Submitted:
06 January 2024
Posted:
08 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Endpoint.
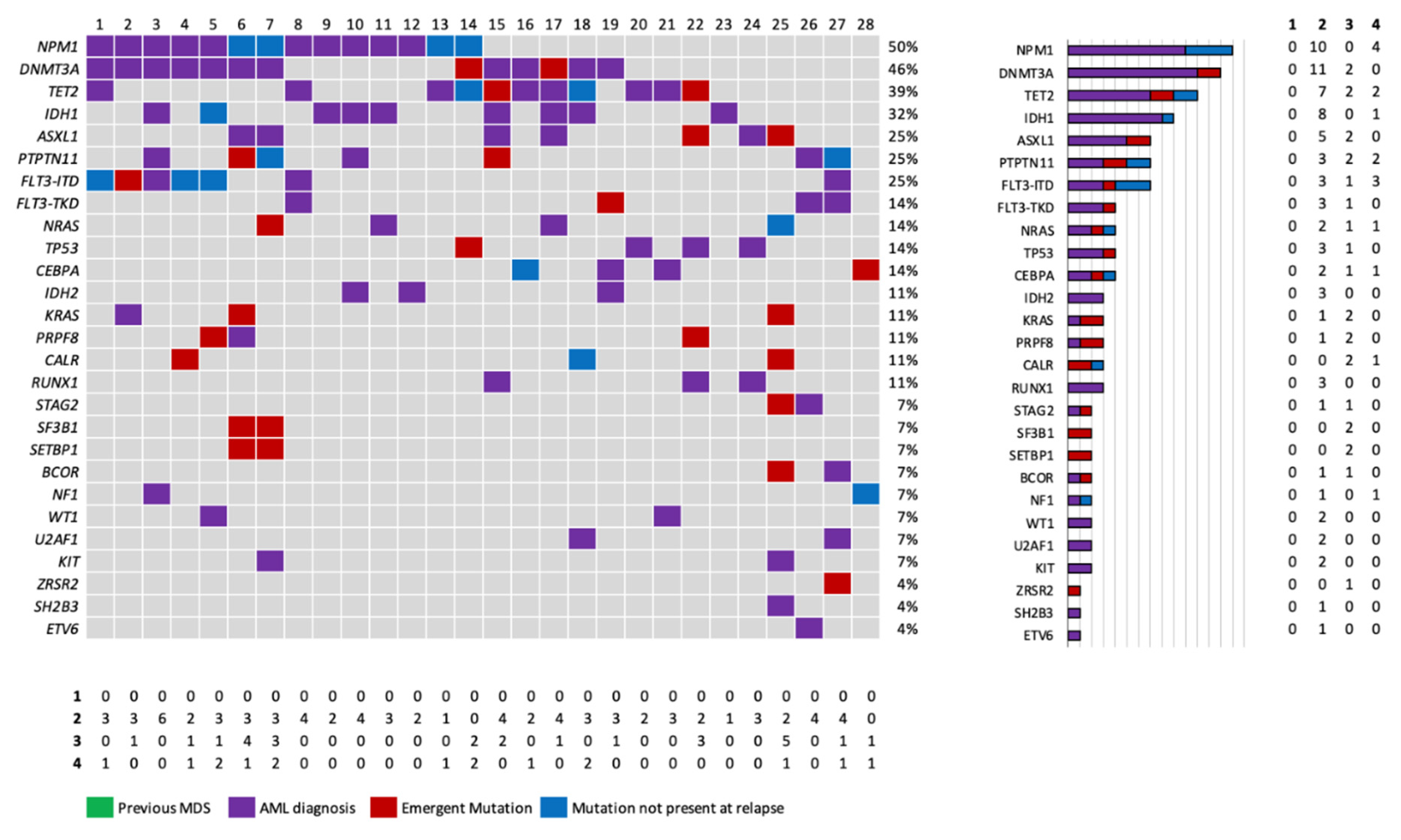
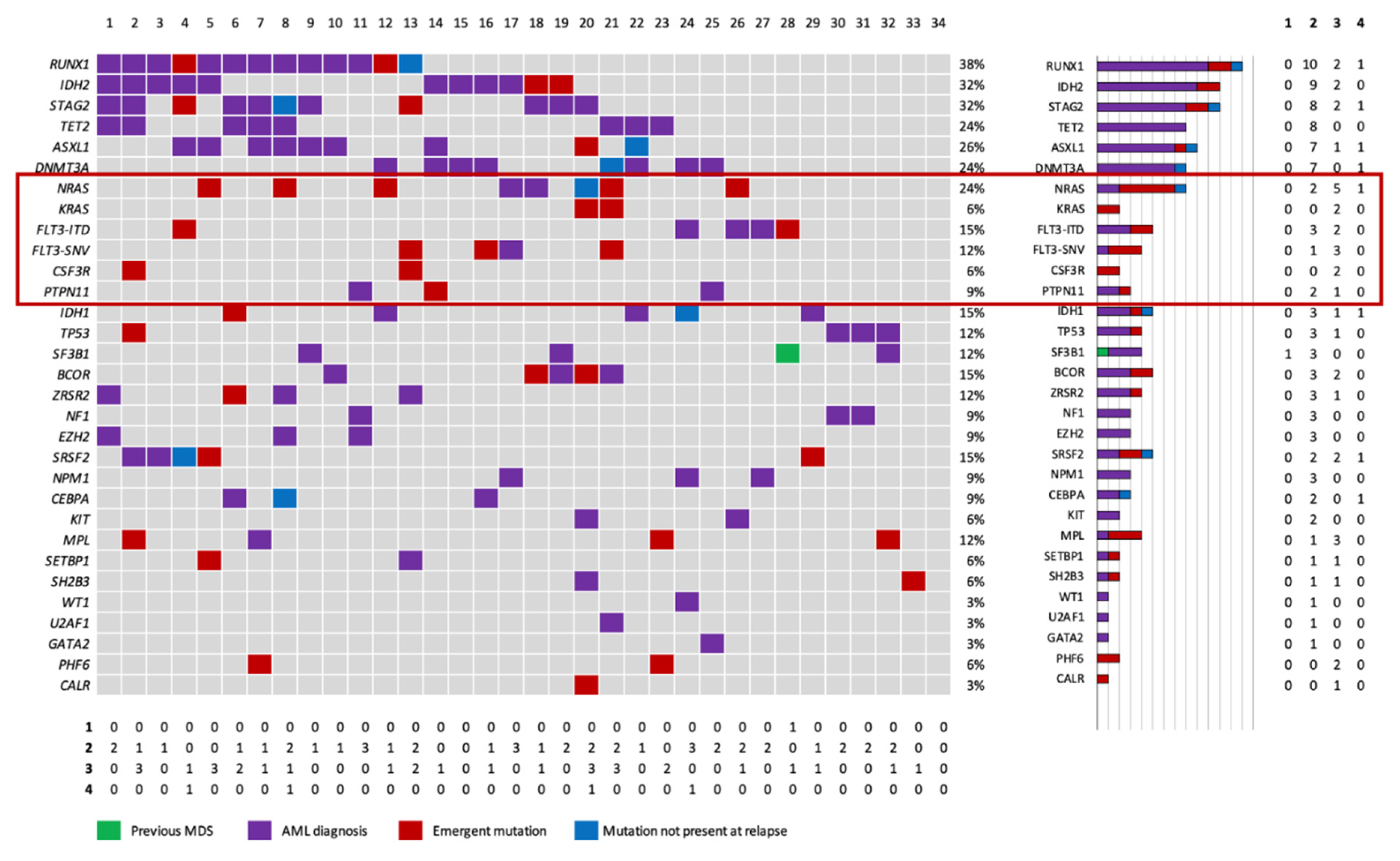
3. Results
3.1. Baseline characteristics.
