Submitted:
08 February 2024
Posted:
09 February 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
3.1. Patients’ Characteristics
3.2. Outcome of co-ultraPEALut and control patients
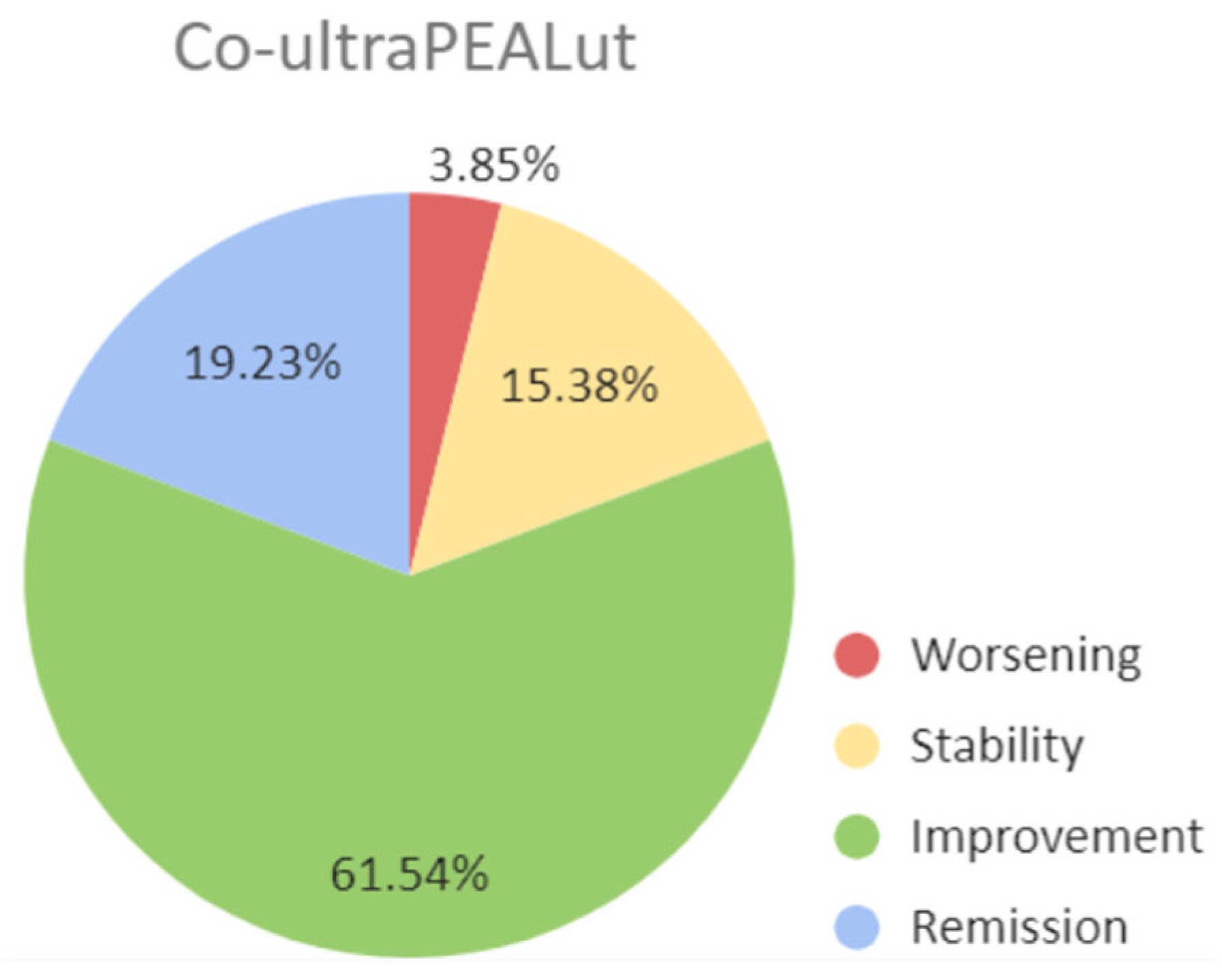
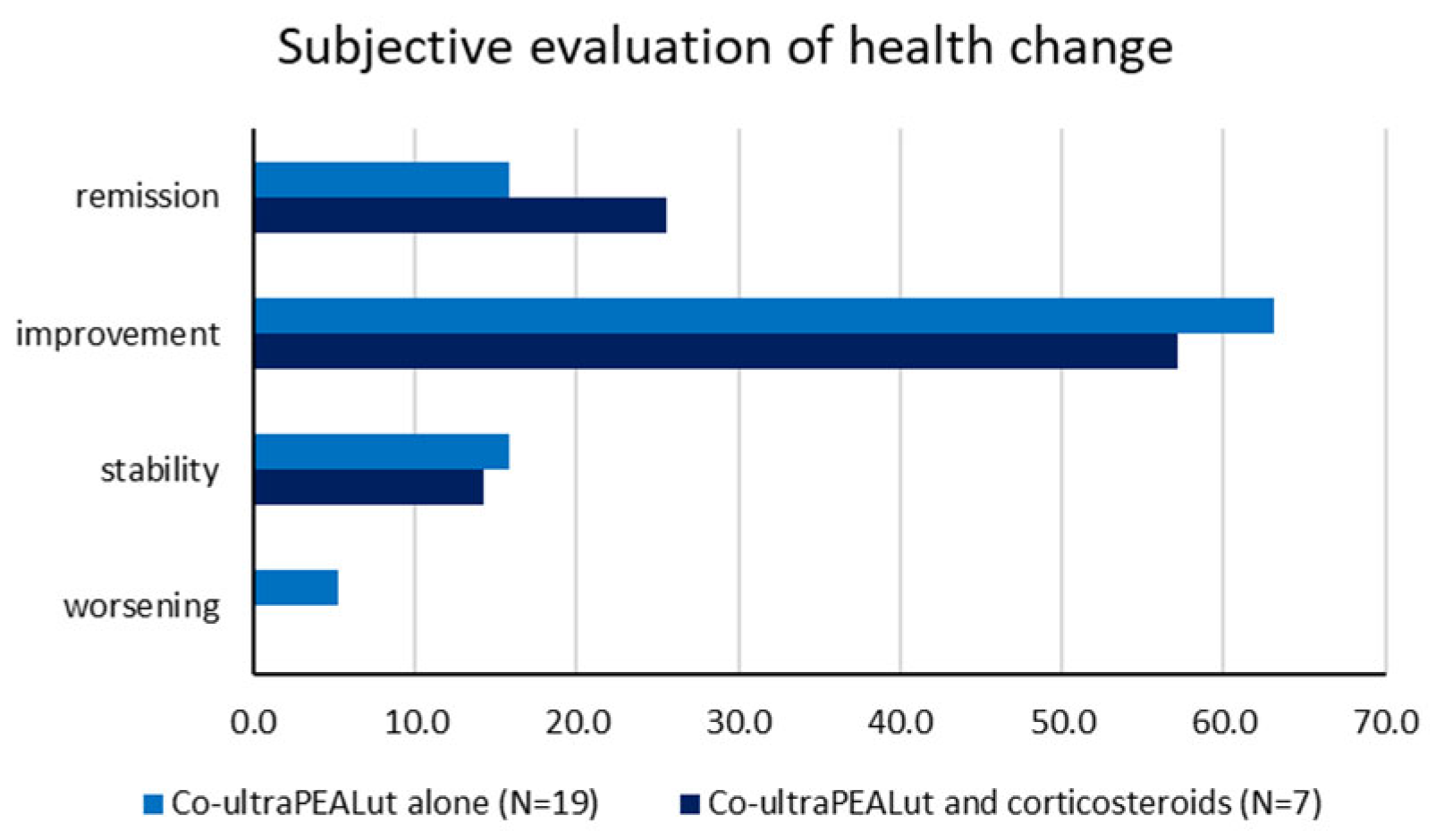
3.3. Outcome of co-ultraPEALut subgroups patients
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Ceban F, Ling S, Lui LMW, et al. (2022) Fatigue and cognitive impairment in Post-COVID-19 Syndrome: A systematic review and meta-analysis. Brain Behav Immun 101:93–135. [CrossRef]
- (2020) Overview | COVID-19 rapid guideline: managing the long-term effects of COVID-19 | Guidance | NICE. https://www.nice.org.uk/guidance/ng188 (accessed on 26 May 2023).
