Submitted:
21 December 2023
Posted:
22 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
3. Discussion
3.1. Technical aspect and clinical model
3.2. Immunomodulation and host-virus interaction in transplant patient
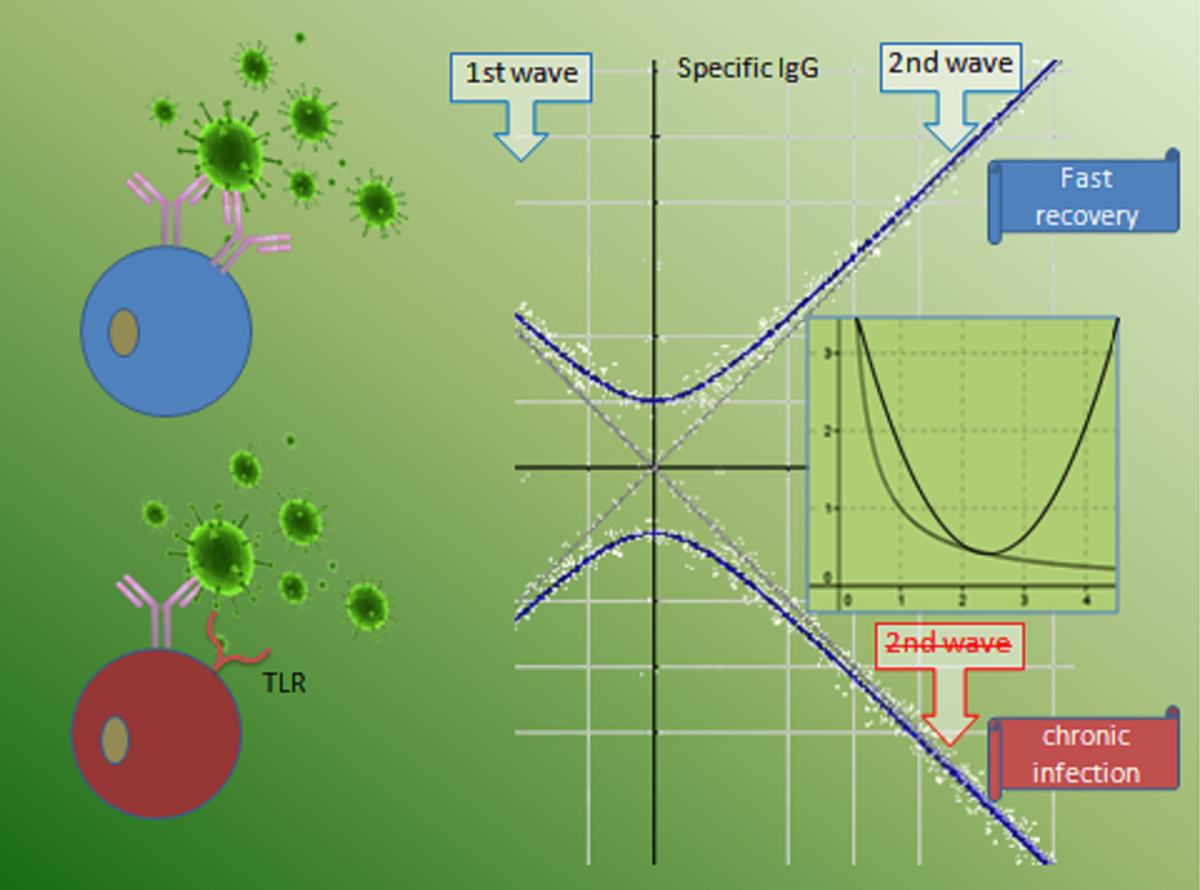
3.3. Host-virus dynamics
- Lag phase—very low/ no antibody is detectable.
- Log phase—the antibody titer increases logarithmically.
- Plateau phase—the antibody titer stabilizes.
- Decline phase—the antibody is catabolized.
