Submitted:
11 December 2023
Posted:
13 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methodology
2.1. PES approximation
2.2. Rate constants estimation
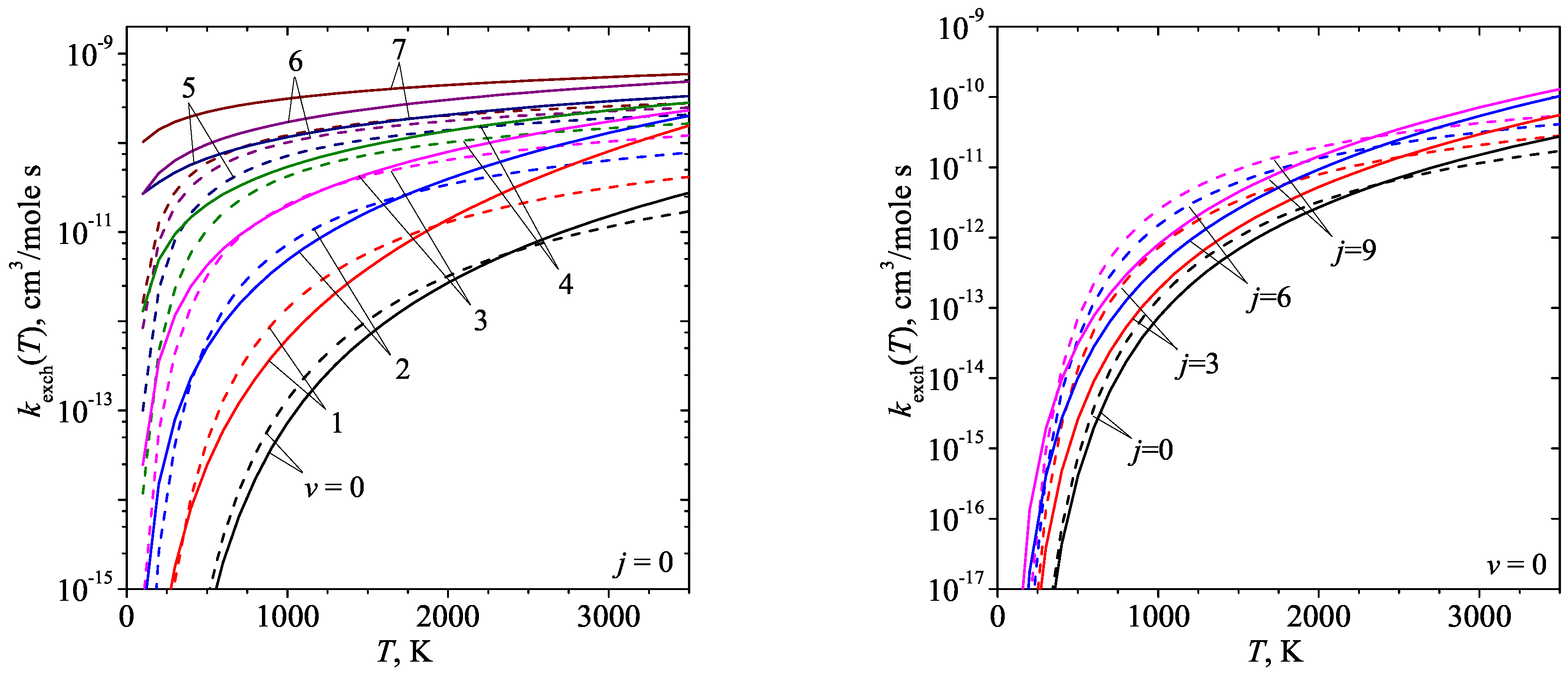
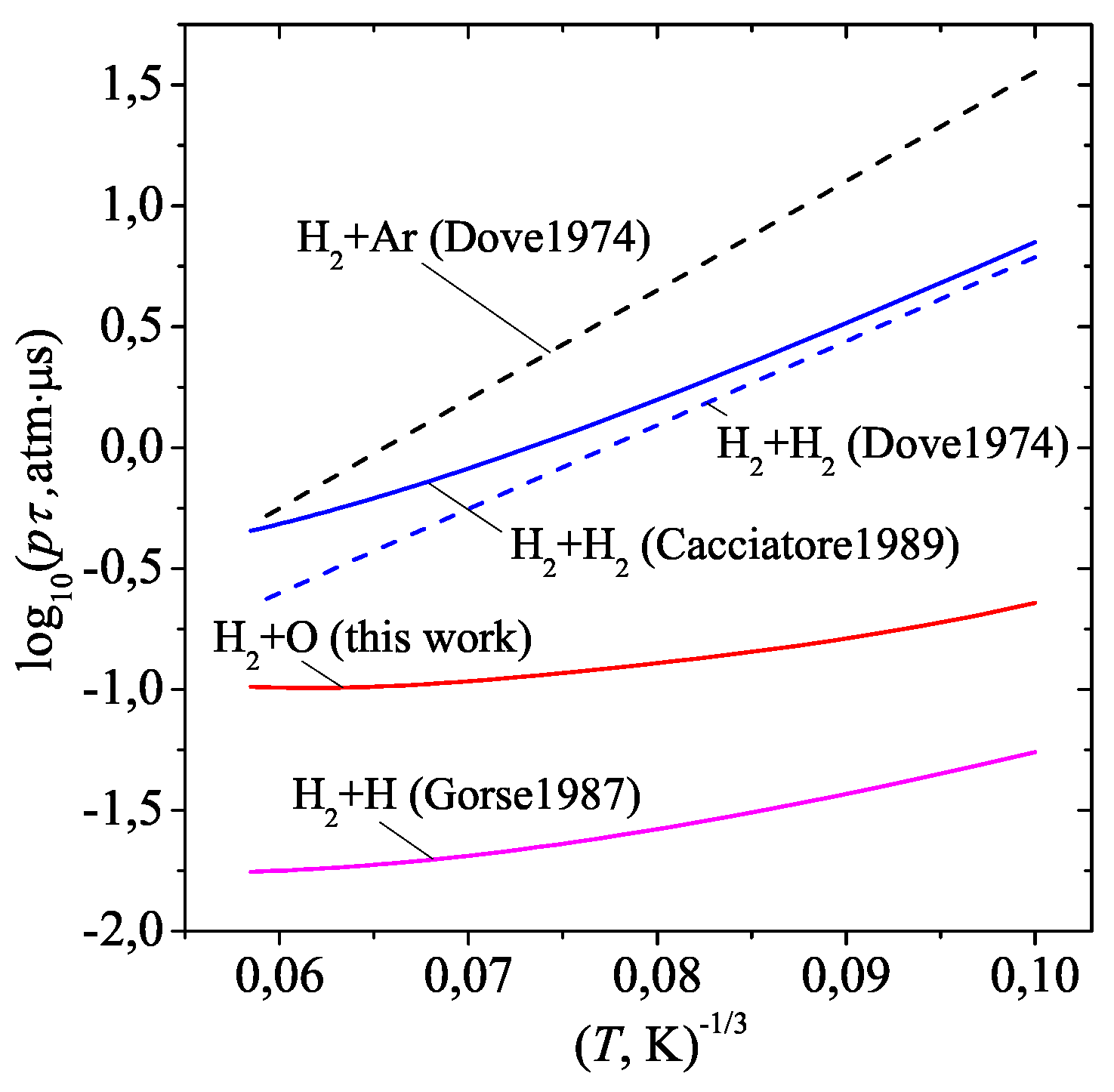
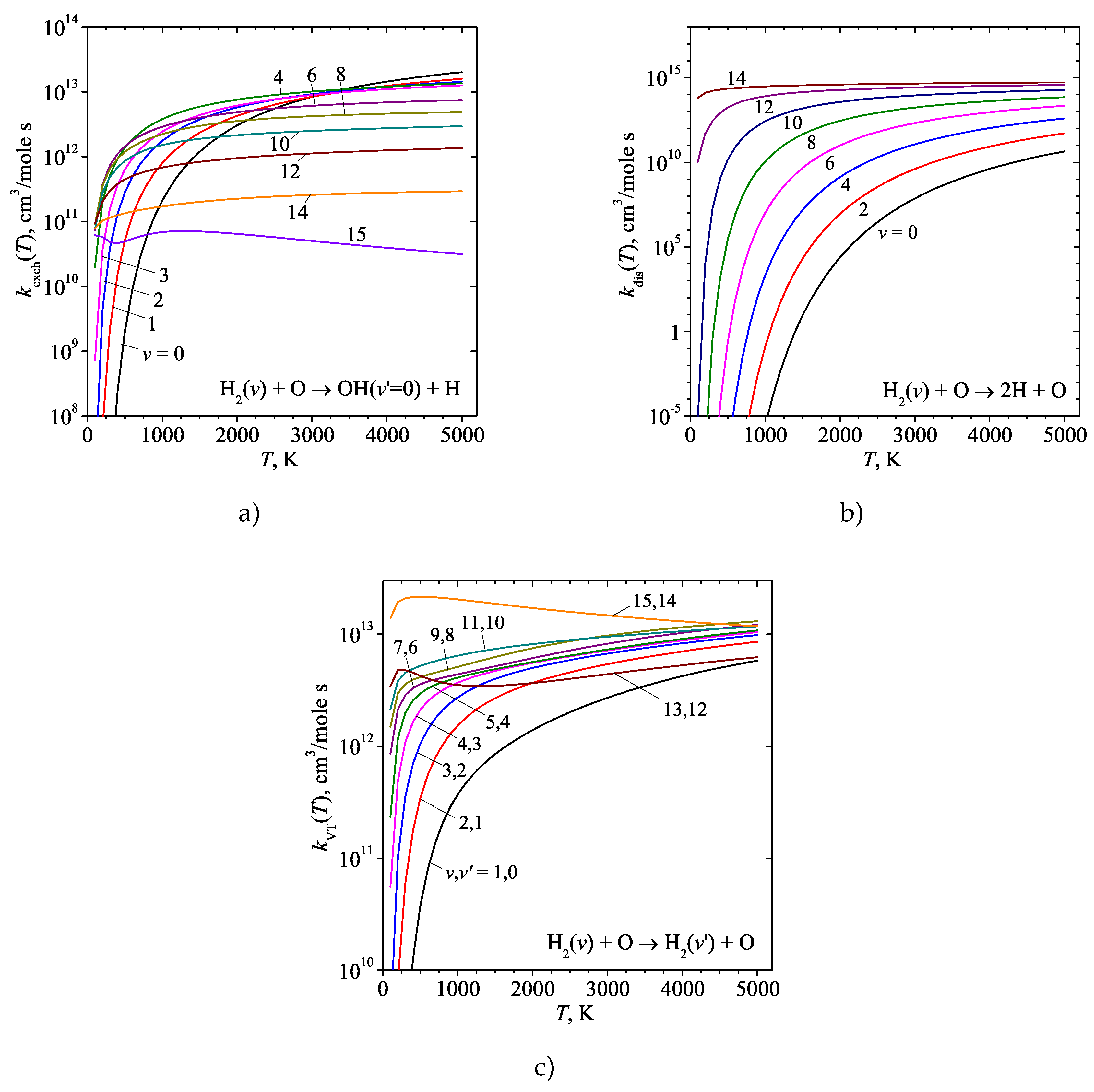
3. Results and discussion
4. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| QCT | quasiclassical trajectory |
| PES | Potential energy surface |
| VT relaxation | vibrational-translational relaxation |
| PIP-NN | permutation invariant polynomials-neural network |
| XMCQDPT2 | extended multi-configuration quasi-degenerate second-order perturbation theory |
| DW-CASSCF | dynamically weighted complete active space self-consistent field method |
References
- Skrebkov, O.V.; Kostenko, S.S.; Smirnov, A.L. Vibrational nonequilibrium in reaction hydrogen with oxygen (review, in press). Technical Physics.
