Submitted:
03 January 2024
Posted:
04 January 2024
You are already at the latest version
Abstract
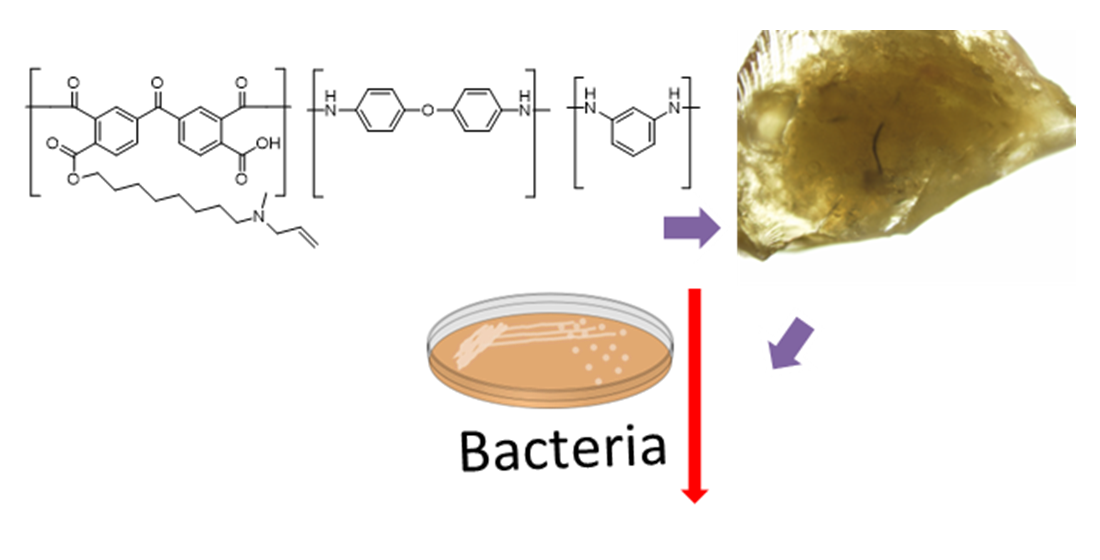
Keywords:
1. Introduction
2. Materials and Methods
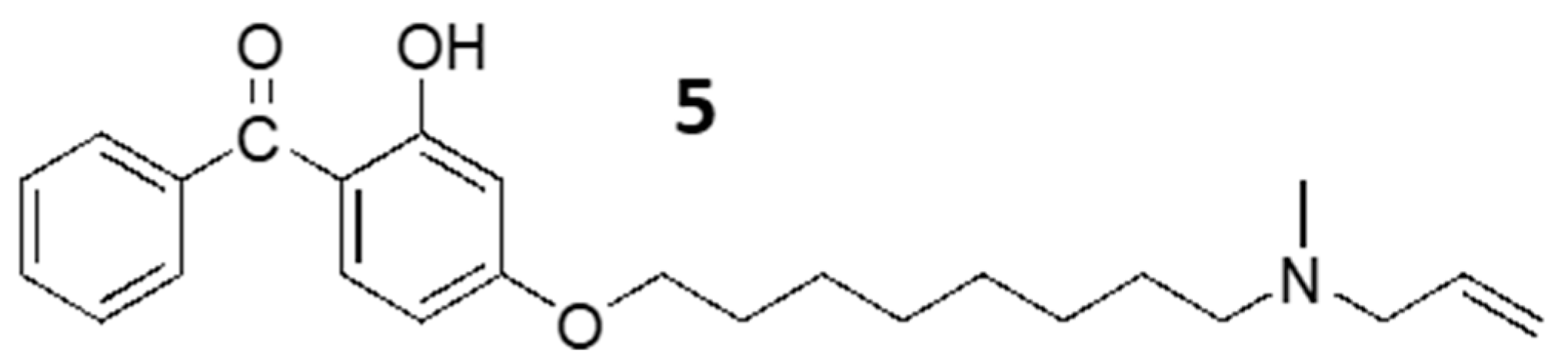
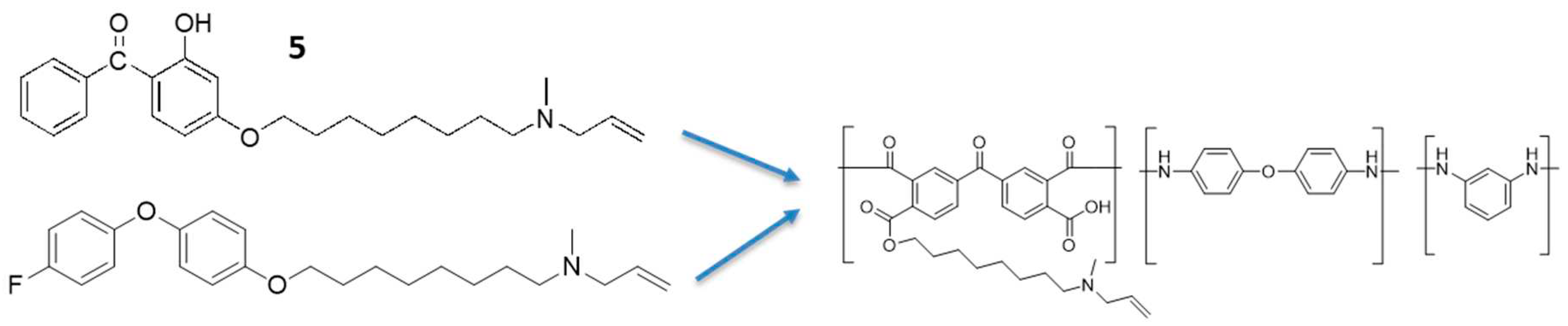
2.1. Synthesis of Benzophenone Derivative, 5
