Submitted:
12 January 2024
Posted:
15 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals and housing
2.2. Compounds and treatment
2.3. Tail Suspension Test (TST)
2.4. Splash Test
2.5. Sucrose Preference Test (SPT)
2.6. Locomotor Activity
2.7. Synaptosome preparation and Western blotting
2.8. Measurement of BDNF concentration
2.9. Statistical analysis
3. Results
3.1. Experiment 1
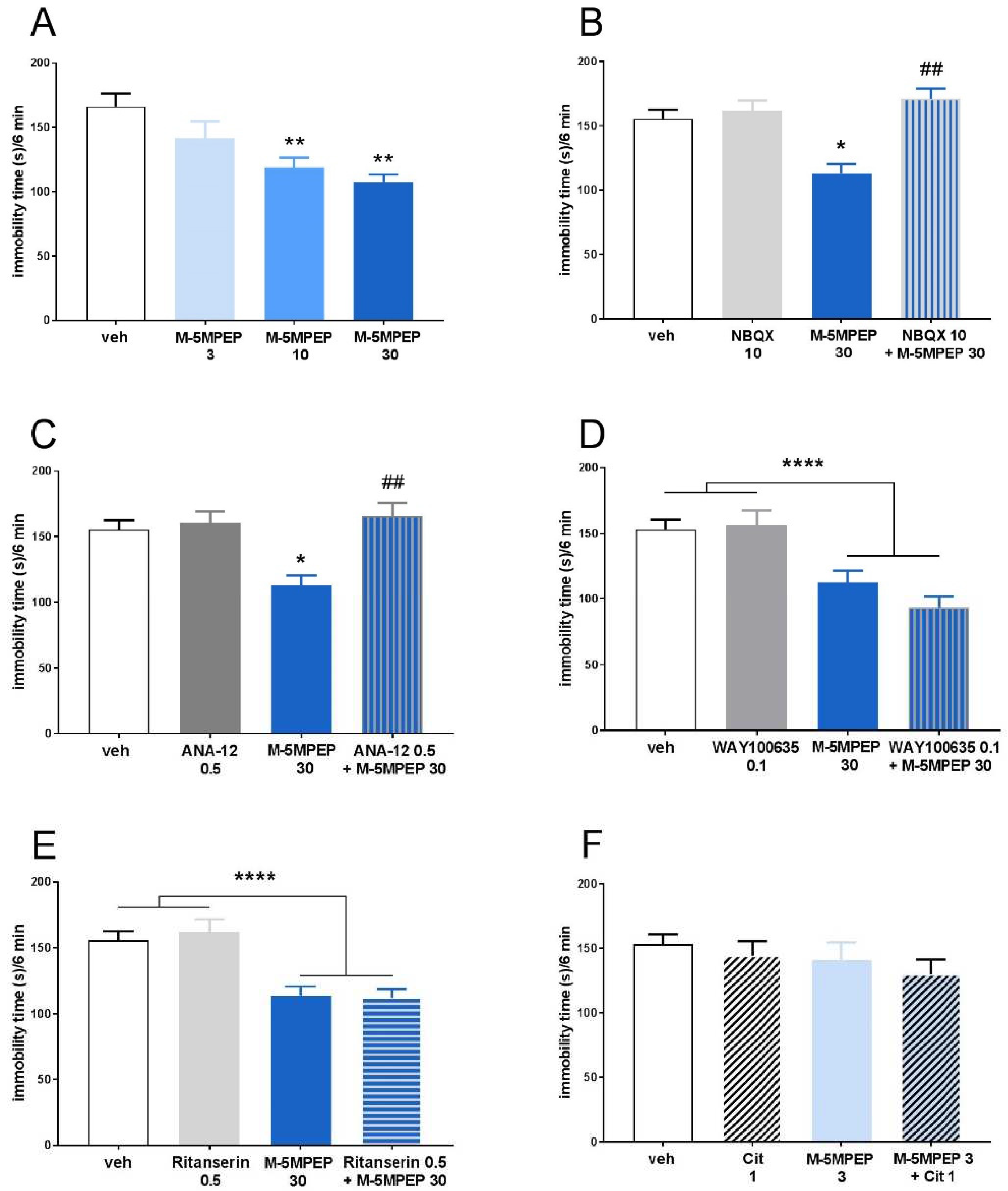
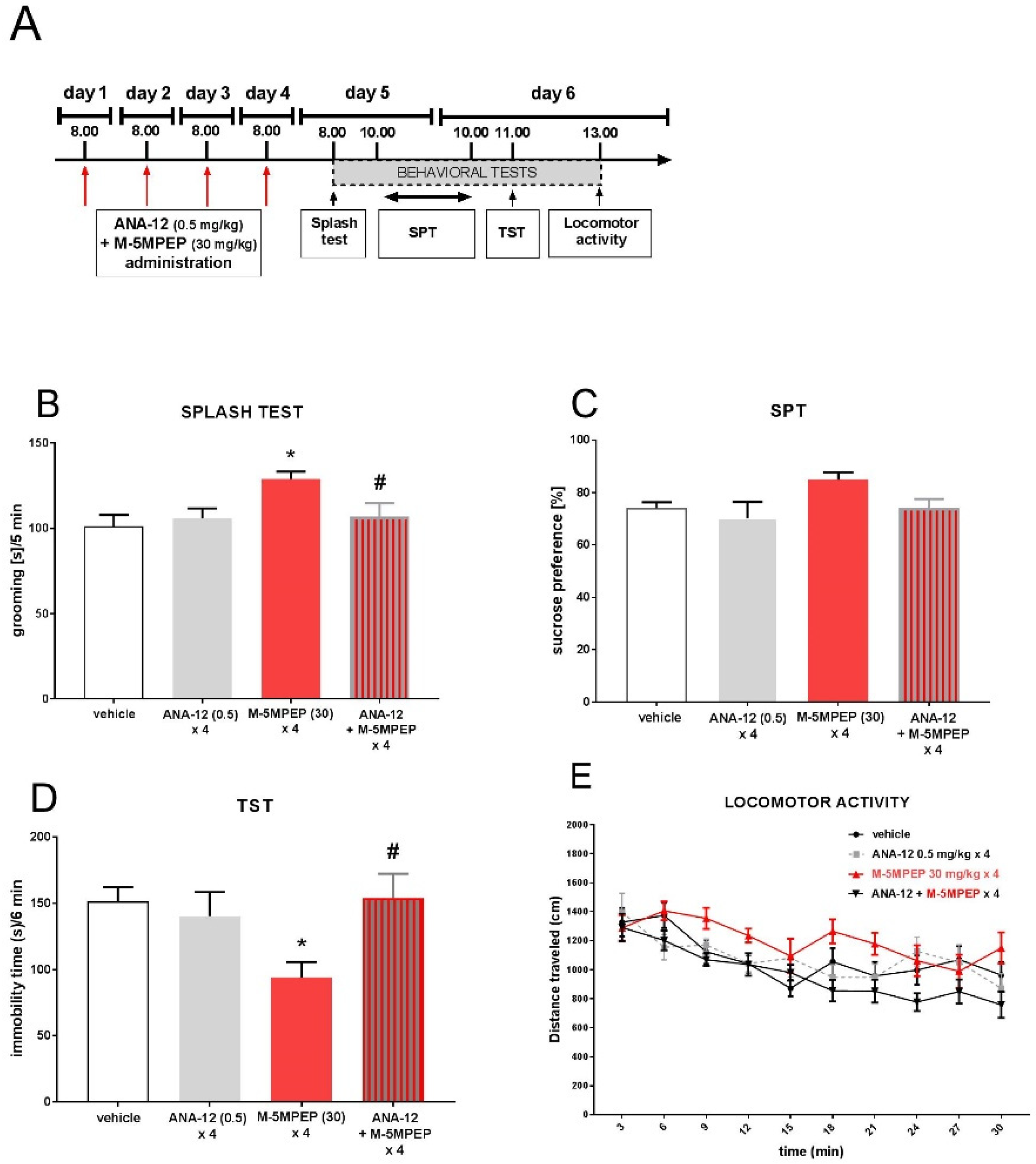
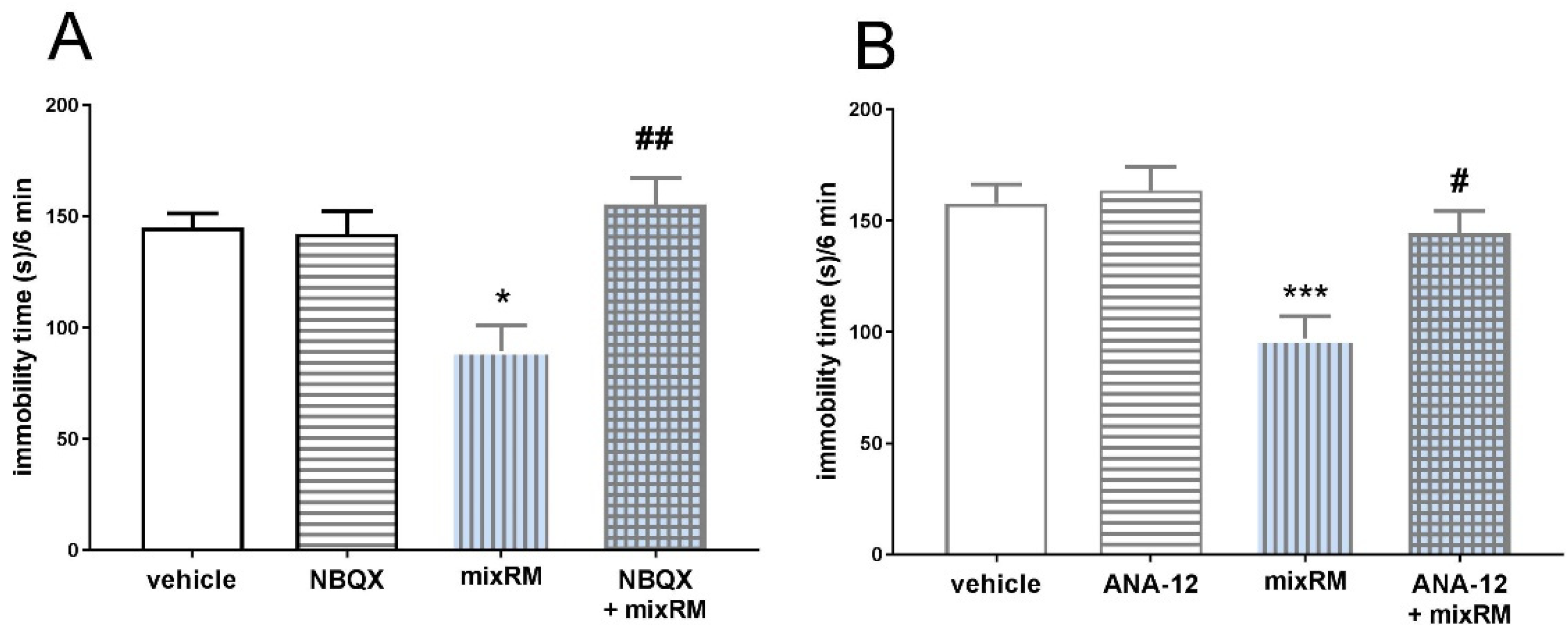
