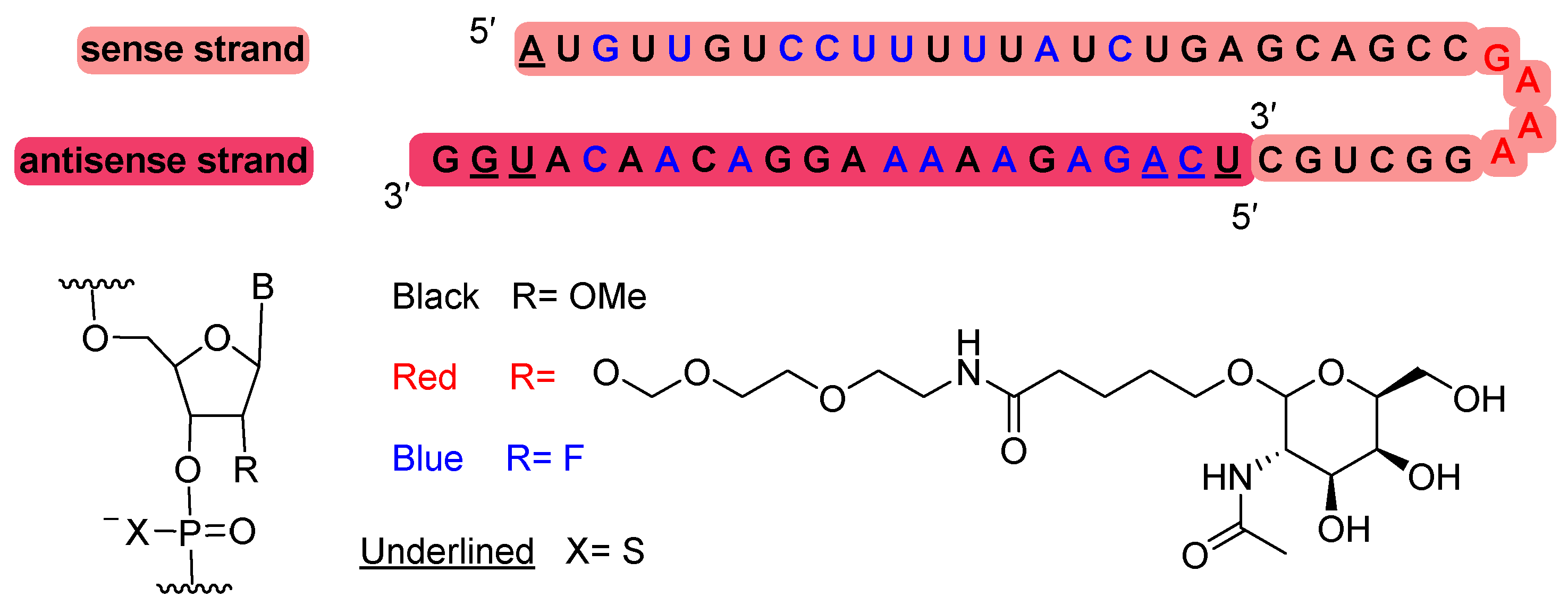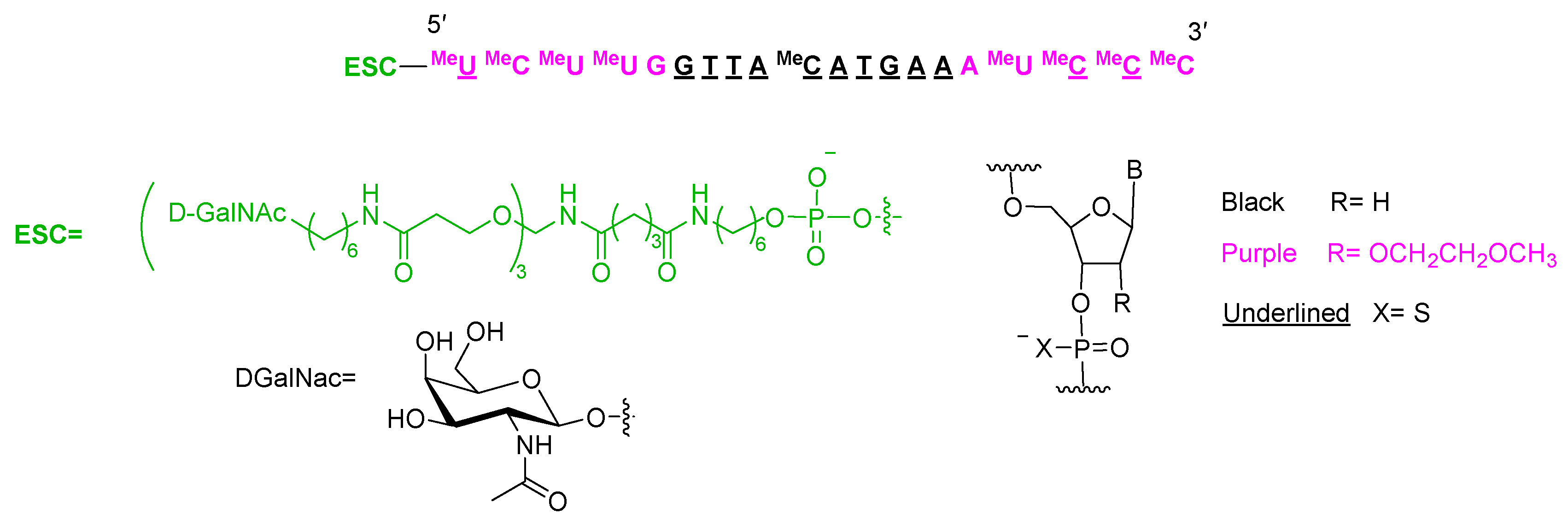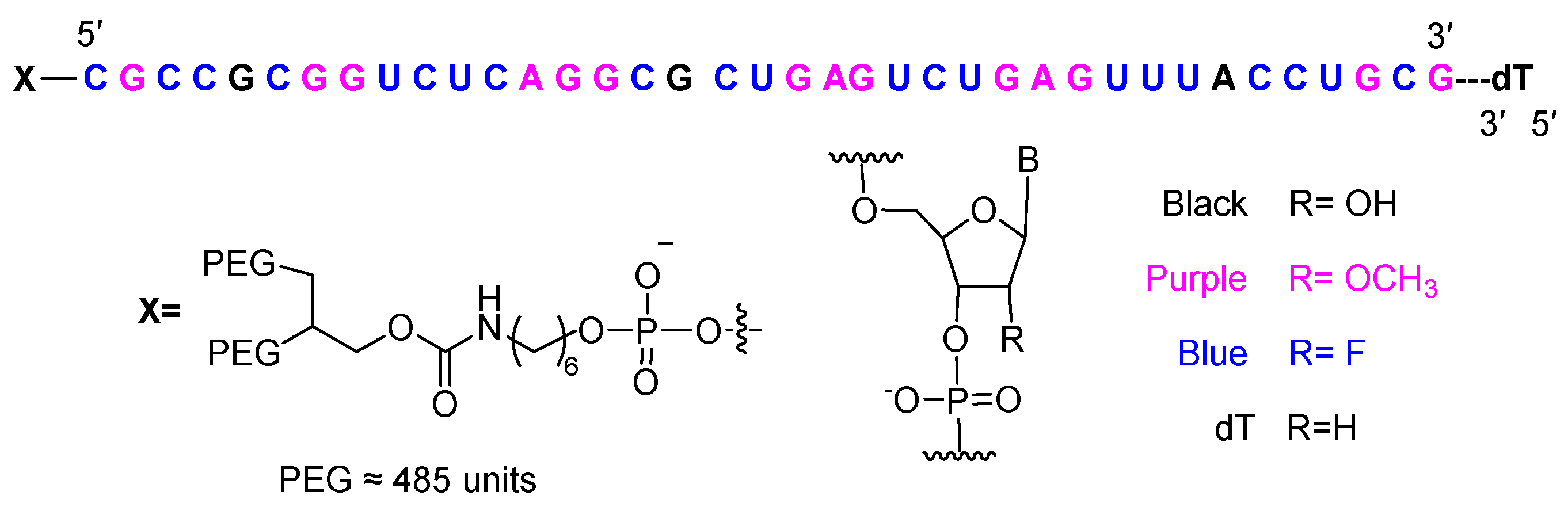2. Discussion
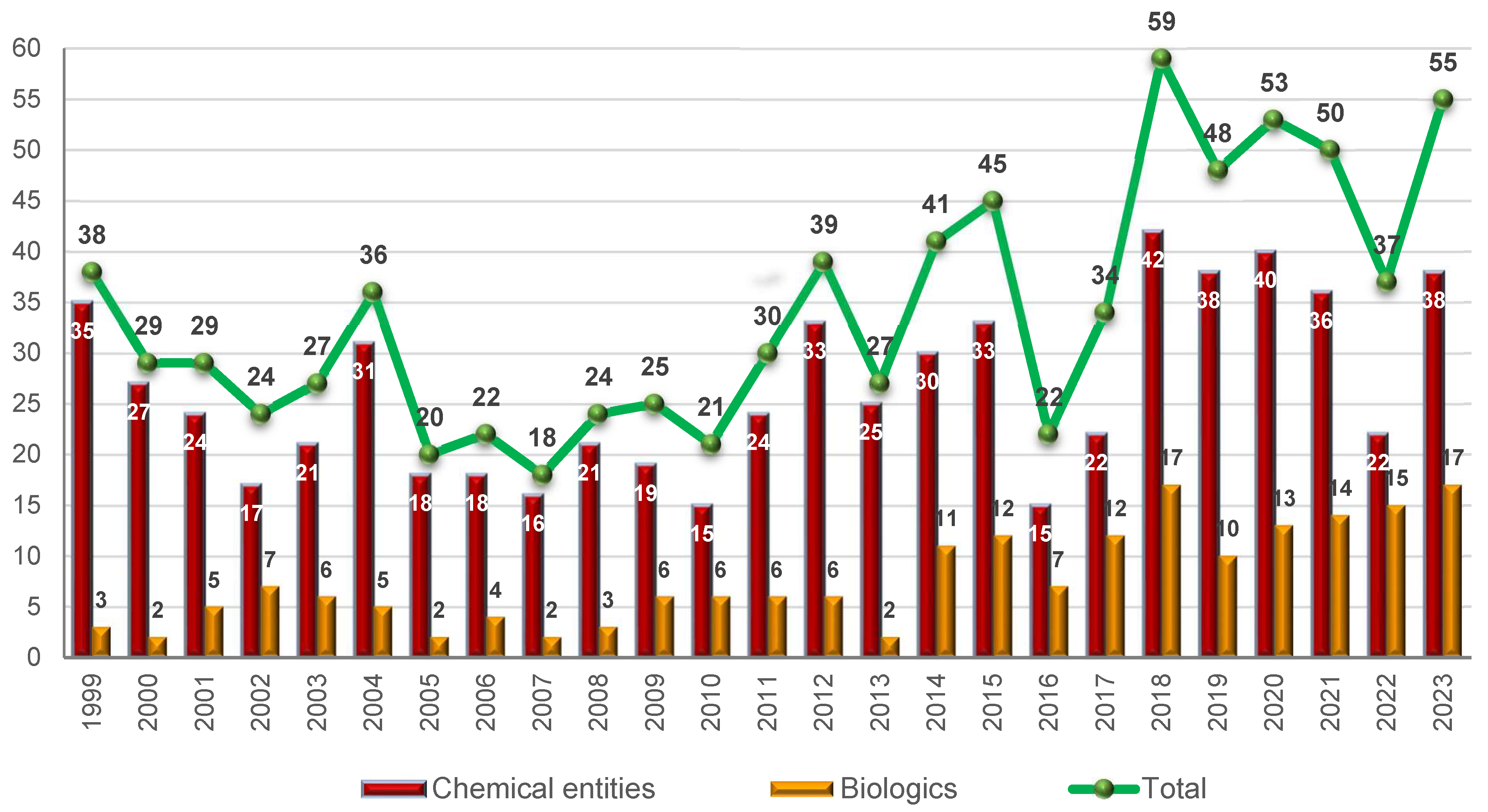
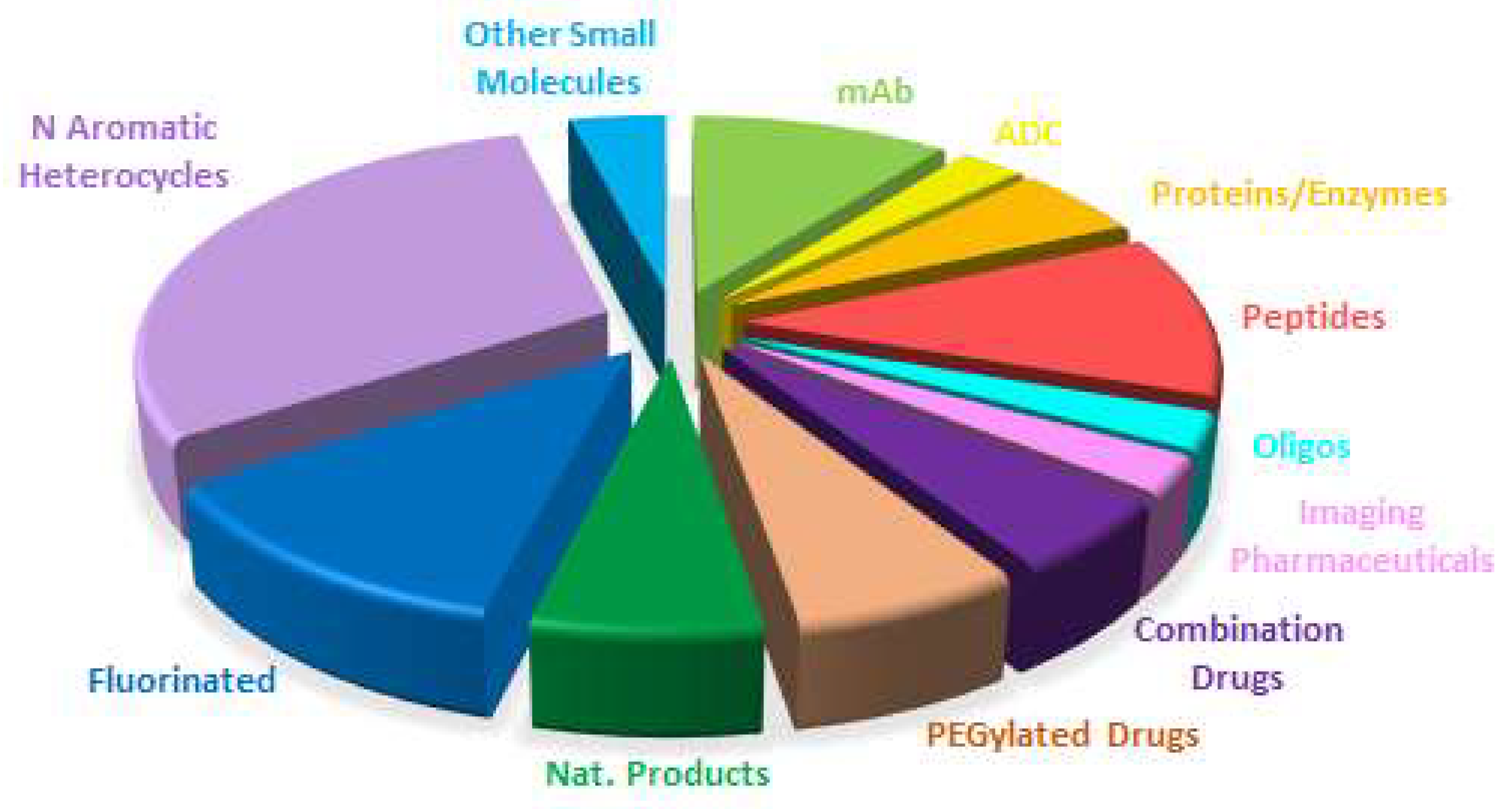
Table 1 shows the 17 biologics approved in 2023, of which 12 are monoclonal antibodies (mAbs), which represents the best year for this kind of drugs, and five enzymes/proteins (
Table 1).
Regarding biologics, and following the trend of recent years, 2023 has seen the approval of two bispecific mAbs. These drugs can simultaneously bind to two distinct epitopes on one antigen or to two different antigens. In this regard, authorization was given to glofitamab-gxbm (Columvi™), a bispecific CD20-directed CD3 T-cell engager, and elranatamab (Elrexfio™), which targets BCMA (a protein on myeloma cells) and CD3 (a protein on the immune system’s T cells). In 2022, three of the biologics approved showed bispecificity [teclistamab-cqyv (TecvayliTM), faricimab-svoa (VabysmoTM), and tebentafusp-tebn (KimmtrakTM)], while only one was approved in 2021 [amivantamab-vmjw (RybrevantTM)], 2018 [emicizumab (HemlibraTM)], and 2014 [blinatumomab (BlincytoTM)].
Last year, we highlighted the controversy around aducanumab-avwa (AduhelmTM), which was approved in 2021 for the treatment of Alzheimer’s disease, and whose effectiveness was under discussion. In 2023, Lecanemab (Leqembi™) has been approved for the same target. This drug has been developed by Biogen—the same company that manufactured aducanumab-avwa—in association with Eisai.
mAbs continue to be the most approved drug class, accounting for 22% (12 vs. 55) of all the drugs accepted by the FDA this year. In 2022, these drugs accounted for 24% (9 vs. 37). As in former years, cancer is the first target for mAbs. However, following the trend of previous years, mAbs have been approved for other targets such as Alzheimer’s disease, psoriasis, respiratory syncytial virus, ulcerative colitis, and myasthenia gravis, thus enlarging the application of these kinds of challenging drugs.