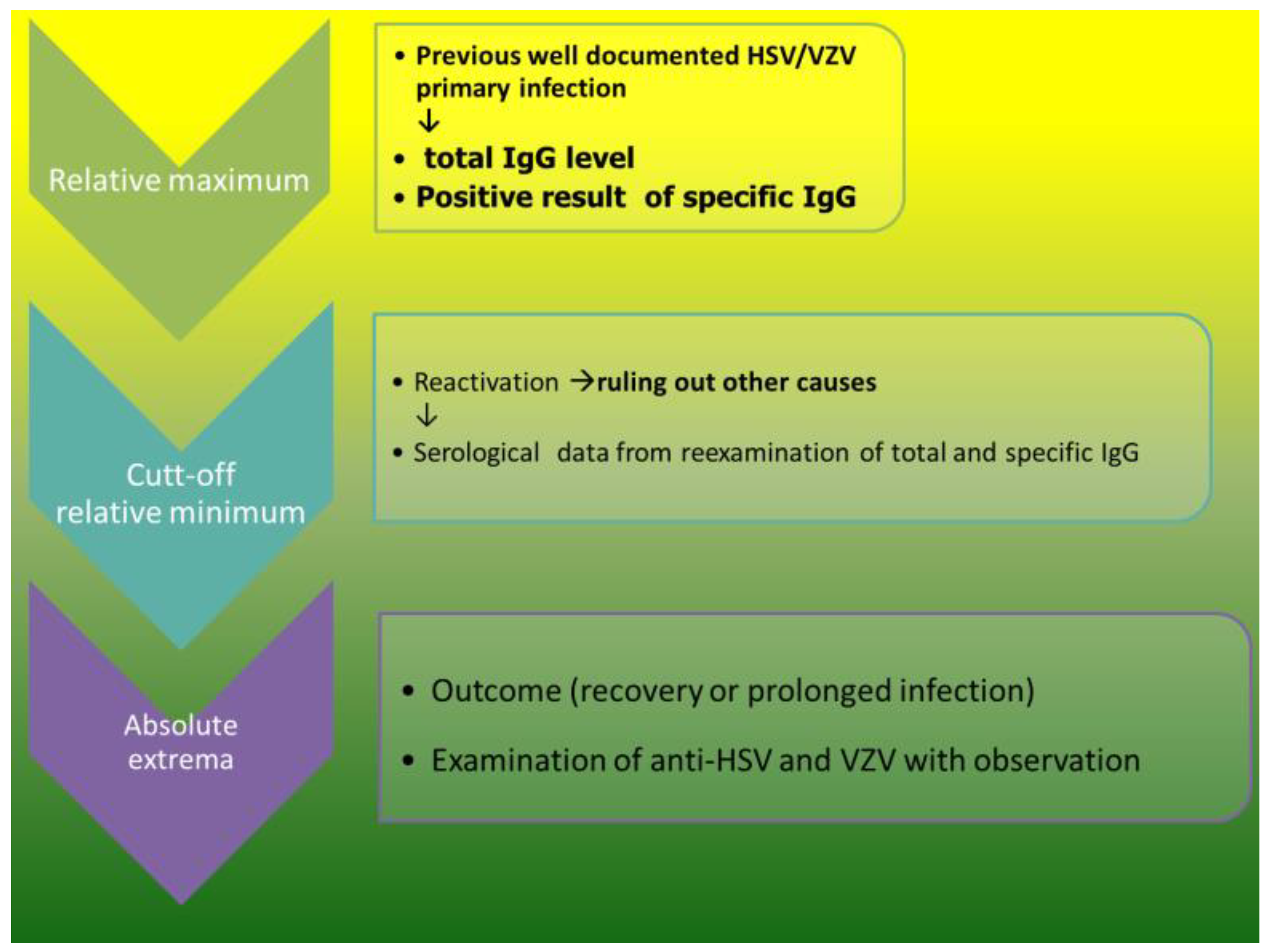
3.3. Humoral compartment in secondary immunodeficiency after HSCT, immunomodulation by conditioning
3.1.2. B cells memory
3.1.4. Two signal and IgG increase
4. Materials and Methods
4.1. Material
4.2. Time-lapse data collection
4.3. Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Pinquier D, Gagneur A, Balu L et al: Prevalence of Anti-Varicella-Zoster Virus Antibodies in French Infants under 15 Months of Age. Clin Vaccine Immunol. 2009;16:484-7.