3.1.1. Evaluation of the mechanism of the rapid antidepressant-like effect of M-5MPEP in the TST
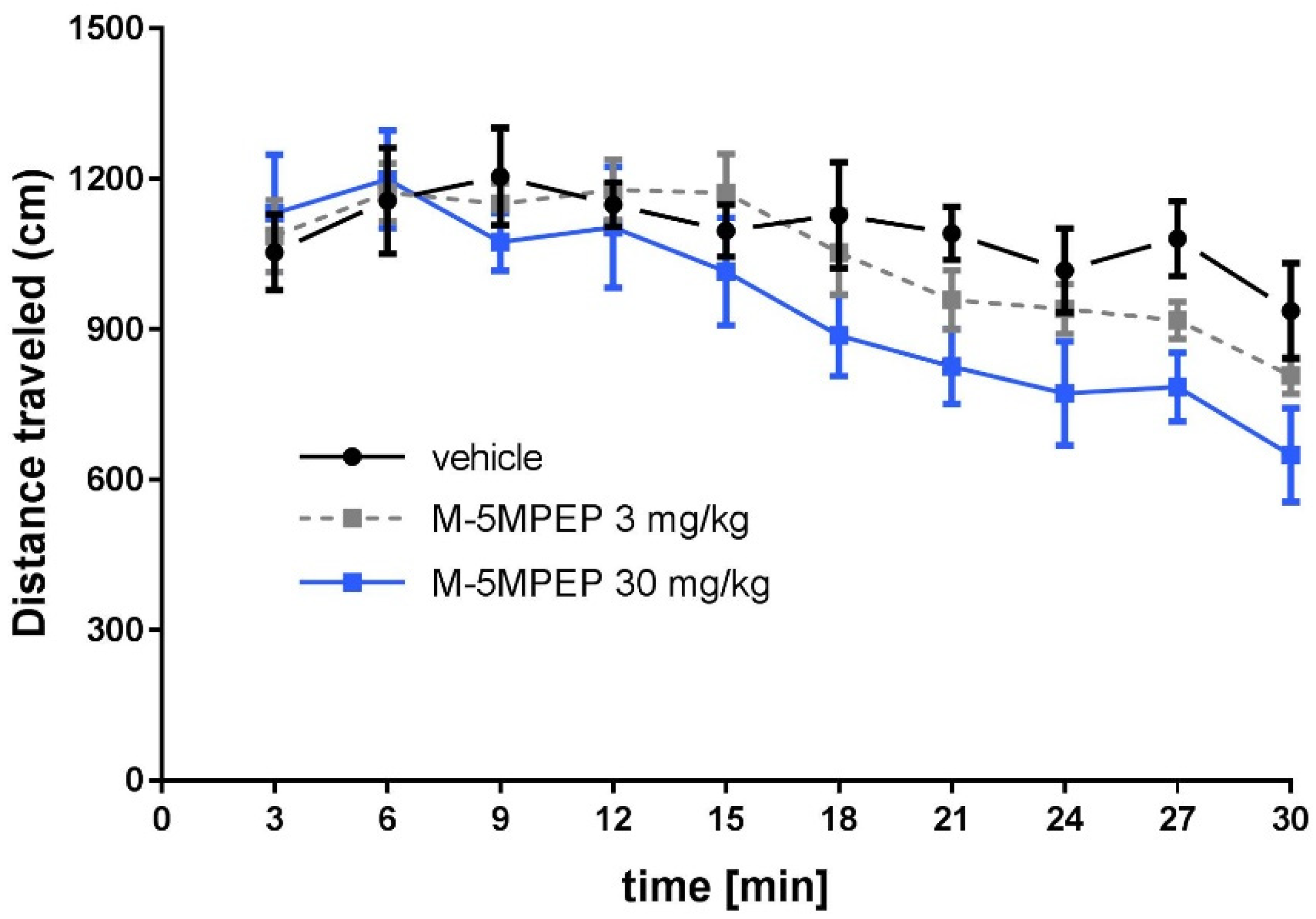
3.1.2. Effect of M-5MPEP on the locomotor activity
3.2. Experiment 2
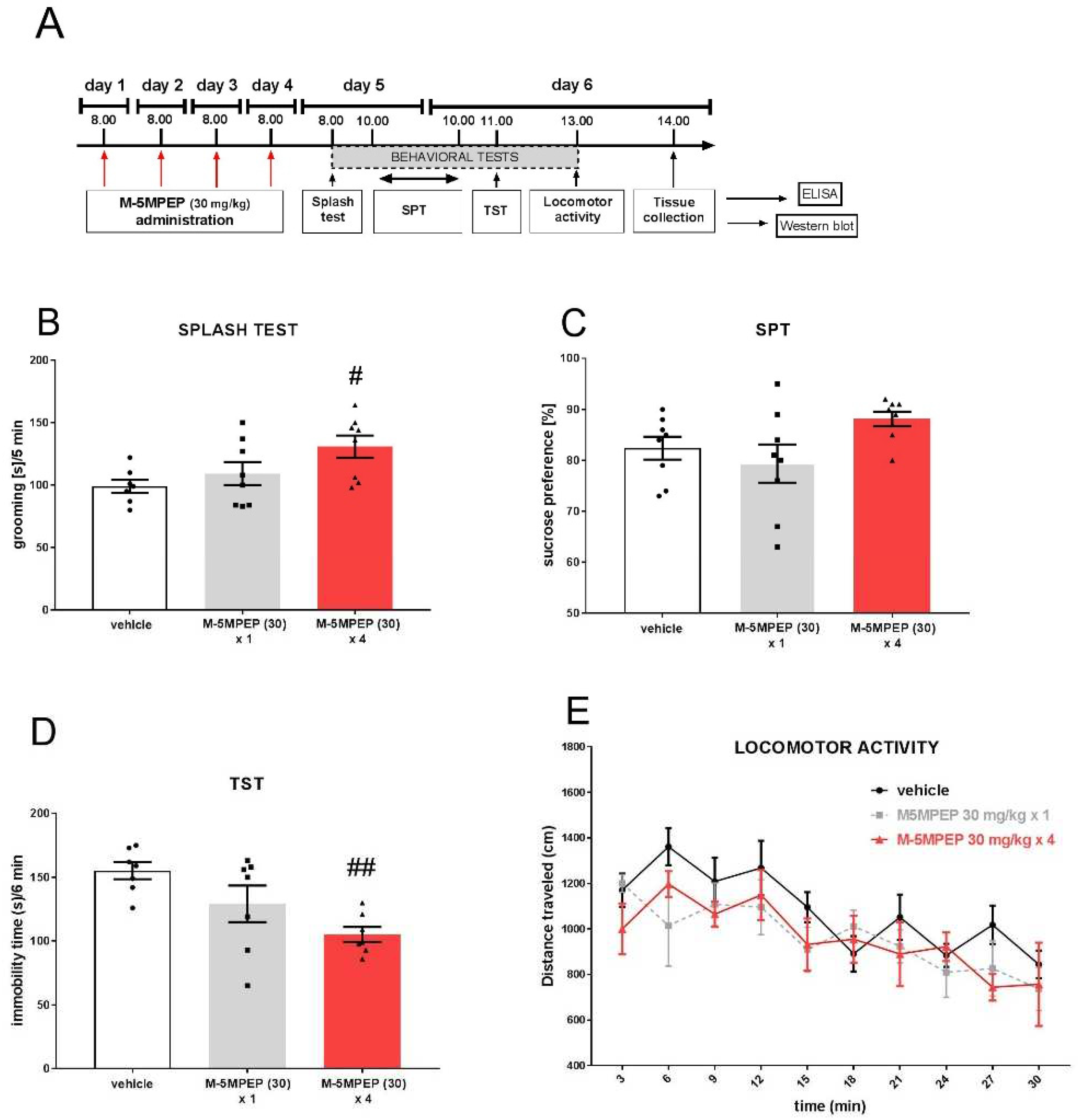