3.2. Standard intensive chemotherapy (ICT)
3.3. Low-intensity treatments
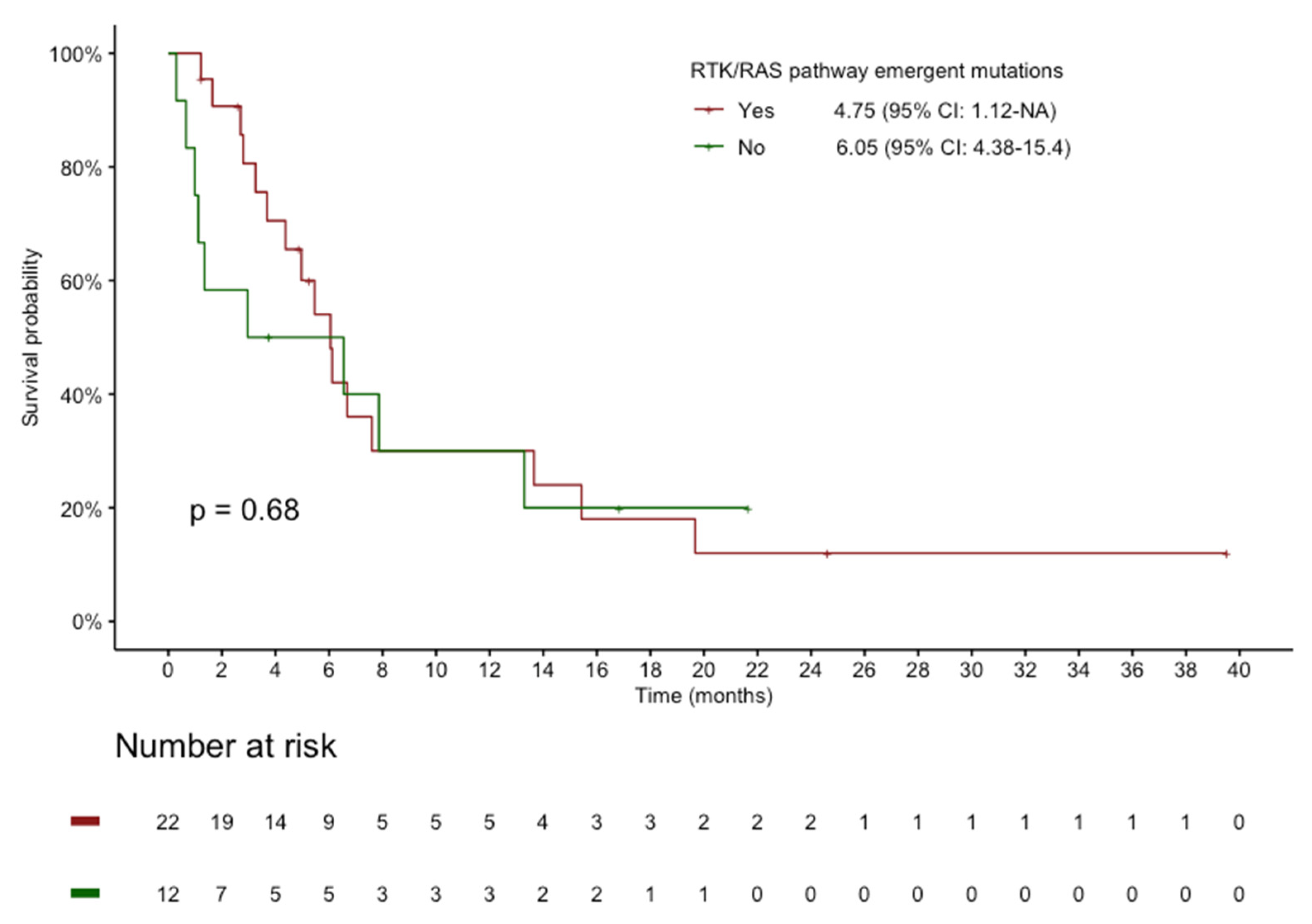
3.4. Subgroup analysis of patients with AML with mutated TP53
4. Discussion
5. Materials and Methods
6. Conclusions
Author Contributions
Conflicts of Interest
Appendix A
Appendix B
References
- Döhner, H., Weisdorf, D. J. & Bloomfield, C. D. Acute Myeloid Leukemia. N. Engl. J. Med. 2015, 373, 1136–1152.
- Döhner, H. et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood vol. 140 (2022).
