Submitted:
22 January 2024
Posted:
23 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Research Goals and Objectives:
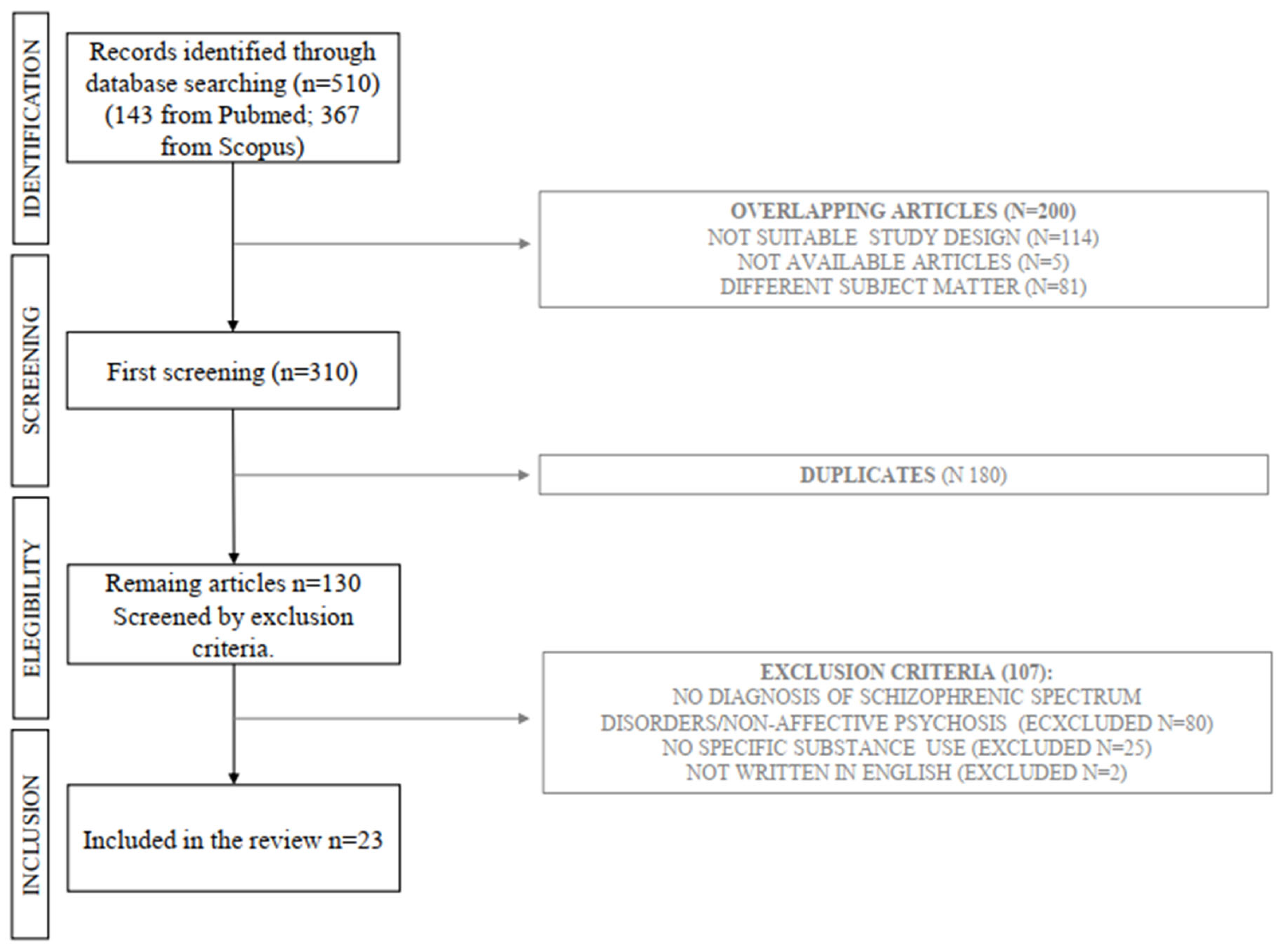
2. Matherials and Methods
2.1. Data Extraction
2.2. Data Synthesis Strategy
3. Results
3.1. Aripirazole
3.2. Cariprazine
3.3. Brexpiprazole
3.4. Lurasidone
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data avalaibility statement
Conflict Interest
References
- Martinotti, G.; De Risio, L.; Vannini, C.; Schifano, F.; Pettorruso, M.; Di Giannantonio, M. Substance-related exogenous psychosis: a postmodern syndrome. CNS Spectrums 2021, 26 (1), 84–91. [CrossRef]
- Orsolini, L.; Chiappini, S.; Papanti, D.; De Berardis, D.; Corkery, J. M.; Schifano, F. The Bridge Between Classical and "Synthetic"/Chemical Psychoses: Towards a Clinical, Psychopathological, and Therapeutic Perspective. Frontiers in Psychiatry 2019, 10, 851. [CrossRef]
- Schifano, F.; Napoletano, F.; Chiappini, S.; Guirguis, A.; Corkery, J. M.; Bonaccorso, S.; Ricciardi, A.; Scherbaum, N.; Vento, A. New/emerging psychoactive substances and associated psychopathological consequences. Psychological Medicine 2021, 51 (1), 30–42. [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed., Text Rev.; American Psychiatric Publishing: Washington, DC, 2022.
