Submitted:
09 February 2024
Posted:
12 February 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
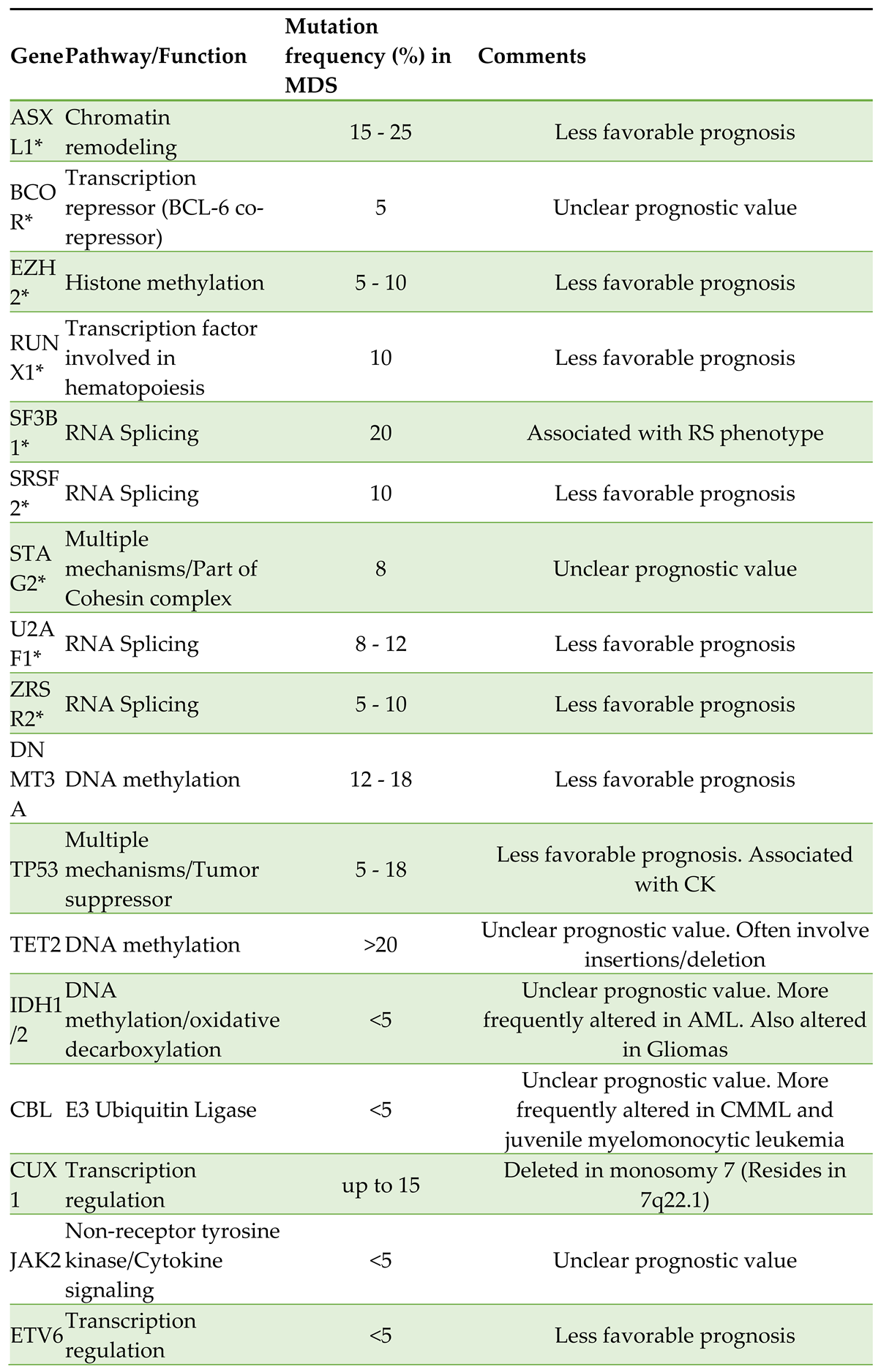
2. Molecular Evolution of MDS from Genotype to Phenotype to Prognostication
3. Prognostication
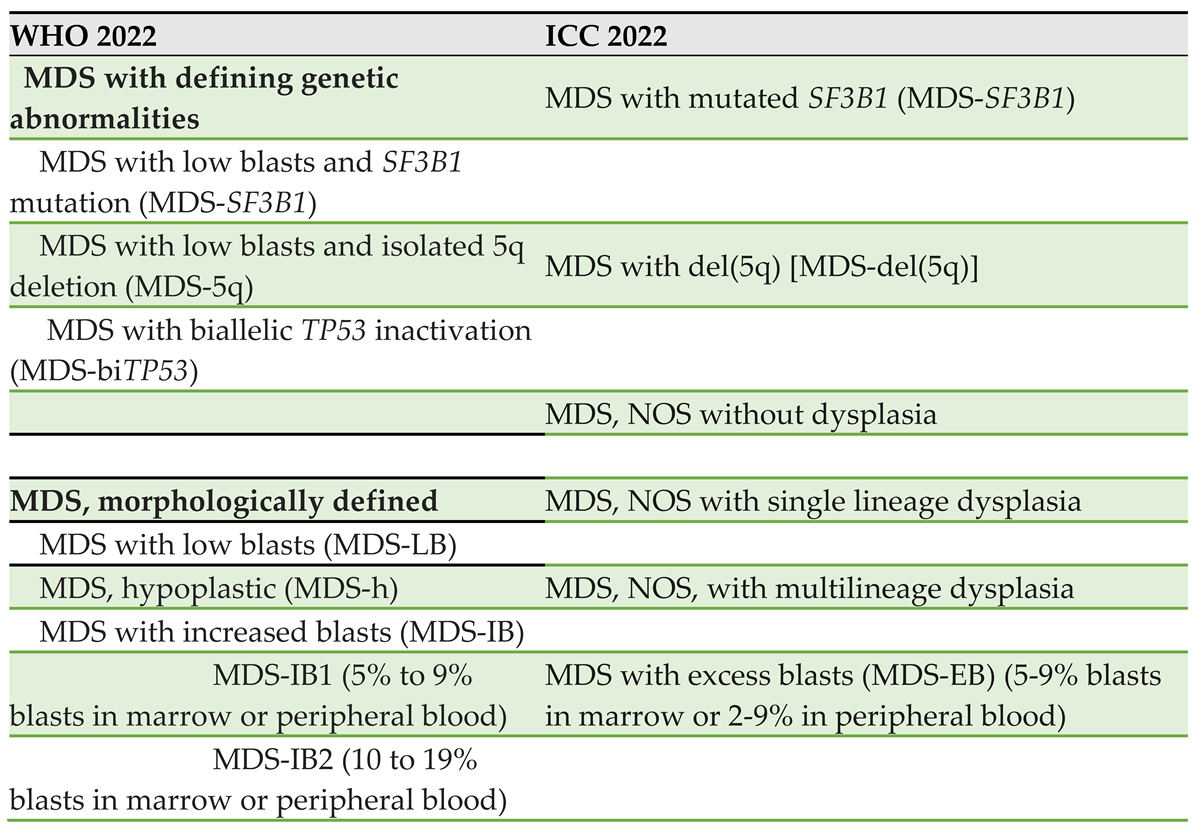
4. Classification Schemes
5. Treatment of Lower Risk Disease
Blood Product Transfusion
Erythropoiesis Stimulating Agents (ESA)
Thrombopoietin Receptor Agonists
Luspatercept
Immunomodulation/Immune Suppression
Hypomethylating Agents (HMA)
Telomerase Inhibition
6. Higher Risk Disease
TPO Mimetics in Higher Risk Disease
HMA
Venetoclax
Chemotherapy
Allogenic Hematopoietic Stem Cell Transplant (Allo HSCT)
IDH Inhibition
Experimental Targeted Agents
Glasdegib
Rigosertib
Pevonedistat
Magrolimab
Sabatolimab
Eprenetapopt (APR-246)
Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Garcia-Manero, G. Myelodysplastic syndromes: 2023 update on diagnosis, risk-stratification, and management. Am J Hematol. 2023 Aug;98, 1307-1325. [CrossRef] [PubMed]
- Ogawa, S. Genetics of MDS. Blood. 2019 Mar 7;133, 1049-1059. PMCID: PMC6587668. [CrossRef] [PubMed]
- Haider, M.; Duncavage, E.J.; Afaneh, K.F.; Bejar, R.; List, A.F. New Insight into the Biology, Risk Stratification, and Targeted Treatment of Myelodysplastic Syndromes. Am Soc Clin Oncol Educ Book. 2017, 37:480-494. [CrossRef] [PubMed]
- Abuhadra, N.; Mukherjee, S.; Al-Issa, K.; Adema, V.; Hirsch, C.M.; Advani, A.; Przychodzen, B.; Makhoul, A.; Awada, H.; Maciejewski, J.P.; Sekeres, M.A.; Nazha, A. BCOR and BCORL1 mutations in myelodysplastic syndromes (MDS): clonal architecture and impact on outcomes. Leuk Lymphoma. 2019 Jun;60, 1587-1590. PMCID: PMC8694070. [CrossRef] [PubMed]
- Katamesh, B.; Nanaa, A.; He, R.; Viswanatha, D.; Nguyen, P.; Greipp, P.; Bessonen, K.; Gangat, N.; Begna, K.; Mangaonkar, A.; Patnaik, M.; Hogan, W.J.; Tefferi, A.; Litzow, M.; Shah, M.V.; Yi, C.A.; Foran, J.; Badar, T.; Alkhateeb, H.B.; Al-Kali, A. Clinical and prognostic impact of STAG2 mutations in myeloid neoplasms: the Mayo Clinic experience. Blood Adv. 2023 Apr 25;7, 1351-1355. PMCID: PMC10139934. [CrossRef] [PubMed]
- AACR Project GENIE Consortium. AACR Project GENIE: Powering Precision Medicine through an International Consortium. Cancer Discov. 2017 Aug;7, 818-831. PMCID: PMC5611790. [CrossRef] [PubMed]
- Aly, M.; Ramdzan, Z.M.; Nagata, Y.; Balasubramanian, S.K.; Hosono, N.; Makishima, H.; Visconte, V.; Kuzmanovic, T.; Adema, V.; Nazha, A.; Przychodzen, B.P.; Kerr, C.M.; Sekeres, M.A.; Abazeed, M.E.; Nepveu, A.; Maciejewski, J.P. Distinct clinical and biological implications of CUX1 in myeloid neoplasms. Blood Adv. 2019 Jul 23;3, 2164-2178. PMCID: PMC6650742. [CrossRef] [PubMed]
- Seethy, A.A.; Pethusamy, K.; Kushwaha, T.; Kumar, G.; Talukdar, J.; Chaubey, R.; Sundaram, U.D.; Mahapatra, M.; Saxena, R.; Dhar, R.; Inampudi, K.K.; Karmakar, S. Alterations of the expression of TET2 and DNA 5-hmC predict poor prognosis in Myelodysplastic Neoplasms. BMC Cancer. 2023 Oct 26;23, 1035. PMCID: PMC10601240. [CrossRef] [PubMed]