3.2.1. Evaluation of sustained anti-depressant-like effects of MPEP after single and four-day administration in the TST
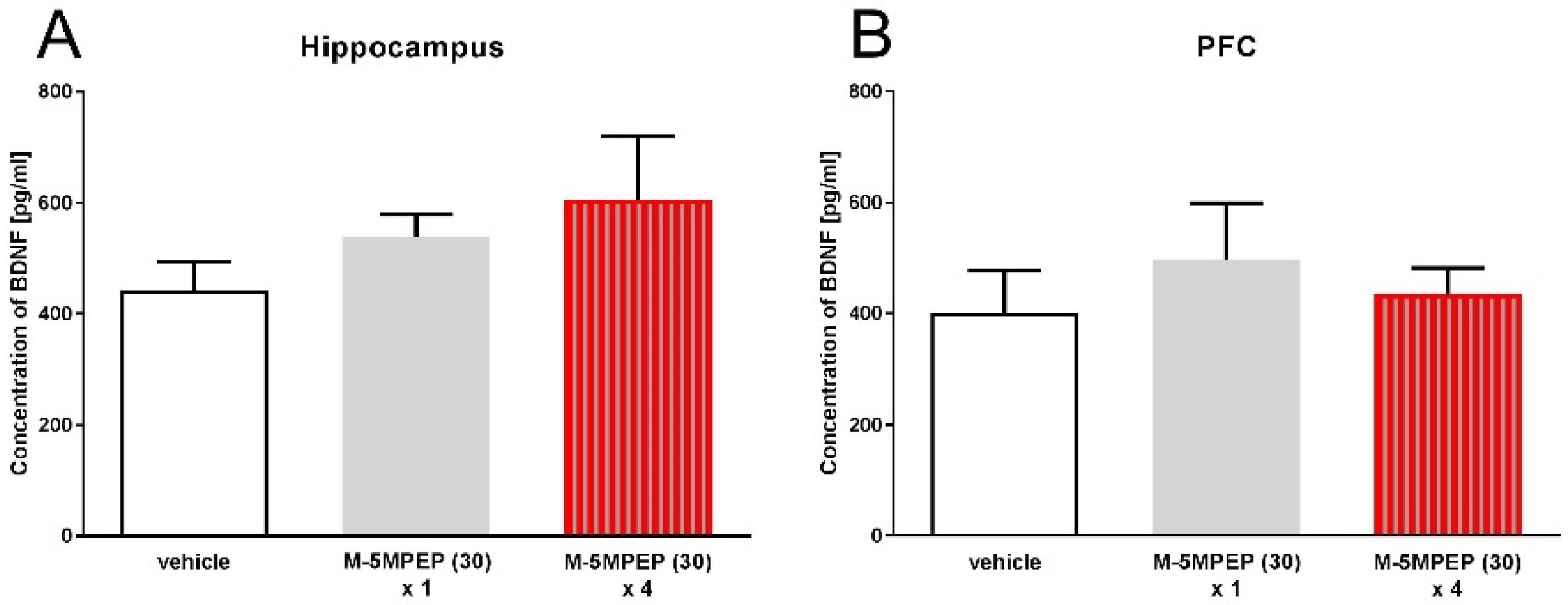
3.2.2. Evaluation of BDNF concentration in the hippocampus and PFC of mice after single and four-day administration of M-5MPEP
