Submitted:
19 January 2024
Posted:
22 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. High Pressure Liquid Chromatography (HPLC)
2.3. Selection of Oils for FT-NE Development
2.4. Preparation of FT-NE Formulations
2.5. FT Solution (FT-S) Formulation
2.6. Measurement of Droplet Size, Zeta Potential (ZP), and Polydispersity Index (PDI)
2.7. Drug Content
2.9. Drug-Excipient Compatibility
3. Scanning Transmission Electron Microscope (STEM) Imaging
3.1. Sterilization and Stability Studies
3.2. In vitro Drug Release
4. Results and Discussion
4.1. Solubility in Selected Oils
4.2. FT-NE Formulation Development
4.3. Measurement of Droplet Size, PDI and ZP
4.4. pH of the FT-NE Formulations
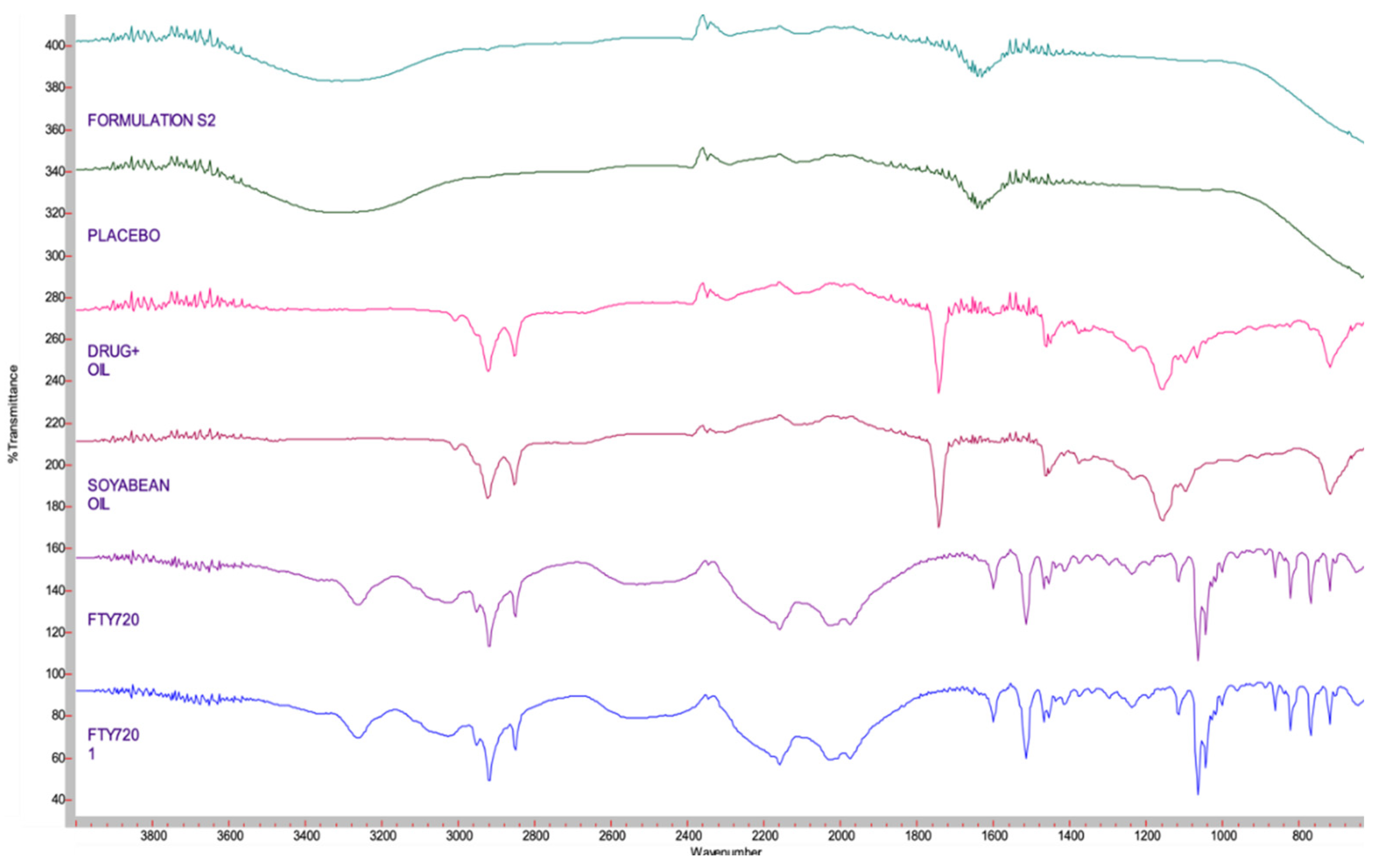
