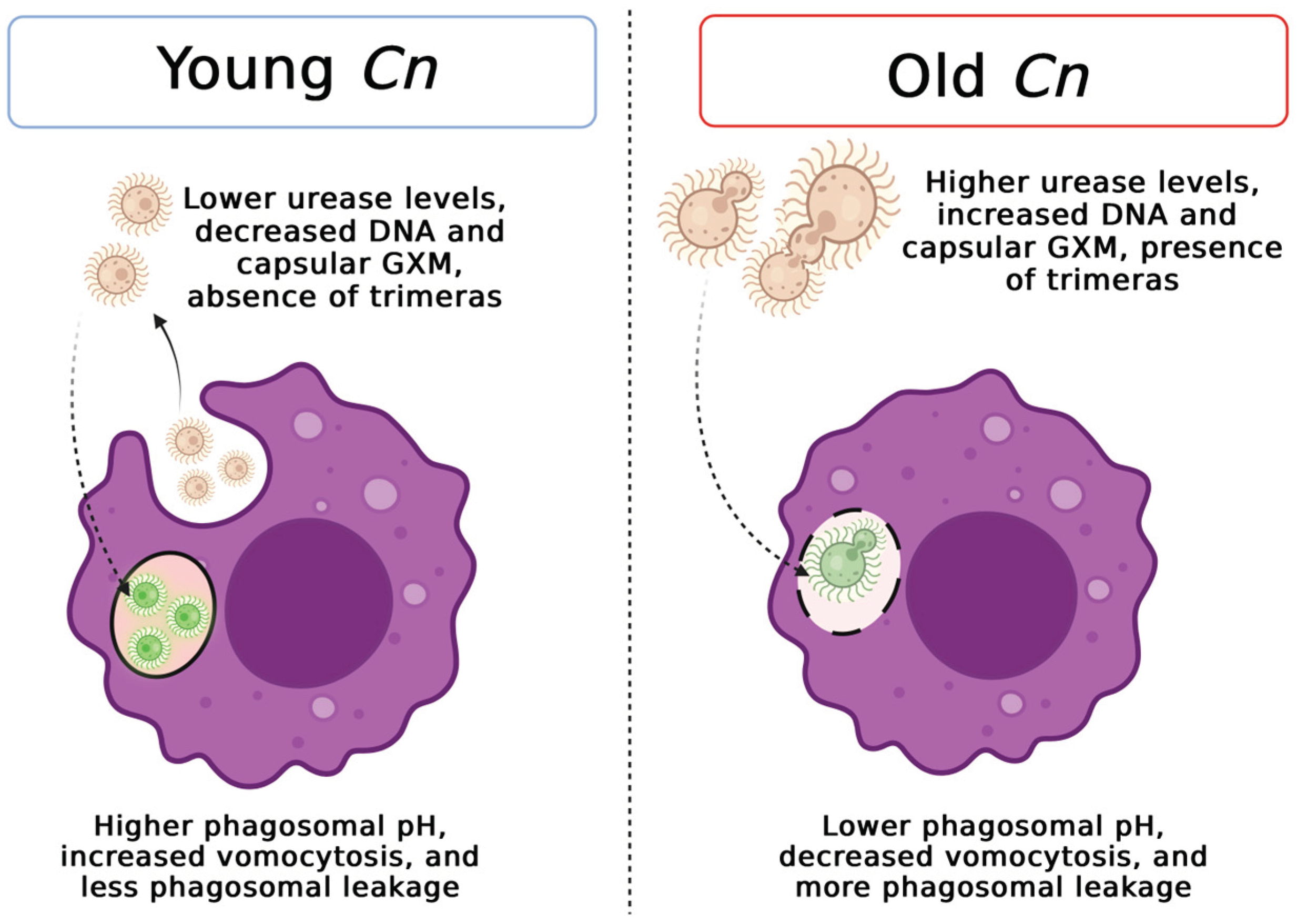
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Casadevall, A.; Fang, F.C. The intracellular pathogen concept. Molecular Microbiology 2020, 113, 541–545. [Google Scholar] [CrossRef]
- Gilbert, A.S.; Seoane, P.I.; Sephton-Clark, P.; Bojarczuk, A.; Hotham, R.; Giurisato, E.; Sarhan, A.R.; Hillen, A.; Velde, G.V.; Gray, N.S.; et al. Vomocytosis of live pathogens from macrophages is regulated by the atypical MAP kinase ERK5. Sci Adv 2017, 3, e1700898. [Google Scholar] [CrossRef]
- Chen, Y.; Shi, Z.W.; Strickland, A.B.; Shi, M. Cryptococcus neoformans Infection in the Central Nervous System: The Battle between Host and Pathogen. Journal of Fungi 2022, 8, 1069. [Google Scholar] [CrossRef]
- Rajasingham, R.; Smith, R.M.; Park, B.J.; Jarvis, J.N.; Govender, N.P.; Chiller, T.M.; Denning, D.W.; Loyse, A.; Boulware, D.R. Global burden of disease of HIV-associated cryptococcal meningitis: An updated analysis. The Lancet Infectious Diseases 2017, 17, 873–881. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.L.; Nosanchuk, J.D. Recognition of fungal priority pathogens: What next? PLoS Negl Trop Dis 2023, 17, e0011136. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.C.; Denning, D.W. The WHO fungal priority pathogens list as a game-changer. Nat Rev Microbiol 2023, 21, 211–212. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.; Cook, E.; Xess, I.; Hasan, F.; Fries, D.; Fries, B.C. Isolation and Characterization of Senescent <i>Cryptococcus neoformans</i> and Implications for Phenotypic Switching and Pathogenesis in Chronic Cryptococcosis. Eukaryotic Cell 2009, 8, 858–866. [Google Scholar] [CrossRef] [PubMed]
- Bouklas, T.; Pechuan, X.; Goldman, D.L.; Edelman, B.; Bergman, A.; Fries, B.C. Old Cryptococcus neoformans Cells Contribute to Virulence in Chronic Cryptococcosis. mBio 2013, 4. [Google Scholar] [CrossRef]
- Orner, E.P.; Zhang, P.; Jo, M.C.; Bhattacharya, S.; Qin, L.; Fries, B.C. High-Throughput Yeast Aging Analysis for Cryptococcus (HYAAC) microfluidic device streamlines aging studies in Cryptococcus neoformans. Communications Biology 2019, 2, 256. [Google Scholar] [CrossRef]
- Silva, V.K.A.; Bhattacharya, S.; Oliveira, N.K.; Savitt, A.G.; Zamith-Miranda, D.; Nosanchuk, J.D.; Fries, B.C. Replicative Aging Remodels the Cell Wall and Is Associated with Increased Intracellular Trafficking in Human Pathogenic Yeasts. mBio 2021, 13, e0019022. [Google Scholar] [CrossRef]