2023 has broken the streak regarding the approval of antibody-drug conjugates (ADCs) as none have been authorized, a situation that contrasts with previous years, in which up to 14 such drugs were accepted by the FDA.
This year, three enzyme replacement therapies have received the green light. Velmanase (Lamzede™) is the first such therapy approved for the treatment of the non-central nervous system manifestations of α-mannosidosis. Pegunigalsidase alfa (Elfabrio™), which is a PEGylated and crosslinked, chemically modified, recombinant human α-galactosidase A enzyme, is indicated for the treatment of Fabry disease. Finally, cipaglucosidase alfa-atga (Pombiliti™) provides an exogenous source of α-glucosidase for the treatment of Pompe disease.
TIDES (oligonucleotides and peptides), although belonging to the class of chemical entities, often show structural complexity similar to that of biologics. TIDES are chemically synthesized and their characterization and the profile of impurities allowed by regulatory agencies are similar to what is required for small molecules, thereby making their introduction into the market a real challenge. In 2023, nine TIDES (five peptides and four oligonucleotides) have been approved, which account for 16% (9 vs 55) of all the drugs authorized this year—almost the same proportion as in 2022 (15%) (5 vs 37), thus reinforcing the importance of these kinds of drugs. As is our custom in this yearly reports, we consider peptidomimetics with a high presence of amino acids or peptide bonds to be peptides.
The last few years have witnessed a double trend in the peptides approved by the FDA and other regulatory agencies. On the one hand, authorization has been given to large linear peptides with more than 30 amino acids, whose best exponent was the approval of tirzepatide (MounjaroTM) in 2022 for the treatment of type 2 diabetes and obesity. On the other hand, cyclic peptides of medium size often contain non-proteinogenic amino acids. In both cases, pending units of fatty acids or/and polyethyleneglycol (PEG) structures are present for increase the drug half-live. In 2023, three cyclic peptides with these characteristics have been approved.