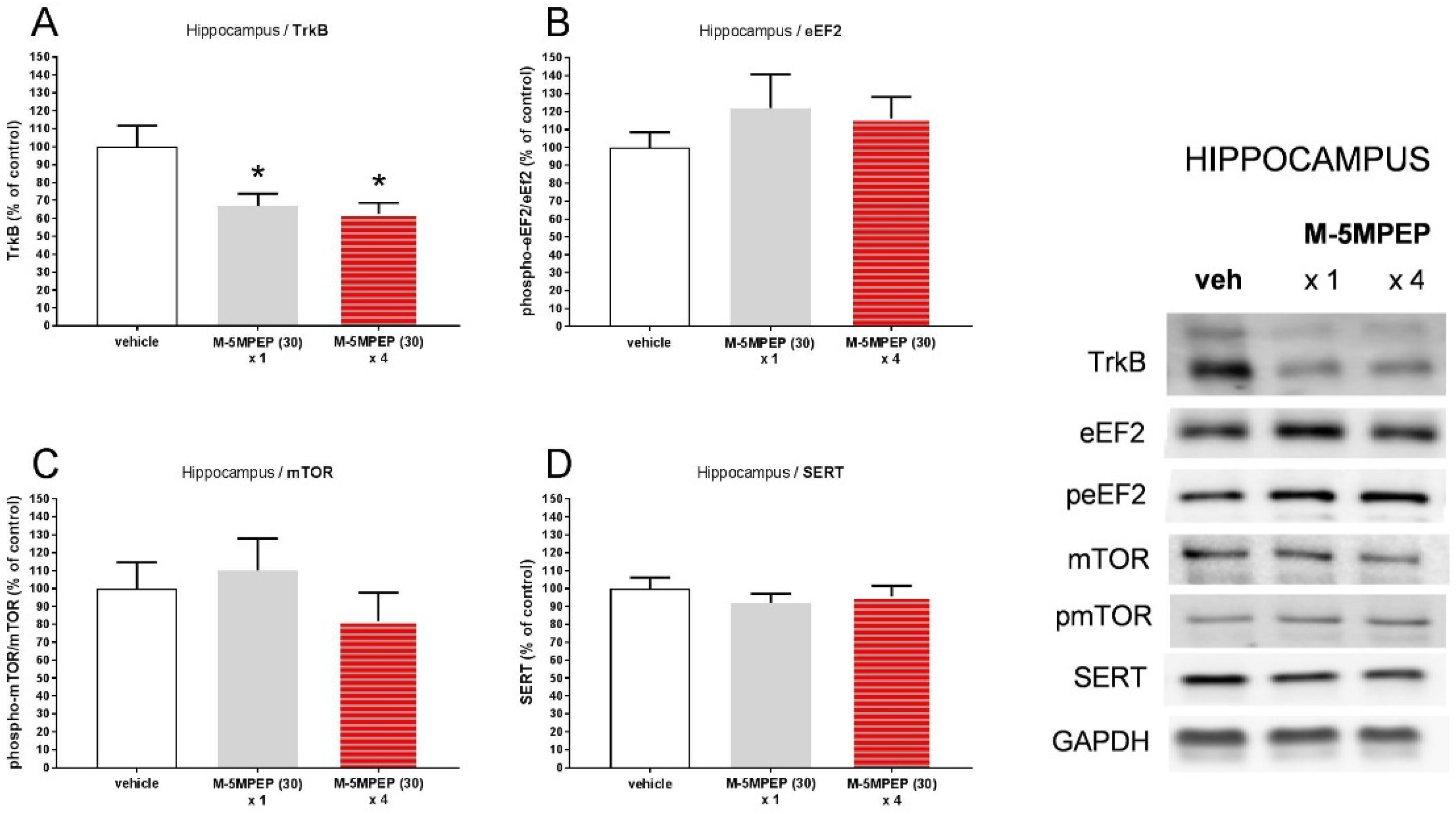
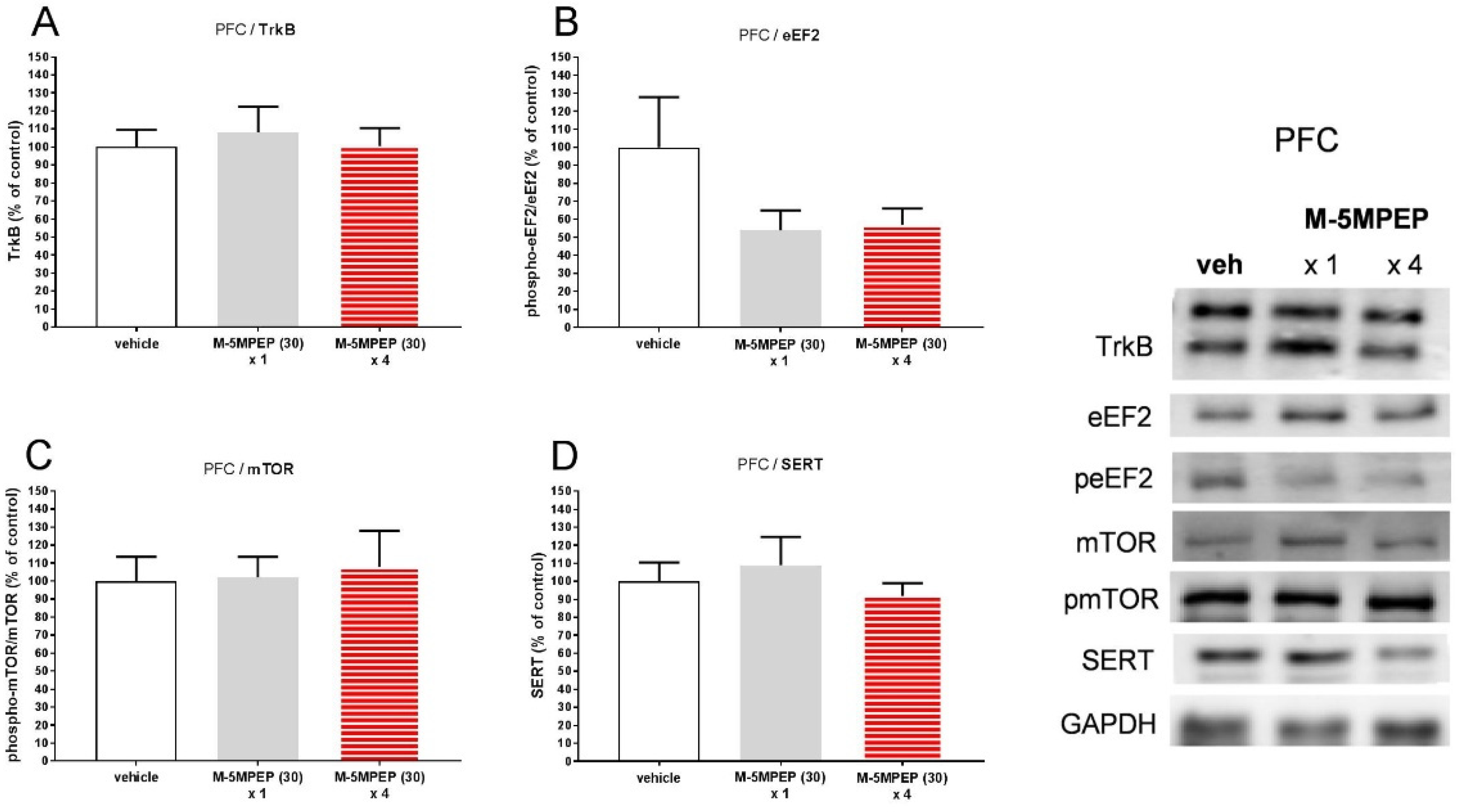
3.2.3. Influence of a single and four-day treatment with M-5MPEP on the TrkB, eEF2, mTOR, and SERT level in the hippocampus and the PFC in mice.