- Park, C. Review of chemical-kinetic problems of future NASA missions. I-Earth entries. Journal of Thermophysics and Heat transfer 1993, 7, 385–398. [Google Scholar] [CrossRef]
- Capitelli, M.; Armenise, I.; Gorse, C. State-to-state approach in the kinetics of air components under re-entry conditions. J. Thermophys. Heat Transfer 1997, 11, 570–578. [Google Scholar] [CrossRef]
- Treanor, C.E.; Adamovich, I.V.; Williams, M.J.; Rich, J.W. Kinetics of nitric oxide formation behind shock waves. Journal of Thermophysics and Heat Transfer 1996, 10, 193–199. [Google Scholar] [CrossRef]
- Kustova, E.V.; Nagnibeda, E.A.; Shevelev, Y.D.; Syzranova, N.G. Comparison of different models for non-equilibrium CO2 flows in a shock layer near a blunt body. Shock Waves 2011, 21, 273–287. [Google Scholar] [CrossRef]
- Surzhikov, S.; Reynier, P.; Seller, G.; Taccogna, F. Radiative aerothermodynamics of entering space vehicles: toward the use of state-to-state approach. Open Plasma Phys. J 2014, 7, 127–154. [Google Scholar] [CrossRef]
- Kadochnikov, I.N.; Loukhovitski, B.I.; Starik, A.M. Kinetics of plasmachemical processes in the expanding flow of nitrogen plasma. Phys. Scr. 2013, 88, 058306 (12pp). [Google Scholar] [CrossRef]
- Gimelshein, S.F.; Wysong, I.J.; Fangman, A.J.; Andrienko, D.A.; Kunova, O.V.; Kustova, E.V.; Morgado, F.; Garbacz, C.; Fossati, M.; Hanquist, K.M. Kinetic and continuum modeling of high-temperature air relaxation. Journal of Thermophysics and Heat Transfer 2022, 36, 870–893. [Google Scholar] [CrossRef]
- Aiken, T.T.; Boyd, I.D. Analysis of Critical Rate Processes for Ionization in Shock-Heated Air. AIAA AVIATION 2023 Forum, 2023, p. 3330.