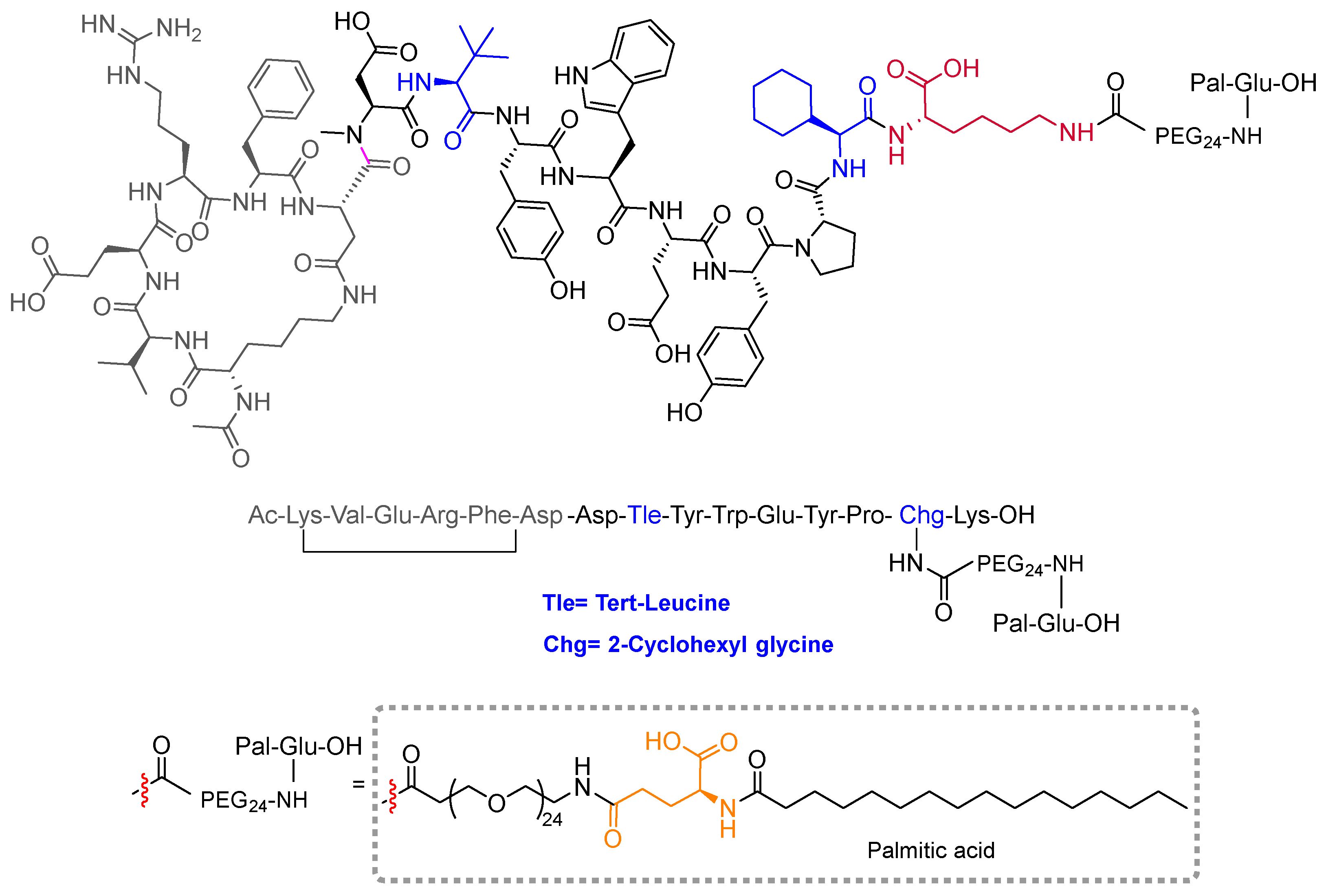
Zilucoplan (Zilbrysq TM) is recommended for the treatment of myasthenia gravis in adults who are anti-acetylcholine receptor antibody-positive. Zilucoplan binds to the protein complement component 5 (C5) and inhibits its cleavage into two fragments. It is a side chain-to-side chain homodetic cyclic peptide with a main backbone of 15 amino acids, all of them with L-configuration. The cyclic structure is at the N-terminal part between the side chains of Lys and Asp residues. A Lys residue situated at the C-terminal part bears a side chain containing a large PEG and a fatty structure (palmitic acid) linked by a γ-Glu residue. The combination of fatty acids linked through a γ-Glu residue to extend the half-life of the molecule in vivo is present in other peptides, including liraglutide, semaglutide, and tirzepatide, all of which were approved in recent years. The backbone of zilucoplan contains several non-proteinogenic amino acids such as NMe-Asp, tert-butylglycine or tert-leucine, aza-tryptophan, and cyclohexylglycine. The N-terminus is acylated and the C-terminus is a free carboxylic group.
Figure 2.
Structure of zilucoplan.
Figure 2.
Structure of zilucoplan.
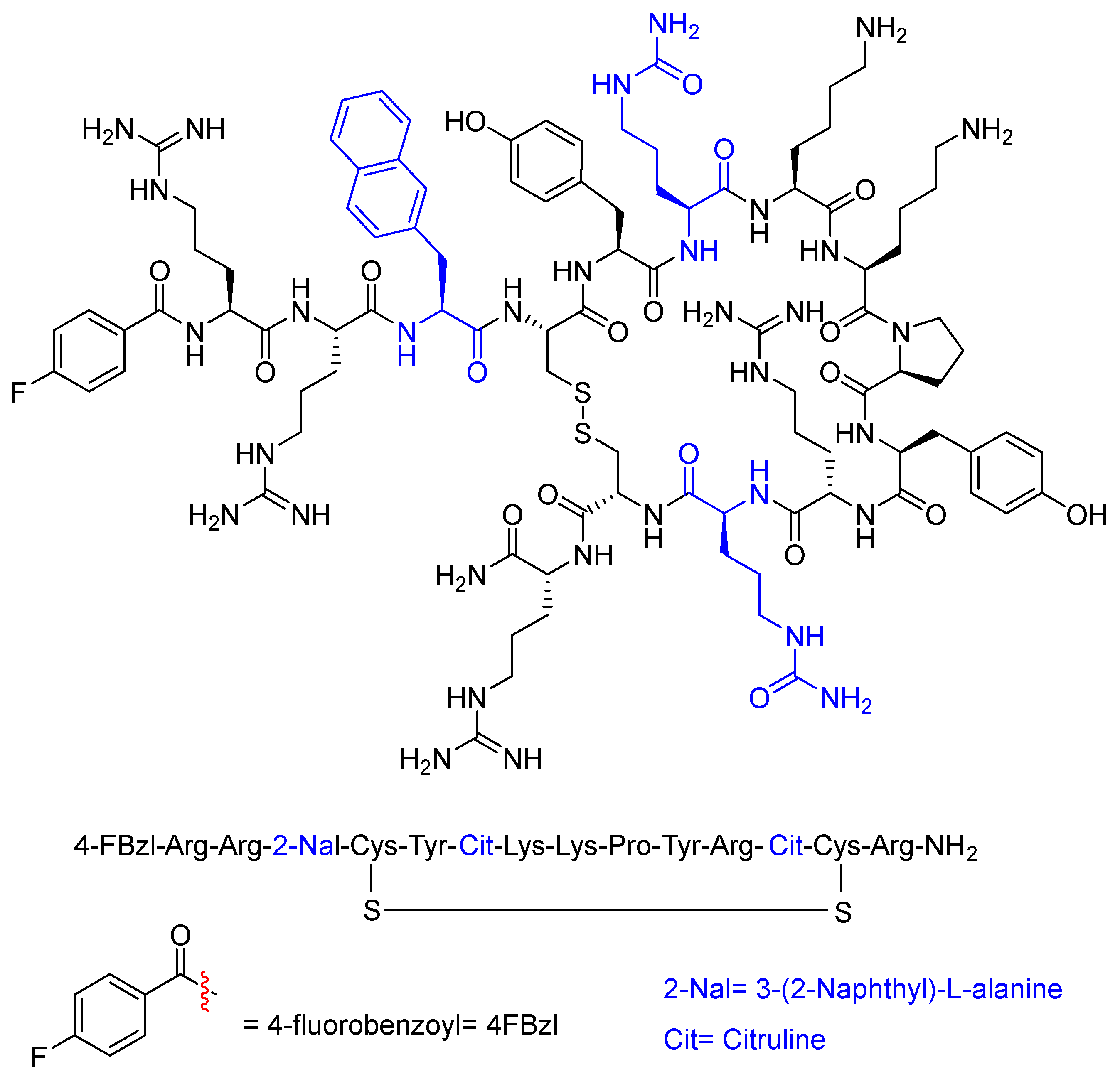
Motixafortide (AphexdaTM) has been approved for the treatment of multiple myeloma. It is a hematopoietic stem cell mobilizer and a CXCR4 antagonist. Motixafortide is a 14-amino acid cyclic heterodetic peptide formed by a disulfide bridge between two Cys residues. It is a cationic peptide with four Arg and two Lys residues. In addition, it contains two Cit residues and one naphthylalanine. The N-terminus is in the form of p-fluorobenzoyl and the C-terminus is amidated.
Figure 3.
Structure of motixafortide.
Figure 3.
Structure of motixafortide.