- Crook H, Raza S, Nowell J, et al. (2021) Long covid—mechanisms, risk factors, and management. BMJ 374:n1648. [CrossRef]
- Michelutti M, Furlanis G, Stella AB, et al. (2022) Sex-dependent characteristics of Neuro-Long-COVID: Data from a dedicated neurology ambulatory service. J Neurol Sci 441:120355. [CrossRef]
- Teodoro T, Chen J, Gelauff J, Edwards MJ (2023) Functional neurological disorder in people with long COVID: A systematic review. Eur J Neurol 30:1505–1514. [CrossRef]
- Van der Feltz-Cornelis CM, Moriarty AS, Strain WD (2023) Neurological Dysfunction in Long COVID Should Not Be Labelled as Functional Neurological Disorder. Viruses 15:783. [CrossRef]
- Nalbandian A, Sehgal K, Gupta A, et al. (2021) Post-acute COVID-19 syndrome. Nat Med 27:601–615. [CrossRef]
- Bortolato B, Carvalho AF, Soczynska JK, et al. (2015) The Involvement of TNF-α in Cognitive Dysfunction Associated with Major Depressive Disorder: An Opportunity for Domain Specific Treatments. Curr Neuropharmacol 13:558–576. [CrossRef]
- Bulla R, Rossi L, Furlanis G, et al. (2023) A likely association between low mannan-binding lectin level and brain fog onset in long COVID patients. Front Immunol 14:1191083. [CrossRef]
- Aloe L, Leon A, Levi-Montalcini R (1993) A proposed autacoid mechanism controlling mastocyte behaviour. Agents Actions 39 Spec No:C145-147. [CrossRef]
- Impellizzeri D, Bruschetta G, Cordaro M, et al. (2014) Micronized/ultramicronized palmitoylethanolamide displays superior oral efficacy compared to nonmicronized palmitoylethanolamide in a rat model of inflammatory pain. J Neuroinflammation 11:136. [CrossRef]
- Petrosino S, Cordaro M, Verde R, et al. (2018) Oral Ultramicronized Palmitoylethanolamide: Plasma and Tissue Levels and Spinal Anti-hyperalgesic Effect. Front Pharmacol 9:249. [CrossRef]
- Di Paola R, Fusco R, Gugliandolo E, et al. (2016) Co-micronized Palmitoylethanolamide/Polydatin Treatment Causes Endometriotic Lesion Regression in a Rodent Model of Surgically Induced Endometriosis. Front Pharmacol 7:382. [CrossRef]
- Peritore AF, Siracusa R, Crupi R, Cuzzocrea S (2019) Therapeutic Efficacy of Palmitoylethanolamide and Its New Formulations in Synergy with Different Antioxidant Molecules Present in Diets. Nutrients 11:2175. [CrossRef]
- Petrosino S, Di Marzo V (2017) The pharmacology of palmitoylethanolamide and first data on the therapeutic efficacy of some of its new formulations. Br J Pharmacol 174:1349–1365. [CrossRef]
- Caltagirone C, Cisari C, Schievano C, et al. (2016) Co-ultramicronized Palmitoylethanolamide/Luteolin in the Treatment of Cerebral Ischemia: from Rodent to Man. Transl Stroke Res 7:54–69. [CrossRef]
- Campolo M, Crupi R, Cordaro M, et al. (2021) Co-Ultra PEALut Enhances Endogenous Repair Response Following Moderate Traumatic Brain Injury. Int J Mol Sci 22:8717. [CrossRef]
- Siracusa R, Impellizzeri D, Cordaro M, et al. (2017) Anti-Inflammatory and Neuroprotective Effects of Co-UltraPEALut in a Mouse Model of Vascular Dementia. Front Neurol 8:233. [CrossRef]
- Aiello EN, Gramegna C, Esposito A, et al. (2022) The Montreal Cognitive Assessment (MoCA): updated norms and psychometric insights into adaptive testing from healthy individuals in Northern Italy. Aging Clin Exp Res 34:375–382. [CrossRef]
- Nasreddine ZS, Phillips NA, Bédirian V, et al. (2005) The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53:695–699. [CrossRef]
- Aiello EN, Fiabane E, Manera MR, et al. (2022) Screening for cognitive sequelae of SARS-CoV-2 infection: a comparison between the Mini-Mental State Examination (MMSE) and the Montreal Cognitive Assessment (MoCA). Neurol Sci Off J Ital Neurol Soc Ital Soc Clin Neurophysiol 43:81–84. [CrossRef]
- Anton HA, Miller WC, Townson AF (2008) Measuring Fatigue in Persons with Spinal Cord Injury. Arch Phys Med Rehabil 89:538–542. [CrossRef]
- Smith G, Della Sala S, Logie RH, Maylor EA (2000) Prospective and retrospective memory in normal ageing and dementia: a questionnaire study. Mem Hove Engl 8:311–321. [CrossRef]
