Submitted:
23 January 2024
Posted:
24 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Aims of the Study
2.2. Research Hypotheses
2.3. Type of Study
2.4. Population
- -
- Patients benefiting from a physiotherapy rehabilitation program at the CHU Liège, site CNRF.
- -
- Patients with a pathological situation justifying functional rehabilitation with the HUBER 360®.
- -
- Patient with any pathology that does not constitute an exclusion criterion.
- -
- Patient with informed consent for the study
- -
- Cardiac, respiratory, neurological or rheumatological disease incompatible with physical activity.
- -
- Arthritis.
- -
- Rheumatic disease in acute phase.
- -
- Recent trauma, infection of the musculoskeletal system.
- -
- Fever.
- -
- Venous thrombosis.
- -
- Acute intervertebral disc disease.
- -
- Neuropsychological problems that do not allow the integration of instructions or other serious psychological problems.
- -
- Cardiovascular disease and any progressive, chronic disease that is not conducive to exercise.
- -
- Major anatomical deformities.
2.5. Randomisation
2.6. Study Design/Intervention Protocol
2.7. Evaluation Criteria
2.8. Statistical Analyses
2.9. Statistical Power
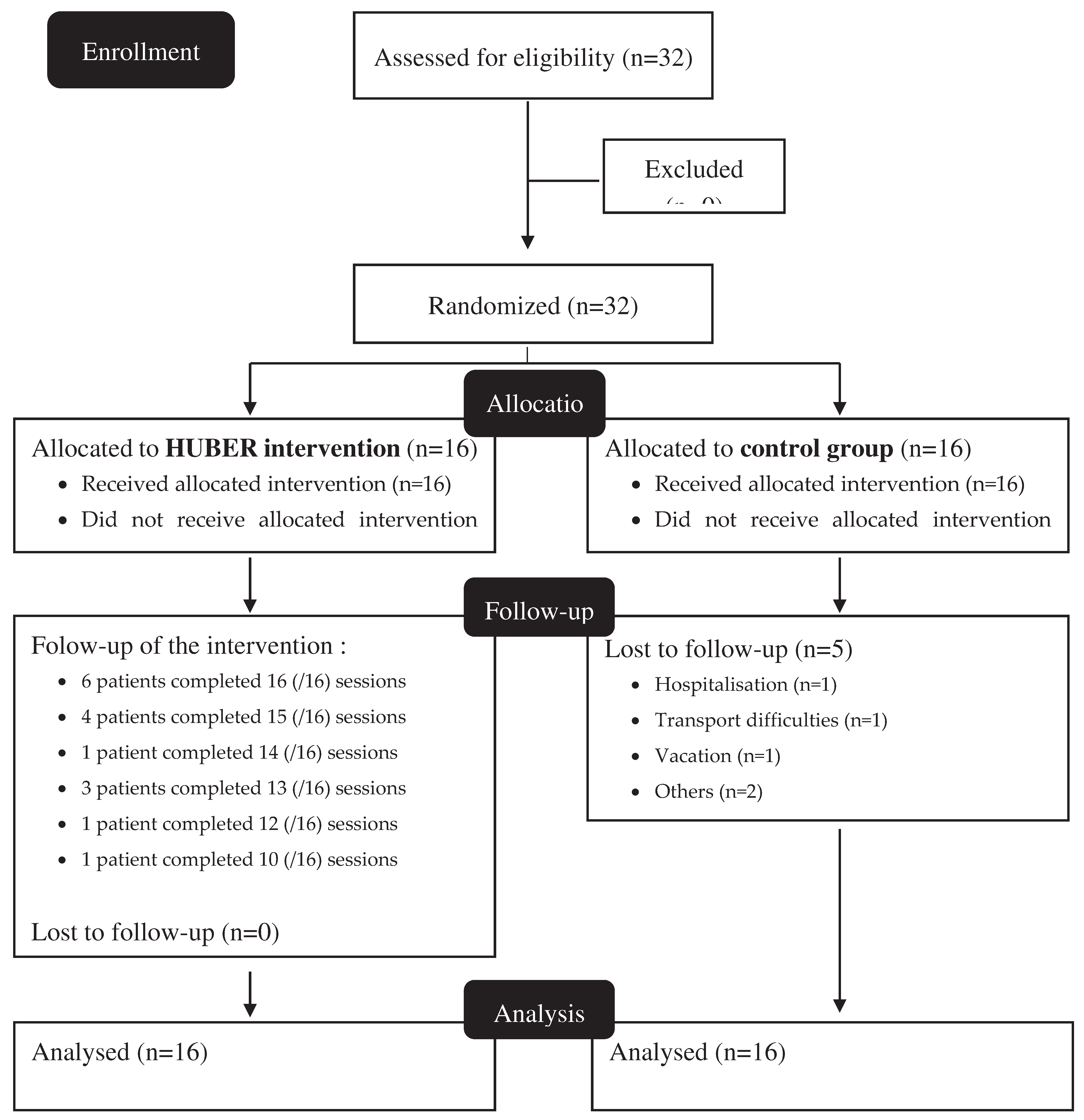
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Dorsey ER, Sherer T, Okun MS, Bloemd BR. The Emerging Evidence of the Parkinson Pandemic. J Parkinsons Dis [Internet]. 2018 [cited 2023 May 1];8(s1):S3–8. Available from:. [CrossRef]
- Bloem BR, Grimbergen YAM, Cramer M, Willemsen M, Zwinderman AH. Prospective assessment of falls in Parkinson’s disease. J Neurol [Internet]. 2001 [cited 2023 May 1];248(11):950–8. Available from: https://pubmed.ncbi.nlm.nih.gov/11757958/. [CrossRef]
- Ashburn A, Stack E, Ballinger C, Fazakarley L, Fitton C. The circumstances of falls among people with Parkinson’s disease and the use of Falls Diaries to facilitate reporting. Disabil Rehabil [Internet]. 2008 [cited 2023 May 1];30(16):1205–12. Available from: https://pubmed.ncbi.nlm.nih.gov/18608387/. [CrossRef]
- Eng JJ, Pang MYC, Ashe MC. Balance, falls, and bone health: role of exercise in reducing fracture risk after stroke. J Rehabil Res Dev [Internet]. 2008 [cited 2023 May 1];45(2):297–314. Available from: https://pubmed.ncbi.nlm.nih.gov/18566947/. [CrossRef]