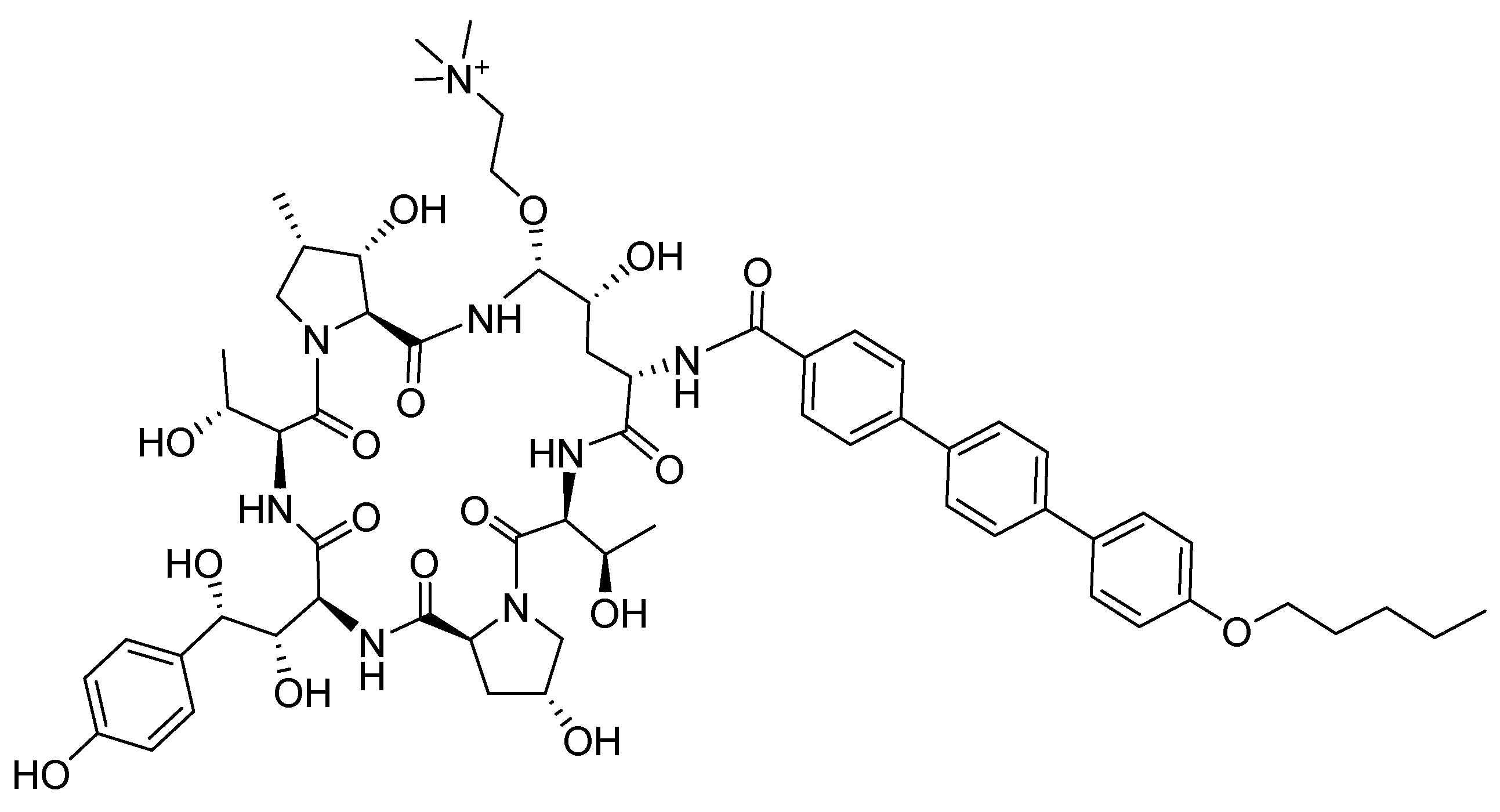
Rezzayo (RezafunginTM) is an antifungal drug of the echinocandin family indicated for the treatment of candidemia and invasive candidiasis. It is a semi-synthetic hexapeptide side chain to tail homodetic cyclic peptide. It is a highly hydroxylated peptide that, in addition to having two Thr residues, contains hydroxyproline (2), hydroxyornitine, and hydroxyhomotyrosine residues. The side chain of the Orn derivative contains a choline moiety. The cycle is formed by the C-terminus of one of the hydroxyprolines and the δ-amino of the Orn. The N-amino is in the form of pentoxyphenylphenylbenzamide.
Figure 4.
Structure of rezzayo.
Figure 4.
Structure of rezzayo.
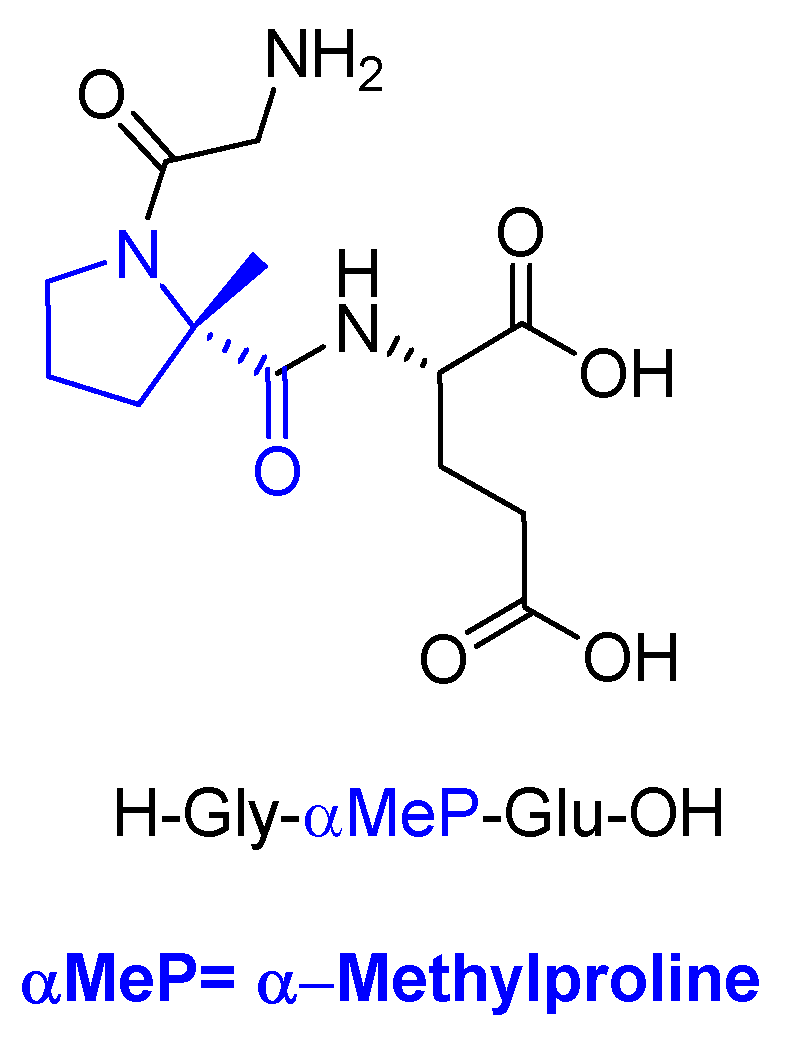
Trofinetide (Daybue TM) is an orally available tripeptide containing two proteinogenic amino acids, Gly and Glu, and a resisue of α-methylproline and it is used for the treatment of Rett syndrome. It is the methylated analog of the N-terminal tripeptide (Gly-Pro-Glu) of the insulin-like growth factor 1 protein.
Figure 4.
Structure of trofinetide.
Figure 4.
Structure of trofinetide.
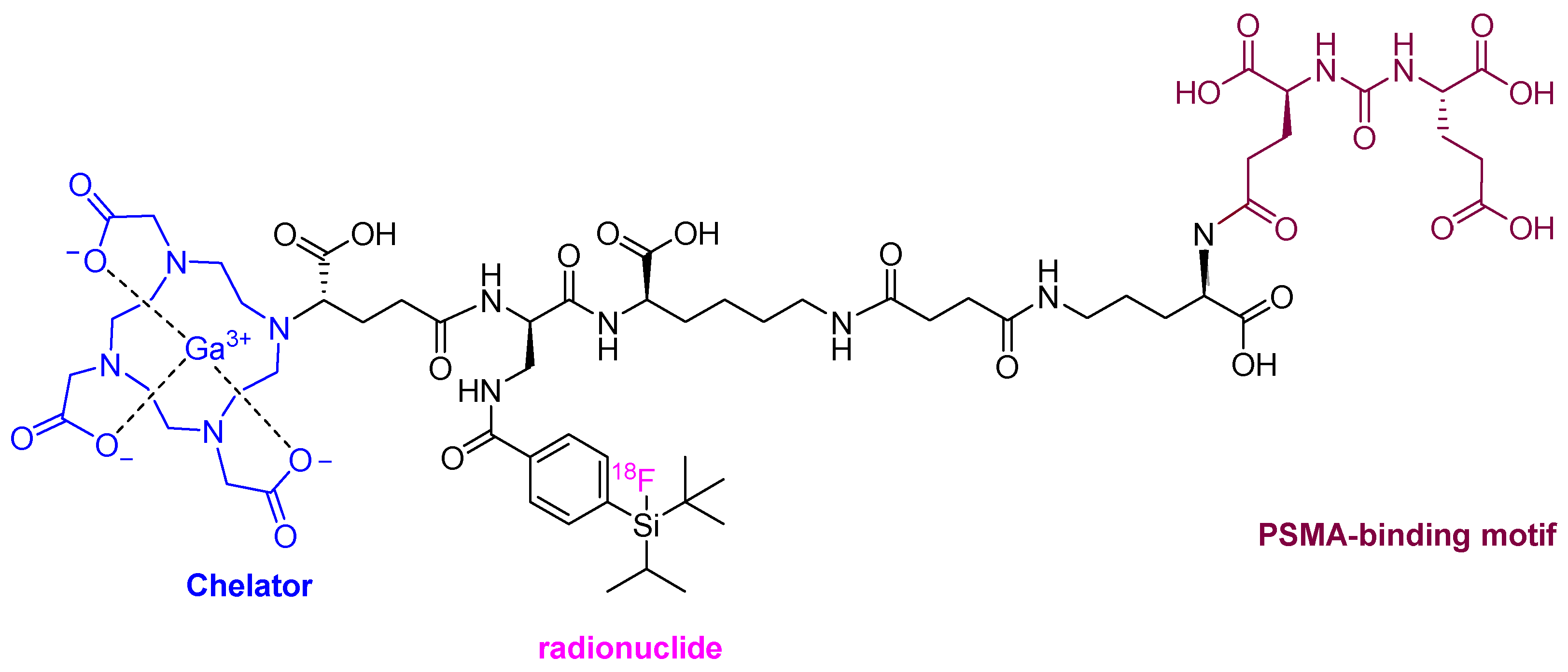
Following last year’s trends, a radiopharmaceutical agent for use as positron emission tomography (PET) imaging for prostate cancer has been approved this year. Flotufolastat F-18 (PoslumaTM) is the fourth prostate-specific membrane antigen (PSMA)-targeted PET imaging drug to be authorized after lutetium (177Lu) vipivotide tetraxetan (PluvictoTM) in 2022, piflufolastat F-18 (PylarifyTM) in 2021, and Ga-68 PSMA-1 in 2020.
The chemical structure of flotufolastat F-18 differs from the previous PSMA-targeted drugs, which were based on the urea of Glu and Lys. In the case of this year’s drug, the urea is formed from the α-amino of two Glu residues that are linked to a DOTAGA complex with non-radioactive Ga and radioactive F-18 covalently bound to a silicon moiety, through a linker formed of four residues: D-Orn, D-Lys, succinic acid and D-diaminopropionic acid (Dap).
Figure 5.
Structure of Flotufolastat F-18.
Figure 5.
Structure of Flotufolastat F-18.
With the FDA approval of four oligonucleotide-based drugs this year, there are now 19 such drugs available, 16 of which have emerged since 2016, the year that marked a new era for oligos as drugs.
Nedosiran (RivflozaTM) is recommended for the treatment of primary hyperoxaluria. It is a double-stranded small interfering RNA (siRNA) containing four units of N-acetyl-D-galactosamine (GalNAc) residues, which are responsible for targeting lactate dehydrogenase A (LDHA) in hepatocytes. Nedosiran is formed by 36 and 22 ribonucleotides for the sense and antisense strands, respectively. It has a total of six tiophosphate linkages. In addition to the GalNAc residues, Nedosiran contains 19 2′-F-ribonucleotide to improve the stability of the double-strand. The remaining ribonucleotides are 2′-methoxy.