- Pinquier D, Lécuyer A, Levy C et al: Inverse correlation between varicella severity and level of anti-Varicella Zoster Virus maternal antibodies in infants below one year of age. Hum Vaccin. 2011;7:534-8;
- Kohl S, West MS, Prober CG, Sullender WM, Loo LS, Arvin AM.: Neonatal antibody-dependent cellular cytotoxic antibody levels are associated with the clinical presentation of neonatal herpes simplex virus infection. J Infect Dis. 1989;160: 770-6. [CrossRef]
- Corey L, Wald A. Maternal and neonatal herpes simplex virus infections.N Engl J Med. 2009;361:1376-85. [CrossRef]
- Drylewicz J, Schellens IM, Gaiser R et al Rapid reconstitution of CD4 T cells and NK cells protects against CMV-reactivation after allogeneic stem cell transplantation. J Transl Med. 2016;14:230. [CrossRef]
- Prelog M, Schönlaub J, Jeller V, Almanzar G, Höfner K, Gruber S, Eiwegger T, Würzner R. Reduced varicella-zoster-virus (VZV)-specific lymphocytes and IgG antibody avidity in solid organ transplant recipients. Vaccine. 2013; 31(20):2420-6. Epub 2013 Apr 10. PMID: 23583889. [CrossRef]
- Mascarenhas K, Teh JB, Peng K, Kim H, Sy A, Forman SJ, Wong FL, Nakamura R, Dadwal SS, Armenian SH. Efficacy of low-dose zoster prophylaxis in patients undergoing allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2020 Aug;55(8):1662-1664. [CrossRef]
- Wehr C, Gennery AR, Lindemans C et al. Multicenter experience in hematopoietic stem cell transplantation for serious complications of common variable immunodeficiency. J Allergy Clin Immunol. 2015;135: 988-97.e6.
- Tenny S, Kerndt CC, Hoffman MR. Case Control Studies. (Updated 2022 Mar 28). In: StatPearls (Internet). Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK448143/.
- Sedgwick P. Bias in observational study designs: case-control studies. BMJ. 2015;350:h560. [CrossRef]
- Solomon, A. R., Rasmussen, J. E., Varani, J. & Pierson, C. L. The Tzanck smear in the diagnosis of cutaneous herpes simplex. JAMA 1984; 251, 633–635 (). [CrossRef]
- Stephanie J. Lee; Classification systems for chronic graft-versus-host disease. Blood 2017; 129 (1): 30–37. [CrossRef]
- Zdziarski P. CMV-specific immune response - new patients, new insight: central role of specific IgG during infancy and long-lasting immune deficiency after allogenic stem cell transplantation. Int J Mol Sci. 2019 Jan 11;20(2):271. [CrossRef]
- Noyan, M.A., Durdu, M. & Eskiocak, A.H. TzanckNet: a convolutional neural network to identify cells in the cytology of erosive-vesiculobullous diseases. Sci Rep 2020; 10, 1831. [CrossRef]
- Turgeon M.L. IMMUNOLOGY & SEROLOGY IN LABORATORY MEDICINE 5th ed.; Mosby, an imprint of Elsevier Inc., St. Louis, Missouri, USA, 2014; pp. 202-203.
- Wu M, Huang F, Jiang X, et al. Herpesvirus-associated central nervous system diseases after allogeneic hematopoietic stem cell transplantation. PLoS One 2013; 8:e77805.
- Andreu-Ballester JC, García-Ballesteros C, Benet-Campos C, Amigó V, Almela-Quilis A, Mayans J, Ballester F. Values for αβ and γδ T-lymphocytes and CD4+, CD8+, and CD56+ subsets in healthy adult subjects: assessment by age and gender. Cytometry Part B 2012; 82B: 238–244. [CrossRef]
- Ardeniz Ö, Unger S, Onay H, Ammann S, Keck C, Cianga C, Gerçeker B, Martin B, Fuchs I, Salzer U, İkincioğulları A, Güloğlu D, Dereli T, Thimme R, Ehl S, Schwarz K, Schmitt-Graeff A, Cianga P, Fisch P, Warnatz K. β2-Microglobulin deficiency causes a complex immunodeficiency of the innate and adaptive immune system. J Allergy Clin Immunol. 2015 Aug;136(2):392-401. [CrossRef]
- Kühn JE, Klaffke K, Munk K, Braun RW. HSV-1 gB and VZV gp-II crossreactive antibodies in human sera. Arch Virol. 1990;112(3-4):203-13. [CrossRef]
- Szomolanyi-Tsuda E., Brehm M.A. Welsh R.M. Acquired immunity against virus infections. In: The immune response to Infection Ed. Kaufmann SHE, Rouse BT, Sacks DL 2011 ASM Press Washington DC p239-67.