- Nilsagard Y, Gunn H, Freeman J, Hoang P, Lord S, Mazumder R, et al. Falls in people with MS--an individual data meta-analysis from studies from Australia, Sweden, United Kingdom and the United States. Mult Scler [Internet]. 2015 Jan 14 [cited 2023 May 1];21(1):92–100. Available from: https://pubmed.ncbi.nlm.nih.gov/24948687/. [CrossRef]
- Callaly EL, Ni Chroinin D, Hannon N, Sheehan O, Marnane M, Merwick A, et al. Falls and fractures 2 years after acute stroke: The North Dublin Population Stroke Study. Age Ageing [Internet]. 2015 Sep 1 [cited 2023 May 1];44(5):882–6. Available from: https://jhu.pure.elsevier.com/en/publications/fallsand-fractures-2-years-after-acute-stroke-the-north-dublin-p-3. [CrossRef]
- Liu TW, Ng GYF, Ng SSM. Effectiveness of a combination of cognitive behavioral therapy and taskoriented balance training in reducing the fear of falling in patients with chronic stroke: Study protocol for a randomized controlled trial. Trials [Internet]. 2018 Mar 7 [cited 2023 May 1];19(1):1–10. Available from: https://trialsjournal.biomedcentral.com/articles/10.1186/s13063-018-2549-z. [CrossRef]
- Paul SS, Sherrington C, Canning CG, Fung VSC, Close JCT, Lord SR. The relative contribution of physical and cognitive fall risk factors in people with Parkinson’s disease: a large prospective cohort study. Neurorehabil Neural Repair [Internet]. 2014 Mar [cited 2023 May 1];28(3):282–90. Available from: https://pubmed.ncbi.nlm.nih.gov/24243915/. [CrossRef]
- Paul SS, Harvey L, Canning CG, Boufous S, Lord SR, Close JCT, et al. Fall-related hospitalization in people with Parkinson’s disease. Eur J Neurol [Internet]. 2017 Jan 24 [cited 2023 May 1];24(3):523–9. Available from: https://europepmc.org/article/MED/28117538. [CrossRef]
- Cameron MH, Nilsagard Y. Balance, gait, and falls in multiple sclerosis. Handb Clin Neurol [Internet]. 2018 Jan 1 [cited 2023 May 1];159:237–50. Available from: https://pubmed.ncbi.nlm.nih.gov/30482317/. [CrossRef]
- Coote S, Comber L, Quinn G, Santoyo-Medina C, Kalron A, Gunn H. Falls in People with Multiple Sclerosis: Risk Identification, Intervention, and Future Directions. Int J MS Care [Internet]. 2020 Nov 1 [cited 2023 May 1];22(6):247–55. Available from: https://pubmed.ncbi.nlm.nih.gov/33424479/. [CrossRef]
- Sherrington C, Michaleff ZA, Fairhall N, Paul SS, Tiedemann A, Whitney J, et al. Exercise to prevent falls in older adults: an updated systematic review and meta-analysis. Br J Sports Med [Internet]. 2017 Dec 1 [cited 2023 May 1];51(24):1749–57. Available from: https://pubmed.ncbi.nlm.nih.gov/27707740/. [CrossRef]
