Submitted:
11 December 2023
Posted:
12 December 2023
You are already at the latest version
Abstract
Keywords:
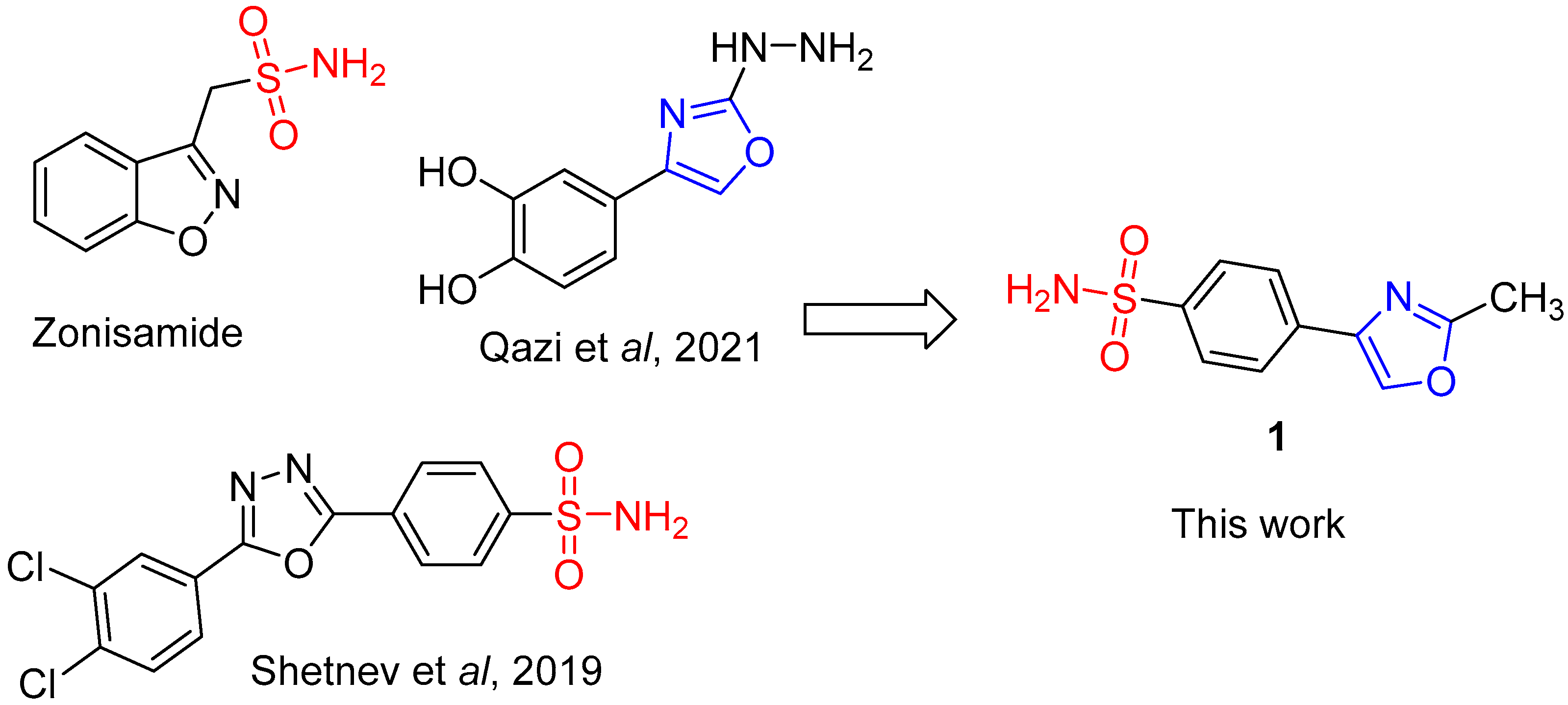
1. Introduction
2. Results
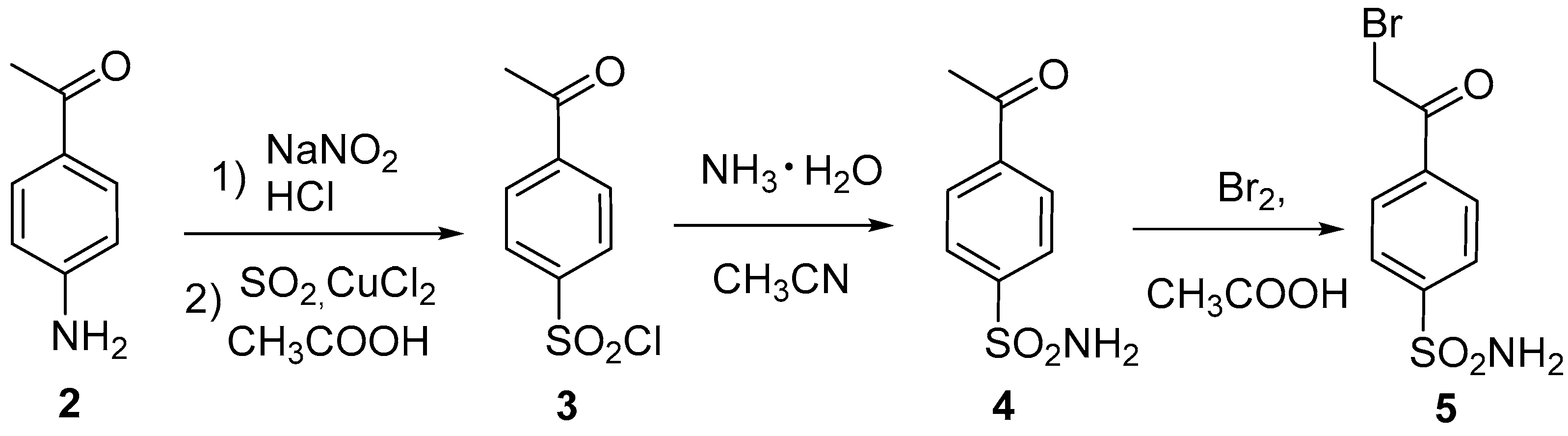
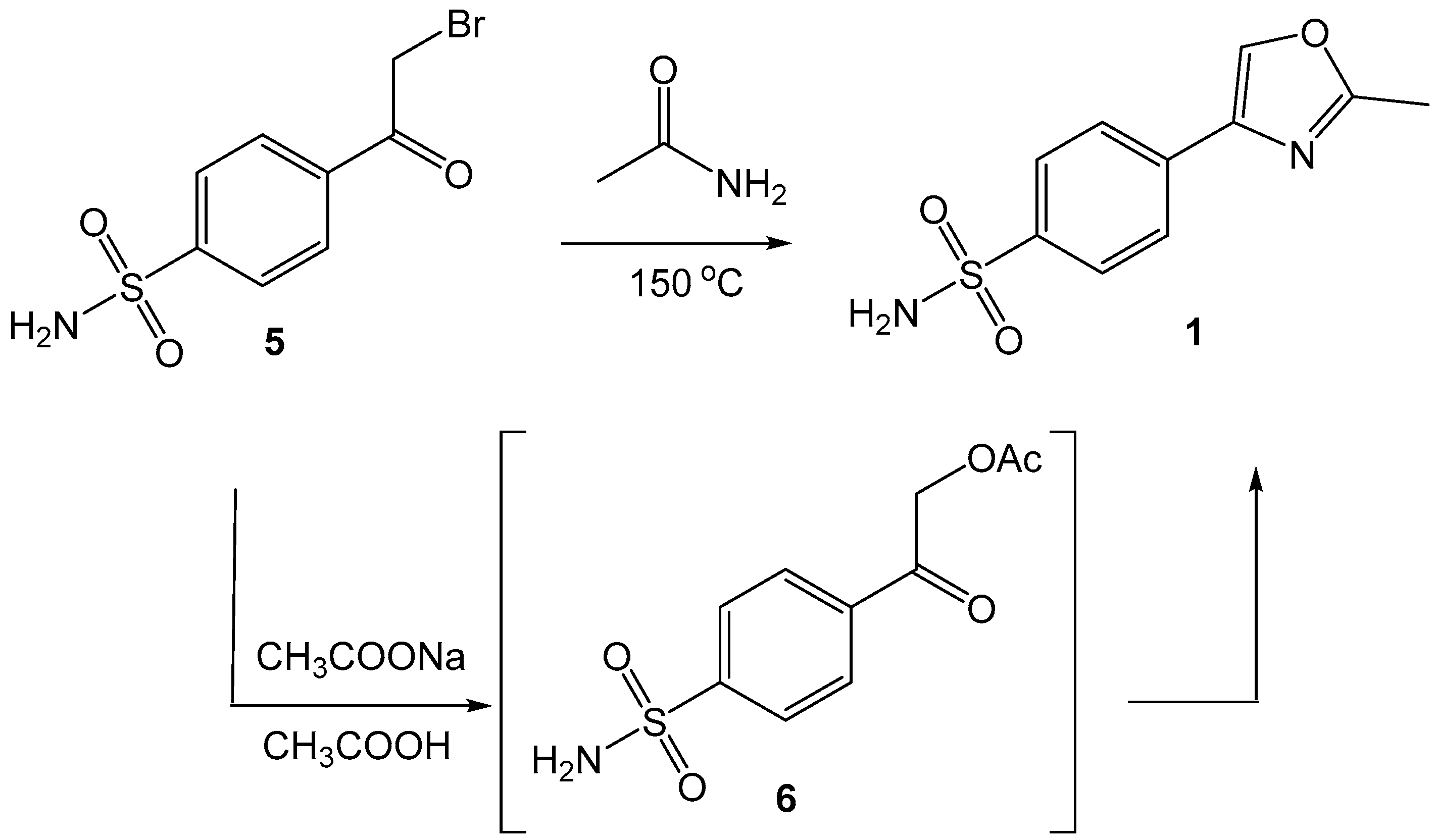
2.1. Chemistry
2.2. MAO inhibition
3. Discussion
4. Materials and Methods
4.1. General
4.2. Procedure for the preparation of 4-(2-bromoacetyl)benzenesulfonamide (5)
4.3. Synthesis and characterization of 4-(2-methyloxazol-4-yl)benzenesulfonamide (1)
4.6. MAO inhibition studies
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tan, Y.Y.; Jenner, P.; Chen, S.D. Monoamine Oxidase-B Inhibitors for the Treatment of Parkinson's Disease: Past, Present, and Future. J Parkinsons Dis. 2022;12(2):477-493. [CrossRef]
- Szökő, É.; Tábi, T.; Riederer, P.; Vécsei, L.; Magyar, K. Pharmacological aspects of the neuroprotective effects of irreversible MAO-B inhibitors, selegiline and rasagiline, in Parkinson’s disease. J Neural Transm. 2018, 125, 1735–1749. [Google Scholar] [CrossRef] [PubMed]
- Ostadkarampour, M.; Putnins, E.E. Monoamine Oxidase Inhibitors: A Review of Their Anti-Inflammatory Therapeutic Potential and Mechanisms of Action. Front Pharmacol. 2021, 12, 676239. [Google Scholar] [CrossRef] [PubMed]