4.5. Compatibility Studies - FTIR
4.6. Scanning Transmission Electronic Microscopy (STEM) Images
4.7. Sterilization and Stability Studies
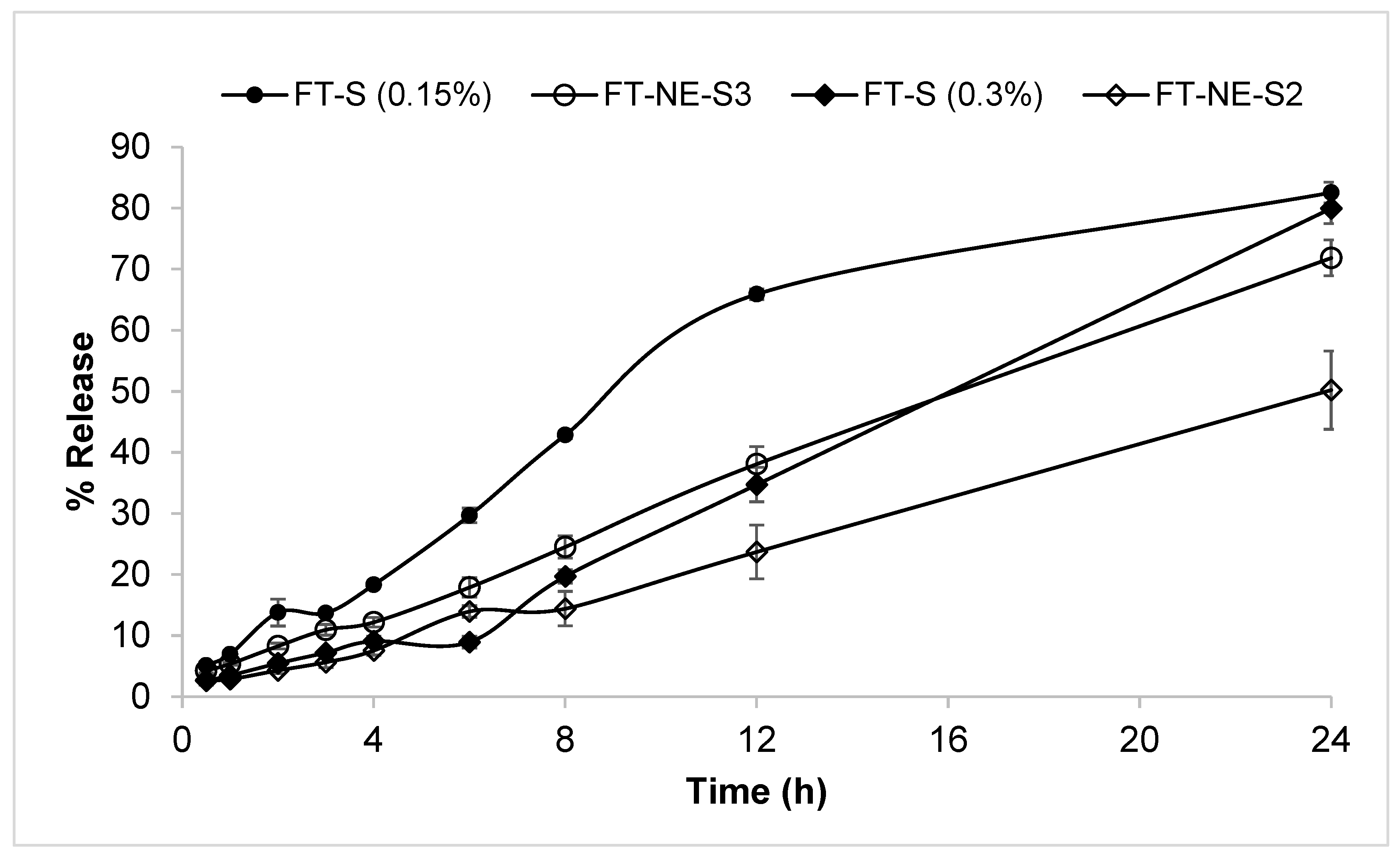
5. In Vitro Release Studies
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Patel, A., Cholkar, K., Agrahari, V., andMitra, A. K. (2013) Ocular drug delivery systems: An overview World J Pharmacol 2, 47-64. [CrossRef]
- Yang, Y., andLockwood, A. (2022) Topical ocular drug delivery systems: Innovations for an unmet need Experimental Eye Research 218, 109006. [CrossRef]
- Jumelle, C., Gholizadeh, S., Annabi, N., andDana, R. (2020) Advances and limitations of drug delivery systems formulated as eye drops Journal of Controlled Release 321, 1-22.