- Ganzel, C. et al. Very poor long-term survival in past and more recent studies for relapsed AML patients: The ECOG-ACRIN experience. Am. J. Hematol. 2018, 93, 1074–1081. [CrossRef]
- DiNardo, C. D. et al. Azacitidine and Venetoclax in Previously Untreated Acute Myeloid Leukemia. N. Engl. J. Med. 2020, 383, 617–629. [CrossRef]
- DiNardo, C. D. et al. 10-day decitabine with venetoclax for newly diagnosed intensive chemotherapy ineligible, and relapsed or refractory acute myeloid leukaemia: a single-centre, phase 2 trial. Lancet Haematol. 2020, 7, e724–e736. [CrossRef]
- DiNardo, C. D. et al. Venetoclax combined with FLAG-IDA induction and consolidation in newly diagnosed acute myeloid leukemia. Am. J. Hematol. 2022, 97, 1035–1043. [CrossRef]
- Chua, C. C. et al. Chemotherapy and Venetoclax in Elderly Acute Myeloid Leukemia Trial (CAVEAT): A Phase Ib Dose-Escalation Study of Venetoclax Combined with Modified Intensive Chemotherapy. J. Clin. Oncol. 2020, 38, 3506–3517. [CrossRef]
- Kadia, T. M. et al. Venetoclax plus intensive chemotherapy with cladribine, idarubicin, and cytarabine in patients with newly diagnosed acute myeloid leukaemia or high-risk myelodysplastic syndrome: a cohort from a single-centre, single-arm, phase 2 trial. Lancet Haematol. 2021, 8, e552–e561. [CrossRef]
- Montesinos, P. et al. Ivosidenib and Azacitidine in IDH1-Mutated Acute Myeloid Leukemia. N. Engl. J. Med. 2022, 386, 1519–1531. [CrossRef]
- DiNardo, C. D. et al. Enasidenib plus azacitidine versus azacitidine alone in patients with newly diagnosed, mutant-<em>IDH2</em> acute myeloid leukaemia (AG221-AML-005): a single-arm, phase 1b and randomised, phase 2 trial. Lancet Oncol. 2021, 22, 1597–1608. [CrossRef]
- Stone, R. M. et al. Midostaurin plus Chemotherapy for Acute Myeloid Leukemia with a FLT3 Mutation. N. Engl. J. Med. 2017, 377, 454–464. [CrossRef]
- Perl, A. E. et al. Gilteritinib or Chemotherapy for Relapsed or Refractory FLT3-Mutated AML. N. Engl. J. Med. 2019, 381, 1728–1740. [CrossRef]
- Juliette Lambert et al. Gemtuzumab ozogamicin for de novo acute myeloid leukemia: final efficacy and safety updates from the open-label, phase III ALFA-0701 trial. Haematologica 2019, 104, 113–119.
- Issa, G. C. et al. The menin inhibitor revumenib in KMT2A-rearranged or NPM1-mutant leukaemia. Nature 2023, 615, 920–924. [CrossRef]
- Christen, F. et al. Genomic landscape and clonal evolution of acute myeloid leukemia with t(8;21): an international study on 331 patients. Blood 2019, 133, 1140–1151. [CrossRef]
- Vosberg, S. Clonal evolution of acute myeloid leukemia from diagnosis to relapse. 839–849 (2019). [CrossRef]
- Greif, P. A. et al. Evolution of Cytogenetically Normal Acute Myeloid Leukemia During Therapy and Relapse: An Exome Sequencing Study of 50 Patients. Clin. Cancer Res. 2018, 24, 1716–1726. [CrossRef]
- Morita, K. et al. Clonal evolution of acute myeloid leukemia revealed by high-throughput single-cell genomics. Nat. Commun. 2020, 11, 1–17. [CrossRef]
- Cocciardi, S. et al. Clonal evolution patterns in acute myeloid leukemia with NPM1 mutation. Nat. Commun. 2019, 10, 2031. [CrossRef]
- Miles, L. A. et al. Single-cell mutation analysis of clonal evolution in myeloid malignancies. Nature 587, (2020). [CrossRef]
- Höllein, A. et al. NPM1 mutated AML can relapse with wild-type NPM1: persistent clonal hematopoiesis can drive relapse. Blood Adv. 2018, 2, 3118–3125. [CrossRef]
- Symeonidou, V. et al. Heterogeneous genetic and non-genetic mechanisms contribute to response and resistance to azacitidine monotherapy. 794–803 (2022). [CrossRef]