3.2.4. Involvement of TrkB receptor activation in the sustained antidepressant-like effects of M-5MPEP in the TST
3.3. Experiment 3
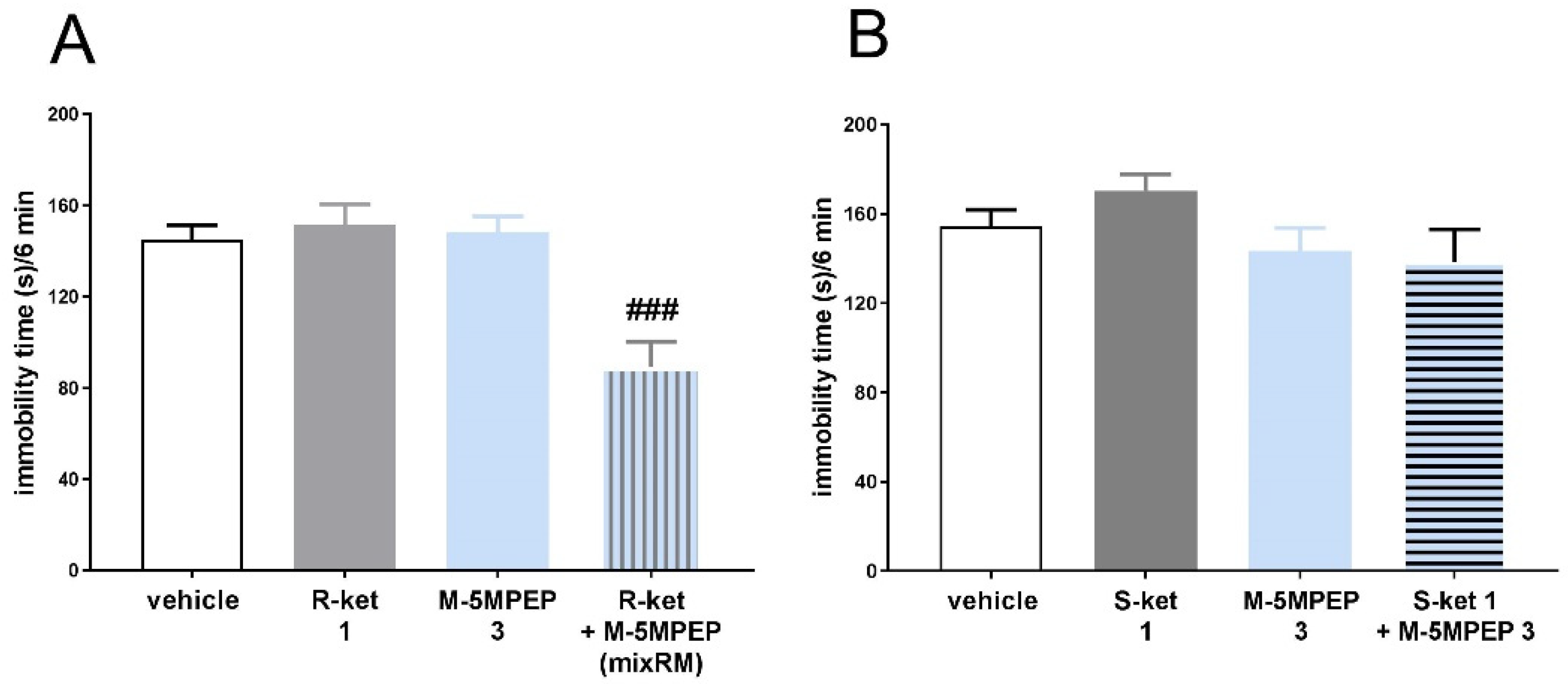
3.3.1. Effect of (R)-ketamine but not (S)-ketamine on the enhancement of the rapid antidepressant-like effect of M-5MPEP in the TST in mice
3.3.2. Evaluation of AMPA and TrkB receptor antagonists on the antidepressant-like effect of M-5MPEP coadministered with (R)-ketamine in the TST in mice
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chaki, S.; Ago, Y.; Pałucha-Poniewiera, A.; Matrisciano, F.; Pilc, A. mGlu2/3 and mGlu5 receptors: potential targets for novel antidepressants. Neuropharmacology 2013, 66, 40–52. [Google Scholar] [CrossRef]
- Nowak, G.; Pomierny-Chamioło, L.; Siwek, A.; Niedzielska, E.; Pomierny, B.; Pałucha-Poniewiera, A.; Pilc, A. Prolonged administration of antidepressant drugs leads to increased binding of [(3)H]MPEP to mGlu5 receptors. Neuropharmacology 2014, 84, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Pałucha, A.; Brański, P.; Szewczyk, B.; Wierońska, J.M.; Kłak, K.; Pilc, A. Potential antidepressant-like effect of MTEP, a potent and highly selective mGluR5 antagonist. Pharmacol. Biochem. Behav. 2005, 81, 901–906. [Google Scholar] [CrossRef]
- Kinney, G.G.; Burno, M.; Campbell, U.C.; Hernandez, L.M.; Rodriguez, D.; Bristow, L.J.; Conn, P.J. Metabotropic glutamate subtype 5 receptors modulate locomotor activity and sensorimotor gating in rodents. J. Pharmacol. Exp. Ther. 2003, 306, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Homayoun, H.; Stefani, M.R.; Adams, B.W.; Tamagan, G.D.; Moghaddam, B. Functional interaction between NMDA and mGlu5 receptors: effects on working memory, instrumental learning, motor behaviors, and dopamine release. Neuropsychopharmacology 2004, 29, 1259–1269. [Google Scholar] [CrossRef] [PubMed]
- Porter, R.H.; Jaeschke, G.; Spooren, W.; Ballard, T.M.; Büttelmann, B.; Kolczewski, S.; Peters, J.U.; Prinssen, E.; Wichmann, J.; Vieira, E.; Mühlemann, A.; Gatti, S.; Mutel, V.; Malherbe, P. Fenobam: a clinically validated nonbenzodiazepine anxiolytic is a potent, selective, and noncompetitive mGlu5 receptor antagonist with inverse agonist activity. J. Pharmacol. Exp. Ther. 2005, 315, 711–721. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, A.L.; Nong, Y.; Sekaran, N.K.; Alagille, D.; Tamagnan, G.D.; Conn, P.J. A close structural analog of 2-methyl-6-(phenylethynyl)-pyridine acts as a neutral allosteric site ligand on metabotropic glutamate receptor subtype 5 and blocks the effects of multiple allosteric modulators. Mol. Pharmacol. 2005, 68, 1793–802. [Google Scholar] [CrossRef]
- Gould, R.W.; Amato, R.J.; Bubser, M.; Joffe, M.E.; Nedelcovych, M.T.; Thompson, A.D.; Nickols, H.H.; Yuh, J.P.; Zhan, X.; Felts, A.S.; Rodriguez, A.L.; Morrison, R.D.; Byers, F.W.; Rook, J.M.; Daniels, J.S.; Niswender, C.M.; Conn, P.J.; Emmitte, K.A.; Lindsley, C.W.; Jones, C.K. Partial mGlu₅ negative allosteric modulators attenuate cocaine-mediated behaviors and lack psychotomimetic-like effects. Neuropsychopharmacology 2016, 41, 1166–1178. [Google Scholar] [CrossRef]
- Holter, K.M.; Lekander, A.D.; LaValley, C.M.; Bedingham, E.G.; Pierce, B.E.; Sands, L.P. 3rd.; Lindsley, C.W.; Jones, C.K.; Gould, R.W. Partial mGlu5 negative allosteric modulator M-5MPEP demonstrates antidepressant-like effects on sleep without affecting cognition or quantitative EEG. Front. Neurosci. 2021, 15, 700822. [Google Scholar] [CrossRef]
- Pałucha-Poniewiera, A. The role of glutamatergic modulation in the mechanism of action of ketamine, a prototype rapid-acting antidepressant drug. Pharmacol. Rep. 2018, 70, 837–846. [Google Scholar] [CrossRef]
- Bobo, W.V.; Vande Voort, J.L.; Croarkin, P.E.; Leung, J.G.; Tye, S.J.; Frye, M.A. Ketamine for treatment-resistant unipolar and bipolar major depression: critical review and implications for clinical practice. Depress. Anxiety 2016, 33, 698–710. [Google Scholar] [CrossRef] [PubMed]
