Submitted:
15 January 2024
Posted:
16 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Experimental animals
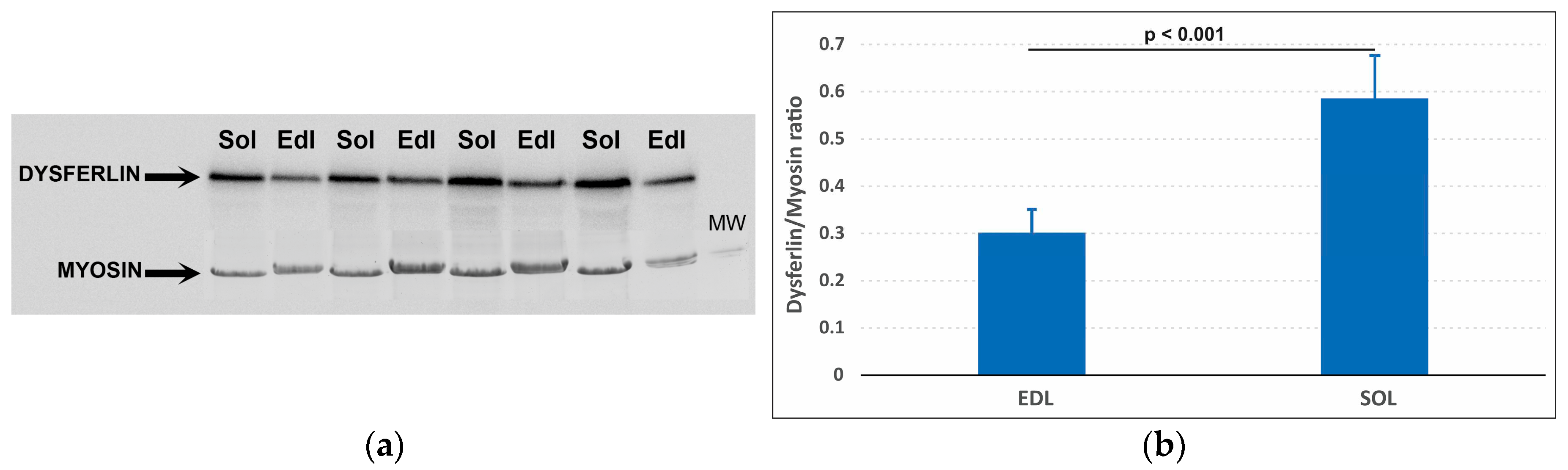
2.2. Western blotting
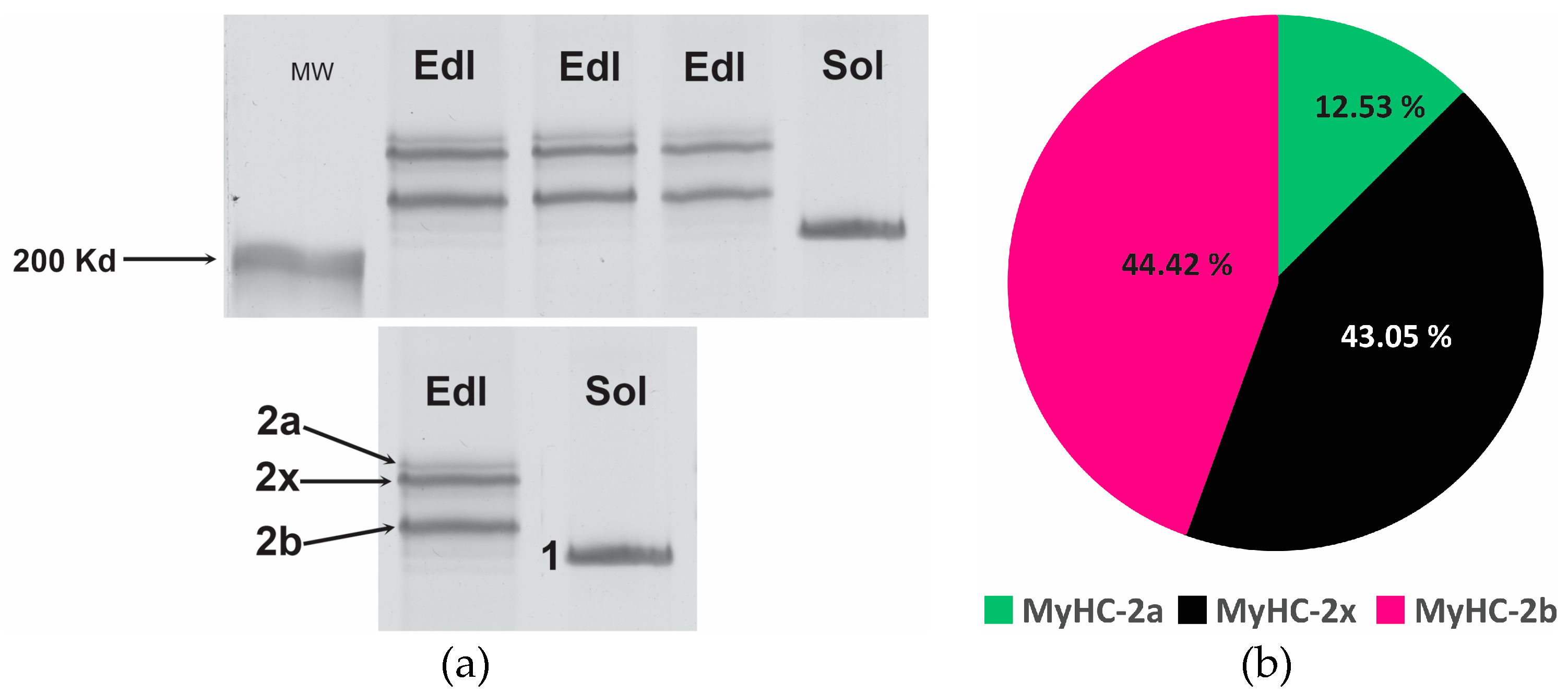
2.3. PAGE of MyHC isoforms
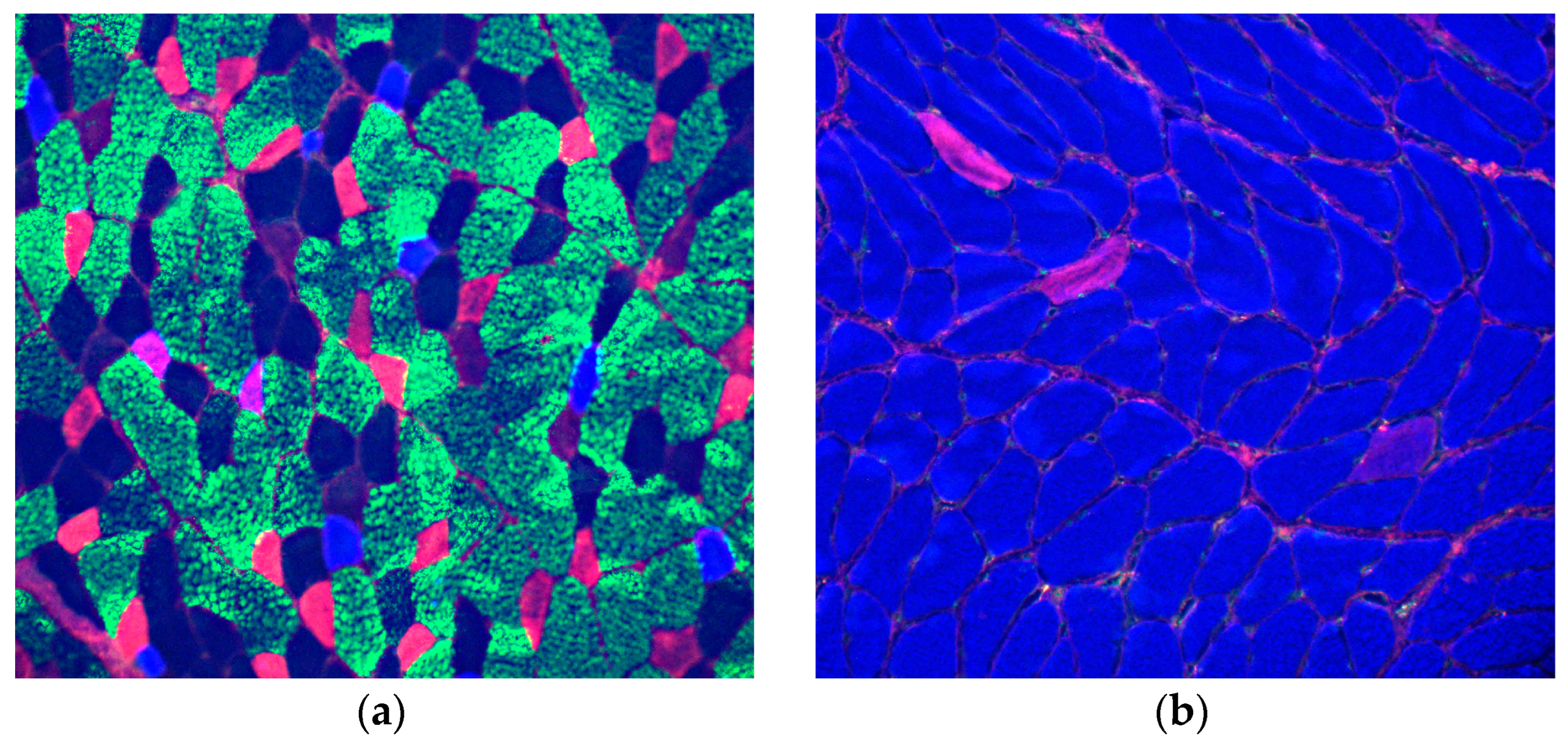
2.4. Immunofluorescence of MyHC isoforms
2.5. Image analysis