2.2. Benzophenone N-Allylamine, 5
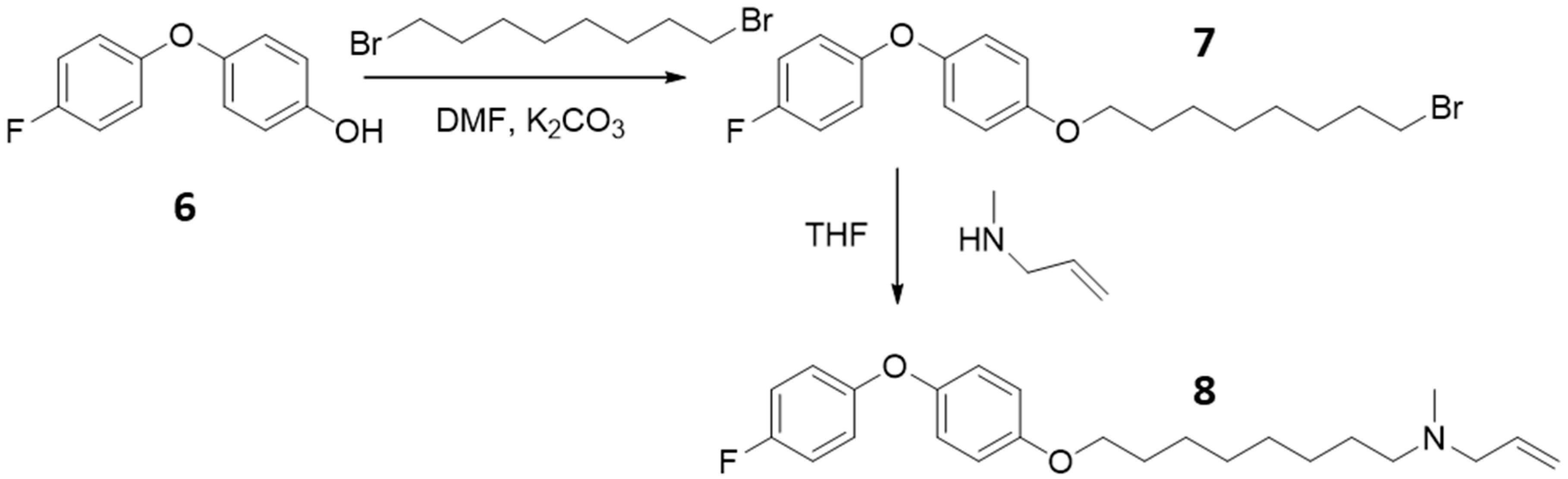
2.3. Synthesis of Alkylated Phenol Bromide Compounds, 7
2.4. Synthesis of Diphenyl Ether N-Allyl Compounds, 8
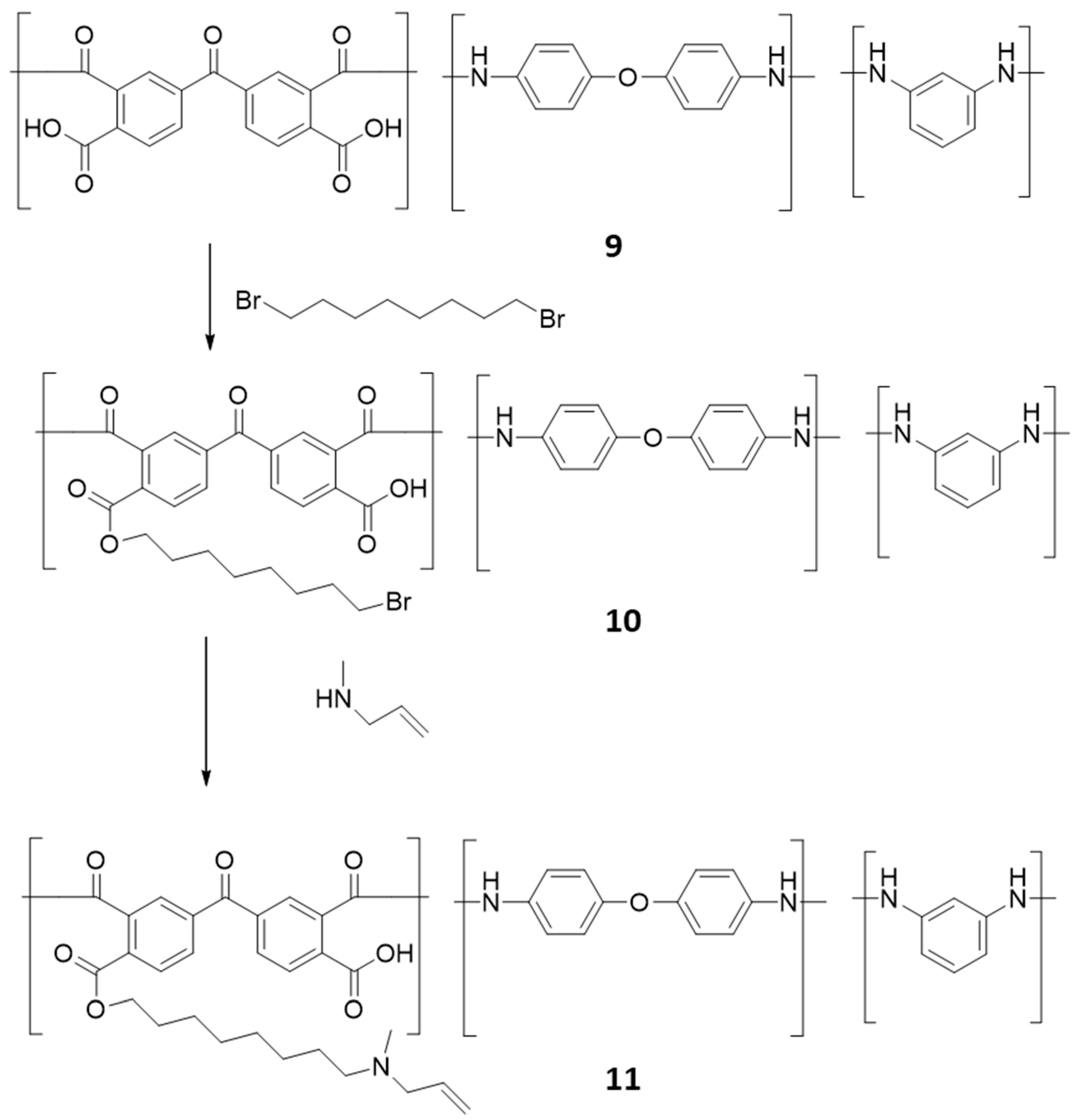
2.5. Synthesis of Polymeric Alkyl Bromide, 10
2.6. Synthesis of Polymeric Alkyl Allyamine, 11
2.7. MIC Determination:
2.8. IC50 Determination:
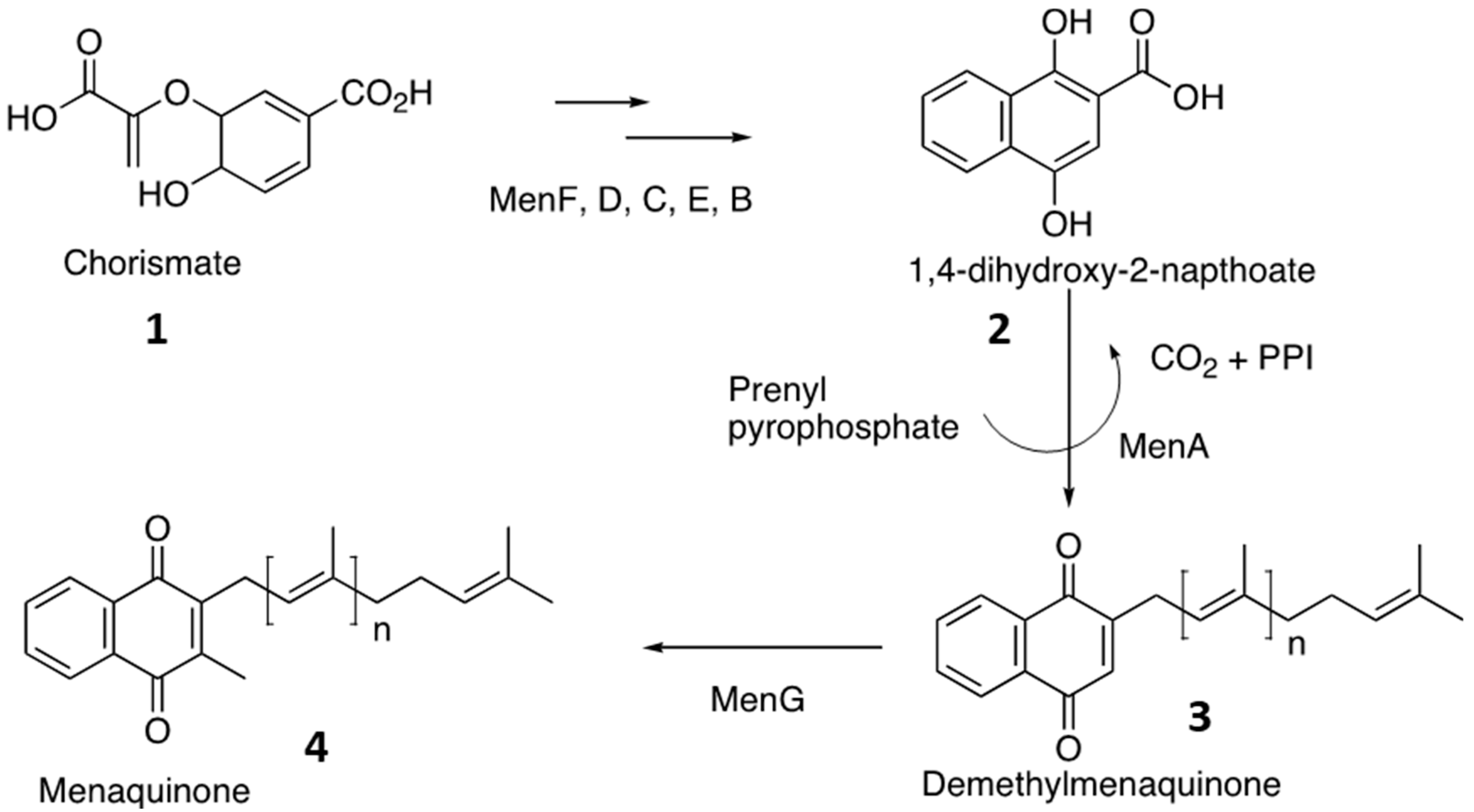
3. Results
4. Discussion
5. Conclusion
Funding
Acknowledgments
Conflicts of Interest
References
- Narayanasamy, P., H. Eoh, P. J. Brennan, and D. C. Crick. 2010. Synthesis of 4-diphosphocytidyl-2-C-methyl-D-erythritol 2-phosphate and kinetic studies of Mycobacterium tuberculosis IspF. Chemistry & biology 17: 117-122. [CrossRef]
- Kurosu, M., P. Narayanasamy, K. Biswas, R. Dhiman, and D. C. Crick. 2007. Discovery of 1,4-dihydroxy-2-naphthoate [corrected] prenyltransferase inhibitors: new drug leads for multidrug-resistant gram-positive pathogens. Journal of medicinal chemistry 50: 3973-3975.