- Campbell, M., andHumphries, P. (2013) The blood-retina barrier: tight junctions and barrier modulation Biology and regulation of blood-tissue barriers 70-84.
- Varela-Fernández, R., Díaz-Tomé, V., Luaces-Rodríguez, A., Conde-Penedo, A., García-Otero, X., Luzardo-Álvarez, A. et al. (2020) Drug delivery to the posterior segment of the eye: biopharmaceutic and pharmacokinetic considerations Pharmaceutics 12, 269.
- Ahmed, S., Amin, M. M., and Sayed, S. (2023) Ocular Drug Delivery: a Comprehensive Review AAPS PharmSciTech 24, 66. [CrossRef]
- Elsayed, I., andSayed, S. (2017) Tailored nanostructured platforms for boosting transcorneal permeation: box–Behnken statistical optimization, comprehensive in vitro, ex vivo and in vivo characterization International Journal of Nanomedicine 12, 7947.
- Kang-Mieler, J. J., Rudeen, K. M., Liu, W., andMieler, W. F. (2020) Advances in ocular drug delivery systems Eye 34, 1371-1379. [CrossRef]
- Kang-Mieler, J. J., Osswald, C. R., andMieler, W. F. (2014) Advances in ocular drug delivery: emphasis on the posterior segment Expert opinion on drug delivery 11, 1647-1660.
- Maulvi, F. A., Shetty, K. H., Desai, D. T., Shah, D. O., andWillcox, M. D. P. (2021) Recent advances in ophthalmic preparations: Ocular barriers, dosage forms and routes of administration International Journal of Pharmaceutics 608, 121105. [CrossRef]
- Liu, S., Dozois, M. D., Chang, C. N., Ahmad, A., Ng, D. L. T., Hileeto, D. et al. (2016) Prolonged Ocular Retention of Mucoadhesive Nanoparticle Eye Drop Formulation Enables Treatment of Eye Diseases Using Significantly Reduced Dosage Molecular Pharmaceutics 13, 2897-2905. [CrossRef]
- Dhahir, R. K., Al-Nima, A. M., andAl-Bazzaz, F. Y. (2021) Nanoemulsions as Ophthalmic Drug Delivery Systems Turk J Pharm Sci 18, 652-664. [CrossRef]
- Ammar, H. O., Salama, H. A., Ghorab, M., andMahmoud, A. A. (2009) Nanoemulsion as a potential ophthalmic delivery system for dorzolamide hydrochloride AAPS PharmSciTech 10, 808-819. [CrossRef]
- Gupta, A. (2020) Nanoemulsions In Nanoparticles for Biomedical Applications, Elsevier, 371-384.
- Youssef AA, Cai C, Dudhipala N, Majumdar S. Design of topical ocular ciprofloxacin nanoemulsion for the management of bacterial keratitis. Pharmaceuticals. 2021 Mar 3;14(3):210.
- Gawin-Mikołajewicz, A., Nartowski, K. P., Dyba, A. J., Gołkowska, A. M., Malec, K., andKarolewicz, B. (2021) Ophthalmic Nanoemulsions: From Composition to Technological Processes and Quality Control Mol Pharm 18, 3719-3740. [CrossRef]
- Senapati S, Youssef AA, Sweeney C, Cai C, Dudhipala N, Majumdar S. Cannabidiol Loaded Topical Ophthalmic Nanoemulsion Lowers Intraocular Pressure in Normotensive Dutch-Belted Rabbits. Pharmaceutics. 2022 Nov 24;14(12):2585.