- Arber, D.A.; Orazi, A.; Hasserjian, R.P.; Borowitz, M.J.; Calvo, K.R.; Kvasnicka, H.M.; Wang, S.A.; Bagg, A.; Barbui, T.; Branford, S.; Bueso-Ramos, C.E.; Cortes, J.E.; Dal Cin, P.; DiNardo, C.D.; Dombret, H.; Duncavage, E.J.; Ebert, B.L.; Estey, E.H.; Facchetti, F.; Foucar, K.; Gangat, N.; Gianelli, U.; Godley, L.A.; Gökbuget, N.; Gotlib, J.; Hellström-Lindberg, E.; Hobbs, G.S.; Hoffman, R.; Jabbour, E.J.; Kiladjian, J.J.; Larson, R.A.; Le Beau, M.M.; Loh, M.L.; Löwenberg, B.; Macintyre, E.; Malcovati, L.; Mullighan, C.G.; Niemeyer, C.; Odenike, O.M.; Ogawa, S.; Orfao, A.; Papaemmanuil, E.; Passamonti, F.; Porkka, K.; Pui, C.H.; Radich, J.P.; Reiter, A.; Rozman, M.; Rudelius, M.; Savona, M.R.; Schiffer, C.A.; Schmitt-Graeff, A.; Shimamura, A.; Sierra, J.; Stock, W.A.; Stone, R.M.; Tallman, M.S.; Thiele, J.; Tien, H.F.; Tzankov, A.; Vannucchi, A.M.; Vyas, P.; Wei, A.H.; Weinberg, O.K.; Wierzbowska, A.; Cazzola, M.; Döhner, H.; Tefferi, A. International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood. 2022 Sep 15;140, 1200-1228. PMCID: PMC9479031. [CrossRef] [PubMed]
- Fenaux, P.; Adès, L. How we treat lower-risk myelodysplastic syndromes. Blood. 2013 May 23;121, 4280-6. [CrossRef] [PubMed]
- Shahjahani, M.; Hadad, E.H.; Azizidoost, S.; Nezhad, K.C.; Shahrabi, S. Complex karyotype in myelodysplastic syndromes: Diagnostic procedure and prognostic susceptibility. Oncol Rev. 2019 Feb 4;13, 389. PMCID: PMC6379782. [CrossRef] [PubMed]
- Bejar, R.; Stevenson, K.; Abdel-Wahab, O.; Galili, N.; Nilsson, B.; Garcia-Manero, G.; Kantarjian, H.; Raza, A.; Levine, R.L.; Neuberg, D.; Ebert, B.L. Clinical effect of point mutations in myelodysplastic syndromes. N Engl J Med. 2011 Jun 30;364, 2496-506. PMCID: PMC3159042. [CrossRef] [PubMed]
- Bersanelli, M.; Travaglino, E.; Meggendorfer, M.; Matteuzzi, T.; Sala, C.; Mosca, E.; Chiereghin, C.; Di Nanni, N.; Gnocchi, M.; Zampini, M.; Rossi, M.; Maggioni, G.; Termanini, A.; Angelucci, E.; Bernardi, M.; Borin, L.; Bruno, B.; Bonifazi, F.; Santini, V.; Bacigalupo, A.; Voso, M.T.; Oliva, E.; Riva, M.; Ubezio, M.; Morabito, L.; Campagna, A.; Saitta, C.; Savevski, V.; Giampieri, E.; Remondini, D.; Passamonti, F.; Ciceri, F.; Bolli, N.; Rambaldi, A.; Kern, W.; Kordasti, S.; Sole, F.; Palomo, L.; Sanz, G.; Santoro, A.; Platzbecker, U.; Fenaux, P.; Milanesi, L.; Haferlach, T.; Castellani, G.; Della Porta, M.G. Classification and Personalized Prognostic Assessment on the Basis of Clinical and Genomic Features in Myelodysplastic Syndromes. J Clin Oncol. 2021 Apr 10;39, 1223-1233. PMCID: PMC8078359. [CrossRef] [PubMed]
- Greenberg, P.L.; Tuechler, H.; Schanz, J.; Sanz, G.; Garcia-Manero, G.; Solé, F.; Bennett, J.M.; Bowen, D.; Fenaux, P.; Dreyfus, F.; Kantarjian, H.; Kuendgen, A.; Levis, A.; Malcovati, L.; Cazzola, M.; Cermak, J.; Fonatsch, C.; Le Beau, M.M.; Slovak, M.L.; Krieger, O.; Luebbert, M.; Maciejewski, J.; Magalhaes, S.M.; Miyazaki, Y.; Pfeilstöcker, M.; Sekeres, M.; Sperr, W.R.; Stauder, R.; Tauro, S.; Valent, P.; Vallespi, T.; van de Loosdrecht, A.A.; Germing, U.; Haase, D. Revised international prognostic scoring system for myelodysplastic syndromes. Blood. 2012 Sep 20;120, 2454-65. PMCID: PMC4425443. [CrossRef] [PubMed]