- De Souto Barreto P, Rolland Y, Vellas B, Maltais M. Association of Long-term Exercise Training With Risk of Falls, Fractures, Hospitalizations, and Mortality in Older Adults: A Systematic Review and Metaanalysis. JAMA Intern Med [Internet]. 2019 Mar 1 [cited 2023 May 1];179(3):394–405. Available from: https://pubmed.ncbi.nlm.nih.gov/30592475/. [CrossRef]
- Cadore EL, Rodríguez-Mañas L, Sinclair A, Izquierdo M. Effects of different exercise interventions on risk of falls, gait ability, and balance in physically frail older adults: a systematic review. Rejuvenation Res [Internet]. 2013 Apr 1 [cited 2023 May 1];16(2):105–14. Available from: https://pubmed.ncbi.nlm.nih.gov/23327448/. [CrossRef]
- De Souto Barreto P, Rolland Y, Vellas B, Maltais M. Association of Long-term Exercise Training With Risk of Falls, Fractures, Hospitalizations, and Mortality in Older Adults: A Systematic Review and Metaanalysis. JAMA Intern Med [Internet]. 2019 Mar 1 [cited 2023 May 1];179(3):394–405. Available from: https://pubmed.ncbi.nlm.nih.gov/30592475/. [CrossRef]
- Pereira CLN, Vogelaere P, Baptista F. Role of physical activity in the prevention of falls and their consequences in the elderly. European Review of Aging and Physical Activity [Internet]. 2008 Apr 23 [cited 2023 May 1];5(1):51–8. Available from: https://eurapa.biomedcentral.com/articles/10.1007/s11556-008-0031-8. [CrossRef]
- Saywell N, Taylor N, Rodgers E, Skinner L, Boocock M. Play-based interventions improve physical function for people with adult-acquired brain injury: a systematic review and meta-analysis of randomised controlled trials. Clin Rehabil [Internet]. 2017 Feb 1 [cited 2023 May 1];31(2):145–57. Available from: https://pubmed.ncbi.nlm.nih.gov/26869595/. [CrossRef]
- McGlinchey MP, James J, McKevitt C, Douiri A, Sackley C. The effect of rehabilitation interventions on physical function and immobility-related complications in severe stroke: a systematic review. BMJ Open [Internet]. 2020 Feb 5 [cited 2023 May 1];10(2). Available from: https://pubmed.ncbi.nlm.nih.gov/32029489/. [CrossRef]
- Laurent C, Beaudart C, Léonard Y, Maertens B, Laurent L, Kaux J-F. Rééducation globale à l’aide du système HUBER® en kinésithérapie : une revue systématique de la littérature. Revue médicale de Liège septembre 2023 :78 (9):490-495.