- Schmalbrock, L. K. et al. Clonal evolution of acute myeloid leukemia with FLT3 -ITD mutation under treatment with midostaurin. 2021, 137, 3093–3104. [CrossRef]
- Mcmahon, C. M. et al. Clonal Selection with RAS Pathway Activation Mediates Secondary Clinical Resistance to Selective FLT3 Inhibition in Acute Myeloid Leukemia. (2019). [CrossRef]
- 25. Chyla, B. et al. Genetic biomarkers of sensitivity and resistance to venetoclax monotherapy in patients with relapsed acute myeloid leukemia. Am. J. Hematol. 2018, 93, E202–E205. [CrossRef]
- DiNardo, C. D. et al. Molecular patterns of response and treatment failure after frontline venetoclax combinations in older patients with AML. Blood 2020, 135, 791–803. [CrossRef]
- Döhner, H. et al. ELN Risk Stratification Is Not Predictive of Outcomes for Treatment-Naïve Patients with Acute Myeloid Leukemia Treated with Venetoclax and Azacitidine. Blood 2022, 140, 1441–1444. [CrossRef]
- Pei, S. et al. Monocytic Subclones Confer Resistance to Venetoclax-Based Therapy in Patients with Acute Myeloid Leukemia. Cancer Discov. 2020, 10, 536–551. [CrossRef]
- Zhang, Q. et al. Activation of RAS/MAPK pathway confers MCL-1 mediated acquired resistance to BCL-2 inhibitor venetoclax in acute myeloid leukemia. Signal Transduct. Target. Ther. 2022, 7, 51.
- Daver, N. et al. Venetoclax Plus Gilteritinib for FLT3-Mutated Relapsed/Refractory Acute Myeloid Leukemia. J. Clin. Oncol. 2022, 40, 4048–4059. [CrossRef]
- Janssen, M. et al. Venetoclax synergizes with gilteritinib in FLT3 wild-type high-risk acute myeloid leukemia by suppressing MCL-1. Blood 2022, 140, 2594–2610. [CrossRef]
- Khoury, J. D. et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia (2022). [CrossRef]
- Arber, D. et al. International Consensus Classification of Myeloid Neoplasms and Acute Leukemia: Integrating Morphological, Clinical, and Genomic Data. Blood (2022).
- Cheson, B. D. et al. Revised Recommendations of the International Working Group for Diagnosis, Standardization of Response Criteria, Treatment Outcomes, and Reporting Standards for Therapeutic Trials in Acute Myeloid Leukemia. J. Clin. Oncol. 2003, 21, 4642–4649. [CrossRef]
- Bataller, A. et al. European LeukemiaNet 2017 risk stratification for acute myeloid leukemia: validation in a risk-adapted protocol. Blood Adv. 2022, 6, 1193–1206. [CrossRef]
| Baseline Characteristics | All patients (N = 62) |
Standard Chemotherapy (n = 28) |
Low-intensity regimens (n = 34) |
p |
|---|---|---|---|---|
| Age, median (range) | 70 (32-85) | 66 (36-76) | 73 (47-82) | 0.08 |
| Sex | 0.85 | |||
| Female | 32 (51.6) | 15 (53.8) | 17 (50) | |
| Male | 30 (48.4) | 13 (46.2) | 17 (50) | |
| ELN 2022 Risk status | 0.009 | |||
| Favorable | 13 (21) | 11 (39.3) | 2 (5.9) | |
| Intermediate | 16 (25.8) | 8 (28.6) | 8 (23.5) | |
| Adverse | 33 (53.2) | 9 (32.1) | 24 (70.6) | |
| Prior treatments to study, n (%) | 0.002 | |||
| Standard chemotherapy | 5 (8) | 0 (0) | 5 (14.7) | |
| Hypomethylating agents | 4 (6.5) | 0 (0) | 4 (6.5) | |
| Treatment-naïve | 50 (82.6) | 29 (100) | 21 (61.8) | |
| Disease status at reevaluation, n (%) | 0.48 | |||
| Primary refractoriness | 10 (16.1) | 3 (10.7) | 7 (20.6) | |
| Relapsed disease | 52 (83.9) | 25 (89.3) | 27 (79.4) | |
| WBC count, median (range) | 4.2 (0.4-72.4) | 9.9 (0.46-72.4) | 3.57 (0.4-30.4) | <0.001 |
| AlloHCT, n (%) | 18 (29) | 12 (42.9) | 6 (17.6) | 0.04 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