- Kewan, T.; Bahaj, W.; Durmaz, A.; Aly, M.M.; Ogbue, O.D.; Carraway, H.E.; Sekeres, M.A.; Visconte, V.; Gurnari, C.; Maciejewski, J.P. Validation of the Molecular International Prognostic Scoring System in patients with myelodysplastic syndromes. Blood. 2023 Jan 30:blood.2022018896. [CrossRef] [PubMed]
- Bernard, E.; Tuechler, H.; Greenberg, P.L.; Hasserjian, R.P.; Arango Ossa, J.E.; Nannya, Y.; Delvin, S.M.; Papaemmanuil, E. Molecular International Prognostic Scoring System for Myelodysplastic Syndromes. June 12, 2022. NEJM Evid 2022;1(7). [CrossRef]
- Sabile, J.M.G.; Kaempf, A.; Tomic, K.; Manu, G.P.; Swords, R.; Migdady, Y. A retrospective validation of the IPSS-M molecular score in primary and therapy-related myelodysplastic syndromes (MDS). Leuk Lymphoma. 2023 Jul 13:1-6. [CrossRef] [PubMed]
- Khoury, J.D.; Solary, E.; Abla, O.; Akkari, Y.; Alaggio, R.; Apperley, J.F.; Bejar, R.; Berti, E.; Busque, L.; Chan, J.K.C.; Chen, W.; Chen, X.; Chng, W.J.; Choi, J.K.; Colmenero, I.; Coupland, S.E.; Cross, N.C.P.; De Jong, D.; Elghetany, M.T.; Takahashi, E.; Emile, J.F.; Ferry, J.; Fogelstrand, L.; Fontenay, M.; Germing, U.; Gujral, S.; Haferlach, T.; Harrison, C.; Hodge, J.C.; Hu, S.; Jansen, J.H.; Kanagal-Shamanna, R.; Kantarjian, H.M.; Kratz, C.P.; Li, X.Q.; Lim, M.S.; Loeb, K.; Loghavi, S.; Marcogliese, A.; Meshinchi, S.; Michaels, P.; Naresh, K.N.; Natkunam, Y.; Nejati, R.; Ott, G.; Padron, E.; Patel, K.P.; Patkar, N.; Picarsic, J.; Platzbecker, U.; Roberts, I.; Schuh, A.; Sewell, W.; Siebert, R.; Tembhare, P.; Tyner, J.; Verstovsek, S.; Wang, W.; Wood, B.; Xiao, W.; Yeung, C.; Hochhaus, A. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia. 2022 Jul;36, 1703-1719. [CrossRef]
- Heptinstall, K.; Myelodysplastic Syndromes Foundation, Inc. Quality of life in myelodysplastic syndromes. A special report from the Myelodysplastic Syndromes Foundation, Inc. Oncology (Williston Park). 2008 Feb;22(2 Suppl Nurse Ed):13-8; discussion 19. [PubMed]
- Platzbecker, U.; Hofbauer, L.C.; Ehninger, G.; Hölig, K. The clinical, quality of life, and economic consequences of chronic anemia and transfusion support in patients with myelodysplastic syndromes. Leuk Res. 2012 May;36, 525-36. [CrossRef] [PubMed]
- Cermak, J.; Kacirkova, P.; Mikulenkova, D.; Michalova, K. Impact of transfusion dependency on survival in patients with early myelodysplastic syndrome without excess of blasts. Leuk Res. 2009 Nov;33, 1469-74. [CrossRef] [PubMed]
- Angelucci, E.; Li, J.; Greenberg, P.; Wu, D.; Hou, M.; Montano Figueroa, E.H. Iron chelation in transfusion-dependent patients with low- to intermediate-1-risk myelodysplastic syndromes: a randomized trial. Ann Intern Med. 2020;172:513–22. [CrossRef]
- National Comprehensive Cancer Network. Myelodysplastic Syndromes (Version 1.2023). Available online: https://www.nccn.org/professionals/physician_gls/pdf/mds.pdf (accessed on 24 July 2023).