- Brennan, P. J., and D. C. Crick. 2007. The cell-wall core of Mycobacterium tuberculosis in the context of drug discovery. Curr Top Med Chem 7: 475-488. [CrossRef]
- Choi, S. R., J. Frandsen, and P. Narayanasamy. 2017. Novel long-chain compounds with both immunomodulatory and MenA inhibitory activities against Staphylococcus aureus and its biofilm. Scientific reports 7: 40077. [CrossRef]
- Choi, S. R., M. A. Larson, S. H. Hinrichs, A. M. Bartling, J. Frandsen, and P. Narayanasamy. 2016. Discovery of bicyclic inhibitors against menaquinone biosynthesis. Future Med Chem 8: 11-16. [CrossRef]
- Choi, S. R., M. A. Larson, S. H. Hinrichs, and P. Narayanasamy. 2016. Development of potential broad spectrum antimicrobials using C-symmetric 9-fluorenone alkyl amine. Bioorganic & medicinal chemistry letters 26: 1997-1999. [CrossRef]
- Choi, S. R., and P. Narayanasamy. 2021. Synthesis, optimization, in vitro and in vivo study of bicyclic substituted amine as MenA inhibitor. Bioorganic & medicinal chemistry letters 47: 128203. [CrossRef]
- Choi, S. R., and P. Narayanasamy. 2023. In Vitro and In Vivo Antimicrobial Activity of an Oxidative Stress-Mediated Bicyclic Menaquinone Biosynthesis Inhibitor against MRSA. ACS Infect Dis 9: 2016-2024. [CrossRef]
- Choi, S. R., Narayanasamy, P. 2024. Investigating Novel IspE Inhibitors of the MEP Pathway in Mycobacterium. Microorganisms 12: 18. [CrossRef]
- Edagwa, B., Y. Wang, and P. Narayanasamy. 2013. Synthesis of azide derivative and discovery of glyoxalase pathway inhibitor against pathogenic bacteria. Bioorganic & medicinal chemistry letters 23: 6138-6140. [CrossRef]
- Edagwa, B. J., and P. Narayanasamy. 2013. Synthesis of chirally pure 1-deoxy-d-xylulose-5-phosphate : A substrate for IspC assay to determine inhibitor. Chemical sciences journal 4.
- Shineberg, B., and I. G. Young. 1976. Biosynthesis of bacterial menaquinones: the membrane-associated 1,4-dihydroxy-2-naphthoate octaprenyltransferase of Escherichia coli. Biochemistry 15: 2754-2758. [CrossRef]
- Kurosu, M., and E. Begari. 2010. Vitamin K2 in electron transport system: are enzymes involved in vitamin K2 biosynthesis promising drug targets? Molecules 15: 1531-1553.
- Bentley, R., and R. Meganathan. 1982. Biosynthesis of vitamin K (menaquinone) in bacteria. Microbiological Reviews 46: 241-280.