- Ricci, V.; Ceci, F.; Di Carlo, F.; Lalli, A.; Ciavoni, L.; Mosca, A.; Sepede, G.; Salone, A.; Quattrone, D.; Fraticelli, S.; Maina, G.; Martinotti, G. Cannabis use disorder and dissociation: A report from a prospective first-episode psychosis study. Drug and Alcohol Dependence 2021, 229 (Pt A), 109118. [CrossRef]
- Schifano, F. Recent Changes in Drug Abuse Scenarios: The New/Novel Psychoactive Substances (NPS) Phenomenon. Brain Sciences 2018, 8 (12), 221. [CrossRef]
- European Monitoring Centre for Drugs and Drug Addiction (EMCDDA). Co-morbid substance use and mental disorders in Europe: A review of the data. 2013; pp. 1-12.
- Chetty, A.; Guse, T.; Malema, M. Integrated vs non-integrated treatment outcomes in dual diagnosis disorders: A systematic review. Health SA = SA Gesondheid 2023, 28, 2094. [CrossRef]
- Han, B.; Compton, W. M.; Blanco, C.; Colpe, L. J. Prevalence, Treatment, And Unmet Treatment Needs Of US Adults With Mental Health And Substance Use Disorders. Health Affairs (Project Hope) 2017, 36 (10), 1739–1747. [CrossRef]
- Hakobyan, S.; Vazirian, S.; Lee-Cheong, S.; Krausz, M.; Honer, W. G.; Schutz, C. G. Concurrent Disorder Management Guidelines. Systematic Review. Journal of Clinical Medicine 2020, 9 (8), 2406. [CrossRef]
- Arnau, F.; Benito, A.; Villar, M.; Ortega, M. E.; López-Peláez, L.; Haro, G. Addressing Dual Disorders in a Medium-Term Admission Unit. Brain Sciences 2021, 12 (1), 24. [CrossRef]
- Murthy, P.; Chand, P. Treatment of dual diagnosis disorders. Current Opinion in Psychiatry 2012, 25 (3), 194–200. [CrossRef]
- Murthy, P.; Mahadevan, J.; Chand, P. K. Treatment of substance use disorders with co-occurring severe mental health disorders. Current Opinion in Psychiatry 2019, 32 (4), 293–299. [CrossRef]
- Janiri, L.; Martinotti, G.; Di Nicola, M. Aripiprazole for relapse prevention and craving in alcohol-dependent subjects: results from a pilot study. Journal of Clinical Psychopharmacology 2007, 27 (5), 519–520. [CrossRef]
- Price, Jobe, L. B.; Mårtensson, S.; Düring, S. W. Polypharmacy in antipsychotic pharmacological treatment among patients with dual diagnosis in Denmark. Nordic Journal of Psychiatry 2023, 1-8. Advance online publication. [CrossRef]
- Temmingh, H. S.; Williams, T.; Siegfried, N.; Stein, D. J. Risperidone versus other antipsychotics for people with severe mental illness and co-occurring substance misuse. The Cochrane Database of Systematic Reviews 2018, 1 (1), CD011057. [CrossRef]
- Grau-López, L.; Roncero, C.; Daigre, C.; Miquel, L.; Barral, C.; Gonzalvo, B.; Collazos, F.; Casas, M. Observational study on medications prescribed to dual-diagnosis outpatients. Journal of Addiction Medicine 2014, 8 (2), 84–89. [CrossRef]
- Grau-López, L.; Szerman, N.; Torrens, M.; Basurte, I.; Vega, P.; Martínez-Raga, J.; Casas, M.; Roncero, C. Professional perception of clozapine use in patients with dual psychosis. Actas Espanolas de Psiquiatria 2020, 48 (3), 99–105.