- Hellström-Lindberg, E. Efficacy of erythropoietin in the myelodysplastic syndromes: a meta-analysis of 205 patients from 17 studies. Br J Haematol. 1995 Jan;89, 67-71. [CrossRef] [PubMed]
- Hellström-Lindberg, E.; Ahlgren, T.; Beguin, Y.; Carlsson, M.; Carneskog, J.; Dahl, I.M.; Dybedal, I.; Grimfors, G.; Kanter-Lewensohn, L.; Linder, O.; Luthman, M.; Löfvenberg, E.; Nilsson-Ehle, H.; Samuelsson, J.; Tangen, J.M.; Winqvist, I.; Oberg, G.; Osterborg, A.; Ost, A. Treatment of anemia in myelodysplastic syndromes with granulocyte colony-stimulating factor plus erythropoietin: results from a randomized phase II study and long-term follow-up of 71 patients. Blood. 1998 Jul 1;92, 68-75. [CrossRef] [PubMed]
- Hellström-Lindberg, E.; Gulbrandsen, N.; Lindberg, G.; Ahlgren, T.; Dahl, I.M.; Dybedal, I.; Grimfors, G.; Hesse-Sundin, E.; Hjorth, M.; Kanter-Lewensohn, L.; Linder, O.; Luthman, M.; Löfvenberg, E.; Oberg, G.; Porwit-MacDonald, A.; Rådlund, A.; Samuelsson, J.; Tangen, J.M.; Winquist, I.; Wisloff, F. ; Scandinavian MDS Group. A validated decision model for treating the anaemia of myelodysplastic syndromes with erythropoietin + granulocyte colony-stimulating factor: significant effects on quality of life. Br J Haematol. 2003 Mar;120, 1037-46. [CrossRef] [PubMed]
- Platzbecker, U.; Symeonidis, A.; Oliva, E.N.; Goede, J.S.; Delforge, M.; Mayer, J.; Slama, B.; Badre, S.; Gasal, E.; Mehta, B.; Franklin, J. A phase 3 randomized placebo-controlled trial of darbepoetin alfa in patients with anemia and lower-risk myelodysplastic syndromes. Leukemia. 2017 Sep;31, 1944-1950. PMCID: PMC5596208. [CrossRef] [PubMed]
- Fenaux, P.; Santini, V.; Spiriti, M.A.A.; Giagounidis, A.; Schlag, R.; Radinoff, A.; Gercheva-Kyuchukova, L.; Anagnostopoulos, A.; Oliva, E.N.; Symeonidis, A.; Berger, M.H.; Götze, K.S.; Potamianou, A.; Haralampiev, H.; Wapenaar, R.; Milionis, I.; Platzbecker, U. A phase 3 randomized, placebo-controlled study assessing the efficacy and safety of epoetin-α in anemic patients with low-risk MDS. Leukemia. 2018 Dec;32, 2648-2658. PMCID: PMC6286328. [CrossRef] [PubMed]
- Kantarjian, H.; Giles, F.; List, A.; Lyons, R.; Sekeres, M.A.; Pierce, S.; Deuson, R.; Leveque, J. The incidence and impact of thrombocytopenia in myelodysplastic syndromes. Cancer. 2007 May 1;109, 1705-14. [CrossRef] [PubMed]
- Oliva, E.N.; Alati, C.; Santini, V.; Poloni, A.; Molteni, A.; Niscola, P.; Salvi, F.; Sanpaolo, G.; Balleari, E.; Germing, U.; Fenaux, P.; Stamatoullas, A.; Palumbo, G.A.; Salutari, P.; Impera, S.; Avanzini, P.; Cortelezzi, A.; Liberati, A.M.; Carluccio, P.; Buccisano, F.; Voso, M.T.; Mancini, S.; Kulasekararaj, A.; Morabito, F.; Bocchia, M.; Cufari, P.; Spiriti, M.A.; Santacaterina, I.; D'Errigo, M.G.; Bova, I.; Zini, G.; Latagliata, R. Eltrombopag versus placebo for low-risk myelodysplastic syndromes with thrombocytopenia (EQoL-MDS): phase 1 results of a single-blind, randomised, controlled, phase 2 superiority trial. Lancet Haematol. 2017 Mar;4, e127-e136. [CrossRef] [PubMed]
- Oliva, E.N.; Riva, M.; Niscola, P.; Santini, V.; Breccia, M.; Giai, V.; Poloni, A.; Patriarca, A.; Crisà, E.; Capodanno, I.; Salutari, P.; Reda, G.; Cascavilla, N.; Ferrero, D.; Guarini, A.; Tripepi, G.; Iannì, G.; Russo, E.; Castelli, A.; Fattizzo, B.; Beltrami, G.; Bocchia, M.; Molteni, A.; Fenaux, P.; Germing, U.; Ricco, A.; Palumbo, G.A.; Impera, S.; Di Renzo, N.; Rivellini, F.; Buccisano, F.; Stamatoullas-Bastard, A.; Liberati, A.M.; Candoni, A.; Delfino, I.M.; Arcadi, M.T.; Cufari, P.; Rizzo, L.; Bova, I.; D'Errigo, M.G.; Zini, G.; Latagliata, R. Eltrombopag for Low-Risk Myelodysplastic Syndromes With Thrombocytopenia: Interim Results of a Phase-II, Randomized, Placebo-Controlled Clinical Trial (EQOL-MDS). J Clin Oncol. 2023 Jun 9:JCO2202699. [CrossRef] [PubMed]
- Vicente, A.; Patel, B.A.; Gutierrez-Rodrigues, F.; Groarke, E.; Giudice, V.; Lotter, J.; Feng, X.; Kajigaya, S.; Weinstein, B.; Barranta, E.; Olnes, M.J.; Parikh, A.R.; Albitar, M.; Wu, C.O.; Shalhoub, R.; Calvo, K.R.; Townsley, D.M.; Scheinberg, P.; Dunbar, C.E.; Young, N.S.; Winkler, T. Eltrombopag monotherapy can improve hematopoiesis in patients with low to intermediate risk-1 myelodysplastic syndrome. Haematologica. 2020 Dec 1;105, 2785-2794. PMCID: PMC7716353. [CrossRef] [PubMed]
- Fenaux, P.; Muus, P.; Kantarjian, H.; Lyons, R.M.; Larson, R.A.; Sekeres, M.A.; Becker, P.S.; Orejudos, A.; Franklin, J. Romiplostim monotherapy in thrombocytopenic patients with myelodysplastic syndromes: long-term safety and efficacy. Br J Haematol. 2017 Sep;178, 906-913. PMCID: PMC5600084. [CrossRef] [PubMed]
- Giagounidis, A.; Mufti, G.J.; Fenaux, P.; Sekeres, M.A.; Szer, J.; Platzbecker, U.; Kuendgen, A.; Gaidano, G.; Wiktor-Jedrzejczak, W.; Hu, K.; Woodard, P.; Yang, A.S.; Kantarjian, H.M. Results of a randomized, double-blind study of romiplostim versus placebo in patients with low/intermediate-1-risk myelodysplastic syndrome and thrombocytopenia. Cancer. 2014 Jun 15;120, 1838-46. PMCID: PMC4298760. [CrossRef] [PubMed]