- Fabre, J.B., Martin, V., Borelli, G., Fritsch, N. , Theurel, J. Effects of a whole-body strength training program on metabolic responses and body composition. Gazzetta Medica Italiana Archivio per le Scienze Mediche 2014 January-February; 173 (1-2):47-56. | Request PDF [Internet]. [cited 2023 May 2]. Available from: https://www.researchgate.net/publication/357484414_Effects_of_a_wholebody_strength_training_program_on_metabolic_responses_and_body_composition_FABRE_JB_ET_AL _Gazzetta_Medica_Italiana_Archivio_per_le_Scienze_Mediche_2014_January-February_173_1-247-56.
- Couillandre A, Duque Ribeiro MJ, Thoumie P, Portero P. Changes in balance and strength parameters induced by training on a motorised rotating platform: a study on healthy subjects. Ann Readapt Med Phys [Internet]. 2008 Mar [cited 2023 May 2];51(2):67–73. Available from: https://pubmed.ncbi.nlm.nih.gov/18207276/. [CrossRef]
- Guiraud T, Labrunée M, Besnier F, Sénard JM, Pillard F, Rivière D, et al. Whole-body strength training with Huber Motion Lab and traditional strength training in cardiac rehabilitation: A randomized controlled study. Ann Phys Rehabil Med [Internet]. 2017 Jan 1 [cited 2023 May 2];60(1):20–6. Available from: https://pubmed.ncbi.nlm.nih.gov/27650531/. [CrossRef]
- Markovic G, Sarabon N, Greblo Z, Krizanic V. Effects of feedback-based balance and core resistance training vs. Pilates training on balance and muscle function in older women: a randomized-controlled trial. Arch Gerontol Geriatr [Internet]. 2015 Sep 1 [cited 2023 May 2];61(2):117–23. Available from: https://pubmed.ncbi.nlm.nih.gov/26036209/. [CrossRef]
- Letafatkar A, Nazarzadeh M, Hadadnezhad M, Farivar N. The efficacy of a HUBER exercise system mediated sensorimotor training protocol on proprioceptive system, lumbar movement control and quality of life in patients with chronic non-specific low back pain. J Back Musculoskelet Rehabil [Internet]. 2017 [cited 2023 May 2];30(4):767–78. Available from: https://pubmed.ncbi.nlm.nih.gov/28453452/. [CrossRef]
- Tantot M, Le Moal V, Mévellec É, Nouy-Trollé I, Lemoine-Josse E, Besnier F, et al. Effects of an Intensive 6-Week Rehabilitation Program with the HUBER Platform in the Treatment of Non-Specific Chronic Low Back Pain: A Pilot Study. Clin Pract [Internet]. 2022 Aug 1 [cited 2023 May 2];12(4):609–18. Available from: https://pubmed.ncbi.nlm.nih.gov/36005067/. [CrossRef]
- D P, S R. The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc [Internet]. 1991 [cited 2023 May 2];39(2):142–8. Available from: https://pubmed.ncbi.nlm.nih.gov/1991946/. [CrossRef]