- Sanford, M. (2014) Fingolimod: A Review of Its Use in Relapsing-Remitting Multiple Sclerosis Drugs 74, 1411-1433. [CrossRef]
- Qi, H., Cole, J., 2nd, Grambergs, R. C., Gillenwater, J. R., Mondal, K., Khanam, S. et al. (2019) Sphingosine Kinase 2 Phosphorylation of FTY720 is Unnecessary for Prevention of Light-Induced Retinal Damage Sci Rep 9, 7771. [CrossRef]
- Aktas, O., Küry, P., Kieseier, B., andHartung, H.-P. (2010) Fingolimod is a potential novel therapy for multiple sclerosis Nature Reviews Neurology 6, 373-382,.
- Masoudipour, E., Kashanian, S., Maleki, N., Karamyan, A., andOmidfar, K. (2018) A novel intracellular pH-responsive formulation for FTY720 based on PEGylated graphene oxide nano-sheets Drug Development and Industrial Pharmacy 44, 99-108. [CrossRef]
- Mehling, M., Johnson, T., Antel, J., Kappos, L., andBar-Or, A. (2011) Clinical immunology of the sphingosine 1-phosphate receptor modulator fingolimod (FTY720) in multiple sclerosis Neurology 76, S20-S27.
- Yang, T., Zha, Z., Yang, X., Kang, Y., Wang, X., Tong, Y. et al. (2021) Neuroprotective Effects of Fingolimod Supplement on the Retina and Optic Nerve in the Mouse Model of Experimental Autoimmune Encephalomyelitis Front Neurosci 15, 663541. [CrossRef]
- Chen, H., Tran, J.-T. A., Eckerd, A., Huynh, T.-P., Elliott, M. H., Brush, R. S. et al. (2013) Inhibition of de novo ceramide biosynthesis by FTY720 protects rat retina from light-induced degeneration Journal of Lipid Research 54, 1616-1629. [CrossRef]
- Stiles, M., Qi, H., Sun, E., Tan, J., Porter, H., Allegood, J. et al. (2016) Sphingolipid profile alters in retinal dystrophic P23H-1 rats and systemic FTY720 can delay retinal degeneration[S] Journal of Lipid Research 57, 818-831.
- Souri, E., Zargarpoor, M., Mottaghi, S., Ahmadkhaniha, R., andKebriaeezadeh, A. (2015) A Stability-Indicating HPLC Method for the Determination of Fingolimod in Pharmaceutical Dosage Forms Sci Pharm 83, 85-93. [CrossRef]
- 52.
- Joshi PH, Youssef AA, Ghonge M, Varner C, Tripathi S, Dudhipala N, Majumdar S. Gatifloxacin Loaded Nano Lipid Carriers for the Management of Bacterial Conjunctivitis. Antibiotics. 2023 Aug 15;12(8):1318.
- Agarwal, P., Craig, J. P., andRupenthal, I. D. (2021) Formulation Considerations for the Management of Dry Eye Disease Pharmaceutics 13. [CrossRef]
- Usón, N., Garcia, M. J., andSolans, C. (2004) Formation of water-in-oil (W/O) nanoemulsions in a water/mixed non-ionic surfactant/oil systems prepared by a low-energy emulsification method Colloids and Surfaces A: Physicochemical and Engineering Aspects 250, 415-421. [CrossRef]
- Sakeena, M. H., Elrashid, S. M., Munavvar, A. S., andAzmin, M. N. (2011) Effects of oil and drug concentrations on droplets size of palm oil esters (POEs) nanoemulsion J Oleo Sci 60, 155-158. [CrossRef]