- Martinotti, G.; Chiappini, S.; Mosca, A.; Miuli, A.; Santovito, M. C.; Pettorruso, M.; Skryabin, V.; Sensi, S. L.; Giannantonio, M. D. Atypical Antipsychotic Drugs in Dual Disorders: Current Evidence for Clinical Practice. Current Pharmaceutical Design 2022, 28 (27), 2241–2259. [CrossRef]
- Clerici, M.; de Bartolomeis, A.; De Filippis, S.; Ducci, G.; Maremmani, I.; Martinotti, G.; Schifano, F. Patterns of Management of Patients With Dual Disorder (Psychosis) in Italy: A Survey of Psychiatrists and Other Physicians Focusing on Clinical Practice. Frontiers in Psychiatry 2018, 9, 575. [CrossRef]
- Abdel-Baki, A.; Thibault, D.; Medrano, S.; Stip, E.; Ladouceur, M.; Tahir, R.; Potvin, S. Long-acting antipsychotic medication as first-line treatment of first-episode psychosis with comorbid substance use disorder. Early Intervention in Psychiatry 2020, 14 (1), 69–79. [CrossRef]
- Cipriani, A.; Geddes, J. Comparison of systematic and narrative reviews: the example of the atypical antipsychotics. Epidemiologia e Psichiatria Sociale 2003, 12 (3), 146-153. [CrossRef]
- Collins, J. A.; Fauser, B. C. Balancing the strengths of systematic and narrative reviews. Human Reproduction Update 2005, 11 (2), 103–104. [CrossRef]
- Lieberman J. A. Dopamine partial agonists: a new class of antipsychotic. CNS Drugs 2004, 18 (4), 251–267. [CrossRef]
- Orsolini, L.; Tomasetti, C.; Valchera, A.; Vecchiotti, R.; Matarazzo, I.; Vellante, F.; Iasevoli, F.; Buonaguro, E. F.; Fornaro, M.; Fiengo, A. L.; Martinotti, G.; Mazza, M.; Perna, G.; Carano, A.; De Bartolomeis, A.; Di Giannantonio, M.; De Berardis, D. An update of safety of clinically used atypical antipsychotics. Expert Opinion on Drug Safety 2016, 15 (10), 1329–1347. [CrossRef]
- Leite, J. V.; Guimarães, F. S.; Moreira, F. A. Aripiprazole, an atypical antipsychotic, prevents the motor hyperactivity induced by psychotomimetics and psychostimulants in mice. European Journal of Pharmacology 2008, 578 (2-3), 222–227. [CrossRef]
- Sørensen, G.; Sager, T. N.; Petersen, J. H.; Brennum, L. T.; Thøgersen, P.; Hee Bengtsen, C.; Thomsen, M.; Wörtwein, G.; Fink-Jensen, A.; Woldbye, D. P. Aripiprazole blocks acute self-administration of cocaine and is not self-administered in mice. Psychopharmacology 2008, 199 (1), 37–46. [CrossRef]
- Natesan, S.; Reckless, G. E.; Nobrega, J. N.; Fletcher, P. J.; Kapur, S. Dissociation between in vivo occupancy and functional antagonism of dopamine D2 receptors: comparing aripiprazole to other antipsychotics in animal models. Neuropsychopharmacology 2006, 31 (9), 1854–1863. [CrossRef]
- Rolland, B.; Geoffroy, P. A.; Jardri, R.; Cottencin, O. Aripiprazole for treating cannabis-induced psychotic symptoms in ultrahigh-risk individuals. Clinical Neuropharmacology 2013, 36 (3), 98–99. [CrossRef]
- Desseilles, M.; Mathot, F.; Desseilles, M. Aripiprazole diminishes cannabis use in schizophrenia. The Journal of Neuropsychiatry and Clinical Neurosciences 2008, 20 (1), 117–118. [CrossRef]
- Feltenstein, M. W.; Altar, C. A.; See, R. E. Aripiprazole blocks reinstatement of cocaine seeking in an animal model of relapse. Biological Psychiatry 2007, 61 (5), 582–590. [CrossRef]