- Kantarjian, H.; Fenaux, P.; Sekeres, M.A.; Becker, P.S.; Boruchov, A.; Bowen, D.; Hellstrom-Lindberg, E.; Larson, R.A.; Lyons, R.M.; Muus, P.; Shammo, J.; Siegel, R.; Hu, K.; Franklin, J.; Berger, D.P. Safety and efficacy of romiplostim in patients with lower-risk myelodysplastic syndrome and thrombocytopenia. J Clin Oncol. 2010 Jan 20;28, 437-44. [CrossRef] [PubMed]
- Sekeres, M.A.; Kantarjian, H.; Fenaux, P.; Becker, P.; Boruchov, A.; Guerci-Bresler, A.; Hu, K.; Franklin, J.; Wang, Y.M.; Berger, D. Subcutaneous or intravenous administration of romiplostim in thrombocytopenic patients with lower risk myelodysplastic syndromes. Cancer. 2011 Mar 1;117, 992-1000. [CrossRef] [PubMed]
- Meng, F.; Chen, X.; Yu, S.; Ren, X.; Liu, Z.; Fu, R.; Li, L. Safety and Efficacy of Eltrombopag and Romiplostim in Myelodysplastic Syndromes: A Systematic Review and Meta-Analysis. Front Oncol. 2020 Nov 26;10:582686. PMCID: PMC7727449. [CrossRef] [PubMed]
- Platzbecker, U.; Germing, U.; Götze, K.S.; Kiewe, P.; Mayer, K.; Chromik, J.; Radsak, M.; Wolff, T.; Zhang, X.; Laadem, A.; Sherman, M.L.; Attie, K.M.; Giagounidis, A. Luspatercept for the treatment of anaemia in patients with lower-risk myelodysplastic syndromes (PACE-MDS): a multicentre, open-label phase 2 dose-finding study with long-term extension study. Lancet Oncol. 2017 Oct;18, 1338-1347. Erratum in: Lancet Oncol. 2017 Oct;18, e562. [CrossRef] [PubMed]
- Platzbecker, U.; Götze, K.S.; Kiewe, P.; Germing, U.; Mayer, K.; Radsak, M.; Wolff, T.; Chromik, J.; Sockel, K.; Oelschlägel, U.; Haase, D.; Illmer, T.; Al-Ali, H.K.; Silling, G.; Reynolds, J.G.; Zhang, X.; Attie, K.M.; Shetty, J.K.; Giagounidis, A. Long-Term Efficacy and Safety of Luspatercept for Anemia Treatment in Patients With Lower-Risk Myelodysplastic Syndromes: The Phase II PACE-MDS Study. J Clin Oncol. 2022 Nov 20;40, 3800-3807. PMCID:PMC9671752. [CrossRef] [PubMed]
- Fenaux, P.; Platzbecker, U.; Mufti, G.J.; Garcia-Manero, G.; Buckstein, R.; Santini, V.; Díez-Campelo, M.; Finelli, C.; Cazzola, M.; Ilhan, O.; Sekeres, M.A.; Falantes, J.F.; Arrizabalaga, B.; Salvi, F.; Giai, V.; Vyas, P.; Bowen, D.; Selleslag, D.; DeZern, A.E.; Jurcic, J.G.; Germing, U.; Götze, K.S.; Quesnel, B.; Beyne-Rauzy, O.; Cluzeau, T.; Voso, M.T.; Mazure, D.; Vellenga, E.; Greenberg, P.L.; Hellström-Lindberg, E.; Zeidan, A.M.; Adès, L.; Verma, A.; Savona, M.R.; Laadem, A.; Benzohra, A.; Zhang, J.; Rampersad, A.; Dunshee, D.R.; Linde, P.G.; Sherman, M.L.; Komrokji, R.S.; List, A.F. Luspatercept in Patients with Lower-Risk Myelodysplastic Syndromes. N Engl J Med. 2020 Jan 9;382, 140-151. [CrossRef] [PubMed]
- Platzbecker, U.; Della Porta, M.G.; Santini, V.; Zeidan, A.M.; Komrokji, R.S.; Shortt, J.; Valcarcel, D.; Jonasova, A.; Dimicoli-Salazar, S.; Tiong, I.S.; Lin, C.C.; Li, J.; Zhang, J.; Giuseppi, A.C.; Kreitz, S.; Pozharskaya, V.; Keeperman, K.L.; Rose, S.; Shetty, J.K.; Hayati, S.; Vodala, S.; Prebet, T.; Degulys, A.; Paolini, S.; Cluzeau, T.; Fenaux, P.; Garcia-Manero, G. Efficacy and safety of luspatercept versus epoetin alfa in erythropoiesis-stimulating agent-naive, transfusion-dependent, lower-risk myelodysplastic syndromes (COMMANDS): interim analysis of a phase 3, open-label, randomised controlled trial. Lancet. 2023 Jul 29;402, 373-385. [CrossRef] [PubMed]
- Fenaux, P.; Giagounidis, A.; Selleslag, D.; Beyne-Rauzy, O.; Mufti, G.; Mittelman, M.; Muus, P.; Te Boekhorst, P.; Sanz, G.; Del Cañizo, C.; Guerci-Bresler, A.; Nilsson, L.; Platzbecker, U.; Lübbert, M.; Quesnel, B.; Cazzola, M.; Ganser, A.; Bowen, D.; Schlegelberger, B.; Aul, C.; Knight, R.; Francis, J.; Fu, T.; Hellström-Lindberg, E. ; MDS-004 Lenalidomide del5q Study Group. A randomized phase 3 study of lenalidomide versus placebo in RBC transfusion-dependent patients with Low-/Intermediate-1-risk myelodysplastic syndromes with del5q. Blood. 2011 Oct 6;118, 3765-76. [CrossRef] [PubMed]
- List, A.; Dewald, G.; Bennett, J.; Giagounidis, A.; Raza, A.; Feldman, E.; Powell, B.; Greenberg, P.; Thomas, D.; Stone, R.; Reeder, C.; Wride, K.; Patin, J.; Schmidt, M.; Zeldis, J.; Knight, R. ; Myelodysplastic Syndrome-003 Study Investigators. Lenalidomide in the myelodysplastic syndrome with chromosome 5q deletion. N Engl J Med. 2006 Oct 5;355, 1456-65. [CrossRef] [PubMed]
- List, A.F.; Bennett, J.M.; Sekeres, M.A.; Skikne, B.; Fu, T.; Shammo, J.M.; Nimer, S.D.; Knight, R.D.; Giagounidis, A. ; MDS-003 Study Investigators. Extended survival and reduced risk of AML progression in erythroid-responsive lenalidomide-treated patients with lower-risk del(5q) MDS. Leukemia. 2014 May;28, 1033-40. Erratum in: Leukemia. 2015 Dec;29, 2452.. PMCID: PMC4017258. [CrossRef] [PubMed]
- Santini, V.; Almeida, A.; Giagounidis, A.; Gröpper, S.; Jonasova, A.; Vey, N.; Mufti, G.J.; Buckstein, R.; Mittelman, M.; Platzbecker, U.; Shpilberg, O.; Ram, R.; Del Cañizo, C.; Gattermann, N.; Ozawa, K.; Risueño, A.; MacBeth, K.J.; Zhong, J.; Séguy, F.; Hoenekopp, A.; Beach, C.L.; Fenaux, P. Randomized Phase III Study of Lenalidomide Versus Placebo in RBC Transfusion-Dependent Patients With Lower-Risk Non-del(5q) Myelodysplastic Syndromes and Ineligible for or Refractory to Erythropoiesis-Stimulating Agents. J Clin Oncol. 2016 Sep 1;34, 2988-96. [CrossRef] [PubMed]
