Submitted:
05 December 2023
Posted:
06 December 2023
You are already at the latest version
Abstract
Keywords:
Introduction
Case presentation
| Diagnostic Test | Normal Value | At Presentation | Hospital Day 2 | Hospital Day 6 | On Discharge | Follow- Up |
|---|---|---|---|---|---|---|
| Hemoglobin, g/dL | 11.6-15.0 | 9.0 | 7.6 | 9.1 | 7.3 | |
| WBC count, ×10(9) /L | 3.4-9.6 | 6.0 | 4.3 | 16.8 | 9.2 | |
| Platelet count, ×10(9) /L | 157-371 | 243 | 207 | 168 | 124 | |
| Serum Urea Nitrogen, mg/dL | 6-21 | 69 | 62 | 55 | 34 | 23 |
| Serum Creatinine, mg/dL | 0.59-1.04 | 10.15 | 9.10 | 8.03 | 5.03 | 1.6 |
| Serum Sodium | 135-145 mmol/l | 136 | 143 | 143 | 144 | 137 |
| Serum Potassium | 3.6-5.2 mmol/l | 2.7 | 3.6 | 3.9 | 3.4 | 3.9 |
| Serum Chloride | 98-107 mmol/l | 95 | 106 | 108 | 109 | 98 |
| Serum Calcium |
8.8-10.2 mg/dl | 9.4 | 8.8 | 8.6 | 8.4 | 9.7 |
| Serum Phosphorus | 2.5 - 4.5 mg/dl | 5.5 | 5.5 | 5.2 | 3.8 | |
| Serum Magnesium | 1.7- 2.3 mg/dl | 2.3 | 2.0 | 1.6 | ||
| Serum Bicarbonate | 22-29 mmol/l | 24 | 21 | 21 | 22 | 26 |