- Thurstone, C.; Loh, R.; Foreman, K.; Thurstone, C. A.; Wolf, C. Program Evaluation to Aid Choice of Aripiprazole or Risperidone for Hospitalized Adolescents with Cannabis Use Disorder and Psychosis. Journal of Child and Adolescent Psychopharmacology 2023, 33 (8), 332–336. [CrossRef]
- Futamura, T.; Akiyama, S.; Sugino, H.; Forbes, A.; McQuade, R. D.; Kikuchi, T. Aripiprazole attenuates established behavioral sensitization induced by methamphetamine. Progress in Neuro-Psychopharmacology & Biological Psychiatry 2010, 34 (6), 1115–1119. [CrossRef]
- Farnia, V.; Shakeri, J.; Tatari, F.; Juibari, T. A.; Yazdchi, K.; Bajoghli, H.; Brand, S.; Abdoli, N.; Aghaei, A. Randomized controlled trial of aripiprazole versus risperidone for the treatment of amphetamine-induced psychosis. The American Journal of Drug and Alcohol Abuse 2014, 40 (1), 10–15. [CrossRef]
- Wang, G.; Zhang, Y.; Zhang, S.; Chen, H.; Xu, Z.; Schottenfeld, R. S.; Hao, W.; Chawarski, M. C. Aripiprazole and Risperidone for Treatment of Methamphetamine-Associated Psychosis in Chinese Patients. Journal of Substance Abuse Treatment 2016, 62, 84–88. [CrossRef]
- Skryabin, V. Y.; Vinnikova, M. A.; Ezhkova, E. V.; Titkov, M. S.; Bulatova, R. A. Atypical Antipsychotics in the Treatment of Patients with a Dual Diagnosis of Schizophrenia Spectrum Disorders and Substance Use Disorders: The Results of a Randomized Comparative Study. Journal of Addictive Diseases 2021, 39 (4), 513–525. [CrossRef]
- Beresford, T. P.; Clapp, L.; Martin, B.; Wiberg, J. L.; Alfers, J.; Beresford, H. F. Aripiprazole in schizophrenia with cocaine dependence: a pilot study. Journal of Clinical Psychopharmacology 2005, 25 (4), 363–366. [CrossRef]
- Sulaiman, A. H.; Gill, J. S.; Said, M. A.; Zainal, N. Z.; Hussein, H. M.; Guan, N. C. A randomized, placebo-controlled trial of aripiprazole for the treatment of methamphetamine dependence and associated psychosis. International Journal of Psychiatry in Clinical Practice 2013, 17 (2), 131–138. [CrossRef]
- Edinoff, A.; Ruoff, M. T.; Ghaffar, Y. T.; Rezayev, A.; Jani, D.; Kaye, A. M.; Cornett, E. M.; Kaye, A. D.; Viswanath, O.; Urits, I. Cariprazine to Treat Schizophrenia and Bipolar Disorder in Adults. Psychopharmacology Bulletin 2020, 50 (4), 83–117. [CrossRef]
- Stahl, S. M. Drugs for psychosis and mood: unique actions at D3, D2, and D1 dopamine receptor subtypes. CNS Spectrums 2017, 22 (5), 375–384. [CrossRef]
- Calabrese, F.; Tarazi, F. I.; Racagni, G.; Riva, M. A. The role of dopamine D3 receptors in the mechanism of action of cariprazine. CNS Spectrums 2020, 25 (3), 343–351. [CrossRef]
- Kiss, B.; Horváth, A.; Némethy, Z.; Schmidt, E.; Laszlovszky, I.; Bugovics, G.; Fazekas, K.; Hornok, K.; Orosz, S.; Gyertyán, I.; Agai-Csongor, E.; Domány, G.; Tihanyi, K.; Adham, N.; Szombathelyi, Z. Cariprazine (RGH-188), a dopamine D(3) receptor-preferring, D(3)/D(2) dopamine receptor antagonist-partial agonist antipsychotic candidate: in vitro and neurochemical profile. The Journal of Pharmacology and Experimental Therapeutics 2010, 333 (1), 328–340. [CrossRef]