- Whery EJ, Klenerman P. Immune response to persistent viruses in: The immune response to Infection Ed. Kaufmann SHE, Rouse BT, Sacks DL 2011 ASM Press Washington DC p.255-265.
- Herold BC, WuDunn D, Soltys N, Spear PG. Glycoprotein C of herpes simplex virus type 1 plays a principal role in the adsorption of virus to cells and in infectivity. J Virol. 1991;65(3):1090–8. [CrossRef]
- Friedman H.M., Wang L., Pangburn M.K., Lambris J.D., Lubinski J. Novel mechanism of antibody-independent complement neutralization of herpes simplex virus type 1. J. Immunol 2000; 165: 4528-4536. [CrossRef]
- Awasthi S, Lubinski JM, Friedman HM. Immunization with HSV-1 glycoprotein C prevents immune evasion from complement and enhances the efficacy of an HSV-1 glycoprotein D subunit vaccine.Vaccine. 2009;27: 6845-53. [CrossRef]
- Rizzi M, Neumann C, Fielding AK, Marks R, Goldacker S, Thaventhiran J, et al. Outcome of allogeneic stem cell transplantation in adults with common variable immunodeficiency. J Allergy Clin Immunol 2011;128:1371-4.e2. [CrossRef]
- Norlin AC, Sairafi D, Mattsson J et al Allogeneic stem cell transplantation: low immunoglobulin levels associated with decreased survival. Bone Marrow Transplant. 2008; 41:267-73.
- Akkaya M, Kwak K, Pierce SK. B cell memory: building two walls of protection against pathogens. Nat Rev Immunol. 2020;20(4):229-238. [CrossRef]