| eGFR | >=60 mL/min/BSA | < 15 | <15 | <15 | <15 | 34 |
| Anion Gap | 7-15 | 17 | 16 | 14 | 13 | 13 |
| Serum Albumin | 3.5-5.0 g/dl | 3.7 | 4.1 |
| Diagnostic Test | Normal Value | Result |
|---|---|---|
| HCV Ab screen, S | Negative | Negative |
| HBs antibody, S | Negative | Negative |
| HBs antigen, S | Non-reactive | Non-reactive |
| HBc total ab w/reflex, S | Negative | Negative |
| Complement, C3 | 75-175 mg/dL | 143 mg/dl |
| Complement, C4 | 14-40 mg/dL | 27 mg/dl |
| Cryoglobulins | Negative | Negative |
| GBM, IgG Ab | <0.1(Negative) U | <0.2 U |
| MPO, Ab | <0.4(Negative) U | <0.2 |
| PR 3, Ab | <0.4(Negative) U | <0.2 |
| SPEP | No monoclonal protein | |
| Haptoglobin, S | 30-200 mg/dl | 297 |
| Kappa Free light Chain, S | 0.3300 - 1.94 mg/dL | 13.7 |
| Lambda free light chain, S | 0.5700 - 2.63 mg/dL | 8.08 |
| Kappa/Lambda FLC Ratio | 0.2600 – 1.65 | 1.70 |
| DNA double stranded Ab, IgG, S | <=4 (Negative) IU/mL | 2 |
| Sm. Ab, IgG, S | <1.0(Negative) U | <0.2 |