- Stahl, M.; DeVeaux, M.; de Witte, T.; Neukirchen, J.; Sekeres, M.A.; Brunner, A.M.; Roboz, G.J.; Steensma, D.P.; Bhatt, V.R.; Platzbecker, U.; Cluzeau, T.; Prata, P.H.; Itzykson, R.; Fenaux, P.; Fathi, A.T.; Smith, A.; Germing, U.; Ritchie, E.K.; Verma, V.; Nazha, A.; Maciejewski, J.P.; Podoltsev, N.A.; Prebet, T.; Santini, V.; Gore, S.D.; Komrokji, R.S.; Zeidan, A.M. The use of immunosuppressive therapy in MDS: clinical outcomes and their predictors in a large international patient cohort. Blood Adv. 2018 Jul 24;2, 1765-1772. [CrossRef]
- Komrokji, R.S.; Mailloux, A.W.; Chen, D.T.; Sekeres, M.A.; Paquette, R.; Fulp, W.J.; Sugimori, C.; Paleveda-Pena, J.; Maciejewski, J.P.; List, A.F.; Epling-Burnette, P.K. A phase II multicenter rabbit anti-thymocyte globulin trial in patients with myelodysplastic syndromes identifying a novel model for response prediction. Haematologica. 2014 Jul;99, 1176-83. [CrossRef]
- Jabbour, E.; Short, N.J.; Montalban-Bravo, G.; Huang, X.; Bueso-Ramos, C.; Qiao, W.; Yang, H.; Zhao, C.; Kadia, T.; Borthakur, G.; Pemmaraju, N.; Sasaki, K.; Estrov, Z.; Cortes, J.; Ravandi, F.; Alvarado, Y.; Komrokji, R.; Sekeres, M.A.; Steensma, D.P.; DeZern, A.; Roboz, G.; Kantarjian, H.; Garcia-Manero, G. Randomized phase 2 study of low-dose decitabine vs low-dose azacitidine in lower-risk MDS and MDS/MPN. Blood. 2017 Sep 28;130, 1514-1522. [CrossRef]
- Garcia-Manero, G.; Santini, V.; Almeida, A.; Platzbecker, U.; Jonasova, A.; Silverman, L.R.; Falantes, J.; Reda, G.; Buccisano, F.; Fenaux, P.; Buckstein, R.; Diez Campelo, M.; Larsen, S.; Valcarcel, D.; Vyas, P.; Giai, V.; Olíva, E.N.; Shortt, J.; Niederwieser, D.; Mittelman, M.; Fianchi, L.; La Torre, I.; Zhong, J.; Laille, E.; Lopes de Menezes, D.; Skikne, B.; Beach, C.L.; Giagounidis, A. Phase III, Randomized, Placebo-Controlled Trial of CC-486 (Oral Azacitidine) in Patients With Lower-Risk Myelodysplastic Syndromes. J Clin Oncol. 2021 May 1;39, 1426-1436. [CrossRef]
- Asai, A.; Oshima, Y.; Yamamoto, Y.; Uochi, T.A.; Kusaka, H.; Akinaga, S.; Yamashita, Y.; Pongracz, K.; Pruzan, R.; Wunder, E.; Piatyszek, M.; Li, S.; Chin, A.C.; Harley, C.B.; Gryaznov, S. A novel telomerase template antagonist (GRN163) as a potential anticancer agent. Cancer Res. 2003 Jul 15;63, 3931-9. [PubMed]
- Steensma, D.P.; Fenaux, P.; Van Eygen, K.; Raza, A.; Santini, V.; Germing, U.; Font, P.; Diez-Campelo, M.; Thepot, S.; Vellenga, E.; Patnaik, M.M.; Jang, J.H.; Varsos, H.; Bussolari, J.; Rose, E.; Sherman, L.; Sun, L.; Wan, Y.; Dougherty, S.; Huang, F.; Feller, F.; Rizo, A.; Platzbecker, U. Imetelstat Achieves Meaningful and Durable Transfusion Independence in High Transfusion-Burden Patients With Lower-Risk Myelodysplastic Syndromes in a Phase II Study. J Clin Oncol. 2021 Jan 1;39, 48-56. [CrossRef] [PubMed]
- Sekeres, M.A.; Cutler, C. How we treat higher-risk myelodysplastic syndromes. Blood. 2014 Feb 6;123, 829-36. [CrossRef] [PubMed]
- Platzbecker, U.; Wong, R.S.; Verma, A.; Abboud, C.; Araujo, S.; Chiou, T.J.; Feigert, J.; Yeh, S.P.; Götze, K.; Gorin, N.C.; Greenberg, P.; Kambhampati, S.; Kim, Y.J.; Lee, J.H.; Lyons, R.; Ruggeri, M.; Santini, V.; Cheng, G.; Jang, J.H.; Chen, C.Y.; Johnson, B.; Bennett, J.; Mannino, F.; Kamel, Y.M.; Stone, N.; Dougherty, S.; Chan, G.; Giagounidis, A. Safety and tolerability of eltrombopag versus placebo for treatment of thrombocytopenia in patients with advanced myelodysplastic syndromes or acute myeloid leukaemia: a multicentre, randomised, placebo-controlled, double-blind, phase 1/2 trial. Lancet Haematol. 2015 Oct;2, e417-26. [CrossRef] [PubMed]
- Mittelman, M.; Platzbecker, U.; Afanasyev, B.; Grosicki, S.; Wong, R.S.M.; Anagnostopoulos, A.; Brenner, B.; Denzlinger, C.; Rossi, G.; Nagler, A.; Garcia-Delgado, R.; Portella, M.S.O.; Zhu, Z.; Selleslag, D. Eltrombopag for advanced myelodysplastic syndromes or acute myeloid leukaemia and severe thrombocytopenia (ASPIRE): a randomised, placebo-controlled, phase 2 trial. Lancet Haematol. 2018 Jan;5, e34-e43. [CrossRef] [PubMed]
- Dickinson, M.; Cherif, H.; Fenaux, P.; Mittelman, M.; Verma, A.; Portella, M.S.O.; Burgess, P.; Ramos, P.M.; Choi, J.; Platzbecker, U. ; SUPPORT study investigators. Azacitidine with or without eltrombopag for first-line treatment of intermediate- or high-risk MDS with thrombocytopenia. Blood. 2018 Dec 20;132, 2629-2638. PMCID: PMC6337824. [CrossRef] [PubMed]
- Fenaux, P.; Mufti, G.J.; Hellstrom-Lindberg, E.; Santini, V.; Finelli, C.; Giagounidis, A.; Schoch, R.; Gattermann, N.; Sanz, G.; List, A.; Gore, S.D.; Seymour, J.F.; Bennett, J.M.; Byrd, J.; Backstrom, J.; Zimmerman, L.; McKenzie, D.; Beach, C.; Silverman, L.R. ; International Vidaza High-Risk MDS Survival Study Group. Efficacy of azacitidine compared with that of conventional care regimens in the treatment of higher-risk myelodysplastic syndromes: a randomised, open-label, phase III study. Lancet Oncol. 2009 Mar;10, 223-32. PMCID: PMC4086808. [CrossRef] [PubMed]