- Lee A, Hong J, Shin DY, Koh Y, Yoon SS, Kim PJ, Kim HG, Kim I, Park HK, Choi Y. Association of HSV-1 and Reduced Oral Bacteriota Diversity with Chemotherapy-Induced Oral Mucositis in Patients Undergoing Autologous Hematopoietic Stem Cell Transplantation. J Clin Med. 2020 Apr 11;9(4):1090. PMID: 32290456; PMCID: PMC7230275. [CrossRef]
- Hong J, Park HK, Park S, Lee A, Lee YH, Shin DY, Koh Y, Choi JY, Yoon SS, Choi Y, Kim I. Strong association between herpes simplex virus-1 and chemotherapy-induced oral mucositis in patients with hematologic malignancies. Korean J Intern Med. 2020 Sep;35(5):1188-1198. Epub 2019 Sep 9. PMID: 31487770; PMCID: PMC7487294. [CrossRef]
- van der Beek MT, Laheij AM, Raber-Durlacher JE, von dem Borne PA, Wolterbeek R, van der Blij-de Brouwer CS, van Loveren C, Claas EC, Kroes AC, de Soet JJ, Vossen AC. Viral loads and antiviral resistance of herpesviruses and oral ulcerations in hematopoietic stem cell transplant recipients. Bone Marrow Transplant. 2012 Sep;47(9):1222-8. Epub 2012 Feb 13. PMID: 22327137. [CrossRef]
- Costa ALF, Santos BA, Torregrossa VR, Miranda ECM, Vigorito AC, Palmieri M, Ricardo ALF, Sarmento DJS, Mamana AC, Tozetto-Mendoza TR, Correa MEP, Braz-Silva PH. Oral shedding of CMV and HSV-1 in hematopoietic stem cell transplantation patients. Oral Dis. 2020 Oct 21. Epub ahead of print. PMID: 33085827. [CrossRef]
- Ochiai K, Maienschein-Cline M, Simonetti G, Chen J, Rosenthal R, Brink R, Chong AS, Klein U, Dinner AR, Singh H, Sciammas R. Transcriptional regulation of germinal center B and plasma cell fates by dynamical control of IRF4. Immunity. 2013 May 23;38(5):918-29. [CrossRef]