- Zarmouh, N.O.; Messeha, S.S.; Mateeva, N.; Gangapuram, M.; Flowers, K.; Eyunni, S.V.K.; Zhang, W.; Redda, K.K.; Soliman, K.F.A. The Antiproliferative Effects of Flavonoid MAO Inhibitors on Prostate Cancer Cells. Molecules. 2020, 25(9), 2257. [Google Scholar] [CrossRef] [PubMed]
- Balu, D.T.; Hoshaw, B.A.; Malberg, J.E.; Rosenzweig-Lipson, S.; Schechter, L.E.; Lucki, I. Brain Res. 2008, 1211, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Sahu, S.; Schachner, M. Pharmacol Biochem Behav. 2018, 171, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Grover, N.D.; Limaye, R.P.; Gokhale, D.V.; Patil, T.R. Zonisamide: a review of the clinical and experimental evidence for its use in Parkinson's disease". Indian Journal of Pharmacology. 2013, 45(6), 547–55. [Google Scholar] [CrossRef] [PubMed]
- Shetnev, A.; Shlenev, R.; Efimova, J.; Ivanovskii, S.; Tarasov, A.; Petzer, A.; Petzer, J.P. 1,3,4-Oxadiazol-2-ylbenzenesulfonamides as privileged structures for the inhibition of monoamine oxidase B. Bioorg. Med. Chem. Lett. 2019, 29, 126677. [Google Scholar] [CrossRef] [PubMed]