- Lübbert, M.; Suciu, S.; Baila, L.; Rüter, B.H.; Platzbecker, U.; Giagounidis, A.; Selleslag, D.; Labar, B.; Germing, U.; Salih, H.R.; Beeldens, F.; Muus, P.; Pflüger, K.H.; Coens, C.; Hagemeijer, A.; Eckart Schaefer, H.; Ganser, A.; Aul, C.; de Witte, T.; Wijermans, P.W. Low-dose decitabine versus best supportive care in elderly patients with intermediate- or high-risk myelodysplastic syndrome (MDS) ineligible for intensive chemotherapy: final results of the randomized phase III study of the European Organisation for Research and Treatment of Cancer Leukemia Group and the German MDS Study Group. J Clin Oncol. 2011 May 20;29, 1987-96. [CrossRef] [PubMed]
- Garcia-Manero, G.; Griffiths, E.A.; Steensma, D.P.; Roboz, G.J.; Wells, R.; McCloskey, J.; Odenike, O.; DeZern, A.E.; Yee, K.; Busque, L.; O'Connell, C.; Michaelis, L.C.; Brandwein, J.; Kantarjian, H.; Oganesian, A.; Azab, M.; Savona, M.R. Oral cedazuridine/decitabine for MDS and CMML: a phase 2 pharmacokinetic/pharmacodynamic randomized crossover study. Blood. 2020 Aug 6;136, 674-683. PMCID: PMC7414597. [CrossRef] [PubMed]
- Ball, B.J.; Famulare, C.A.; Stein, E.M.; Tallman, M.S.; Derkach, A.; Roshal, M.; Gill, S.I.; Manning, B.M.; Koprivnikar, J.; McCloskey, J.; Testi, R.; Prebet, T.; Al Ali, N.H.; Padron, E.; Sallman, D.A.; Komrokji, R.S.; Goldberg, A.D. Venetoclax and hypomethylating agents (HMAs) induce high response rates in MDS, including patients after HMA therapy failure. Blood Adv. 2020 Jul 14;4, 2866-2870. PMCID: PMC7362378. [CrossRef] [PubMed]
- Kadia, T.M.; Reville, P.K.; Borthakur, G.; Yilmaz, M.; Kornblau, S.; Alvarado, Y.; Dinardo, C.D.; Daver, N.; Jain, N.; Pemmaraju, N.; Short, N.; Wang, S.A.; Tidwell, R.S.S.; Islam, R.; Konopleva, M.; Garcia-Manero, G.; Ravandi, F.; Kantarjian, H.M. Venetoclax plus intensive chemotherapy with cladribine, idarubicin, and cytarabine in patients with newly diagnosed acute myeloid leukaemia or high-risk myelodysplastic syndrome: a cohort from a single-centre, single-arm, phase 2 trial. Lancet Haematol. 2021 Aug;8, e552-e561. [CrossRef]
- DeFilipp, Z.; Ciurea, S.O.; Cutler, C.; Robin, M.; Warlick, E.D.; Nakamura, R.; Brunner, A.M.; Dholaria, B.; Walker, A.R.; Kröger, N.; Bejanyan, N.; Atallah, E.; Tamari, R.; Solh, M.M.; Percival, M.E.; de Lima, M.; Scott, B.; Oran, B.; Garcia-Manero, G.; Hamadani, M.; Carpenter, P.; DeZern, A.E. Hematopoietic Cell Transplantation in the Management of Myelodysplastic Syndrome: An Evidence-Based Review from the American Society for Transplantation and Cellular Therapy Committee on Practice Guidelines. Transplant Cell Ther. 2023 Feb;29, 71-81. [CrossRef] [PubMed]
- DiNardo, C.D.; Jabbour, E.; Ravandi, F.; Takahashi, K.; Daver, N.; Routbort, M.; Patel, K.P.; Brandt, M.; Pierce, S.; Kantarjian, H.; Garcia-Manero, G. IDH1 and IDH2 mutations in myelodysplastic syndromes and role in disease progression. Leukemia. 2016 Apr;30, 980-4. PMCID: PMC4733599. [CrossRef] [PubMed]
- Lin, C.C.; Hou, H.A.; Chou, W.C.; Kuo, Y.Y.; Liu, C.Y.; Chen, C.Y.; Lai, Y.J.; Tseng, M.H.; Huang, C.F.; Chiang, Y.C.; Lee, F.Y.; Liu, M.C.; Liu, C.W.; Tang, J.L.; Yao, M.; Huang, S.Y.; Ko, B.S.; Wu, S.J.; Tsay, W.; Chen, Y.C.; Tien, H.F. IDH mutations are closely associated with mutations of DNMT3A, ASXL1 and SRSF2 in patients with myelodysplastic syndromes and are stable during disease evolution. Am J Hematol. 2014 Feb;89, 137-44. [CrossRef] [PubMed]
- Sebert, M.; Cluzeau, T.; Beyne Rauzy, O. Ivosidenib monotherapy is effective in patients with IDH1 mutated myelodysplastic syndrome (MDS): the IDIOME phase 2 study by the GFM group. Blood. 2021;138:62. [CrossRef]
- Stein, E.M.; DiNardo, C.D.; Fathi, A.T.; Pollyea, D.A.; Stone, R.M.; Altman, J.K.; Roboz, G.J.; Patel, M.R.; Collins, R.; Flinn, I.W.; Sekeres, M.A.; Stein, A.S.; Kantarjian, H.M.; Levine, R.L.; Vyas, P.; MacBeth, K.J.; Tosolini, A.; VanOostendorp, J.; Xu, Q.; Gupta, I.; Lila, T.; Risueno, A.; Yen, K.E.; Wu, B.; Attar, E.C.; Tallman, M.S.; de Botton, S. Molecular remission and response patterns in patients with mutant-IDH2 acute myeloid leukemia treated with enasidenib. Blood. 2019 Feb 14;133, 676-687. [CrossRef]
- DiNardo, C.D.; Venugopal, S.; Lachowiez, C.; Takahashi, K.; Loghavi, S.; Montalban-Bravo, G.; Wang, X.; Carraway, H.; Sekeres, M.; Sukkur, A.; Hammond, D.; Chien, K.; Maiti, A.; Masarova, L.; Sasaki, K.; Alvarado, Y.; Kadia, T.; Short, N.J.; Daver, N.; Borthakur, G.; Ravandi, F.; Kantarjian, H.M.; Patel, B.; Dezern, A.; Roboz, G.; Garcia-Manero, G. Targeted therapy with the mutant IDH2 inhibitor enasidenib for high-risk IDH2-mutant myelodysplastic syndrome. Blood Adv. 2023 Jun 13;7, 2378-2387. PMCID: PMC10220255. [CrossRef] [PubMed]
- Sallman, D.A.; Komrokji, R.S.; Sweet, K.L.; Mo, Q.; McGraw, K.L.; Duong, V.H.; Zhang, L.; Nardelli, L.A.; Padron, E.; List, A.F.; Lancet, J.E. A phase 2 trial of the oral smoothened inhibitor glasdegib in refractory myelodysplastic syndromes (MDS). Leuk Res. 2019 Jun;81:56-61. PMCID: PMC7787349. [CrossRef] [PubMed]