- Kiss, B.; Krámos, B.; Laszlovszky, I. Potential Mechanisms for Why Not All Antipsychotics Are Able to Occupy Dopamine D3 Receptors in the Brain in vivo. Frontiers in Psychiatry 2022, 13, 785592. [CrossRef]
- Wesołowska, A.; Partyka, A.; Jastrzębska-Więsek, M.; Kołaczkowski, M. The preclinical discovery and development of cariprazine for the treatment of schizophrenia. Expert Opinion on Drug Discovery 2018, 13 (8), 779–790. [CrossRef]
- Legros, C.; Rojas, A.; Dupré, C.; Brasseur, C.; Riest-Fery, I.; Muller, O.; Ortuno, J. C.; Nosjean, O.; Guenin, S. P.; Ferry, G.; Boutin, J. A. Approach to the specificity and selectivity between D2 and D3 receptors by mutagenesis and binding experiments part I: Expression and characterization of D2 and D3 receptor mutants. Protein Science: A Publication of the Protein Society 2022, 31 (12), e4459. [CrossRef]
- Legg, D. R. K.; Dubroff, J. G.; Labban, K. J.; Mach, R. H. Selectivity of probes for PET imaging of dopamine D3 receptors. Neuroscience Letters 2019, 691, 18–25. [CrossRef]
- Lacroix, Weerasinghe, D. K.; Hodge, J. M.; Pasco, J. A.; Samarasinghe, R. M.; Azimi Manavi, B.; Williams, L. J. Antipsychotic-induced bone loss: the role of dopamine, serotonin and adrenergic receptor signalling. Frontiers in Cell and Developmental Biology 2023, 11, 1184550. [CrossRef]
- Huang, M.; Panos, J. J.; Kwon, S.; Oyamada, Y.; Rajagopal, L.; Meltzer, H. Y. Comparative effect of lurasidone and blonanserin on cortical glutamate, dopamine, and acetylcholine efflux: role of relative serotonin (5-HT)2A and DA D2 antagonism and 5-HT1A partial agonism. Journal of Neurochemistry 2014, 128 (6), 938–949. [CrossRef]
- Girgis, R. R.; Slifstein, M.; D'Souza, D.; Lee, Y.; Periclou, A.; Ghahramani, P.; Laszlovszky, I.; Durgam, S.; Adham, N.; Nabulsi, N.; Huang, Y.; Carson, R. E.; Kiss, B.; Kapás, M.; Abi-Dargham, A.; Rakhit, A. Preferential binding to dopamine D3 over D2 receptors by cariprazine in patients with schizophrenia using PET with the D3/D2 receptor ligand [(11)C]-(+)-PHNO. Psychopharmacology 2016, 233 (19-20), 3503–3512. [CrossRef]
- Huang, M.; He, W.; Kiss, B.; Farkas, B.; Adham, N.; Meltzer, H. Y. The Role of Dopamine D3 Receptor Partial Agonism in Cariprazine-Induced Neurotransmitter Efflux in Rat Hippocampus and Nucleus Accumbens. The Journal of Pharmacology and Experimental Therapeutics 2019, 371 (2), 517–525. [CrossRef]
- Yun, S.; Yang, B.; Anair, J. D.; Martin, M. M.; Fleps, S. W.; Pamukcu, A.; Yeh, N. H.; Contractor, A.; Kennedy, A.; Parker, J. G. Antipsychotic drug efficacy correlates with the modulation of D1 rather than D2 receptor-expressing striatal projection neurons. Nature Neuroscience 2023, 26 (8), 1417–1428. [CrossRef]
- Sanson, A.; Riva, M. A. Anti-Stress Properties of Atypical Antipsychotics. Pharmaceuticals (Basel, Switzerland) 2020, 13 (10), 322. [CrossRef]
- Németh, G.; Laszlovszky, I.; Czobor, P.; Szalai, E.; Szatmári, B.; Harsányi, J.; Barabássy, Á.; Debelle, M.; Durgam, S.; Bitter, I.; Marder, S.; Fleischhacker, W. W. Cariprazine versus risperidone monotherapy for treatment of predominant negative symptoms in patients with schizophrenia: a randomised, double-blind, controlled trial. Lancet (London, England) 2017, 389 (10074), 1103–1113. [CrossRef]