| CRP | <=8.0 mg/L | < 3.0 mg/l |
| Bilirubin, Total | <=1.2 mg/L | 0.8 |
| ALT, S | 7 - 45 U/L | 11 |
| AST, S | 8 - 43 U/L | 17 |
| Alkaline phosphatase | 35 – 104 U/L | 60 |
| Protein, total | 6.3 – 7.9 g/dL | 6.9 g/dl |
| Albumin | 3.5-5.0 g/dL | 3.7 g/dl |
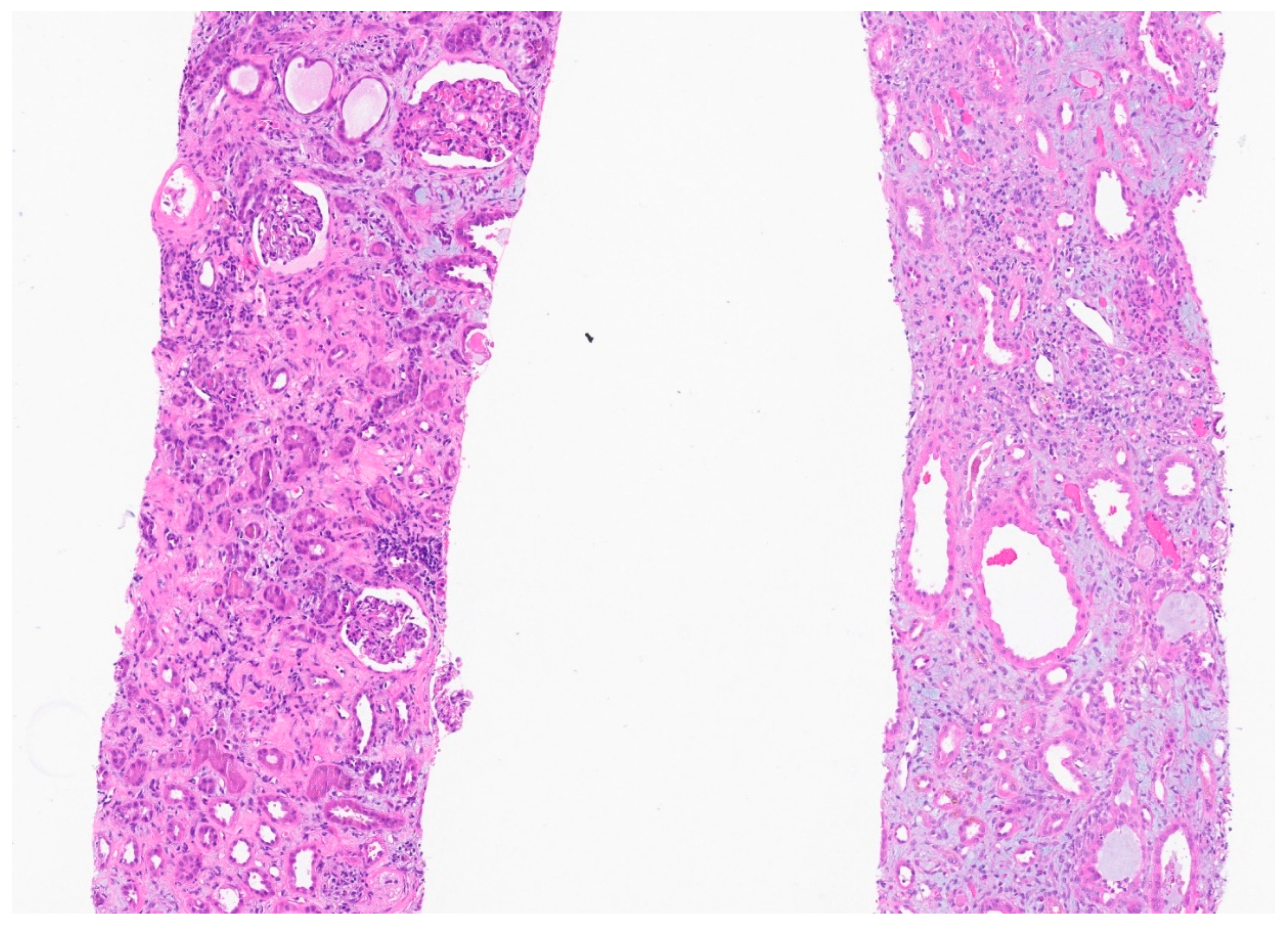
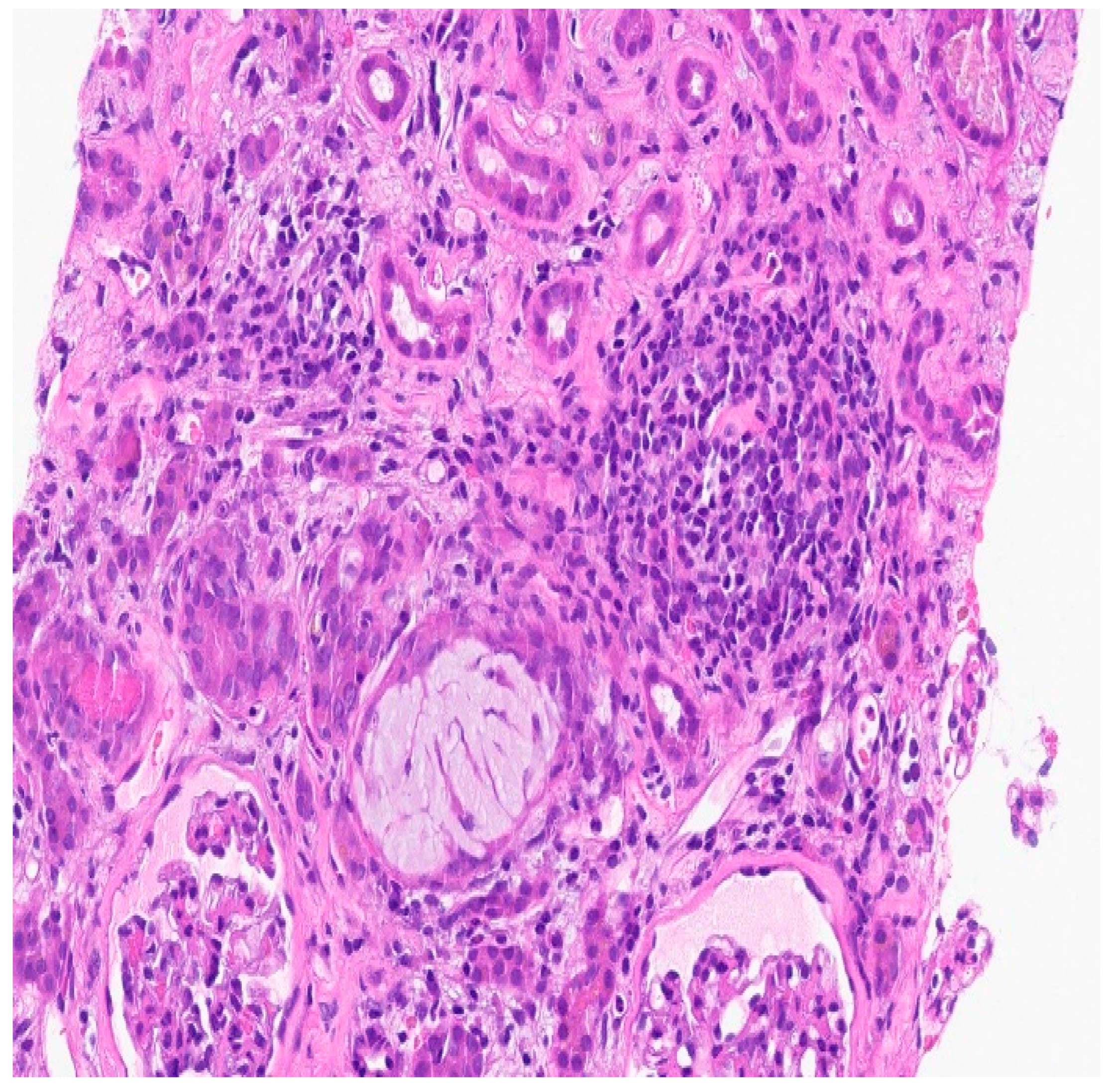
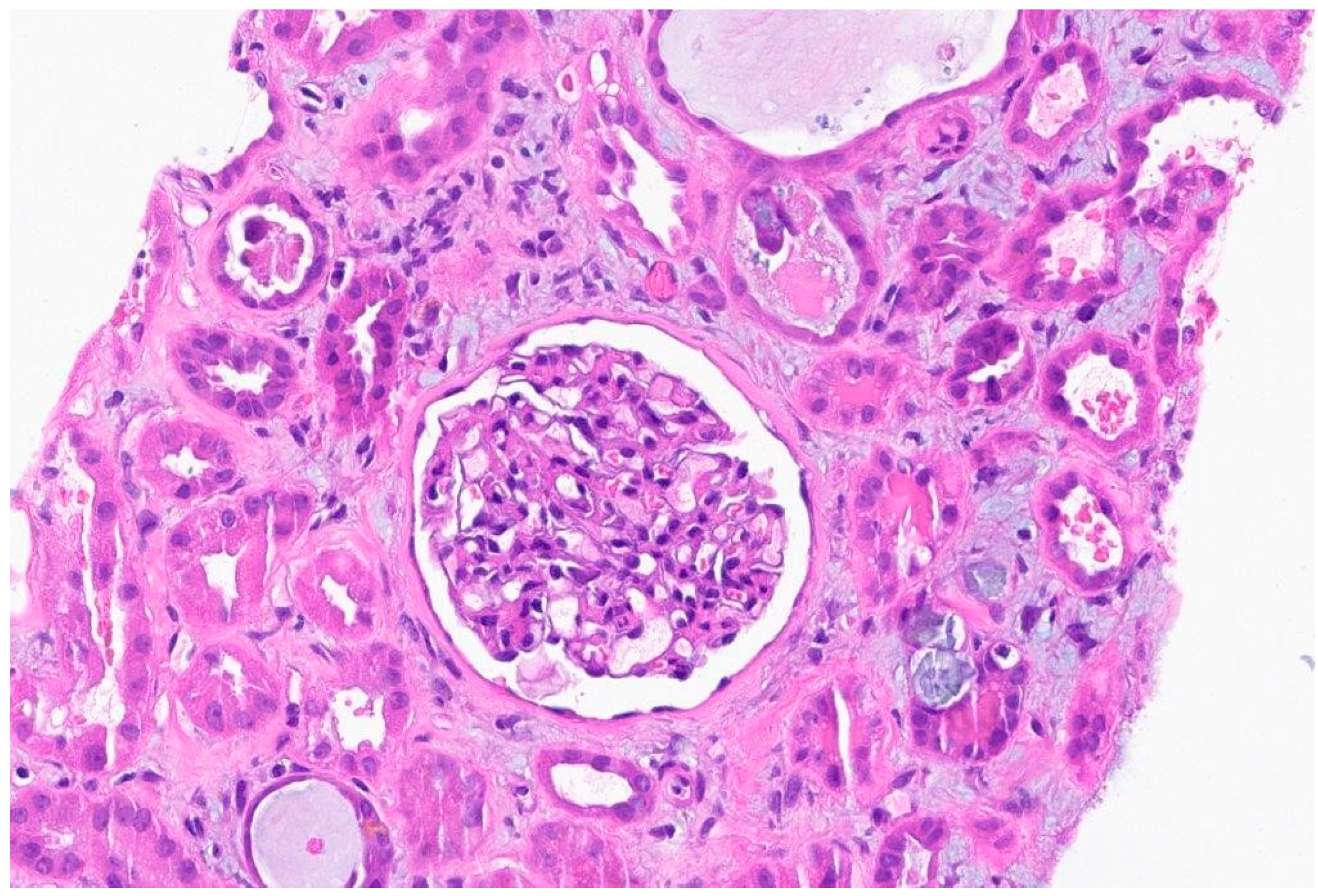
Biopsy report:
Discussion:
Author Contributions
Funding
Ethics approval
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Magil, A.B.; et al. Acute interstitial nephritis associated with thiazide diuretics. Clinical and pathologic observations in three cases. Am J Med 1980, 69, 939–43. [Google Scholar] [CrossRef] [PubMed]
- Perazella, M.A.; Rosner, M.H. Drug-Induced Acute Kidney Injury. Clin J Am Soc Nephrol 2022, 17, 1220–1233. [Google Scholar] [CrossRef] [PubMed]
- Yarlagadda, S.G.; Perazella, M.A. Drug-induced crystal nephropathy: an update. Expert Opin Drug Saf 2008, 7, 147–58. [Google Scholar] [CrossRef] [PubMed]