- Cortes, J.E.; Heidel, F.H.; Hellmann, A.; Fiedler, W.; Smith, B.D.; Robak, T.; Montesinos, P.; Pollyea, D.A.; DesJardins, P.; Ottmann, O.; Ma, W.W.; Shaik, M.N.; Laird, A.D.; Zeremski, M.; O'Connell, A.; Chan, G.; Heuser, M. Randomized comparison of low dose cytarabine with or without glasdegib in patients with newly diagnosed acute myeloid leukemia or high-risk myelodysplastic syndrome. Leukemia. 2019 Feb;33, 379-389. PMCID: PMC6365492. [CrossRef] [PubMed]
- Garcia-Manero, G.; Fenaux, P.; Al-Kali, A.; Baer, M.R.; Sekeres, M.A.; Roboz, G.J.; Gaidano, G.; Scott, B.L.; Greenberg, P.; Platzbecker, U.; Steensma, D.P.; Kambhampati, S.; Kreuzer, K.A.; Godley, L.A.; Atallah, E.; Collins RJr Kantarjian, H.; Jabbour, E.; Wilhelm, F.E.; Azarnia, N.; Silverman, L.R. ; ONTIME study investigators. Rigosertib versus best supportive care for patients with high-risk myelodysplastic syndromes after failure of hypomethylating drugs (ONTIME): a randomised, controlled, phase 3 trial. Lancet Oncol. 2016 Apr;17, 496-508. [CrossRef] [PubMed]
- de Carvalho, L.G.A.; Komoto, T.T.; Moreno, D.A.; Goes, J.V.C.; de Oliveira, R.T.G.; de Lima Melo, M.M.; Roa, M.E.G.V.; Gonçalves, P.G.; Montefusco-Pereira, C.V.; Pinheiro, R.F.; Ribeiro Junior, H.L. USP15-USP7 Axis and UBE2T Differential Expression May Predict Pathogenesis and Poor Prognosis in De Novo Myelodysplastic Neoplasm. Int J Mol Sci. 2023 Jun 13;24, 10058. PMCID: PMC10298103. [CrossRef] [PubMed]
- Soucy, T.A.; Smith, P.G.; Milhollen, M.A.; Berger, A.J.; Gavin, J.M.; Adhikari, S.; Brownell, J.E.; Burke, K.E.; Cardin, D.P.; Critchley, S.; Cullis, C.A.; Doucette, A.; Garnsey, J.J.; Gaulin, J.L.; Gershman, R.E.; Lublinsky, A.R.; McDonald, A.; Mizutani, H.; Narayanan, U.; Olhava, E.J.; Peluso, S.; Rezaei, M.; Sintchak, M.D.; Talreja, T.; Thomas, M.P.; Traore, T.; Vyskocil, S.; Weatherhead, G.S.; Yu, J.; Zhang, J.; Dick, L.R.; Claiborne, C.F.; Rolfe, M.; Bolen, J.B.; Langston, S.P. An inhibitor of NEDD8-activating enzyme as a new approach to treat cancer. Nature. 2009 Apr 9;458, 732-6. [CrossRef] [PubMed]
- Sekeres, M.A.; Watts, J.; Radinoff, A.; Sangerman, M.A.; Cerrano, M.; Lopez, P.F.; Zeidner, J.F.; Campelo, M.D.; Graux, C.; Liesveld, J.; Selleslag, D.; Tzvetkov, N.; Fram, R.J.; Zhao, D.; Bell, J.; Friedlander, S.; Faller, D.V.; Adès, L. Randomized phase 2 trial of pevonedistat plus azacitidine versus azacitidine for higher-risk MDS/CMML or low-blast AML. Leukemia. 2021 Jul;35, 2119-2124. Erratum in: Leukemia. 2021 Dec;35, 3637. PMCID: PMC8257476. [CrossRef] [PubMed]
- Sallman, D.A.; Al Malki, M.M.; Asch, A.S.; Wang, E.S.; Jurcic, J.G.; Bradley, T.J.; Flinn, I.W.; Pollyea, D.A.; Kambhampati, S.; Tanaka, T.N.; Zeidner, J.F.; Garcia-Manero, G.; Jeyakumar, D.; Komrokji, R.; Lancet, J.; Kantarjian, H.M.; Gu, L.; Zhang, Y.; Tan, A.; Chao, M.; O'Hear, C.; Ramsingh, G.; Lal, I.; Vyas, P.; Daver, N.G. Magrolimab in Combination With Azacitidine in Patients With Higher-Risk Myelodysplastic Syndromes: Final Results of a Phase Ib Study. J Clin Oncol. 2023 May 20;41, 2815-2826. [CrossRef] [PubMed]
- Magrolimab + azacitidine versus azacitidine + placebo in untreated participants with myelodysplastic syndrome (MDS) (ENHANCE). ClinicalTrials.gov identifier: NCT04313881. Updated September 25, 2023. Available online: https://classic.clinicaltrials.gov/ct2/show/NCT04313881 (accessed on 1 December 2023).
- Zeidan, A.M.; Giagounidis, A.; Sekeres, M.A.; Xiao, Z.; Sanz, G.F.; Hoef, M.V.; Ma, F.; Hertle, S.; Santini, V. STIMULUS-MDS2 design and rationale: a phase III trial with the anti-TIM-3 sabatolimab (MBG453) + azacitidine in higher risk MDS and CMML-2. Future Oncol. 2023 Mar;19, 631-642. [CrossRef] [PubMed]
- Zeidan, A.M.; Esteve, J.; Giagounidis, A. The STIMULUS program: clinical trials evaluating sabatolimab (MBG453) combination therapy in patients (pts) with higher-risk myelodysplastic syndromes (HR-MDS) or acute myeloid leukemia (AML). Blood. 2020;136(suppl 1)45-46. [CrossRef]
- Maslah, N.; Salomao, N.; Drevon, L.; Verger, E.; Partouche, N.; Ly, P.; Aubin, P.; Naoui, N.; Schlageter, M.H.; Bally, C.; Miekoutima, E.; Rahmé, R.; Lehmann-Che, J.; Ades, L.; Fenaux, P.; Cassinat, B.; Giraudier, S. Synergistic effects of PRIMA-1Met (APR-246) and 5-azacitidine in TP53-mutated myelodysplastic syndromes and acute myeloid leukemia. Haematologica. 2020 Jun;105, 1539-1551. PMCID: PMC7271596. [CrossRef] [PubMed]
- Sallman, D.A.; DeZern, A.E.; Garcia-Manero, G.; Steensma, D.P.; Roboz, G.J.; Sekeres, M.A.; Cluzeau, T.; Sweet, K.L.; McLemore, A.; McGraw, K.L.; Puskas, J.; Zhang, L.; Yao, J.; Mo, Q.; Nardelli, L.; Al Ali, N.H.; Padron, E.; Korbel, G.; Attar, E.C.; Kantarjian, H.M.; Lancet, J.E.; Fenaux, P.; List, A.F.; Komrokji, R.S. Eprenetapopt (APR-246) and Azacitidine in TP53-Mutant Myelodysplastic Syndromes. J Clin Oncol. 2021 May 10;39, 1584-1594. [CrossRef] [PubMed]
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