- Voelkel, S.; Masselot, D.; Varghese, P.L.; Raman, V. Analysis of Hydrogen-Air Detonation Waves with Vibrational Nonequilibrium. AIP Conf. Proc. 2016, 1786, 070015. [Google Scholar]
- Shi, L.; Shen, H.; Zhang, P.; Zhang, D.; Wen, C. Assessment of vibrational non-equilibrium effect on detonation cell size. Combustion Science and Technology 2017, 189, 841–853. [Google Scholar] [CrossRef]
- Kadochnikov, I.N.; Arsentiev, I.V. Modelling of vibrational nonequilibrium effects on the H2–air mixture ignition under shock wave conditions in the state-to-state and mode approximations. Shock Waves 2020, 30, 491–504. [Google Scholar] [CrossRef]
- Skrebkov, O.V.; Kostenko, S.S.; Smirnov, A.L. Vibrational nonequilibrium and reaction heat effect in diluted hydrogen-oxygen mixtures behind reflected shock waves at 1000<T<1300 K. International Journal of Hydrogen Energy 2020, 45, 3251–3262. [Google Scholar]
- Smirnov, V.N.; Vlasov, P.A. Effects of hydrocarbon impurities, vibrational relaxation, and boundary-layer-induced pressure rise on the ignition of H2–O2–Ar mixtures behind reflected shock waves. International Journal of Hydrogen Energy 2021, 46, 9580–9594. [Google Scholar] [CrossRef]
- Colonna, G.; Capitelli, M. Self-consistent model of chemical, vibrational, electron kinetics in nozzle expansion. Journal of thermophysics and heat transfer 2001, 15, 308–316. [Google Scholar] [CrossRef]
- Guy, A.; Bourdon, A.; Perrin, M.Y. Consistent multi-internal-temperatures models for nonequilibrium nozzle flows. Chemical Physics 2013, 420, 15–24. [Google Scholar] [CrossRef]
- Zidane, A.; Haoui, R.; Sellam, M.; Bouyahiaoui, Z. Numerical study of a nonequilibrium H2-O2 rocket nozzle flow. International Journal of Hydrogen Energy 2019, 44, 4361–4373. [Google Scholar] [CrossRef]
- Starikovskiy, A.; Aleksandrov, N. Plasma-assisted ignition and combustion. Prog. Energy Combust. Sci. 2013, 39, 61–110. [Google Scholar] [CrossRef]
- Popov, N.A. Kinetics of plasma-assisted combustion: effect of non-equilibrium excitation on the ignition and oxidation of combustible mixtures. Plasma Sources Sci. Technol. 2016, 25, 043002(31pp). [Google Scholar] [CrossRef]
- Pietanza, L.D.; Colonna, G.; Capitelli, M. Activation of vibrational-induced CO2 dissociation in cold non-equilibrium plasma. Plasma Physics and Controlled Fusion 2023, 65, 044004. [Google Scholar] [CrossRef]
- Kadochnikov, I.N.; Arsentiev, I.V.; Loukhovitski, B.I.; Sharipov, A.S. State-to-state vibrational kinetics of diatomic molecules in laser-induced ignition of a syngas-air mixture: Modeling study. Chemical Physics 2022, 562, 111669. [Google Scholar] [CrossRef]
- Mankelevich, Y.A.; Rakhimova, T.V.; Voloshin, D.G.; Chukalovskii, A.A. Vibrationally Excited Ozone in Kinetics of O/N/Ar Mixtures after Ozone Photolysis. Russian Journal of Physical Chemistry A 2023, 97, 1033–1045. [Google Scholar] [CrossRef]