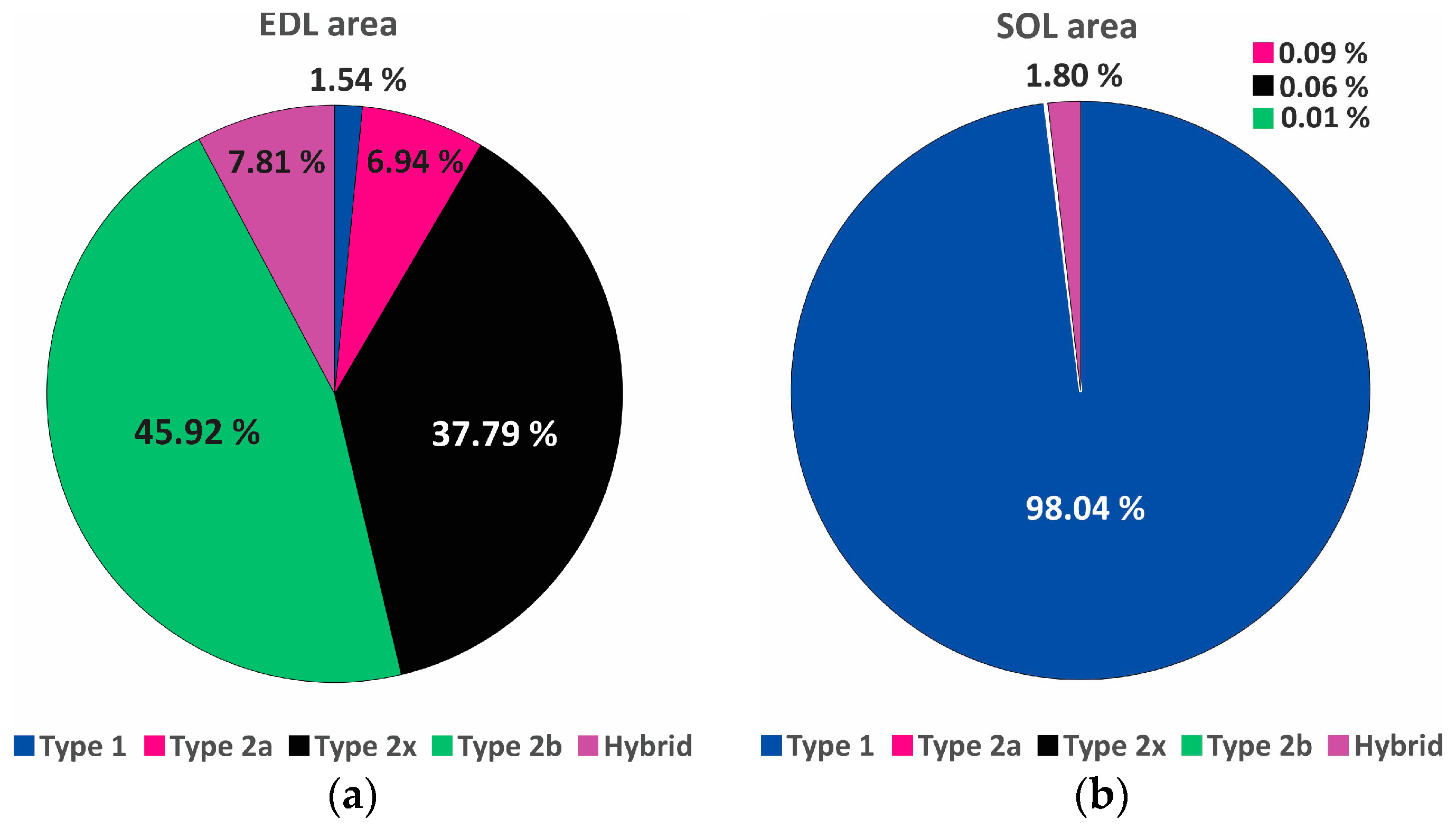
2.6. Histochemistry
2.7. Statistical analysis
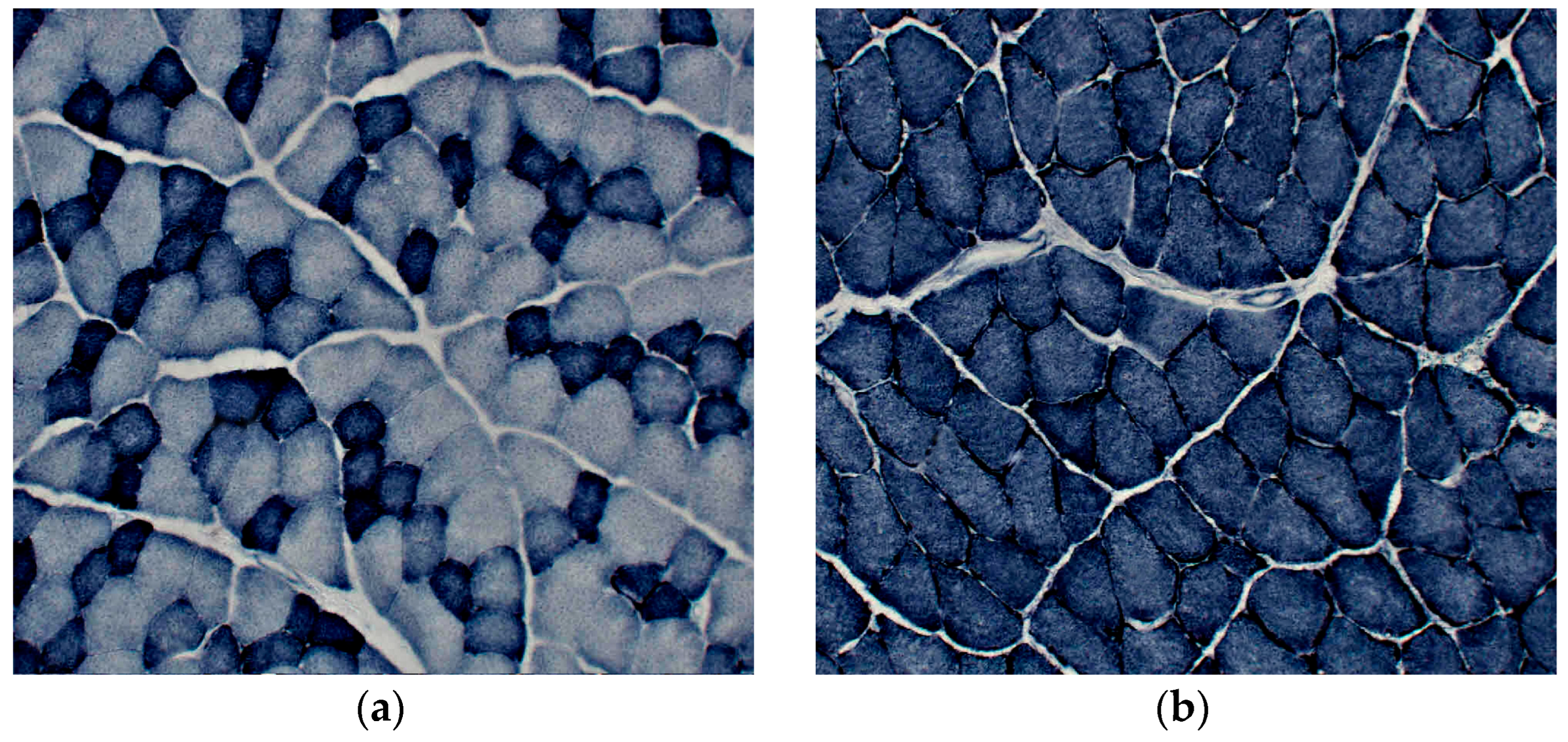
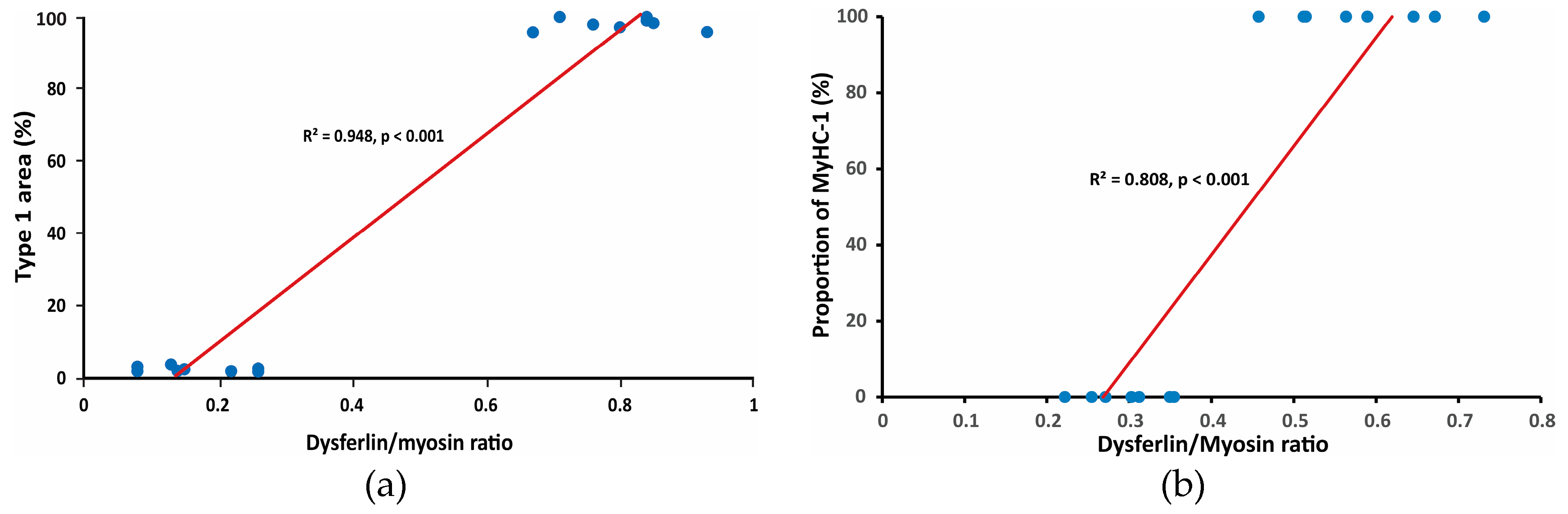
3. Results
3.1. Dysferlin Western blotting
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Posey, A.D.; Demonbreun A.; McNally E.M. Ferlin proteins in myoblast fusion and muscle growth. Curr Top Dev Biol 2011, 96, 203-230. [CrossRef]