- Schutz, M., M. Brugna, E. Lebrun, F. Baymann, R. Huber, K.-O. Stetter, G. Hauska, R. Toci, D. Lemesle-Meunier, P. Tron, C. Schmidt, and W. Nitschke. 2000. Early evolution of cytochrome bc complexes. Journal of Molecular Biology 300: 663-675. [CrossRef]
- Unden, G., and J. Bongaerts. 1997. Alternative respiratory pathways of Escherichia coli: energetics and transcriptional regulation in response to electron acceptors. Biochimica et Biophysica Acta, Bioenergetics 1320: 217-234. [CrossRef]
- Dhiman, R. K., S. Mahapatra, R. A. Slayden, M. E. Boyne, A. Lenaerts, J. C. Hinshaw, S. K. Angala, D. Chatterjee, K. Biswas, P. Narayanasamy, M. Kurosu, and D. C. Crick. 2009. Menaquinone synthesis is critical for maintaining mycobacterial viability during exponential growth and recovery from non-replicating persistence. Molecular microbiology 72: 85-97. [CrossRef]
- Honaker, R. W., R. K. Dhiman, P. Narayanasamy, D. C. Crick, and M. I. Voskuil. 2010. DosS responds to a reduced electron transport system to induce the Mycobacterium tuberculosis DosR regulon. Journal of bacteriology 192: 6447-6455. [CrossRef]
- Li, X., N. Liu, H. Zhang, S. E. Knudson, H.-J. Li, C.-T. Lai, C. Simmerling, R. A. Slayden, and P. J. Tonge. 2011. CoA Adducts of 4-Oxo-4-phenylbut-2-enoates: Inhibitors of MenB from the M. tuberculosis Menaquinone Biosynthesis Pathway. ACS Medicinal Chemistry Letters 2: 818-823.
- Lu, X., H. Zhang, J. Tonge Peter, and S. Tan Derek. 2008. Mechanism-based inhibitors of MenE, an acyl-CoA synthetase involved in bacterial menaquinone biosynthesis. Bioorganic & medicinal chemistry letters 18: 5963-5966.
- Dhiman, R. K., S. Mahapatra, R. A. Slayden, M. E. Boyne, A. Lenaerts, J. C. Hinshaw, S. K. Angala, D. Chatterjee, K. Biswas, P. Narayanasamy, M. Kurosu, and D. C. Crick. 2009. Menaquinone synthesis is critical for maintaining mycobacterial viability during exponential growth and recovery from nonreplicating persistence. Molecular Microbiology 72: 85-97. [CrossRef]
- Lu, X., R. Zhou, I. Sharma, X. Li, G. Kumar, S. Swaminathan, J. Tonge Peter, and S. Tan Derek. 2012. Stable analogues of OSB-AMP: potent inhibitors of MenE, the o-succinylbenzoate-CoA synthetase from bacterial menaquinone biosynthesis. Chembiochem : a European journal of chemical biology 13: 129-136.
- Kurosu, M., P. Narayanasamy, K. Biswas, R. Dhiman, and D. C. Crick. 2007. Discovery of 1,4-Dihydroxy-2-naphthoate Prenyltransferase Inhibitors: New Drug Leads for Multidrug-Resistant Gram-Positive Pathogens. J. Med. Chem. 50: 3973-3975. [CrossRef]
- Debnath, J., S. Siricilla, B. Wan, C. Crick Dean, J. Lenaerts Anne, G. Franzblau Scott, and M. Kurosu. 2012. Discovery of selective menaquinone biosynthesis inhibitors against Mycobacterium tuberculosis. Journal of medicinal chemistry 55: 3739-3755.