- Román, V.; Gyertyán, I.; Sághy, K.; Kiss, B.; Szombathelyi, Z. Cariprazine (RGH-188), a D₃-preferring dopamine D₃/D₂ receptor partial agonist antipsychotic candidate demonstrates anti-abuse potential in rats. Psychopharmacology 2013, 226 (2), 285–293. [CrossRef]
- Jordan, C. J.; He, Y.; Bi, G. H.; You, Z. B.; Cao, J.; Xi, Z. X.; Newman, A. H. (±)VK4-40, a novel dopamine D3 receptor partial agonist, attenuates cocaine reward and relapse in rodents. British Journal of Pharmacology 2020, 177 (20), 4796–4807. [CrossRef]
- Rodriguez Cruz, J.; Sahlsten Schölin, J.; Hjorth, S. Case Report: Cariprazine in a Patient With Schizophrenia, Substance Abuse, and Cognitive Dysfunction. Frontiers in Psychiatry 2021, 12, 727666. [CrossRef]
- Ricci V, Di Salvo G, Maina G. Remission of persistent methamphetamine-induced psychosis after cariprazine therapy: presentation of a case report. J Addict Dis 2022, 40(1), 145-148. [CrossRef]
- Truong, T. T.; Li, B. Case Series: Cariprazine for treatment of methamphetamine use disorder. The American Journal on Addictions 2022, 31(1), 85–88. [CrossRef]
- Montes, J. M.; Montes, P.; Hernández-Huerta, D. Cariprazine in Three Acute Patients with Schizophrenia: A Real-World Experience. Neuropsychiatric Disease and Treatment 2021, 17, 291–296. [CrossRef]
- Vannucchi, T.; Taddeucci, C.; Tatini, L. Case Report: Functional and Symptomatic Improvement With Cariprazine in Various Psychiatric Patients: A Case Series. Frontiers in Psychiatry 2022, 13, 878889. [CrossRef]
- Gentile, A.; Marini, S.; Matarazzo, I.; De Berardis, D.; Ventriglio, A. Cariprazine in the treatment of psychosis with comorbid cannabis use: A case report (2022). Psychiatry Research Communications 2022, 2 (2), art. no. 100048. [CrossRef]
- Maeda, K.; Sugino, H.; Akazawa, H.; Amada, N.; Shimada, J.; Futamura, T.; Yamashita, H.; Ito, N.; McQuade, R. D.; Mørk, A.; Pehrson, A. L.; Hentzer, M.; Nielsen, V.; Bundgaard, C.; Arnt, J.; Stensbøl, T. B.; Kikuchi, T. Brexpiprazole I: in vitro and in vivo characterization of a novel serotonin-dopamine activity modulator. The Journal of Pharmacology and Experimental Therapeutics 2014, 350 (3), 589–604. [CrossRef]
- Maeda, K.; Lerdrup, L.; Sugino, H.; Akazawa, H.; Amada, N.; McQuade, R. D.; Stensbøl, T. B.; Bundgaard, C.; Arnt, J.; Kikuchi, T. Brexpiprazole II: antipsychotic-like and procognitive effects of a novel serotonin-dopamine activity modulator. The Journal of Pharmacology and Experimental Therapeutics 2014, 350 (3), 605–614. [CrossRef]
- Stahl, S. M. Mechanism of action of brexpiprazole: comparison with aripiprazole. CNS Spectrums 2016, 21 (1), 1–6. [CrossRef]
- Edinoff, A. N.; Wu, N. W.; Maxey, B. S.; Ren, A. L.; Leethy, K. N.; Girma, B.; Odisho, A.; Kaye, J. S.; Kaye, A. J.; Kaye, A. M.; Kaye, A. D.; Mychaskiw, G.; Viswanath, O.; Urits, I. Brexpiprazole for the Treatment of Schizophrenia and Major Depressive Disorder: A Comprehensive Review of Pharmacological Considerations in Clinical Practice. Psychopharmacology Bulletin 2021, 51 (2), 69–95.