- Capitelli, M.; Ferreira, C.M.; Gordiets, B.F.; Osipov, A.I. Plasma kinetics in atmospheric gases; Vol. 31, Springer Series on Atomic, Optical, and Plasma Physics, Springer-Verlag, Berlin, 2000.
- Truhlar, D.G.; Muckerman, J.T. Reactive scattering cross sections III: Quasiclassical and semiclassical methods. In Atom-Molecule Collision Theory: A Guide for the Experimentalist; Springer, 1979; pp. 505–566.
- Gray, S.K.; Balint-Kurti, G.G. Quantum dynamics with real wave packets, including application to three-dimensional (J=0) D+H2=HD+H reactive scattering. The Journal of chemical physics 1998, 108, 950–962. [Google Scholar] [CrossRef]
- Gorse, C.; Capitelli, M.; Bacal, M.; Bretagne, J.; Laganà, A. Progress in the non-equilibrium vibrational kinetics of hydrogen in magnetic multicusp H-ion sources. Chemical physics 1987, 117, 177–195. [Google Scholar] [CrossRef]
- Pogosbekian, M.J.; Sergievskaia, A.L.; Losev, S.A. Verification of theoretical models of chemical exchange reactions on the basis of quasiclassical trajectory calculations. Chemical Physics 2006, 328, 371–378. [Google Scholar] [CrossRef]
- Esposito, F.; Armenise, I.; Capitelli, M. N–N2 state to state vibrational-relaxation and dissociation rates based on quasiclassical calculations. Chemical Physics 2006, 331, 1–8. [Google Scholar] [CrossRef]
- Bender, J.D.; Valentini, P.; Nompelis, I.; Paukku, Y.; Varga, Z.; Truhlar, D.G.; Schwartzentruber, T.; Candler, G.V. An improved potential energy surface and multi-temperature quasiclassical trajectory calculations of N2+N2 dissociation reactions. The Journal of chemical physics 2015, 143. [Google Scholar] [CrossRef] [PubMed]
- Andrienko, D.A.; Boyd, I.D. State-specific dissociation in O2–O2 collisions by quasiclassical trajectory method. Chemical Physics 2017, 491, 74–81. [Google Scholar] [CrossRef]
- Macdonald, R.L.; Jaffe, R.L.; Schwenke, D.W.; Panesi, M. Construction of a coarse-grain quasi-classical trajectory method. I. Theory and application to N2–N2 system. The Journal of chemical physics 2018, 148. [Google Scholar] [CrossRef]
- Vargas, J.; Monge-Palacios, M.; Lacoste, D.A. State-Specific Dissociation and Inelastic Rate Constants for Collisions of H2 with H and He. Journal of Thermophysics and Heat Transfer 2023. pp. 1–12. [Google Scholar]
- Kuntz, P.J.; Nemeth, E.M.; Polanyi, J.C.; Rosner, S.D.; Young, C.E. Energy distribution among products of exothermic reactions. II. Repulsive, mixed, and attractive energy release. The Journal of Chemical Physics 1966, 44, 1168–1184. [Google Scholar] [CrossRef]
- Sharipov, A.S.; Loukhovitski, B.I. Energy disposal into the vibrational degrees of freedom of bimolecular reaction products: Key factors and simple model. Chemical Physics 2021, 544, 111098. [Google Scholar] [CrossRef]
- Dawes, R.; Thompson, D.L.; Guo, Y.; Wagner, A.F.; Minkoff, M. Interpolating moving least-squares methods for fitting potential energy surfaces: Computing high-density potential energy surface data from low-density ab initio data points. The Journal of chemical physics 2007, 126. [Google Scholar] [CrossRef]