- Zarate, C.A. Jr.; Singh, J.B.; Carlson, P.J.; Brutsche, N.E.; Ameli, R.; Luckenbaugh, D.A.; Charney, D.S.; Manji, H.K. A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch. Gen. Psychiatry 2006, 63, 856–864. [Google Scholar] [CrossRef] [PubMed]
- Mahase, E. Esketamine is approved in Europe for treating resistant major depressive disorder. B.M.J. 2019, 367, l7069. [Google Scholar] [CrossRef] [PubMed]
- Fukumoto, K.; Iijima, M.; Chaki, S. Serotonin-1A receptor stimulation mediates effects of a metabotropic glutamate 2/3 receptor antagonist, 2S-2-amino-2-(1S,2S-2-carboxycycloprop-1-yl)-3-(xanth-9-yl)propanoic acid (LY341495), and an N-methyl-D-aspartate receptor antagonist, ketamine, in the novelty-suppressed feeding test. Psychopharmacology 2014, 231, 2291–2298. [Google Scholar] [CrossRef] [PubMed]
- Fukumoto, K.; Iijima, M.; Chaki, S. The antidepressant effects of an mGlu2/3 receptor antagonist and ketamine require AMPA receptor stimulation in the mPFC and subsequent activation of the 5-HT neurons in the DRN. Neuropsychopharmacology 2016, 41, 1046–1056. [Google Scholar] [CrossRef] [PubMed]
- Pałucha-Poniewiera, A. The role of mGlu2/3 receptor antagonists in the enhancement of the antidepressant-like effect of ketamine. Pharmacol. Biochem. Behav. 2022, 220, 173454. [Google Scholar] [CrossRef]
- Witkin, J.M. mGlu2/3 receptor antagonism: A mechanism to induce rapid antidepressant effects without ketamine-associated side-effects. Pharmacol. Biochem. Behav. 2020, 190, 172854. [Google Scholar] [CrossRef] [PubMed]
- Pałucha-Poniewiera, A.; Podkowa, K.; Rafało-Ulińska, A. The group II mGlu receptor antagonist LY341495 induces a rapid antidepressant-like effect and enhances the effect of ketamine in the chronic unpredictable mild stress model of depression in C57BL/6J mice. Prog. Neuropsychopharmacol. Biol. Psychiatry 2021, 109, 110239. [Google Scholar] [CrossRef]
- Steru, L.; Chermat, R.; Thierry, B.; Simon, P. The tail suspension test: A new method for screening antidepressants in mice. Psychopharmacology 1985, 85, 367–370. [Google Scholar] [CrossRef]
- Pałucha-Poniewiera, A.; Podkowa, K.; Rafało-Ulińska, A.; Brański, P.; Burnat, G. The influence of the duration of chronic unpredictable mild stress on the behavioural responses of C57BL/6J mice. Behav. Pharmacol. 2020, 31, 574–582. [Google Scholar] [CrossRef]
- Krystal, J.H.; Karper, L.P.; Seibyl, J.P.; Freeman, G.K.; Delaney, R.; Bremner, J.D.; Heninger, G.R.; Bowers, M.B. Jr.; Charney, D.S. Subanesthetic effects of the noncompetitive NMDA antagonist, ketamine, in humans. Psychotomimetic, perceptual, cognitive, and neuroendocrine responses. Arch. Gen. Psychiatry 1994, 51, 199–192. [Google Scholar] [CrossRef] [PubMed]
- Iijima, M.; Fukumoto, K.; Chaki, S. Acute and sustained effects of a metabotropic glutamate 5 receptor antagonist in the novelty-suppressed feeding test. Behav. Brain Res. 2012, 235, 287–292. [Google Scholar] [CrossRef]
- Pałucha-Poniewiera, A.; Szewczyk, B.; Pilc, A. Activation of the mTOR signaling pathway in the antidepressant-like activity of the mGlu5 antagonist MTEP and the mGlu7 agonist AMN082 in the FST in rats. Neuropharmacology 2014, 82, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; Mombereau, C.; Vassout, A. The tail suspension test as a model for assessing antidepressant activity: review of pharmacological and genetic studies in mice. Neurosci. Biobehav. Rev. 2005, 29, 571–625. [Google Scholar] [CrossRef] [PubMed]
- Autry, A.E.; Adachi, M.; Nosyreva, E.; Na, E.S.; Los, M.F.; Cheng, P.F.; Kavalali, E.T.; Monteggia, L.M. NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature 2011, 475, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Koike, H.; Iijima, M.; Chaki, S. Involvement of AMPA receptor in both the rapid and sustained antidepressant-like effects of ketamine in animal models of depression. Behav. Brain Res. 2011, 224, 107–111. [Google Scholar] [CrossRef]