Figure 6.
Structure of nedosiran.
Figure 6.
Structure of nedosiran.
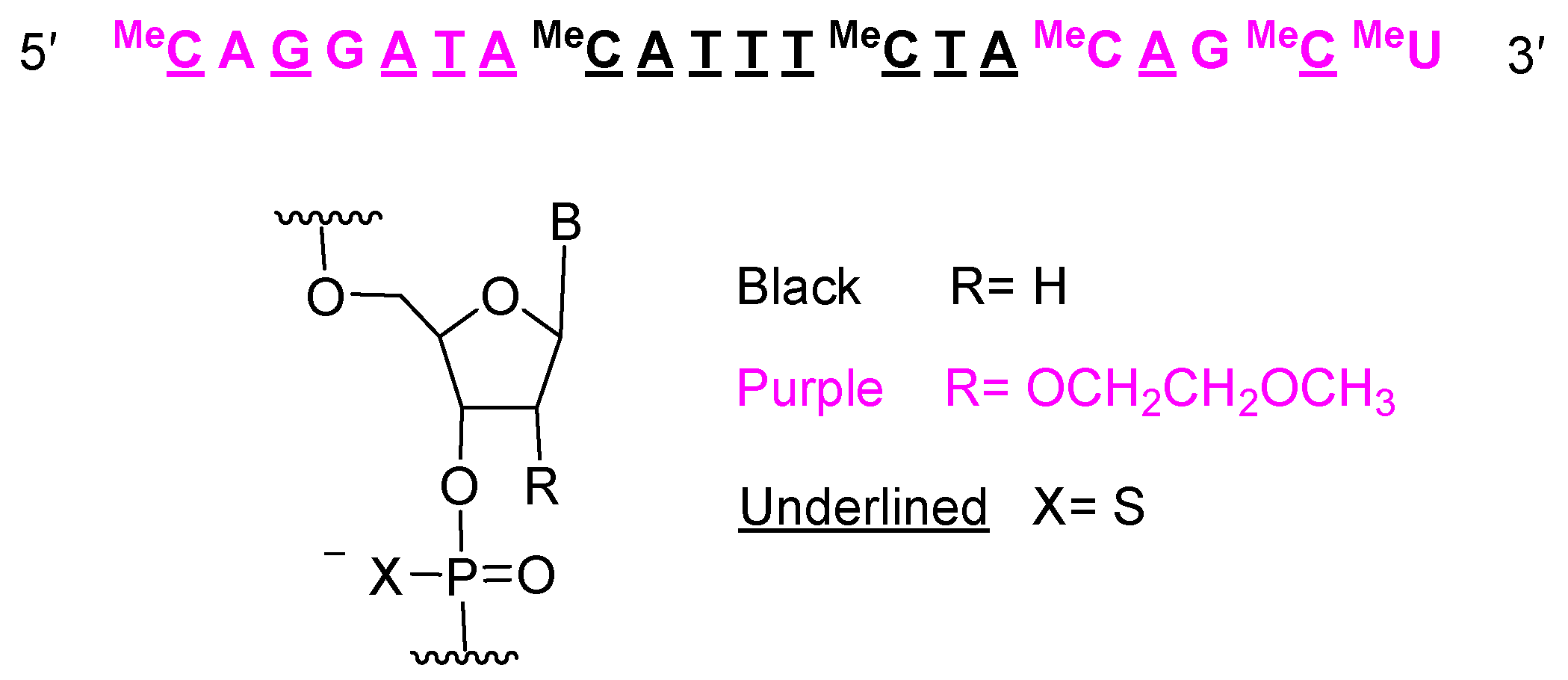
The remaining oligonucleotides approved in 2023 are single-strain antisense oligonucleotides. Tofersen (QalsodyTM) is used for the treatment of amyotrophic lateral sclerosis (ALS). It contains 15 tiophosphate linkages and is formed by 20 nucleotides: 10 metoxyethoxy and 10 deoxy.
Figure 7.
Structure of tofersen.
Figure 7.
Structure of tofersen.
Eplontersen (Wainua TM) is indicated for the treatment of transthyretin-mediated amyloidosis. It contains 11 tiophosphate linkages and is formed again by 20 nucleotides (10 metoxyethoxy and 10 deoxy) and it is terminated by a trivalent N-acetylgalactosamine (GalNAc) [Enhanced Stabilization Chemistry (ESC)], which mediates the binding and internalization of the drug by hepatocytes. This system is present in many oligonucleotides that have recently reached the market.
Figure 8.
Structure of eplontersen.
Figure 8.
Structure of eplontersen.
Finally, avacincaptad pegol (IzervayTM) is recommended for the treatment of age-related macular degeneration. Containing 39 nucleotides, this modified aptamer incorporates an inverted thymidine nucleotide to cap the 3′ end, 2′-fluorine (21), and O-methyl (14) modified nucleotides, together with hydroxy ones (3). It is terminated in two PEG chains, each with approximately 485 units. These modifications extend the half-life of the drug in vivo. Avacincaptad pegol functions as a chemical antibody against complement C5 and inhibits the cleavage of complement C5 into its two fragments—a similar mode of action to that of the peptide zilucoplan, which has also been approved this year.
Figure 9.
Structure of avacincaptad pegol.
Figure 9.
Structure of avacincaptad pegol.
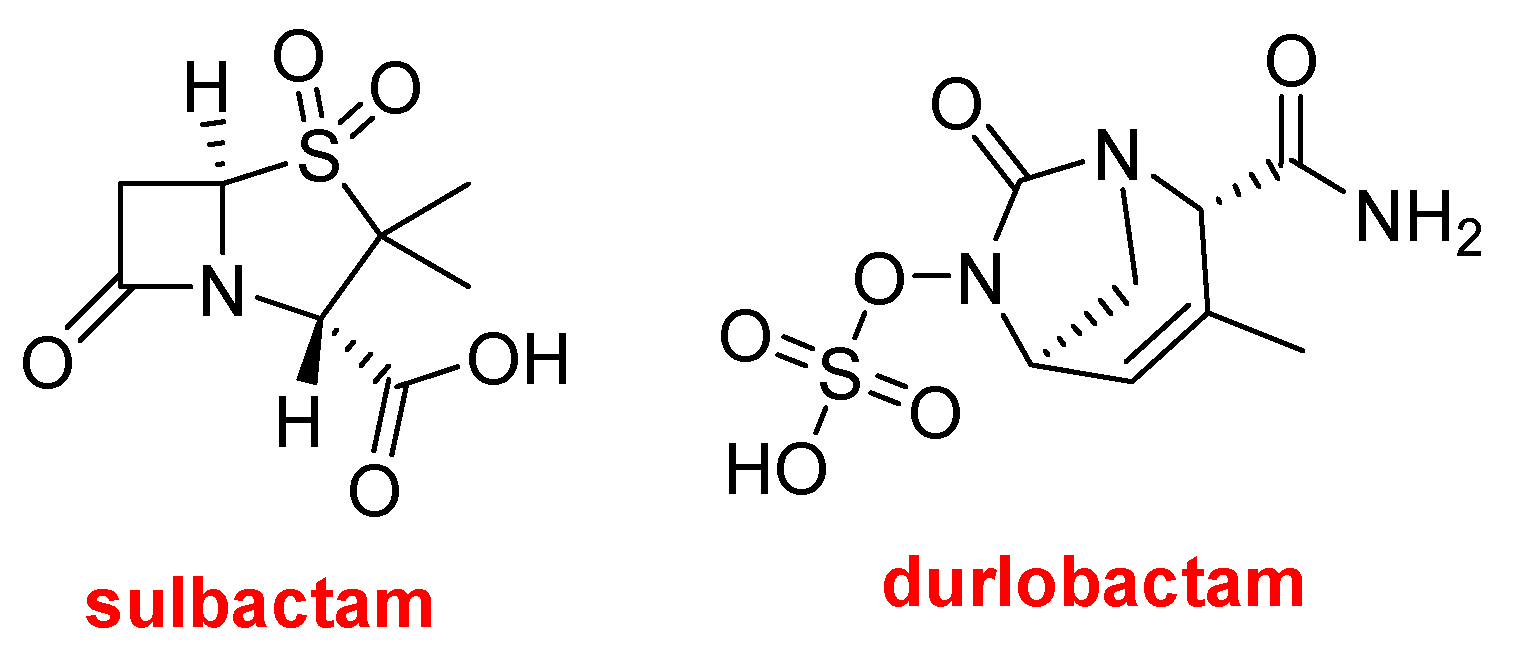
This year, the FDA has also authorized three drugs that contain more than one active pharmaceutical ingredient (API). XacduroTM, which contains sulbactam and durlobactam, is used for the treatment of bacterial pneumonia caused by Acinetobacter baumannii-calcoaceticus complex. Sulbactam is a β-lactam antibacterial and β-lactamase inhibitor, and durlobactam is a β-lactamase inhibitor.
Figure 10.
Structures of sulbactam and durlobactam.
Figure 10.
Structures of sulbactam and durlobactam.
Sulbactam was first approved in 1986 and is already used in combination with ampicillin (UnasynTM) and cefoperazone (SulperazonTM).