- Alvarez, M.; Casadevall, A. Phagosome extrusion and host-cell survival after Cryptococcus neoformans phagocytosis by macrophages. Curr Biol 2006, 16, 2161–2165. [Google Scholar] [CrossRef]
- Fu, M.S.; Coelho, C.; De Leon-Rodriguez, C.M.; Rossi, D.C.P.; Camacho, E.; Jung, E.H.; Kulkarni, M.; Casadevall, A. Cryptococcus neoformans urease affects the outcome of intracellular pathogenesis by modulating phagolysosomal pH. PLoS Pathog 2018, 14, e1007144. [Google Scholar] [CrossRef]
- De Leon-Rodriguez, C.M.; Fu, M.S.; Çorbali, M.O.; Cordero, R.J.B.; Casadevall, A. The Capsule of Cryptococcus neoformans Modulates Phagosomal pH through Its Acid-Base Properties. mSphere 2018, 3. [Google Scholar] [CrossRef]
- Altamirano, S.; Li, Z.; Fu, M.S.; Ding, M.; Fulton, S.R.; Yoder, J.M.; Tran, V.; Nielsen, K. The Cyclin Cln1 Controls Polyploid Titan Cell Formation following a Stress-Induced G(2) Arrest in Cryptococcus. mBio 2021, 12, e0250921. [Google Scholar] [CrossRef] [PubMed]
- Santiago-Burgos, E.J.; Stuckey, P.V.; Santiago-Tirado, F.H. Real-time visualization of phagosomal pH manipulation by Cryptococcus neoformans in an immune signal-dependent way. Front Cell Infect Microbiol 2022, 12, 967486. [Google Scholar] [CrossRef] [PubMed]
- Roberts, G.D.; Horstmeier, C.D.; Land, G.A.; Foxworth, J.H. Rapid urea broth test for yeasts. J Clin Microbiol 1978, 7, 584–588. [Google Scholar] [CrossRef] [PubMed]
- Feder, M.J.; Akyel, A.; Morasko, V.J.; Gerlach, R.; Phillips, A.J. Temperature-dependent inactivation and catalysis rates of plant-based ureases for engineered biomineralization. Engineering Reports 2021, 3, e12299. [Google Scholar] [CrossRef]
- Casadevall, A.; Cleare, W.; Feldmesser, M.; Glatman-Freedman, A.; Goldman, D.L.; Kozel, T.R.; Lendvai, N.; Mukherjee, J.; Pirofski, L.A.; Rivera, J.; et al. Characterization of a murine monoclonal antibody to Cryptococcus neoformans polysaccharide that is a candidate for human therapeutic studies. Antimicrob Agents Chemother 1998, 42, 1437–1446. [Google Scholar] [CrossRef]
- Rodrigues, J.; Ramos, C.L.; Frases, S.; Godinho, R.M.d.C.; Fonseca, F.L.; Rodrigues, M.L. Lack of chitin synthase genes impacts capsular architecture and cellular physiology in Cryptococcus neoformans. The Cell Surface 2018, 2, 14–23. [Google Scholar] [CrossRef]