- Montejo, Brand, B. A.; Willemse, E. J. M.; Hamers, I. M. H.; Sommer, I. E. Evidence-Based Recommendations for the Pharmacological Treatment of Women with Schizophrenia Spectrum Disorders. Current Psychiatry Reports 2023, 25 (11), 723–733. [CrossRef]
- Mauri, M. C.; Paletta, S.; Di Pace, C.; Reggiori, A.; Cirnigliaro, G.; Valli, I.; et al. Clinical Pharmacokinetics of Atypical Antipsychotics: An Update. Clinical Pharmacokinetics 2018, 57. Springer International Publishing. [Internet].
- Das, S.; Barnwal, P.; Winston, A. B.; Mondal, S.; Saha, I. Brexpiprazole: so far so good. Therapeutic Advances in Psychopharmacology 2016, 6(1), 39–54. [CrossRef]
- Correll, C. U.; Skuban, A.; Ouyang, J.; Hobart, M.; Pfister, S.; McQuade, R. D.; Nyilas, M.; Carson, W. H.; Sanchez, R.; Eriksson, H. Efficacy and safety of brexpiprazole for the treatment of acute schizophrenia: a 6-week randomized, double-blind, placebo-controlled trial. Am J Psychiatry 2015, 172, 870–880. [CrossRef]
- Kane, J. M.; Skuban, A.; Ouyang, J.; Hobart, M.; Pfister, S.; McQuade, R. D.; Nyilas, M.; Carson, W. H.; Sanchez, R.; Eriksson, H. A multicenter, randomized, double-blind, controlled phase 3 trial of fixed-dose brexpiprazole for the treatment of adults with acute schizophrenia. Schizophr Res 2015, 164, 127–135. [CrossRef]
- Fleischhacker, W. W.; Hobart, M.; Ouyang, J.; Forbes, A.; Pfister, S.; McQuade, R. D.; Carson, W. H.; Sanchez, R.; Nyilas, M.; Weiller, E. Efficacy and safety of brexpiprazole (OPC-34712) as maintenance treatment in adults with schizophrenia: a randomized, double-blind, placebo-controlled study. Int J Neuropsychopharmacol 2017, 20, 11–21. [CrossRef]
- Siwek, M.; Wojtasik-Bakalarz, K.; Krupa, A. J.; Chrobak, A. A. Brexpiprazole-Pharmacologic Properties and Use in Schizophrenia and Mood Disorders. Brain Sciences 2023, 13 (3), 397. [CrossRef]
- Nickols, J. E. R.; Dursun, S. M.; Taylor, A. M. W. Preclinical evidence for the use of the atypical antipsychotic, brexpiprazole, for opioid use disorder. Neuropharmacology 2023, 233, 109546. [CrossRef]
- Kung, F. H.; Lin, H. Y.; Tai, Y. M.; et al. Brexpiprazole in the Treatment of Cannabis Withdrawal Psychotic Disorder. Am J Ther 2022, 29(4), 492-493. [CrossRef]
- Ishibashi, T.; Horisawa, T.; Tokuda, K.; Ishiyama, T.; Ogasa, M.; Tagashira, R.; Matsumoto, K.; Nishikawa, H.; Ueda, Y.; Toma, S.; Oki, H.; Tanno, N.; Saji, I.; Ito, A.; Ohno, Y.; Nakamura, M. Pharmacological profile of lurasidone, a novel antipsychotic agent with potent 5-hydroxytryptamine 7 (5-HT7) and 5-HT1A receptor activity. J Pharmacol Exp Ther 2010, 334(1), 171–181. [CrossRef]
- Miura, I.; Horikoshi, S.; Ichinose, M.; Suzuki, Y.; Watanabe, K. Lurasidone for the Treatment of Schizophrenia: Design, Development, and Place in Therapy. Drug Design, Development and Therapy 2023, 17, 3023–3031. [CrossRef]
- Sunovion. Latuda (Lurasidone HCl) Tablets: Highlights of Prescribing Information. Sunovion, 2013. http://www.latuda.com/LatudaPrescribingInformation.pdf.