- Kurosu, M., and D. C. Crick. 2009. MenA is a promising drug target for developing novel lead molecules to combat Mycobacterium tuberculosis. Medicinal Chemistry 5: 197-207. [CrossRef]
- Choi, S. R., B. E. Britigan, D. M. Moran, and P. Narayanasamy. 2017. Gallium nanoparticles facilitate phagosome maturation and inhibit growth of virulent Mycobacterium tuberculosis in macrophages. PLoS ONE 12: e0177987. [CrossRef]
- Choi, S. R., B. E. Britigan, and P. Narayanasamy. 2017. Ga(III) Nanoparticles Inhibit Growth of both Mycobacterium tuberculosis and HIV and Release of Interleukin-6 (IL-6) and IL-8 in Coinfected Macrophages. Antimicrobial agents and chemotherapy 61: e02505-02516. [CrossRef]
- Choi, S. R., B. E. Britigan, and P. Narayanasamy. 2019. Iron/Heme Metabolism-Targeted Gallium(III) Nanoparticles Are Active against Extracellular and Intracellular Pseudomonas aeruginosa and Acinetobacter baumannii. Antimicrobial agents and chemotherapy 63: e02643-02618. [CrossRef]
- Choi, S. R., B. E. Britigan, and P. Narayanasamy. 2019. Treatment of Virulent Mycobacterium tuberculosis and HIV Coinfected Macrophages with Gallium Nanoparticles Inhibits Pathogen Growth and Modulates Macrophage Cytokine Production. mSphere 4. [CrossRef]
- Choi, S. R., B. E. Britigan, and P. Narayanasamy. 2022. Synthesis and in vitro analysis of novel gallium tetrakis(4-methoxyphenyl)porphyrin and its long-acting nanoparticle as a potent antimycobacterial agent. Bioorganic & medicinal chemistry letters 62: 128645. [CrossRef]
- Choi, S. R., B. E. Britigan, B. Switzer, T. Hoke, D. Moran, and P. Narayanasamy. 2018. In Vitro Efficacy of Free and Nanoparticle Formulations of Gallium(III) meso-Tetraphenylporphyrine against Mycobacterium avium and Mycobacterium abscessus and Gallium Biodistribution in Mice. Molecular pharmaceutics 15: 1215-1225. [CrossRef]
- Choi, S. R., G. A. Talmon, B. E. Britigan, and P. Narayanasamy. 2021. Nanoparticulate beta-Cyclodextrin with Gallium Tetraphenylporphyrin Demonstrates in Vitro and in Vivo Antimicrobial Efficacy against Mycobacteroides abscessus and Mycobacterium avium. ACS Infect Dis.
- Sundararajan, G., and N. Prabagaran. 2001. A new polymer-anchored chiral catalyst for asymmetric Michael addition reactions. Org Lett 3: 389-392. [CrossRef]
- Gruppo, V., C. M. Johnson, K. S. Marietta, H. Scherman, E. E. Zink, D. C. Crick, L. B. Adams, I. M. Orme, and A. J. Lenaerts. 2006. Rapid microbiologic and pharmacologic evaluation of experimental compounds against Mycobacterium tuberculosis. Antimicrobial agents and chemotherapy 50: 1245-1250. [CrossRef]
- Dhiman, R. K., V. Pujari, J. M. Kincaid, M. A. Ikeh, T. Parish, and D. C. Crick. 2019. Characterization of MenA (isoprenyl diphosphate:1,4-dihydroxy-2-naphthoate isoprenyltransferase) from Mycobacterium tuberculosis. PLoS One 14: e0214958. [CrossRef]
- Crick, D. C., M. C. Schulbach, E. E. Zink, M. Macchia, S. Barontini, G. S. Besra, and P. J. Brennan. 2000. Polyprenyl phosphate biosynthesis in Mycobacterium tuberculosis and Mycobacterium smegmatis. Journal of bacteriology 182: 5771-5778. [CrossRef]
- Leppik, R. A., P. Stroobant, B. Shineberg, I. G. Young, and F. Gibson. 1976. Membrane-associated reactions in ubiquinone biosynthesis. 2-Octaprenyl-3-methyl-5-hydroxy-6-methoxy-1,4-benzoquinone methyltransferase. Biochim Biophys Acta 428: 146-156. [CrossRef]
- Narayanasamy, P., and D. C. Crick. 2008. Enantiomeric Synthesis of 2-C-Methyl-D-Erythritol 2, 4-Cyclodiphosphate. Heterocycles 76: 243-247. [CrossRef]
- Narayanasamy, P., H. Eoh, and D. C. Crick. 2008. Chemoenzymatic synthesis of 4-diphosphocytidyl-2-C-methyl-D-erythritol: A substrate for IspE. Tetrahedron letters 49: 4461-4463. [CrossRef]

| MIC (µg/mL) | IC50 | ||
|---|---|---|---|
| Compound | M.tb | M.tb MenA | |
| 8 | 6.2 | 12.9 | |
| MIC (µg/mL) | IC50 | ||
|---|---|---|---|
| Compound | M.tb | M.tb MenA | |
| 11 | 1.0 | 1.0 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