- Qazi, S.U.; Naz, A.; Hameed, A.; Osra, F.A.; Jalil, S.; Iqbal, J.; Shah, S.A.; Mirza, A.Z. Semicarbazones, thiosemicarbazone, thiazole and oxazole analogues as monoamine oxidase inhibitors: Synthesis, characterization, biological evaluation, molecular docking, and kinetic studies. Bioorg Chem. 2021, 115, 105209. [Google Scholar] [CrossRef] [PubMed]
- Shetnev, A.; Kotov, A.; Kunichkina, A.; Proskurina, I.; Baykov, S.; Korsakov, M.; Petzer, A.; Petzer, J.P. Monoamine oxidase inhibition properties of 2,1-benzisoxazole derivatives. Mol Divers 2023. [Google Scholar] [CrossRef] [PubMed]
- Tzvetkov, N.T.; Antonov, L. Subnanomolar indazole-5-carboxamide inhibitors of monoamine oxidase B (MAO-B) continued: indications of iron binding, experimental evidence for optimised solubility and brain penetration. J. Enzyme Inhib. Med. Chem. 2017, 32, 960–967. [Google Scholar] [CrossRef] [PubMed]
- Efimova, J.A.; Shetnev, A.A.; Baykov, S.V.; Petzer, A.; Petzer, J.P. 3-(3,4-Dichlorophenyl)-5-(1H-indol-5-yl)-1,2,4-oxadiazole. Molbank 2023, M1552. [Google Scholar]