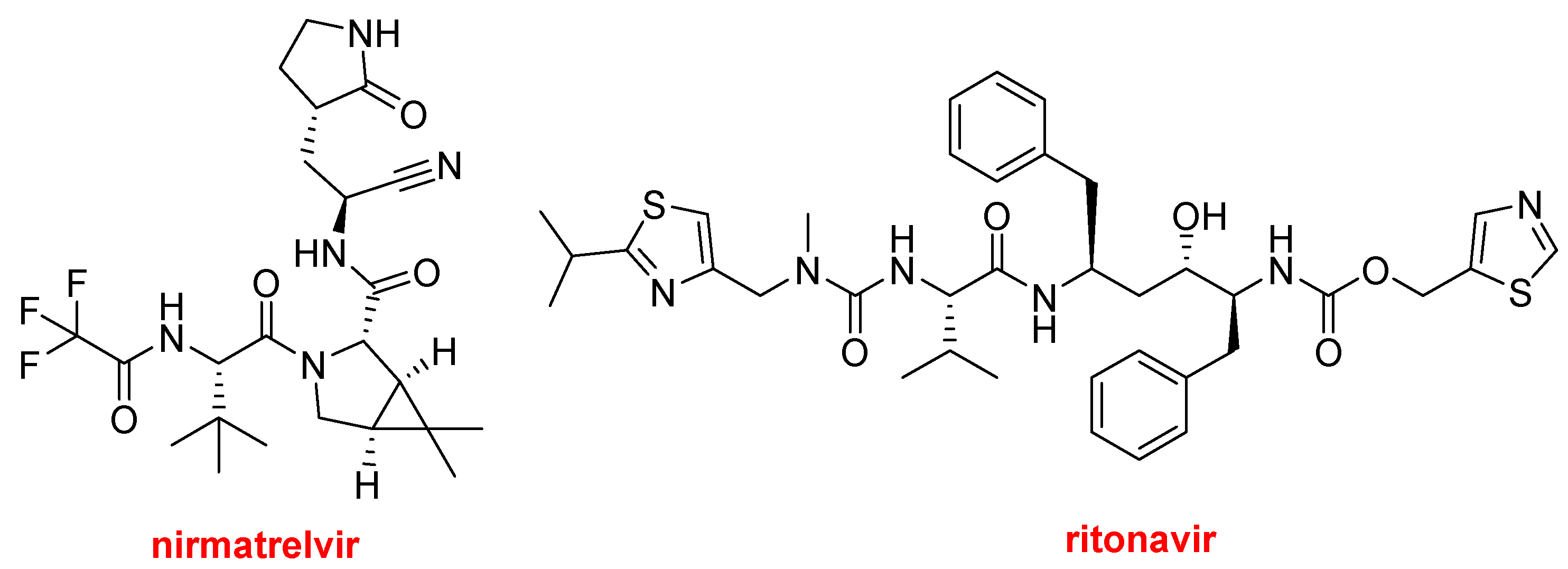
PaxlovidTM, which contains nirmatrelvir and ritonavir, has been officially approved this year to treat mild-to-moderate COVID-19 to avoid progression to a severe disease. This drug already received emergency authorization in 2021 and 2022 in the USA and other countries, respectively, for this purpose. There has been some controversy regarding its efficacy and the COVID-19 variants most likely to be susceptible to treatment.
Figure 11.
Structures of nirmatrelvir and ritonavir.
Figure 11.
Structures of nirmatrelvir and ritonavir.
Nirmatrelvir is a protease inhibitor used as an antiviral drug. Ritonavir (Norvir TM), approved in 1996, is also a protease inhibitor that is used as an antiretroviral in combination with other drugs to treat HIV/AIDS and hepatitis C, among others. In 2014 and 2015, ritonavir, ombitasvir, and paritaprevir, in combination with dasabuvir or ribavirin, were approved to treat the hepatitis C virus.
In PaxlovidTM, ritonavir slows the metabolism of nirmatrelvir via cytochrome enzyme inhibition and therefore reinforces the role of nirmatrelvir, which is the main drug.
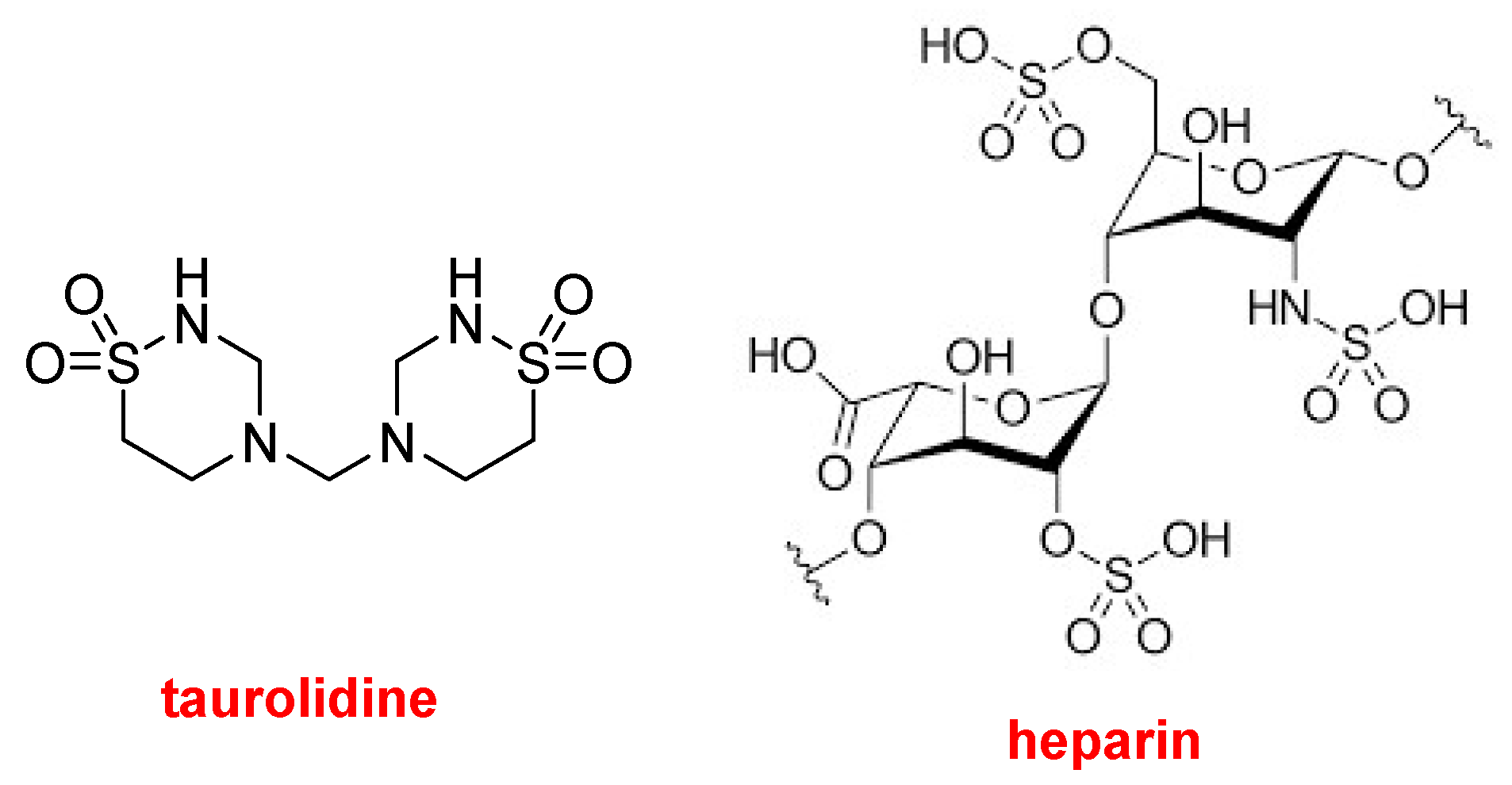
DefencathTM contains taurolidine, a thiadiazinane antimicrobial compound, and the glycosaminoglycan polymer heparin, an anticoagulant. It is used as a catheter lock solution for central venous catheter instillation. Taurolidine is derived from taurine, which is an endogenous amino acid.
Figure 12.
Structures of taurolidine and heparin.
Figure 12.
Structures of taurolidine and heparin.
Twenty-two combination drugs have been approved by the FDA between 2016 and 2023. This figure illustrates the relevance of this strategy for drug discovery, which often takes advantage of “old” drugs already approved by regulatory agencies.
Natural products are one of the most important sources of inspiration for developing new drugs. In addition to the biologics, TIDES, sulbactam, taurolidine, and heparin cited above, six more drugs approved this year by the FDA found their roots in the natural product universe.
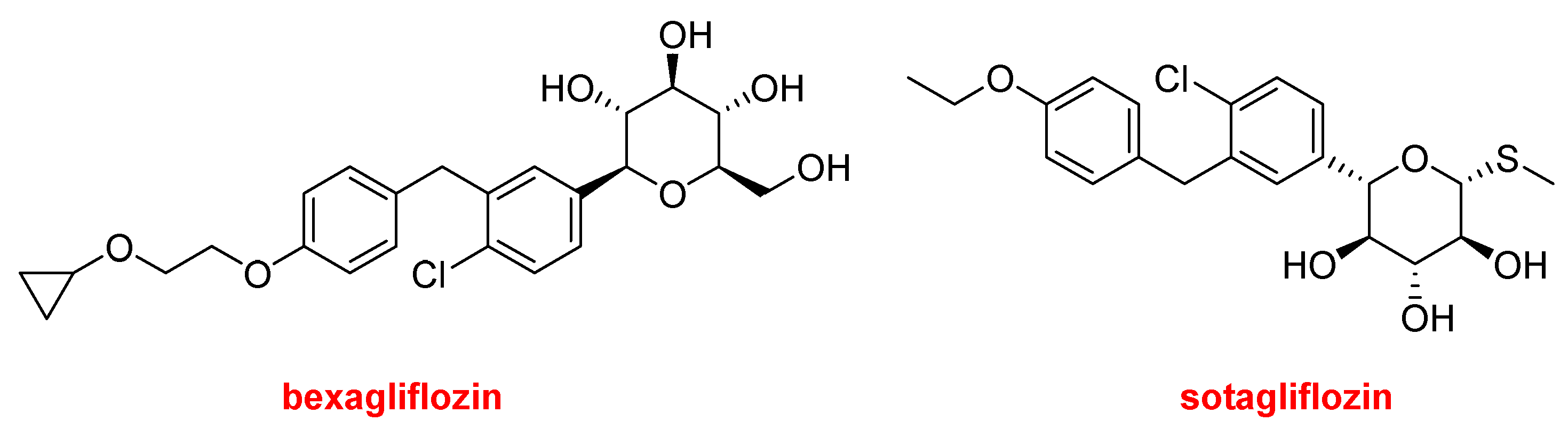
Bexagliflozin (BrenzavvyTM) and Sotagliflozin (Inpefa TM) are two related inhibitors of sodium-glucose cotransporter 2 (SGLT2).
Figure 14.
Structures of bexagliflozin and sotagliflozin.
Figure 14.
Structures of bexagliflozin and sotagliflozin.
The former has been authorized for improving glycemic control in adults with type 2 diabetes in combination with exercise and the corresponding diet. In contrast, sotagliflozin has been approved only this year by the FDA for reducing the risk of death due to heart failure. However, in Europe, sotagliflozin (ZynquistaTM) was approved in 2019 for the treatment of type 1 diabetes, but it was withdrawn in 2022. The FDA has refused its approval in combination with insulin for the treatment of type 1 diabetes.