- Nasr, S.H.; et al. Triamterene crystalline nephropathy. Am J Kidney Dis 2014, 63, 148–52. [Google Scholar] [CrossRef] [PubMed]
- Farge, D.; et al. Dyazide-induced reversible acute renal failure associated with intracellular crystal deposition. Am J Kidney Dis 1986, 8, 445–9. [Google Scholar] [CrossRef] [PubMed]
- Ansari, F.A.; et al. A Rare Case of Acute Kidney Injury Due to Levofloxacin-induced Crystal Nephropathy. Indian J Nephrol 2019, 29, 424–426. [Google Scholar] [CrossRef] [PubMed]
- Garneau, A.P.; Riopel, J.; Isenring, P. Acute Methotrexate-Induced Crystal Nephropathy. N Engl J Med 2015, 373, 2691–3. [Google Scholar] [CrossRef] [PubMed]
- Goli, R.; et al. Acute Ciprofloxacin-Induced Crystal Nephropathy with Granulomatous Interstitial Nephritis. Indian J Nephrol 2017, 27, 231–233. [Google Scholar] [CrossRef] [PubMed]
- Geraghty, R.; Wood, K.; Sayer, J.A. Calcium oxalate crystal deposition in the kidney: identification, causes and consequences. Urolithiasis 2020, 48, 377–384. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