- European Medicines Agency. Latuda: EPAR - Product Information. Annex I: Summary of Product Characteristics. EMA, 2014. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/002713/WC500164683.pdf.
- Preskorn, S.; Ereshefsky, L.; Chiu, Y. Y.; Poola, N.; Loebel, A. Effect of food on the pharmacokinetics of lurasidone: results of two randomized, open-label, crossover studies. Human psychopharmacology 2013, 28 (5), 495–505. [CrossRef]
- Chiu, Y. Y.; Ereshefsky, L.; Preskorn, S. H.; Poola, N.; Loebel, A. Lurasidone drug-drug interaction studies: a comprehensive review. Drug metabolism and drug interactions 2014, 29 (3), 191–202. [CrossRef]
- Sanford, M. Lurasidone: In the Treatment of Schizophrenia. CNS Drugs 2013, 27, 67–80.
- Ng-Mak, D.; Tongbram, V.; Ndirangu, K.; Rajagopalan, K.; Loebel, A. Efficacy and Metabolic Effects of Lurasidone Versus Brexpiprazole in Schizophrenia: A Network Meta-Analysis. J. Comp. Eff. Res. 2018. [CrossRef]
- Corponi, F.; Fabbri, C.; Bitter, I.; Montgomery, S.; Vieta, E.; Kasper, S.; Pallanti, S.; Serretti, A. Novel Antipsychotics Specificity Profile: A Clinically Oriented Review of Lurasidone, Brexpiprazole, Cariprazine, and Lumateperone. Eur. Neuropsychopharmacol. 2019, 29 (9), 971–985. [CrossRef]
- Zheng W, Cai DB, Yang XH, et al. Short-term efficacy and tolerability of lurasidone in the treatment of acute schizophrenia: a meta-analysis of randomized controlled trials. J Psychiatr Res 2018, 103, 244–251. [CrossRef]
- Citrome L, Cucchiaro J, Sarma K, et al. Long-term safety and tolerability of lurasidone in schizophrenia: a 12-month, double-blind, active-controlled study. Int Clin Psychopharmacol 2012, 27(3), 165–176. [CrossRef]
- Stahl SM, Cucchiaro J, Simonelli D, Hsu J, Pikalov A, Loebel A. Effectiveness of lurasidone for patients with schizophrenia following 6 weeks of acute treatment with lurasidone, olanzapine, or placebo: a 6-month, open-label, extension study. J Clin Psychiatry 2013, 74 (05), 507–515. [CrossRef]
- Wei, Y. M.; Wang, X. J.; Yang, X. D.; Wang, C. S.; Wang, L. L.; Xu, X. Y.; Zhao, G. J.; Li, B.; Zhu, D. M.; Wu, Q.; Shen, Y. F. Safety and effectiveness of lurasidone in the treatment of Chinese schizophrenia patients: An interim analysis of post-marketing surveillance. World journal of psychiatry 2023, 13(11), 937–948. [CrossRef]
- Correll CU, Cucchiaro J, Silva R, Hsu J. Long-term safety and effectiveness of lurasidone in schizophrenia: a 22-month, open-label extension study. CNS Spectr 2016, 21(5), 393–402. [CrossRef]
- Higuchi T, Ishigooka J, Iyo M, Hagi K. Safety and effectiveness of lurasidone for the treatment of schizophrenia in Asian patients: results of a 26-week open-label extension study. Asia Pac Psychiatry 2020, 12(1), e12377. [CrossRef]
- Ricci, V.; Martinotti, G.; De Berardis, D.; Maina, G. Lurasidone use in Cannabis-Induced Psychosis: A Novel Therapeutic Strategy and Clinical Considerations in Four Cases Report. International journal of environmental research and public health 2022, 19(23), 16057. [CrossRef]
- Mole, T. B.; Furlong, Y.; Clarke, R. J.; Rao, P.; Moore, J. K.; Pace, G.; Van Odyck, H.; Chen, W. Lurasidone for Adolescents With Complex Mental Disorders: A Case Series. Journal of pharmacy practice 2022, 35(5), 800–804. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