- Akkaya M, et al. Toll-like receptor 9 antagonizes antibody affinity maturation. Nat. Immunol. 2018;19:255–266. [CrossRef]
- Zyzak J, Mitkiewicz M, Leszczyńska E, Reniewicz P, Moynagh PN, Siednienko J. HSV-1/TLR9-Mediated IFNβ and TNFα Induction Is Mal-Dependent in Macrophages. J Innate Immun. 2020;12(5):387-398. Epub 2019 Dec 18. PMID: 31851971; PMCID: PMC7506264. [CrossRef]
- Yu HR, Huang HC, Kuo HC, Sheen JM, Ou CY, Hsu TY, Yang KD. IFN-α production by human mononuclear cells infected with varicella-zoster virus through TLR9-dependent and -independent pathways. Cell Mol Immunol. 2011 Mar;8(2):181-8. Epub 2011 Feb 14. PMID: 21317915; PMCID: PMC4003143. [CrossRef]
- Black AP, Jones L, Malavige GN, Ogg GS. Immune evasion during varicella zoster virus infection of keratinocytes. Clin Exp Dermatol. 2009; 34(8): e941-4. [CrossRef]
- Liang F, Glans H, Enoksson SL, Kolios AGA, Loré K, Nilsson J. Recurrent Herpes Zoster Ophthalmicus in a Patient With a Novel Toll-Like Receptor 3 Variant Linked to Compromised Activation Capacity in Fibroblasts. J Infect Dis. 2020;221(8):1295-1303. PMID: 31268141. [CrossRef]
- Kawamura K, Wada H, Yamasaki R, Ishihara Y, Sakamoto K, Ashizawa M, Sato M, Machishima T, Terasako K, Kimura S, Kikuchi M, Nakasone H, Yamazaki R, Kanda J, Kako S, Tanihara A, Nishida J, Kanda Y. Prophylactic role of long-term ultra-low-dose acyclovir for varicella zoster virus disease after allogeneic hematopoietic stem cell transplantation. Int J Infect Dis. 2014; 19: 26-32. [CrossRef]