- Sarheed, O., Dibi, M., andRamesh, K. (2020) Studies on the Effect of Oil and Surfactant on the Formation of Alginate-Based O/W Lidocaine Nanocarriers Using Nanoemulsion Template Pharmaceutics 12. [CrossRef]
- Sweeney, C., Dudhipala, N., Thakkar, R., Mehraj, T., Marathe, S., Gul, W. et al. (2021) Effect of surfactant concentration and sterilization process on intraocular pressure-lowering activity of Δ(9)-tetrahydrocannabinol-valine-hemisuccinate (NB1111) nanoemulsions Drug Deliv Transl Res 11, 2096-2107. [CrossRef]

| Oil | Solubility of FT (µg/mL) |
|---|---|
| Castor Oil | 3.9 ± 0.07 |
| Soybean Oil | 13.8 ± 0.03 |
| Cottonseed Oil | 0.3 ± 0.002 |
| Sesame Oil | 0.3 ± 0.008 |
| Ingredients (%w/v) | Formulation composition | |
|---|---|---|
| FT-NE-S2 | FT-NE-S3 | |
| Soybean oil | 5.0 | 5.0 |
| Fingolimod HCl | 0.3 | 0.15 |
| d-α-tocopheryl succinate | 0.002 | 0.002 |
| Tween 80 | 2 | 2 |
| Poloxamer 188 | 0.2 | 0.2 |
| Glycerin | 2.25 | 2.25 |
| Water (mL) | QS 10ml | QS 10ml |
| Parameter | Physicochemical characteristics (mean ± SD, n = 3) | |
| Droplet size (nm) | 195.3±3.6 | 181.1±1.1 |
| PDI | 0.24±0.02 | 0.24±0.02 |
| ZP (mV) | 53.8±1.4 | 41.4±1.7 |
| Assay (%) | 104.5±2.8 | 104.6±4.1 |
| pH* | 6.5 ± 0.5 | 6.5 ± 0.5 |
| Time (day) | Droplet size (nm) | PDI | ZP (mV) | Assay (%) |
|---|---|---|---|---|
| Pre-filtration | ||||
| Initial | 186.3±2.9 | 0.25±0.01 | 50.6±0.6 | 102.0±7.3 |
| Post-filtration at 4 oC | ||||
| Initial | 169.8±1.2 | 0.23±0.002 | 47.7±2.8 | 100.9±3.0 |
| 30 | 167.5±2.4 | 0.24±0.05 | 36.6±1.9 | 108.9±3.1 |
| 60 | 166.3±3.2 | 0.22±0.6 | 35.8±1.7 | 104.9±6.2 |
| 90 | 165.8±2.3 | 0.21±0.01 | 34.1±0.45 | 100.3±3.5 |
| 180 | 167.1±2.8 | 0.24±0.01 | 44.6±1.9 | 94.4±3.7 |
| Post-filtration at 25 oC | ||||
| Initial | 166.4±2.5 | 0.25±0.01 | 39.9±0.1 | 109.5±8.5 |
| 30 | 167.3±1.3 | 0.23±0.01 | 30.6±2.3 | 101.2±2.5 |
| Post-filtration at 40 oC | ||||
| Initial | 162.2±0.3 | 0.23±0.01 | 35.8±0.7 | 100.3±3.5 |
| 30 | 160.7±1.3 | 0.26±0.07 | 27.6±2 | 80.3± 8.2 |
| Time (day) | Droplet size (nm) | PDI | ZP (mV) | Assay (%) |
|---|---|---|---|---|
| Pre-filtration | ||||
| Initial | 181.8±1.7 | 0.22±0.01 | 35.2±0.5 | 112.3±0.5 |
| Post-filtration at 4 oC | ||||
| Initial | 177.9±1.4 | 0.24±0.02 | 37.8±0.4 | 112.8±0.7 |
| 30 | 175.8±1.8 | 0.23±0.02 | 33.4±0.8 | 102.2±6.5 |
| 60 | 177.8±1.2 | 0.19±0.001 | 31.6±0.6 | 108.3±4.0 |
| 90 | 171.0±0.6 | 0.22±0.005 | 36.1±1.2 | 103.2±3.6 |
| 180 | 172.3±1.4 | 0.25±0.007 | 37.1±0.2 | 100.4± 3.3 |
| Post-filtration at 25 oC | ||||
| Initial | 174.9±1.8 | 0.22±0.01 | 34.0±1.3 | 107.1±5.1 |
| 30 | 173.2±0.9 | 0.23±0.04 | 27.6±2.3 | 108.2±2.4 |
| Post-filtration at 40 oC | ||||
| Initial | 171.9±1.6 | 0.23±0.01 | 32.3±0.7 | 80.6± 9.0 |
| 30 | 169.4±1.7 | 0.24±0.04 | 23.6±1.4 | 80.14±2.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