- Jiang, B.; Li, J.; Guo, H. Potential energy surfaces from high fidelity fitting of ab initio points: The permutation invariant polynomial-neural network approach. International Reviews in Physical Chemistry 2016, 35, 479–506. [Google Scholar] [CrossRef]
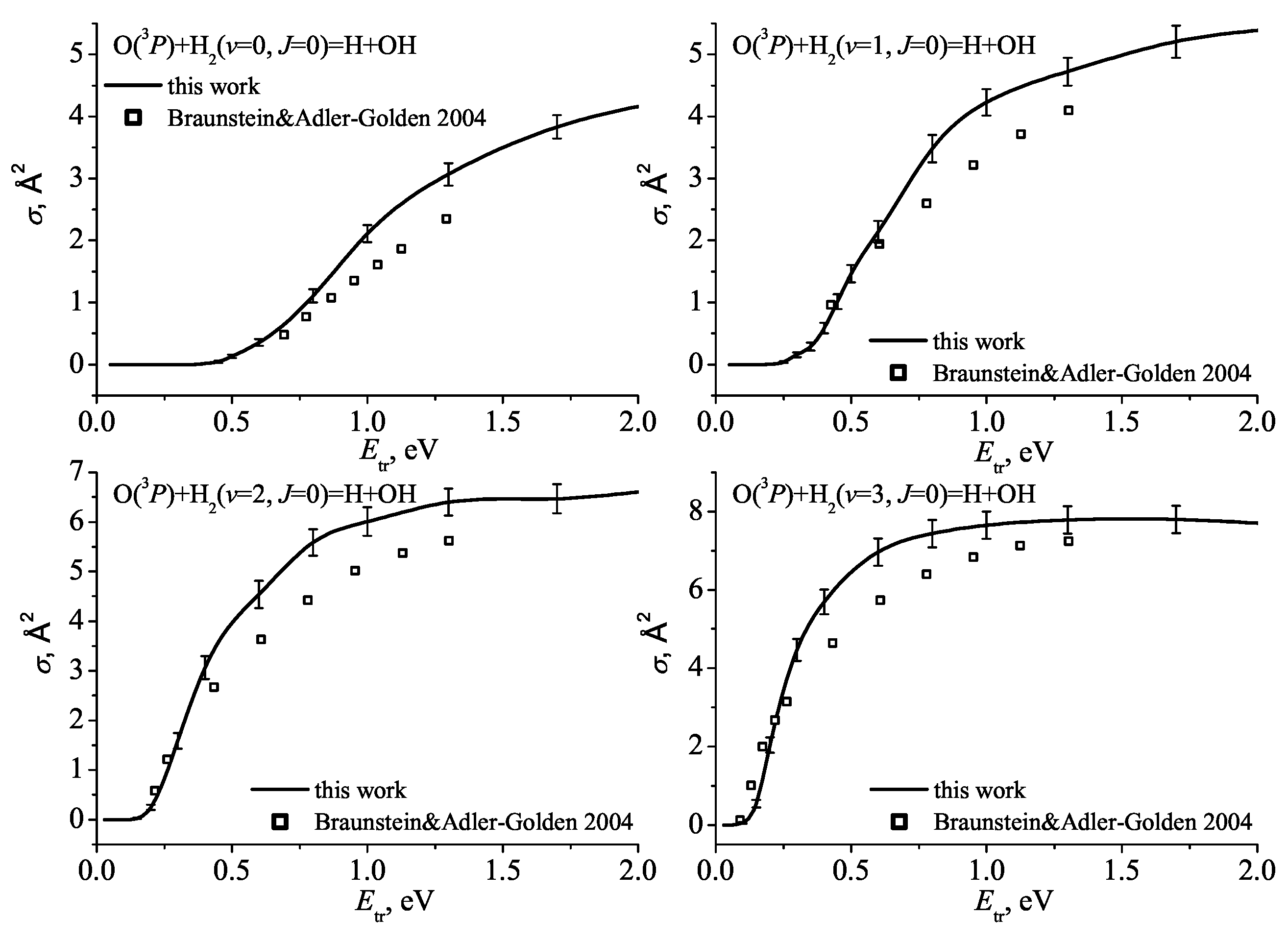
- Braunstein, M.; Adler-Golden, S.; Maiti, B.; Schatz, G. Quantum and classical studies of the O(3P)+H2(v=0-3, j=0)→OH+H reaction using benchmark potential surface. Technical Report, 2003; AIR FORCE RESEARCH 116 LAB EDWARDS AFB CA SPACE AND MISSILE PROPULSION DIV. [Google Scholar]
- Braunstein, M.; Adler-Golden, S.; Maiti, B.; Schatz, G. Quantum and classical studies of the O(3P)+H2(v=0–3, j=0)→OH+H reaction using benchmark potential surfaces. The Journal of chemical physics 2004, 120, 4316–4323. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Rosa, C.; Brandão, J. Theoretical studies on the O(3P)+H2→OH+H reaction. Chemical physics letters 2006, 418, 250–254. [Google Scholar] [CrossRef]
- Xu, Z.H.; Zong, F.J.; Han, B.R.; Dong, S.H.; Liu, J.Q.; Ji, F. Effects of a reagent’s rotational and vibrational excitations on reaction O(3P)+H2(n= 0, 3, j= 0, 3, 5, 7, 9, 12, 15)→OH+H. Chinese Physics B 2012, 21, 093103. [Google Scholar] [CrossRef]
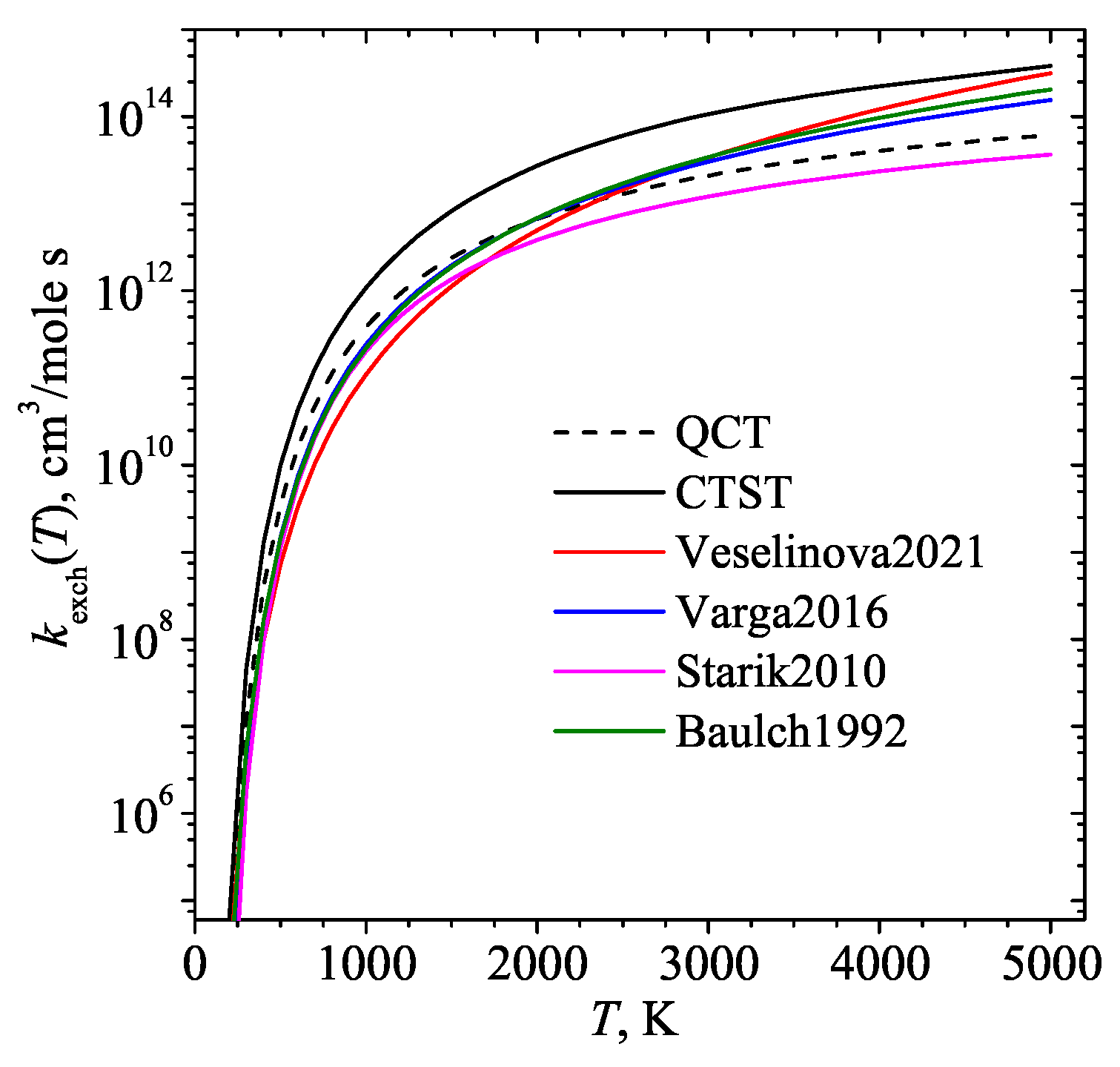
- Veselinova, A.; Agúndez, M.; Goicoechea, J.R.; Menéndez, M.; Zanchet, A.; Verdasco, E.; Jambrina, P.; Aoiz, F.J. Quantum study of reaction O(3P)+H2(v,j)→OH+H: OH formation in strongly UV-irradiated gas. Astronomy & Astrophysics 2021, 648, A76. [Google Scholar]
- Granovsky, A.A. Extended Multi-Configuration Quasi-Degenerate Perturbation Theory: The New Approach to Multi-state Multi-Reference Perturbation Theory. J. Chem. Phys. 2011, 134, 214113. [Google Scholar] [CrossRef] [PubMed]
- Deskevich, M.P.; Nesbitt, D.J. Dynamically weighted multiconfiguration self-consistent field: Multistate calculations for F+H2O→HF+OH reaction paths. J. Chem. Phys. 2004, 120, 7281. [Google Scholar] [CrossRef] [PubMed]
- Pelevkin, A.V.; Loukhovitski, B.I.; Sharipov, A.S. Reaction of the N Atom with Electronically Excited O2 Revisited: A Theoretical Study. The Journal of Physical Chemistry A 2021, 125, 8294–8312. [Google Scholar] [CrossRef]
- Kendall, R.A.; Dunning Jr., T. H.; Harrison, R.J. Electron Affinities of the First-Row Atoms Revisited. Systematic Basis Sets and Wave Functions. J. Chem. Phys. 1992, 96, 6796–6806. [Google Scholar] [CrossRef]
- Lesiuk, M.; Jeziorski, B. Complete basis set extrapolation of electronic correlation energies using the Riemann zeta function. Journal of Chemical Theory and Computation 2019, 15, 5398–5403. [Google Scholar] [CrossRef] [PubMed]
- Granovsky, A.A. Firefly V. 8.2.0. http://classic.chem.msu.su/gran/firefly/index.html, 2016.