- Todd, R.T.; Braverman, A.L.; Selmecki, A. Flow Cytometry Analysis of Fungal Ploidy. Current Protocols in Microbiology 2018, 50, e58. [Google Scholar] [CrossRef]
- Samantaray, S.; Correia, J.N.; Garelnabi, M.; Voelz, K.; May, R.C.; Hall, R.A. Novel cell-based in vitro screen to identify small-molecule inhibitors against intracellular replication of Cryptococcus neoformans in macrophages. Int J Antimicrob Agents 2016, 48, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Jamil, K.; Polyak, M.J.; Feehan, D.D.; Surmanowicz, P.; Stack, D.; Li, S.S.; Ogbomo, H.; Olszewski, M.; Ganguly, A.; Mody, C.H. Phagosomal F-Actin Retention by Cryptococcus gattii Induces Dendritic Cell Immunoparalysis. mBio 2020, 11. [Google Scholar] [CrossRef]
- Yoneda, A.; Doering, T.L. A eukaryotic capsular polysaccharide is synthesized intracellularly and secreted via exocytosis. Mol Biol Cell 2006, 17, 5131–5140. [Google Scholar] [CrossRef] [PubMed]
- García-Rodas, R.; Cordero, R.J.; Trevijano-Contador, N.; Janbon, G.; Moyrand, F.; Casadevall, A.; Zaragoza, O. Capsule growth in Cryptococcus neoformans is coordinated with cell cycle progression. mBio 2014, 5, e00945-00914. [Google Scholar] [CrossRef]
- Stukes, S.; Casadevall, A. Visualizing non-lytic exocytosis of Cryptococcus neoformans from macrophages using digital light microscopy. J Vis Exp 2014, e52084. [Google Scholar] [CrossRef]
- Dragotakes, Q.; Jacobs, E.; Ramirez, L.S.; Yoon, O.I.; Perez-Stable, C.; Eden, H.; Pagnotta, J.; Vij, R.; Bergman, A.; D’Alessio, F.; et al. Bet-hedging antimicrobial strategies in macrophage phagosome acidification drive the dynamics of Cryptococcus neoformans intracellular escape mechanisms. PLOS Pathogens 2022, 18, e1010697. [Google Scholar] [CrossRef] [PubMed]
- Dragotakes, Q.; Stouffer, K.M.; Fu, M.S.; Sella, Y.; Youn, C.; Yoon, O.I.; De Leon-Rodriguez, C.M.; Freij, J.B.; Bergman, A.; Casadevall, A. Macrophages use a bet-hedging strategy for antimicrobial activity in phagolysosomal acidification. The Journal of Clinical Investigation 2020, 130, 3805–3819. [Google Scholar] [CrossRef]
- Davis, M.J.; Eastman, A.J.; Qiu, Y.; Gregorka, B.; Kozel, T.R.; Osterholzer, J.J.; Curtis, J.L.; Swanson, J.A.; Olszewski, M.A. Cryptococcus neoformans-induced macrophage lysosome damage crucially contributes to fungal virulence. J Immunol 2015, 194, 2219–2231. [Google Scholar] [CrossRef]