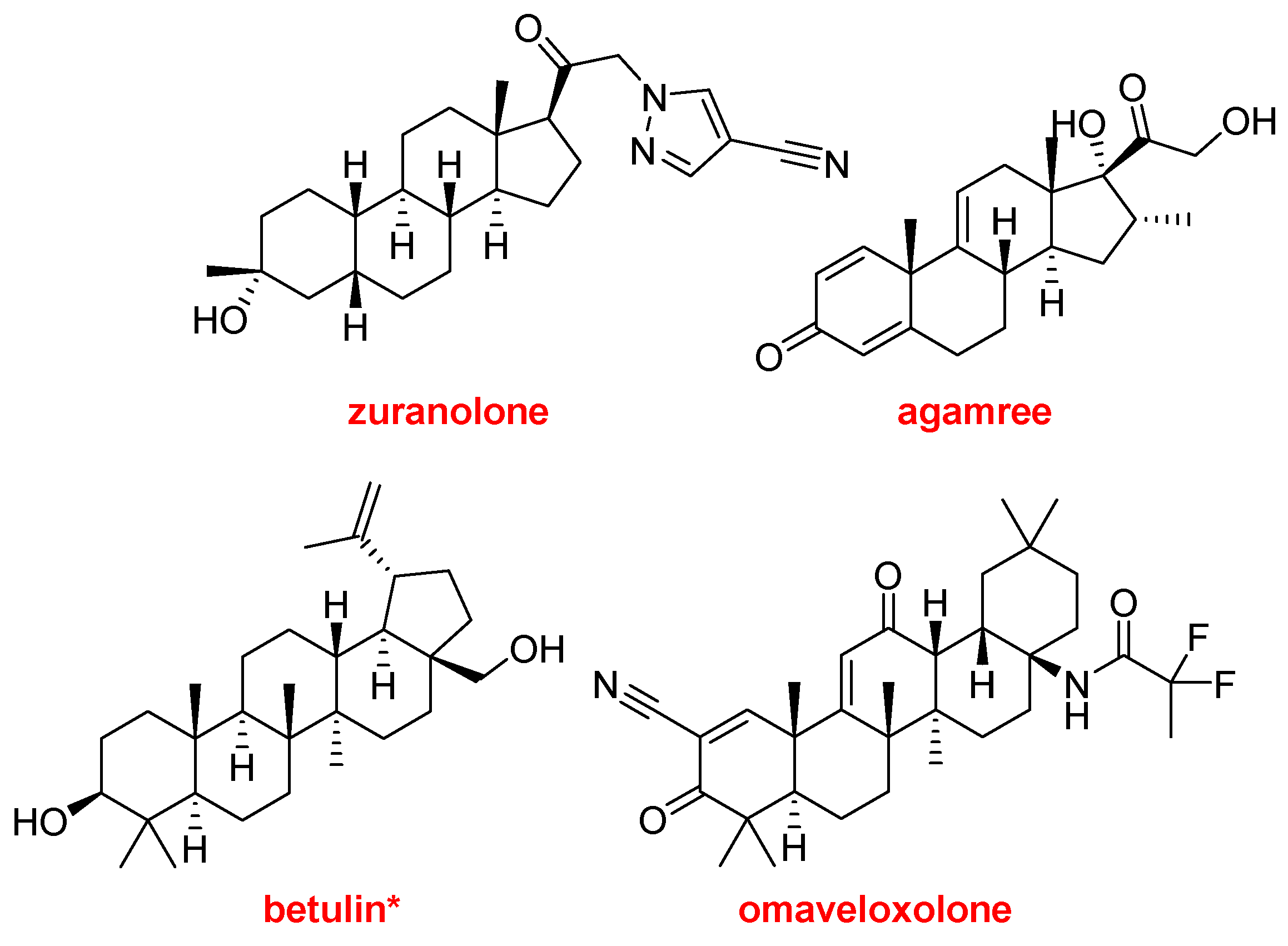
Zuranolone (ZurzuvaeTM) and agamree (VamoroloneTM) belong to the steroid family and have been approved for the treatment of postpartum depression and Duchenne muscular dystrophy, respectively.
Birch triterpenes (FilsuvezTM) could be considered the typical natural product drug, because it is a botanical drug substance composed of a mixture of pentacyclic triterpenes: botulin (72-88%), lupeol (2.4-5.7%), betulinic acid (2.6-4.2%), erythrodiol (0.5-1.2%), oleanolic acid (0.3-0.8%). It is used as a topical gel indicated for the treatment of partial-thickness wounds with junctional and dystrophic epidermolysis bullosa (JEB and DEB). This is the first treatment approved for wounds associated with JEB, which is considered a rare disease.
Omaveloxolone (Skyclarys
TM) is recommended for the treatment of Friedrich’s ataxia. The FDA has granted Orphan Drug. Fast Track, Priority Review, and Rare Pediatric Disease designations to omaveloxolone [
5].
Steroids and triterpenes are the most important class of small molecule-based drugs, as is reflected by their approval year after year.
Figure 15.
Structures of zuranolone, agamree, betulin -main component of birch triterpenes-, and omaveloxolone.
Figure 15.
Structures of zuranolone, agamree, betulin -main component of birch triterpenes-, and omaveloxolone.
Every year, we highlight the importance of F in the drug discovery arena, and 2023 has been no exception. Thus, in addition to two peptides (motixafortide and flotufolastat F-18), two oligonucleotides (nedosiran and avacincaptad pegol), nirmatrelvir as part of PaxlovidTM, and omaveloxolone, eight more APIs containing F have been authorized. Thus, 25% (14 vs. 55) of all approved drugs contain F, and if biologics are omitted from the calculation, this figure increases to 37% (14 vs. 38).
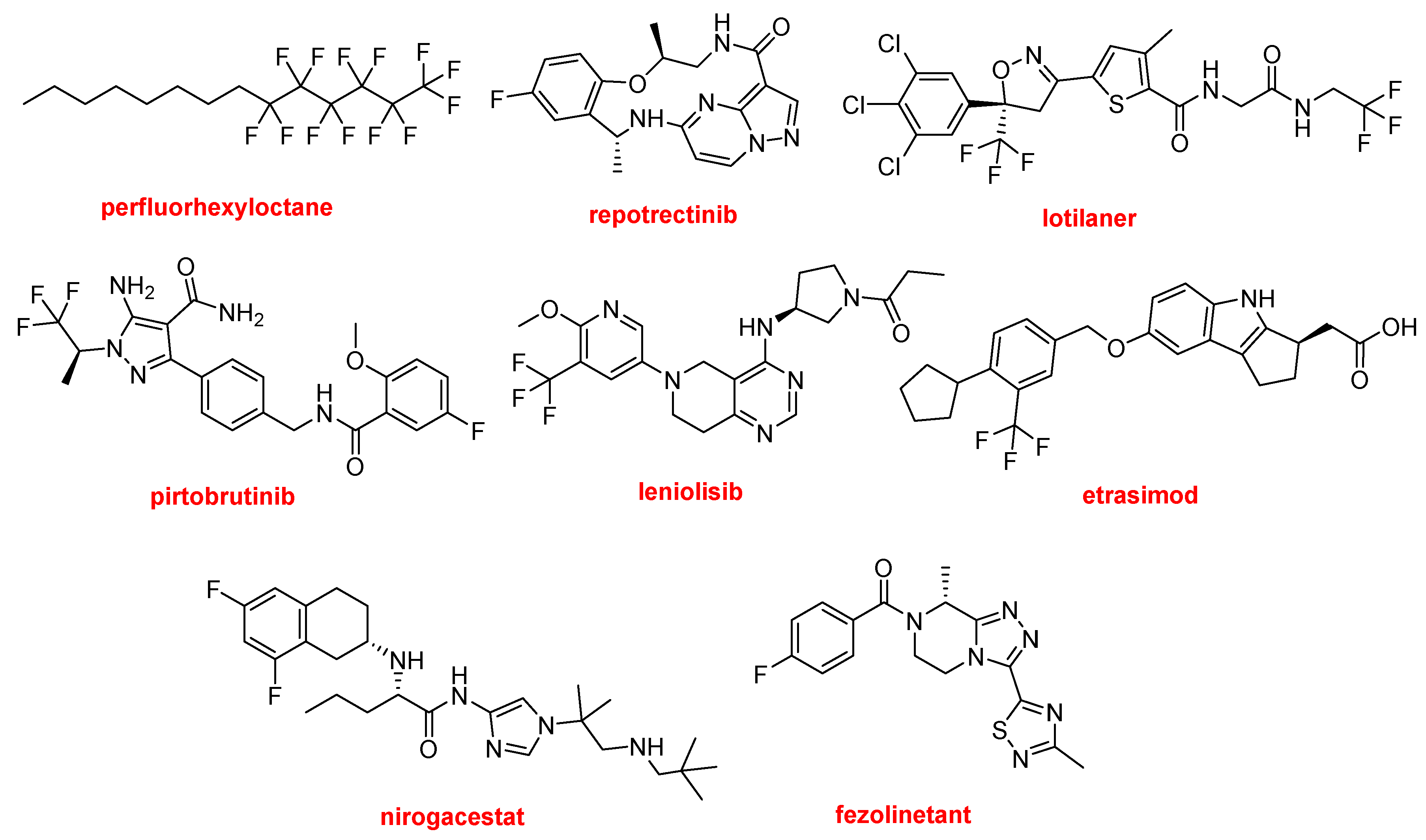
Possibly the simplest but also one of the most interesting F-containing drugs is perfluorhexyloctane (MieboTM). This semi-fluorinated alkane is used to treat dry eye disease. The presence of F makes an alkane druggable.
Repotrectinib (Augtyro TM) is the only macrocycle approved this year for the treatment of non-small cell lung cancer. It is an inhibitor of proto-oncogene tyrosine-protein kinase ROS1 and of the tropomyosin receptor tyrosine kinases (TRKs) TRKA, TRKB, and TRKC.
Lotilaner (XdemvyTM) is an antiparasitic drug used to treat inflammation of the eyelid caused by infestation by the tiny mites Demodex. It contains two CF3 groups in an extended polyheterocyclic structure, which is a common feature of other F-containing drugs.