| P1 | P2 | |||
|---|---|---|---|---|
| Gender | F | M | ||
| Underlying disease | AML | SAA | ||
| Conditioning | Bu-Cy | Flu-Cy-ATG | ||
|
Graft versus Host Stage Acute (chronic) |
0 (1) |
0 (0) |
||
| Age | 38 | 27 | ||
| initial | reactivation | initial | reactivation | |
|
Complement C4 (mg/dL) |
15.20 | 25.2 | 10.2 | 15.0 |
| β2microglobulin (mg/l) | 1.3 | 3.23 | 1.8 | 4.0 |
| CsA (ng/ml) | ND* | 137 | ND | 155 |
| WBC (/ul) | 3500 | 2530 | 7200 | 6500 |
| %lymphocytes (cells/μl) | ||||
| CD3 | 75.3(642) | 78.1(↓444) | ND | 60.0(752) |
| CD4 | ↓25.6(218) | ↓↓15.1(86) | ND | ↓25.3(317) |
| CD8 | 46.1(393) | 64.3(366) | ND | 28.6(359) |
| CD20 | 12.3(↓104) | ↓↓2.5(14) | ND | ↓5.2(65) |
| CD20+27+ | ↓1.2(10) | ↓↓<0.5%* | ND | ↓↓0.7(9) |
| CD20+23+ | 11.2 (96) | 1.6 (9) | ND | 3.5(44) |
| CD16+ | 8.1 (69) | 8(45) | ND | 7,7 (97) |
| CD16+56+ | 7.5 (64) | 3.8 (21) | ND | 6.7(84) |
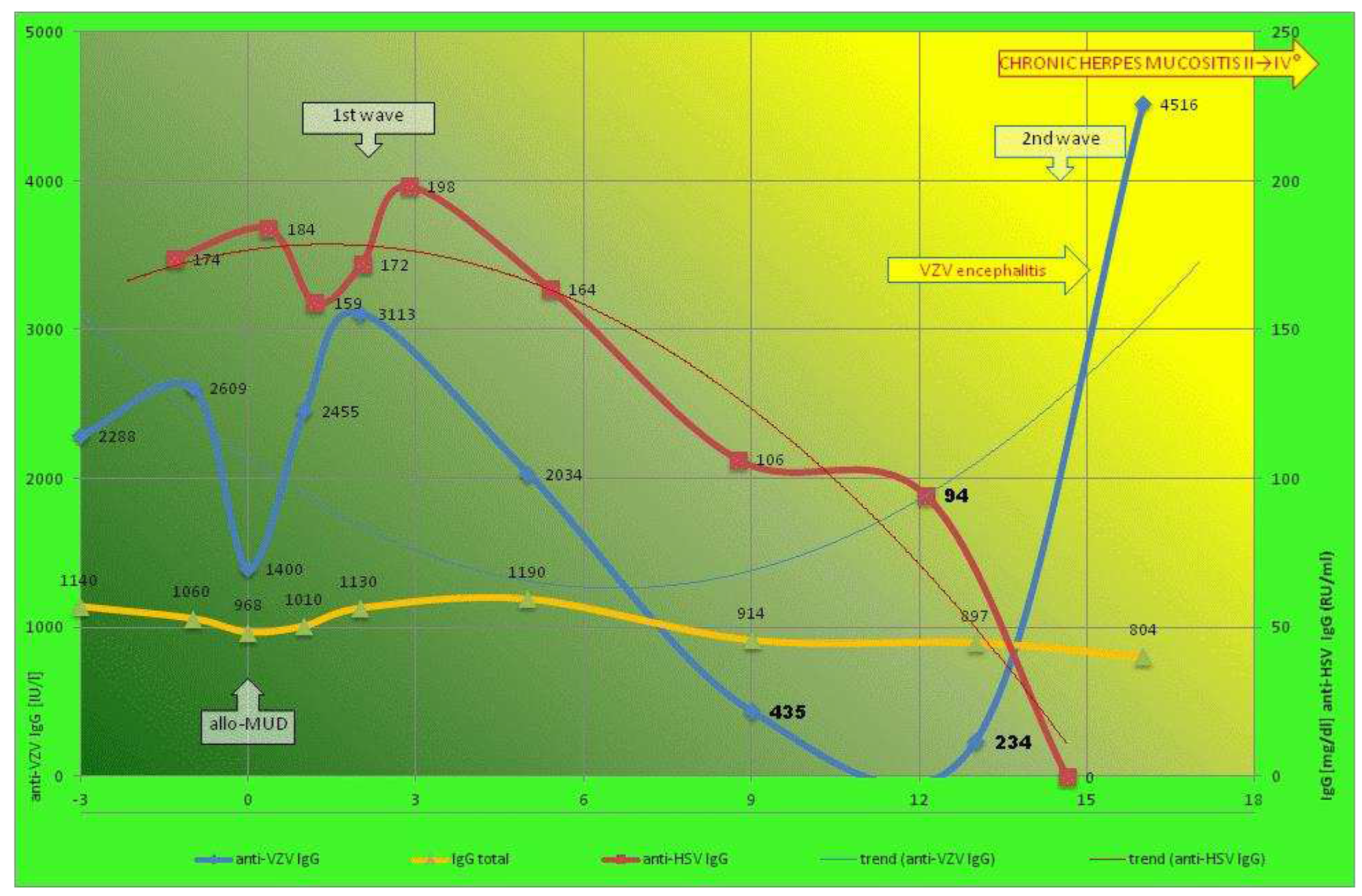
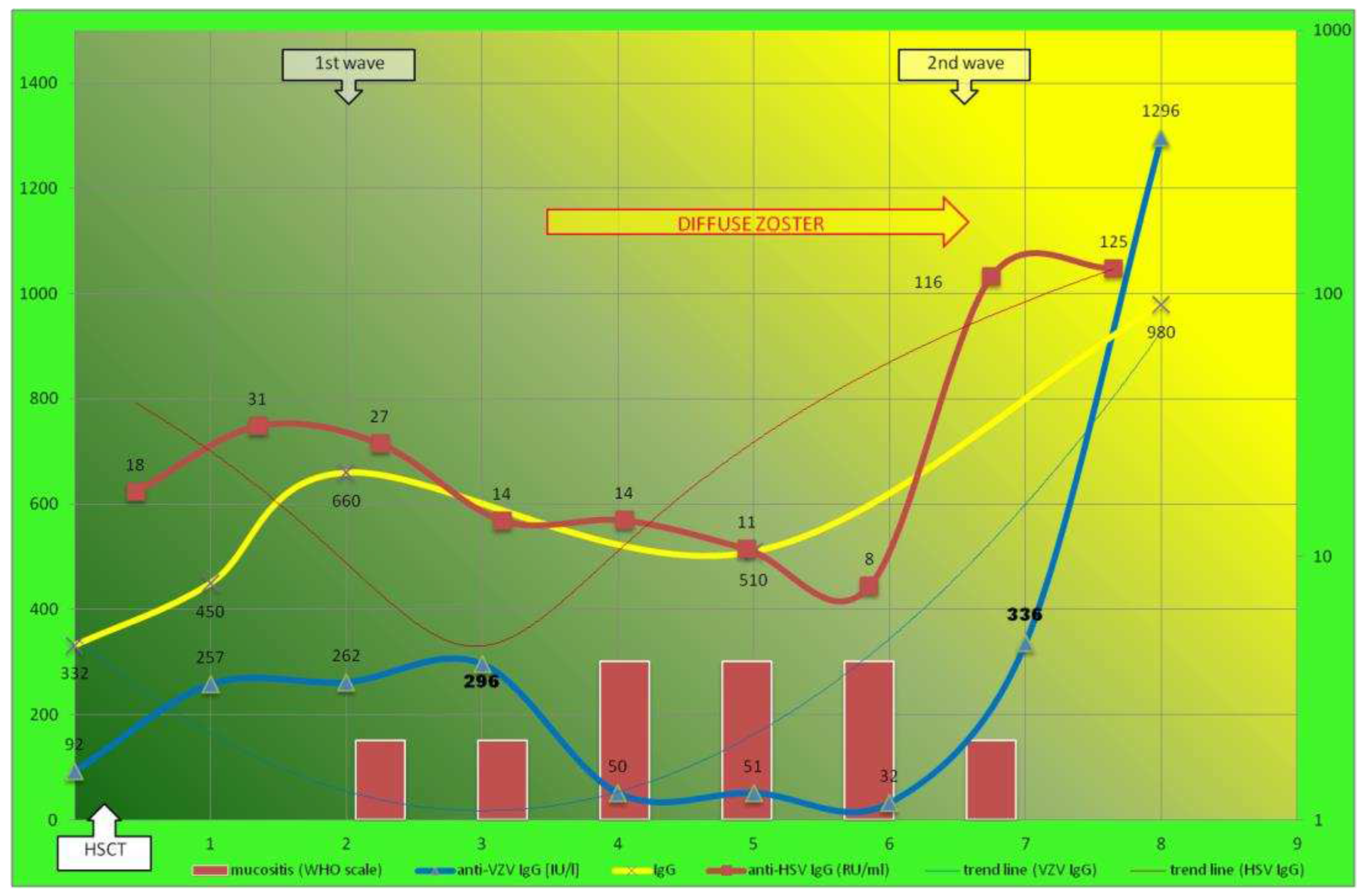
| Level | P1 | P2 |
|---|---|---|
| IgG total | 804-1190 | 332-980 |
| IgG anti-EBV (Ru/ml) | >200 | >200 |
| IgG anti-CMV (RU/ml) | >200 | >200 |
|
IgG anti-HSV* (RU/ml) (avidity) |
178→0 (40→NA) |
7,73→124 (ND) |
|
IgG anti-VZV (IU/l) (avidity ) |
234→4515 (78) |
32→1295 (72) |
| IgG anti-HbsAg** | 660 | 160 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