- Lyra e Silva NM, Barros-Aragão FGQ, De Felice FG, Ferreira ST (2022) Inflammation at the crossroads of COVID-19, cognitive deficits and depression. Neuropharmacology 209:109023. [CrossRef]
- Sutter R, Hert L, De Marchis GM, et al. (2021) Serum Neurofilament Light Chain Levels in the Intensive Care Unit: Comparison between Severely Ill Patients with and without Coronavirus Disease 2019. Ann Neurol 89:610–616. [CrossRef]
- Figueiredo CP, Barros-Aragão FGQ, Neris RLS, et al. (2019) Zika virus replicates in adult human brain tissue and impairs synapses and memory in mice. Nat Commun 10:3890. [CrossRef]
- Burton MD, Johnson RW (2012) Interleukin-6 trans-signaling in the senescent mouse brain is involved in infection-related deficits in contextual fear conditioning. Brain Behav Immun 26:732–738. [CrossRef]
- Buoite Stella A, Furlanis G, Frezza NA, et al. (2022) Autonomic dysfunction in post-COVID patients with and witfhout neurological symptoms: a prospective multidomain observational study. J Neurol 269:587–596. [CrossRef]
- Mazza MG, Palladini M, De Lorenzo R, et al. (2021) Persistent psychopathology and neurocognitive impairment in COVID-19 survivors: Effect of inflammatory biomarkers at three-month follow-up. Brain Behav Immun 94:138–147. [CrossRef]
- Fudim M, Qadri YJ, Ghadimi K, et al. (2020) Implications for Neuromodulation Therapy to Control Inflammation and Related Organ Dysfunction in COVID-19. J Cardiovasc Transl Res 13:894–899. [CrossRef]
- Ajčević M, Iscra K, Furlanis G, et al. (2023) Cerebral hypoperfusion in post-COVID-19 cognitively impaired subjects revealed by arterial spin labeling MRI. Sci Rep 13:5808. [CrossRef]
- Hosp JA, Dressing A, Blazhenets G, et al. (2021) Cognitive impairment and altered cerebral glucose metabolism in the subacute stage of COVID-19. Brain awab009. [CrossRef]
- Furlanis G, Buoite Stella A, Biaduzzini F, et al. (2023) Cognitive deficit in post-acute COVID-19: an opportunity for EEG evaluation? Neurol Sci 44:1491–1498. [CrossRef]
- Manganotti P, Michelutti M, Furlanis G, et al. (2023) Deficient GABABergic and glutamatergic excitability in the motor cortex of patients with long-COVID and cognitive impairment. Clin Neurophysiol 151:83–91. [CrossRef]
| Co-ultraPEALut (n = 26) |
Control (n = 15) |
|
|---|---|---|
| Age (years) | 54.69 ± 11.26 | 55.8 ± 14.84 |
| Sex Females Males |
19 (73.08%) 7 (26.92%) |
7 (46.67%) 8 (53.33%) |
| Years of education | 14.10 ± 3.05 | 14.33 ± 3.46 |
| COVID-19 severity Paucisymptomatic infection, at-home management Hospitalization without ventilation ICU admission with ventilation |
22 (84.62%) 4 (15.38%) 0 |
14 (93.33%) 1 (6.67%) 0 |
| NS-T0 interval (months) | 7.33 ± 4.75 | 10.43 ± 5.12 |
| T0-T1 interval (months) | 9.92 ± 5.37 | 7.54 ± 2.36 |
| Co-ultraPEALut (n = 26) | |||
| Test | T0 | T1 | p value |
| MoCA (raw score) | 25.76 ± 2.3 | 27.2 ± 2 | .0261* |
| MoCA (adjusted score) | 24.34 ± 2.58 | 25.59 ± 2.39 | .0958 |
| FSS | 5.03 ± 1.69 | 4.52 ± 1.63 | .3188 |
| PRMQ | 51.94 ± 10.55 | 39.67 ± 13.02 | <.00001* |
| Control (n = 15) | |||
| Test | T0 | T1 | p value |
| MoCA (raw score) | 25.93 ± 2.6 | - | - |
| MoCA (adjusted score) | 24.74 ± 3.26 | - | - |
| FSS | 4.95 ± 1.57 | 4.06 ± 1.47 | .1352 |
| PRMQ | 45.77 ± 13.47 | 42.33 ± 16.86 | .2051 |
| Co-ultraPEALut alone (n = 19) | |||
|---|---|---|---|
| Test | T0 | T1 | p value |
| MoCA (raw score) | 25.93 ± 2.52 | 27.11 ± 1.82 | .1417 |
| MoCA (adjusted score) | 24.54 ± 2.82 | 25.33 ± 2.03 | .8488 |
| FSS | 4.54 ± 1.54 | 4.29 ± 1.68 | .6803 |
| PRMQ | 53.38 ± 9.92 | 40.24 ± 13.3 | .0067* |
| Co-ultraPEALut and corticosteroids (n = 7) | |||
| Test | T0 | T1 | p value |
| MoCA (raw score) | 25.33 ± 1.21 | 27.43 ± 2.3 | .0709 |
| MoCA (adjusted score) | 24.47 ± 2.2 | 26.27 ± 3.11 | .2609 |
| FSS | 6.8 ± 0.23 | 5.24 ± 1.32 | .6251 |
| PRMQ | 47.25 ± 11.35 | 38.29 ± 12.88 | .2782 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