- Gaylord, E.A.; Choy, H.L.; Doering, T.L. Dangerous Liaisons: Interactions of Cryptococcus neoformans with Host Phagocytes. Pathogens 2020, 9, 891. [Google Scholar] [CrossRef]
- Artavanis-Tsakonas, K.; Love, J.C.; Ploegh, H.L.; Vyas, J.M. Recruitment of CD63 to <i>Cryptococcus neoformans</i> phagosomes requires acidification. Proceedings of the National Academy of Sciences 2006, 103, 15945–15950. [Google Scholar] [CrossRef]
- Baker, R.P.; Casadevall, A. Reciprocal modulation of ammonia and melanin production has implications for cryptococcal virulence. Nat Commun 2023, 14, 849. [Google Scholar] [CrossRef] [PubMed]
- Orner, E.P.; Bhattacharya, S.; Kalenja, K.; Hayden, D.; Del Poeta, M.; Fries, B.C. Cell Wall-Associated Virulence Factors Contribute to Increased Resilience of Old Cryptococcus neoformans Cells. Front Microbiol 2019, 10, 2513. [Google Scholar] [CrossRef]
- Bouklas, T.; Jain, N.; Fries, B.C. Modulation of Replicative Lifespan in Cryptococcus neoformans: Implications for Virulence. Frontiers in Microbiology 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Weber, S.M.; Levitz, S.M. Chloroquine antagonizes the proinflammatory cytokine response to opportunistic fungi by alkalizing the fungal phagolysosome. J Infect Dis 2001, 183, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Tucker, S.C.; Casadevall, A. Replication of Cryptococcus neoformans in macrophages is accompanied by phagosomal permeabilization and accumulation of vesicles containing polysaccharide in the cytoplasm. Proc Natl Acad Sci U S A 2002, 99, 3165–3170. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Morris, I.R.; Wickes, B.L. The Production of Monokaryotic Hyphae by Cryptococcus neoformans Can Be Induced by High Temperature Arrest of the Cell Cycle and Is Independent of Same-Sex Mating. PLOS Pathogens 2013, 9, e1003335. [Google Scholar] [CrossRef]
- Harrison, B.D.; Hashemi, J.; Bibi, M.; Pulver, R.; Bavli, D.; Nahmias, Y.; Wellington, M.; Sapiro, G.; Berman, J. A tetraploid intermediate precedes aneuploid formation in yeasts exposed to fluconazole. PLoS Biol 2014, 12, e1001815. [Google Scholar] [CrossRef]
- Cruz-Acuña, M.; Pacifici, N.; Lewis, J.S. Vomocytosis: Too Much Booze, Base, or Calcium? mBio 2019, 10. [Google Scholar] [CrossRef]
| Golgi Morphology | Young | Old |
|---|---|---|
| Peripherical | 62.85% | 60% |
| Central | 37.15% | 40% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