- Schmidt, M.W.; Baldridge, K.K.; Boatz, J.A.; Elbert, S.T.; Gordon, M.S.; Jensen, J.H.; Koseki, S.; Matsunaga, N.; Nguyen, K.A.; Su, S.; Windus, T.L.; Dupuis, M.; Montgomery, J.A. General Atomic and Molecular Electronic Structure System. J. Comput. Chem. 1993, 14, 1347–1363. [Google Scholar] [CrossRef]
- Abadi, M.; Agarwal, A.; Barham, P.; Brevdo, E.; Chen, Z.; Citro, C.; Corrado, G.S.; Davis, A.; Dean, J.; Devin, M.; Ghemawat, S.; Goodfellow, I.; Harp, A.; Irving, G.; Isard, M.; Jia, Y.; Jozefowicz, R.; Kaiser, L.; Kudlur, M.; Levenberg, J.; Mané, D.; Monga, R.; Moore, S.; Murray, D.; Olah, C.; Schuster, M.; Shlens, J.; Steiner, B.; Sutskever, I.; Talwar, K.; Tucker, P.; Vanhoucke, V.; Vasudevan, V.; Viégas, F.; Vinyals, O.; Warden, P.; Wattenberg, M.; Wicke, M.; Yu, Y.; Zheng, X. TensorFlow: Large-Scale Machine Learning on Heterogeneous Systems, 2015. Software available from tensorflow.org.
- Paszke, A.; Gross, S.; Chintala, S.; Chanan, G.; Yang, E.; DeVito, Z.; Lin, Z.; Desmaison, A.; Antiga, L.; Lerer, A. Automatic differentiation in PyTorch. NIPS-W, 2017.
- Chollet, Francois and others. Keras. https://github.com/fchollet/keras, 2015. GitHub.
- Braams, B.J.; Bowman, J.M. Permutationally invariant potential energy surfaces in high dimensionality. International Reviews in Physical Chemistry 2009, 28, 577–606. [Google Scholar] [CrossRef]
- Bosma, W.; Cannon, J.; Playoust, C. The Magma algebra system I: The user language. Journal of Symbolic Computation 1997, 24, 235–265. [Google Scholar] [CrossRef]
- Decker, W.; Greuel, G.M.; Pfister, G.; Schönemann, H. Singular 4-1-2—A computer algebra system for polynomial computations. http://www. singular. uni-kl. de, 2019. [Google Scholar]
- Polak, L.S.; Goldenberg, M.J.; Levickij, A.A. Vychislitel’nye metody v himicheskoj kinetike [Computational methods in chemical kinetics]; Nauka, 1984.