- Schiaffino, S.; Reggiani, C. Fiber types in mammalian skeletal muscles. Physiol Rev 2011, 91, 1447-1531. [CrossRef]
- Schiaffino, S.; Muscle fiber type diversity revealed by anti-myosin heavy chain antibodies. FEBS J 2018, 285, 3688-3694. [CrossRef]
- Edström, L.; Kugelberg E. Histochemical composition, distribution of fibres and fatiguability of single motor units. Anterior tibial muscle of the rat. J Neurol Neurosurg Psychiatry 1968, 31, 424-433. [CrossRef]
- Close, R. Properties of motor units in fast and slow skeletal muscles of the rat. J Physiol 1967, 193, 45-55. [CrossRef]
- Schiaffino, S.; Hanzlíková V.; Pierobon S. Relations between structure and function in rat skeletal muscle fibers. J Cell Biol 1970, 47, 107-119. [CrossRef]
- Murgia, M.; Nogara, L.; Baraldo, M.; Reggiani, C.; Mann, M.; Schiaffino, S. Protein profile of fiber types in human skeletal muscle: a single-fiber proteomics study. Skelet Muscle 2021, 11, 24. [CrossRef]
- Fanin, M.; Angelini, C. Progress and challenges in diagnosis of dysferlinopathy. Muscle Nerve 2016, 54, 821-835.