- Panova, V.A.; Filimonov, S.I.; Chirkova, Z.V.; Kabanova, M.V.; Shetnev, A.A.; Korsakov, M.K.; Petzer, A.; Petzer, J.P.; Suponitsky, K.Y. Investigation of pyrazolo[1,5-a]quinoxalin-4-ones as novel monoamine oxidase inhibitors. Bioorg. Chem. 2021, 108, 104563. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.W.; Leadbetter, M.R.; Bell, N.; Koo-McCoy, S.; Carreras, C.W.; He, L.; Kohler, J.; Kozuka, K.; Labonté, E.D.; Navre, M.; Spencer, A.G.; Charmot, D. Discovery of tenapanor: A first-in-class minimally systemic inhibitor of intestinal na+/h+ exchanger isoform 3. ACS Med. Chem. Lett 2022, 13(7), 1043–1051. [Google Scholar] [CrossRef] [PubMed]
- Denney, D.B.; Pastor, S.D. Structures in solution of adducts of hexamethylphosphorus triamide and substituded benzils. Phosphorus and Sulfur and the Related Elements. 1983, 16(3), 239–246. [Google Scholar] [CrossRef]
- Mostert, S.; Petzer, A.; Petzer, J.P. Indanones as high-potency reversible inhibitors of monoamine oxidase. Chem. Med. Chem. 2015, 10, 862–873. [Google Scholar] [CrossRef] [PubMed]
| Structure | IC50 (μM ± SD)1 | |
|---|---|---|
| MAO-A | MAO-B | |
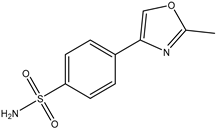 |
43.3 ± 7.12 | 3.47 ± 0.31 |
| Curcumin2 | 5.02 ± 0.45 | 2.56 ± 0.21 |
| Toloxatone [13]2 | 3.92 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