- Abdel-Halim, H.M.; Jaafreh, S.M. Reaction rate constants from classical trajectories of atom-diatomic molecule collisions. Zeitschrift für Naturforschung A 2008, 63, 159–169. [Google Scholar] [CrossRef]
- Le Roy, R.J. LEVEL: A computer program for solving the radial Schrödinger equation for bound and quasibound levels. Journal of Quantitative Spectroscopy and Radiative Transfer 2017, 186, 167–178. [Google Scholar] [CrossRef]
- Varga, T.; Olm, C.; Nagy, T.; Zsély, I.G.; Valkó, É.; Pálvölgyi, R.; Curran, H.J.; Turányi, T. Development of a joint hydrogen and syngas combustion mechanism based on an optimization approach. International journal of chemical kinetics 2016, 48, 407–422. [Google Scholar] [CrossRef]
- Starik, A.M.; Sharipov, A.S.; Titova, N.S. Intensification of syngas ignition through the excitation of CO molecule vibrations: a numerical study. Journal of Physics D: Applied Physics 2010, 43, 245501. [Google Scholar] [CrossRef]
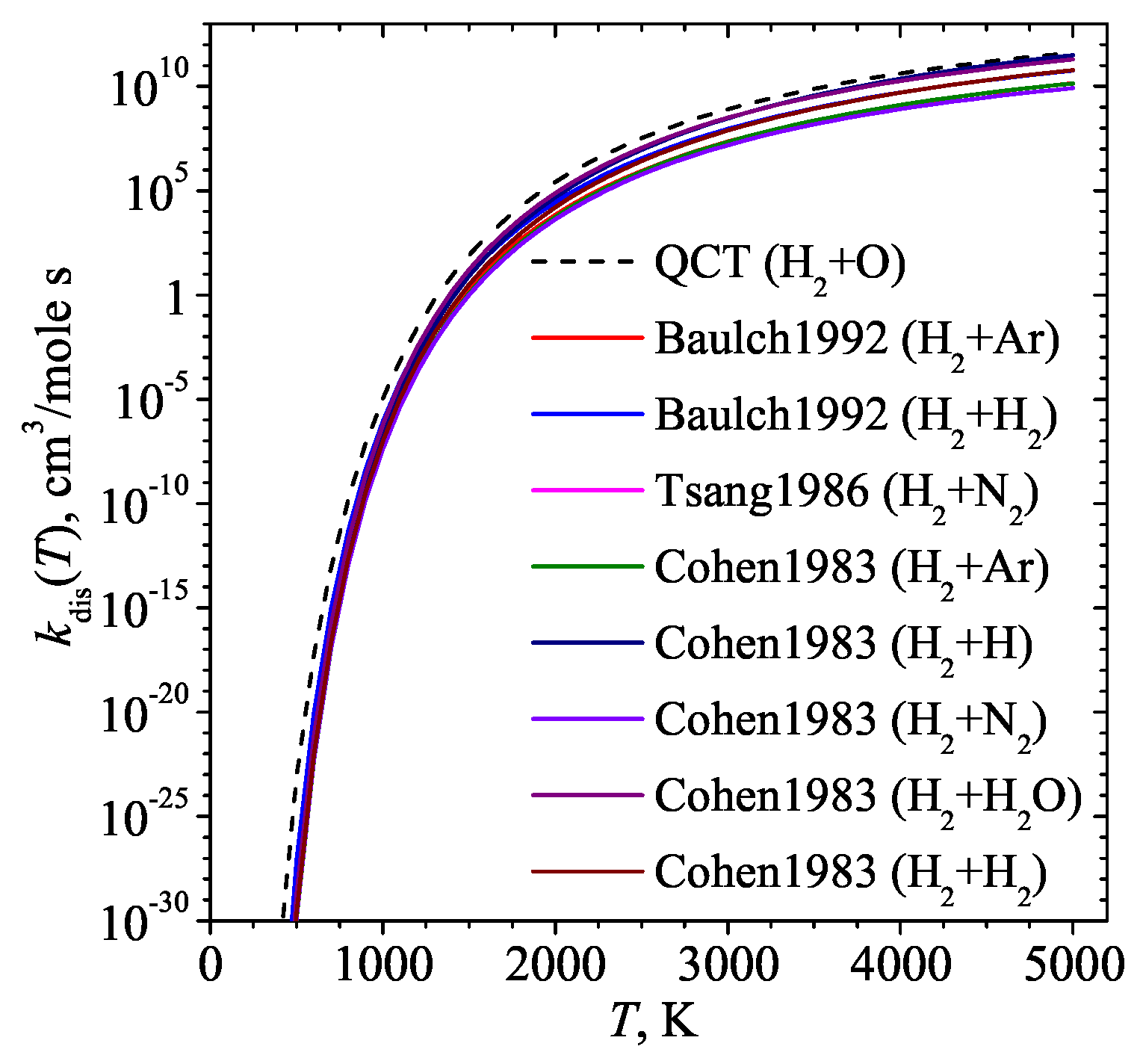
- Atkinson, R.; Baulch, D.L.; Cox, R.A.; Hampson, J.R.F.; Kerr, J.A.; Troe, J. Evaluated Kinetic and Photochemical Data for Atmospheric Chemistry. Supplement IV. J. Phys. Chem. Ref. Data 1992, 21, 1125–568. [Google Scholar] [CrossRef]
- Tsang, W.; Hampson, R.F. Chemical Kinetic Data Base for Combustion Chemistry. J. Phys. Chem. Ref. Data. 1986, 15, 1087–1280. [Google Scholar] [CrossRef]
- Cohen, N.; Westberg, K.R. Chemical kinetic data sheets for high-temperature chemical reactions. Journal of physical and chemical reference data 1983, 12, 531–590. [Google Scholar] [CrossRef]
- Dove, J.E.; Teitelbaum, H. The vibrational relaxation of H2. I. Experimental measurements of the rate of relaxation by H2, He, Ne, Ar, and Kr. Chemical Physics 1974, 6, 431–444. [Google Scholar] [CrossRef]
- Cacciatore, M.; Capitelli, M.; Billing, G. Vibration-to-translation energy exchanges in H2 colliding with highly vibrationally excited H2 molecules. Chemical physics letters 1989, 157, 305–308. [Google Scholar] [CrossRef]
- Millikan, R.C.; White, D.R. Systematics of vibrational relaxation. J. Chem. Phys. 1963, 39, 3209–3213. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