- Pavasini R, Guralnik J, Brown JC, di Bari M, Cesari M, Landi F, et al. Short Physical Performance Battery and all-cause mortality: systematic review and meta-analysis. BMC Med [Internet]. 2016 Dec 22 [cited 2023 May 2];14(1). Available from: https://pubmed.ncbi.nlm.nih.gov/28003033/. [CrossRef]
- Ware JE, Gandek B. Overview of the SF-36 Health Survey and the International Quality of Life Assessment (IQOLA) Project. J Clin Epidemiol [Internet]. 1998 Nov [cited 2023 May 2];51(11):903–12. Available from: https://pubmed.ncbi.nlm.nih.gov/9817107/. [CrossRef]

| HUBER (n=16) | Control (n=16) | p-value between groups∆ | |
|---|---|---|---|
| Sex Women |
7 (43.7%) |
5 (31.2%) |
0.46 |
| Age (years) | 65.4 ± 8.44 | 57.4 ± 10.9 | 0.03 |
| BMI (kg/m²) | 26.1 ± 3.26 | 28.9 ± 6.41 | 0.13 |
| Pathologies Multiple Sclerosis Parkinson Stroke Other (PNP, CT) |
2 (12.5) 0 (0.00) 8 (50.0) 6 (37.5) |
3 (18.5) 2 (12.5) 3 (18.5) 8 (50.0) |
0.31 |
| Walking aid Yes |
5 (31.2%) |
5 (31.2%) |
1.00 |
| Baseline | 4 weeks‡ | 8 Weeks‡ | Change between groups over time (pvalue)∆ | |
|---|---|---|---|---|
| SPPB Balance HUBER |
3.69 ± 0.60 |
3.75 ± 0.58 |
3.81 ± 0.54 |
0.81 |
| Control P-value |
3.44 ± 1.03 0.41 |
3.560 ± 1.03 0.53 |
3.56 ± 1.03 0.39 |
|
| SPPB Gait HUBER Control P-value |
3.5 ± 1.09 3.69 ± 0.87 0.59 |
3.56 ± 0.97 3.38 ± 1.20 0.63 |
3.50 ± 1.09 3.38 ± 1.15 0.75 |
0.15 |
| SPPB CRT HUBER Control P-value |
1.81 ± 1.28 2.44 ± 1.55 0.22 |
2.25 ± 1.34 2.44 ± 1.41 0.70 |
2.44 ± 1.21* 2.38 ± 1.36 0.89 |
0.021 |
| SPPB Total HUBER Control P-value |
9.00 ± 2.45 9.56 ± 3.03 0.57 |
9.56 ± 2.39 9.38 ± 3.12 0.85 |
9.75 ± 2.46* 9.31 ± 2.96 0.65 |
0.003 |
| TUG (seconds) HUBER Control P-value |
16.5 ± 16.2 21.18 ± 38.7 0.66 |
15.6 ± 15.7 16.6 ± 23.6 0.89 |
13.5 ± 11.5 16.7 ± 23.6 0.63 |
0.6 |
| Baseline | 4 weeks | 8 Weeks | Change between groups over time (pvalue)∆ | |
|---|---|---|---|---|
| EQ5D utility score HUBER Control P-value |
0.61 ± 0.23 0.61 ± 0.22 0.98 |
0.71 ± 0.18* 0.59 ± 0.29 0.23 |
0.76 ± 0.13* 0.65 ± 0.17 0.042 |
0.063 |
| EQVAS HUBER Control P-value |
70.0 ± 13.17 64.1 ± 10.99 0.18 |
70.53 ± 12.1 66.2 ± 11.9 0.32 |
78.1 ± 10.9* 68.7 ± 9.04* 0.013 |
0.21 |
| SF-36 PCS HUBER Control P-value |
38.6 ± 8.18 36.2 ± 9.70 0.46 |
41.5 ± 8.3 34.0 ± 7.85 0.014 |
42.3 ± 8.54 35.9 ± 8.84 0.046 |
0.18 |
| SF-36 MCS HUBER Control P-value |
45.5 ± 9.01 45.9 ± 9.76 0.91 |
47.8 ± 10.0 43.6 ± 11.8 0.29 |
50.8 ± 8.29* 45.3 ± 10.6 0.11 |
0.16 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