- Li, N.; Lee, B.; Liu, R.J.; Banasr, M.; Dwyer, J.M.; Iwata, M.; Li, X.Y.; Aghajanian, G.; Duman, R.S. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science 2010, 329, 959–964. [Google Scholar] [CrossRef]
- Pomierny-Chamioło, L.; Poleszak, E.; Pilc, A.; Nowak, G. NMDA but not AMPA glutamatergic receptors are involved in the antidepressant-like activity of MTEP during the forced swim test in mice. Pharmacol. Rep. 2010, 62, 1186–1190. [Google Scholar] [CrossRef]
- Sun, H.L.; Zhou, Z.Q.; Zhang, G.F.; Yang, C.; Wang, X.M.; Shen, J.C.; Hashimoto, K.; Yang, J.J. Role of hippocampal p11 in the sustained antidepressant effect of ketamine in the chronic unpredictable mild stress model. Transl. Psychiatry 2016, 6, e741. [Google Scholar] [CrossRef]
- Koike, H.; Fukumoto, K.; Iijima, M.; Chaki, S. Role of BDNF/TrkB signaling in antidepressant-like effects of a group II metabotropic glutamate receptor antagonist in animal models of depression. Behav. Brain Res. 2013, 238, 48–52. [Google Scholar] [CrossRef]
- du Jardin, K.G.; Liebenberg, N.; Cajina, M.; Müller, H.K.; Elfving, B.; Sanchez, C.; Wegener, G. S-ketamine mediates its acute and sustained antidepressant-like activity through a 5-HT1B receptor dependent mechanism in a genetic rat model of depression. Front. Pharmacol. 2018, 8, 97. [Google Scholar] [CrossRef]
- du Jardin, K.G.; Liebenberg, N.; Müller, H.K.; Elfving, B.; Sanchez, C.; Wegener, G. Differential interaction with the serotonin system by S-ketamine, vortioxetine, and fluoxetine in a genetic rat model of depression. Psychopharmacology 2016, 233, 2813–2825. [Google Scholar] [CrossRef]
- Pałucha-Poniewiera, A.; Brański, P.; Wierońska, J.M.; Stachowicz, K.; Sławińska, A.; Pilc, A. The antidepressant-like action of mGlu5 receptor antagonist, MTEP, in the tail suspension test in mice is serotonin dependent. Psychopharmacology 2014, 231, 97–107. [Google Scholar] [CrossRef]
- Fukumoto, K.; Chaki, S. Involvement of serotonergic system in the effect of a metabotropic glutamate 5 receptor antagonist in the novelty-suppressed feeding test. J. Pharmacol. Sci. 2015, 127, 57–61. [Google Scholar] [CrossRef]
- Gigliucci, V.; O'Dowd, G.; Casey, S.; Egan, D.; Gibney, S.; Harkin, A. Ketamine elicits sustained antidepressant-like activity via a serotonin-dependent mechanism. Psychopharmacology 2013, 228, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Sun, C.; Zhai, L.; Dong, W. A selective M1 and M3 receptor antagonist, penehyclidine hydrochloride, exerts antidepressant-like effect in mice. Neurochem. Res. 2019, 44, 2723–2732. [Google Scholar] [CrossRef]
- Rafało-Ulińska, A.; Brański, P.; Pałucha-Poniewiera, A. Combined administration of (R)-ketamine and the mGlu2/3 receptor antagonist LY341495 induces rapid and sustained effects in the CUMS model of depression via a TrkB/BDNF-dependent mechanism. Pharmaceuticals 2022, 15, 125. [Google Scholar] [CrossRef] [PubMed]
- Rafało-Ulińska, A.; Pałucha-Poniewiera, A. The effectiveness of (R)-ketamine and its mechanism of action differ from those of (S)-ketamine in a chronic unpredictable mild stress model of depression in C57BL/6J mice. Behav. Brain Res. 2022, 418, 113633. [Google Scholar] [CrossRef]
- Wu, C.; Wang, Y.; He, Y.; Wu, S.; Xie, Z.; Zhang, J.; Shen, J.; Wang, Z.; He, L. Sub-anesthetic and anesthetic ketamine produce different long-lasting behavioral phenotypes (24 h post-treatment) via inducing different brain-derived neurotrophic factor (BDNF) expression level in the hippocampus. Neurobiol. Learn. Mem. 2020, 167, 107136. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Zhang, K.; Pu, Y.; Qu, Y.; Wang, S.M.; Xiong, Z.; Ren, Q.; Dong, C.; Fujita, Y.; Hashimoto, K. Comparison of antidepressant and side effects in mice after intranasal administration of (R,S)-ketamine, (R)-ketamine, and (S)-ketamine. Pharmacol. Biochem. Behav. 2019, 181, 53–59. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