- Fanin, M.; Nascimbeni, A.C.; Fulizio, L.; Trevisan, C.P.; Meznaric-Petrusa, M., Angelini, C. Loss of calpain-3 autocatalytic activity in LGMD2A patients with normal protein expression. Am J Pathol 2003, 163, 1929-1936. [CrossRef]
- Talmadge, R.J.; Roy, R.R. Electrophoretic separation of rat skeletal muscle myosin heavy-chain isoforms. J Appl Physiol (1985) 1993, 75, 2337-2340. [CrossRef]
- Mizunoya, W.; Wakamatsu, J.; Tatsumi, R.; Ikeuchi, Y. Protocol for high-resolution separation of rodent myosin heavy chain isoforms in a mini-gel electrophoresis system. Anal Biochem 2008, 377, 111-113. [CrossRef]
- Blum, H., Beier, H.; Gross, H. Improved silver staining of plant proteins, RNA and DNA in polyacrylamide gels. Electrophoresis 1987, 8, 93-99 doi:org/10.1002/elps.1150080203.
- Smerdu, V.; Soukup, T. Demonstration of myosin heavy chain isoforms in rat and humans: the specificity of seven available monoclonal antibodies used in immunohistochemical and immunoblotting methods. Eur J Histochem 2008, 52, 179-190. [CrossRef]
- Meznaric, M.; Eržen, I.; Karen, P.; Cvetko, E. Effect of ageing on the myosin heavy chain composition of the human sternocleidomastoid muscle. Ann Anat 2018, 216, 95-99. [CrossRef]
- Meznaric, M.; Čarni A., Characterisation of flexor digitorum profundus, flexor digitorum superficialis and extensor digitorum communis by electrophoresis and immunohistochemical analysis of myosin heavy chain isoforms in older men. Ann Anat 2020, 227, 151412. [CrossRef]
- Čebašek, V.; Ribarič, S. Changes in local capillarity of pure and hybrid MyHC muscle fiber types after nerve injury in rat extensor digitorum longus muscle (EDL). Histochem Cell Biol 2019, 152, 89-107.
- Karen, P., Števanec, M.; Smerdu, V.; Cvetko, E.; Kubínová, L.; Eržen, I. Software for muscle fibre type classification and analysis. Eur J Histochem 2009, 53, 87-95. [CrossRef]
- Dubowitz, V.; Sewry, C. Histological and histochemical stains and reactions. In Muscle Biopsy A Practical Approach, 3rd ed.; Dubowitz, V., Editor; Saunders Elsevier: China, 2007; 21-39.
- Larsson, L.; Moss, R.L. Maximum velocity of shortening in relation to myosin isoform composition in single fibres from human skeletal muscles. J Physiol, 1993, 472, 595-614. [CrossRef]
- Stauber, W.T. Eccentric action of muscles: physiology, injury, and adaptation. Exerc Sport Sci Rev 1989, 17, 157-185.
- McNeil, P.L.; Khakee, R. Disruptions of muscle fiber plasma membranes. Role in exercise-induced damage. Am J Pathol 1992, 140, 1097-1109.
- Petrof, B.J.; Stedman, H.H.; Shrager, J.B.; Eby, J.; Sweeney, H.L.; Kelly, A.M. Dystrophin protects the sarcolemma from stresses developed during muscle contraction. Proc Natl Acad Sci U S A 1993, 90, 3710-3714. [CrossRef]
- Polgar, J.; Johnson M.A.; Weightman, D.; Appleton, D. Data on fibre size in thirty-six human muscles. An autopsy study. J Neurol Sci 1973, 19, 307-318. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