There are three drugs containing a CF3 moiety. Pirtobrutinib (JaypircaTM) is indicated to treat mantle cell lymphoma. It is a Bruton’s tyrosine kinase (BTK) inhibitor, acting on B cell lymphocyte proliferation and survival. In addition to the CF3 group, pirtobrutinib contains a F phenyl moiety. Leniolisib (JoenjaTM) is another kinase inhibitor used to treat activated phosphoinositide 3-kinase delta syndrome. Etrasimod (VelsipityTM) is an immune modulator recommended for the treatment of ulcerative colitis. Nirogacestat (OgsiveoTM), which contains a difluorophenyl moiety, is a selective γ-secretase inhibitor used to treat desmoid tumors. Finally, fezolinetant (VeozahTM) is indicated for the treatment of vasomotor symptoms (hot flashes) caused by menopause.
Figure 16.
Structures of perfluorhexyloctane, repotrectinib, lotilaner, pirtobrutinib, leniolisib, etrasimod, nirogacestat, and fezolinetant, all F-containing APIs.
Figure 16.
Structures of perfluorhexyloctane, repotrectinib, lotilaner, pirtobrutinib, leniolisib, etrasimod, nirogacestat, and fezolinetant, all F-containing APIs.
Six of the seven F-containing drugs, as well as nirmatrelvir and ritonavir present in PaxlovidTM, could be considered extended polyheterocyclics with N as major hetereoatom. In addition to these nine APIs, there are nine more from the same group, which represents 33% (18 vs. 55) of all approved drugs.
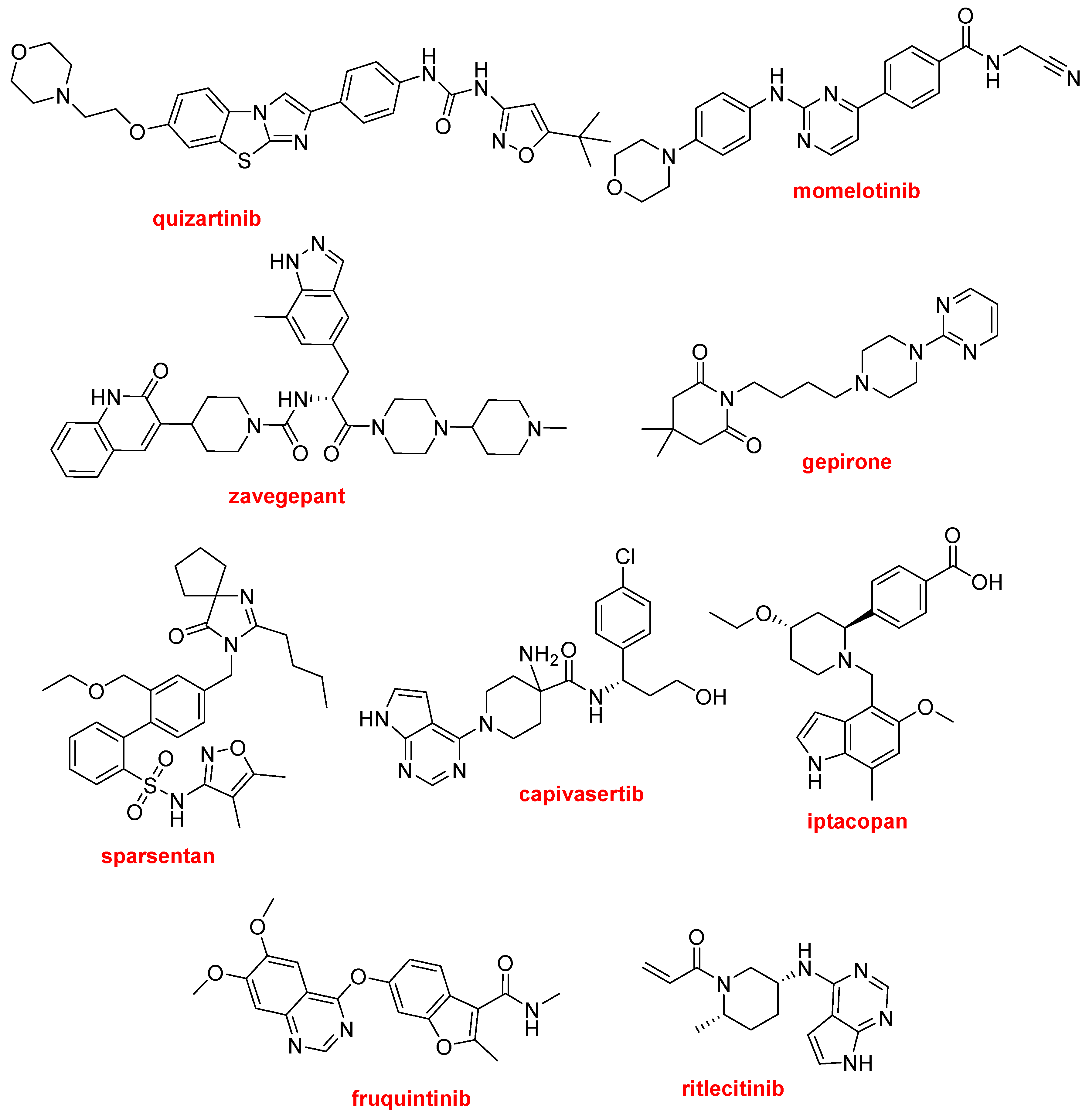
Quizartinib (Vanflyta TM), a clear example of this kind of molecule, is indicated for the treatment of acute myeloid leukemia. It is a tyrosine kinase inhibitor, targeting the proto-oncogene FLT3 or CD135.
Momelotinib (OjjaaraTM) is a Janus kinase inhibitor used for the treatment of myelofibrosis. Like the previous drug, it has a morpholine moiety at one end.
Zavegepant (ZavzpretTM) is a calcitonin gene-related peptide receptor antagonist indicated for the treatment of migraine.
Gepirone (Exxua TM) is a clear example of the difficulty of marketing a new drug. It was first synthesized in 1986 and was rejected three times by the FDA before its approval in 2023 for the treatment of major depressive disorder. Gepirone is a partial agonist of the serotonin 5-HT1A receptor and one of its metabolites is an α2-adrenergic receptor antagonist.
Sparsentan (FilspariTM), the only sulfonamide approved this year, is indicated for the treatment of primary immunoglobulin A nephropathy. Sparsentan is an endothelin and angiotensin II receptor antagonist.
Capivasertib (TruqapTM) is used in combination with fulvestrant (FaslodexTM) for the treatment patients with hormone receptor-positive, human epidermal growth factor receptor 2-negative breast cancer with metastatic disease.
Iptacopan (FabhaltaTM), a complement factor B inhibitor, is approved for paroxysmal nocturnal hemoglobinuria.
Fruquintinib (FruzaqlaTM) is a kinase inhibitor indicated for the treatment of metastatic colorectal cancer.
Ritlecitinib (LitfuloTM) is a kinase inhibitor, acting on Janus kinase 3 and tyrosine kinase, used for the treatment of severe hair loss (alopecia areata).
Figure 17.
Structures of quizartinib, momelotinib, zavegepant, gepirone, sparsentan, capivasertib, iptacopan, fruquintinib, and ritlecitinib, extended polyheterocyclics with N as major hetereoatom.
Figure 17.
Structures of quizartinib, momelotinib, zavegepant, gepirone, sparsentan, capivasertib, iptacopan, fruquintinib, and ritlecitinib, extended polyheterocyclics with N as major hetereoatom.
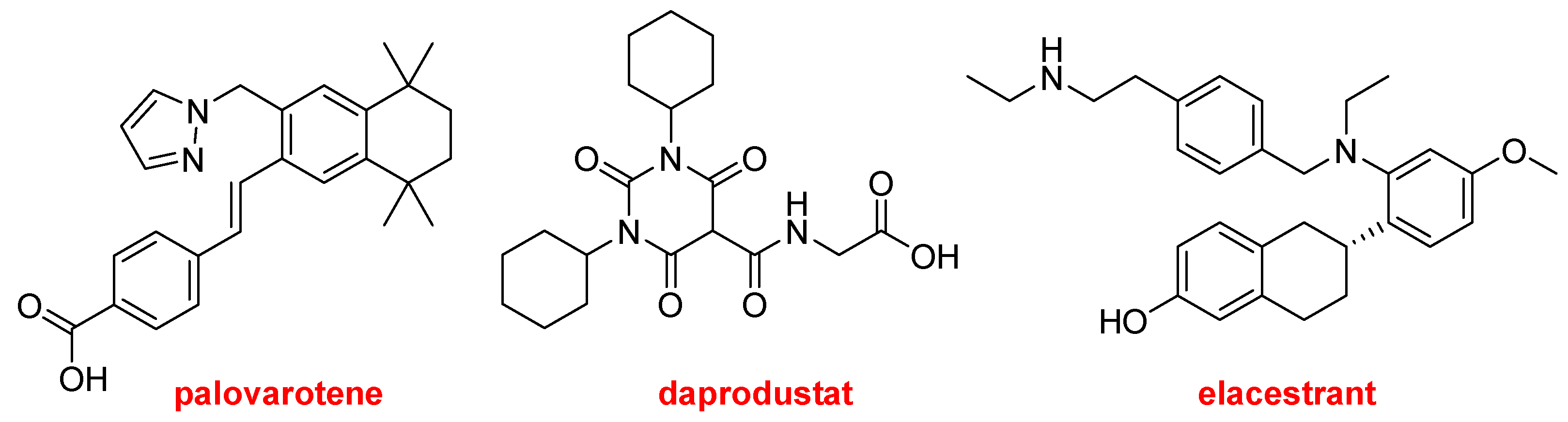
Palovarotene (SohonosTM) and daprodustat (Duvroq TM) also contain at least one N-heterocycle. The former is indicated to reduce the volume of new heterotopic ossification in adults and pediatric patients with fibrodysplasia ossificans progressiva. It is a highly selective retinoic acid receptor gamma (RARγ) agonist. Daprodustat, a hypoxia-inducible factor prolyl hydroxylase inhibitor, is indicated for the treatment of anemia resulting from chronic kidney disease.
Elacestrant (OrserduTM) is an estrogen receptor antagonist approved for the treatment of breast cancer.
Figure 18.
Structures of palovarotene, daprodustat, and elacestrant.
Figure 18.
Structures of palovarotene, daprodustat, and elacestrant.