Submitted:
01 November 2023
Posted:
02 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental
2.1. Materials
2.1.1. Materials for Preparation and Characterization of PET NPs
2.1.2. Materials for Cellular Assays
2.2. Methods
2.2.1. Preparation of PET NPs
2.2.2. NPs Size Separation and Removal of Impurities
2.2.3. Preparation of NPs for NMR Analysis
2.2.4. Determination of Concentration of NPs in Water Suspensions
2.2.5. Determination of NPs Size
2.2.6. Determination of Zeta Potential of NPs
2.3. Effects of NPs on Cells
2.3.1. Isolation of Peripheral Blood Mononuclear Cells (PBMCs)
2.3.2. MTT Assay
2.3.3. Annexin V Assay
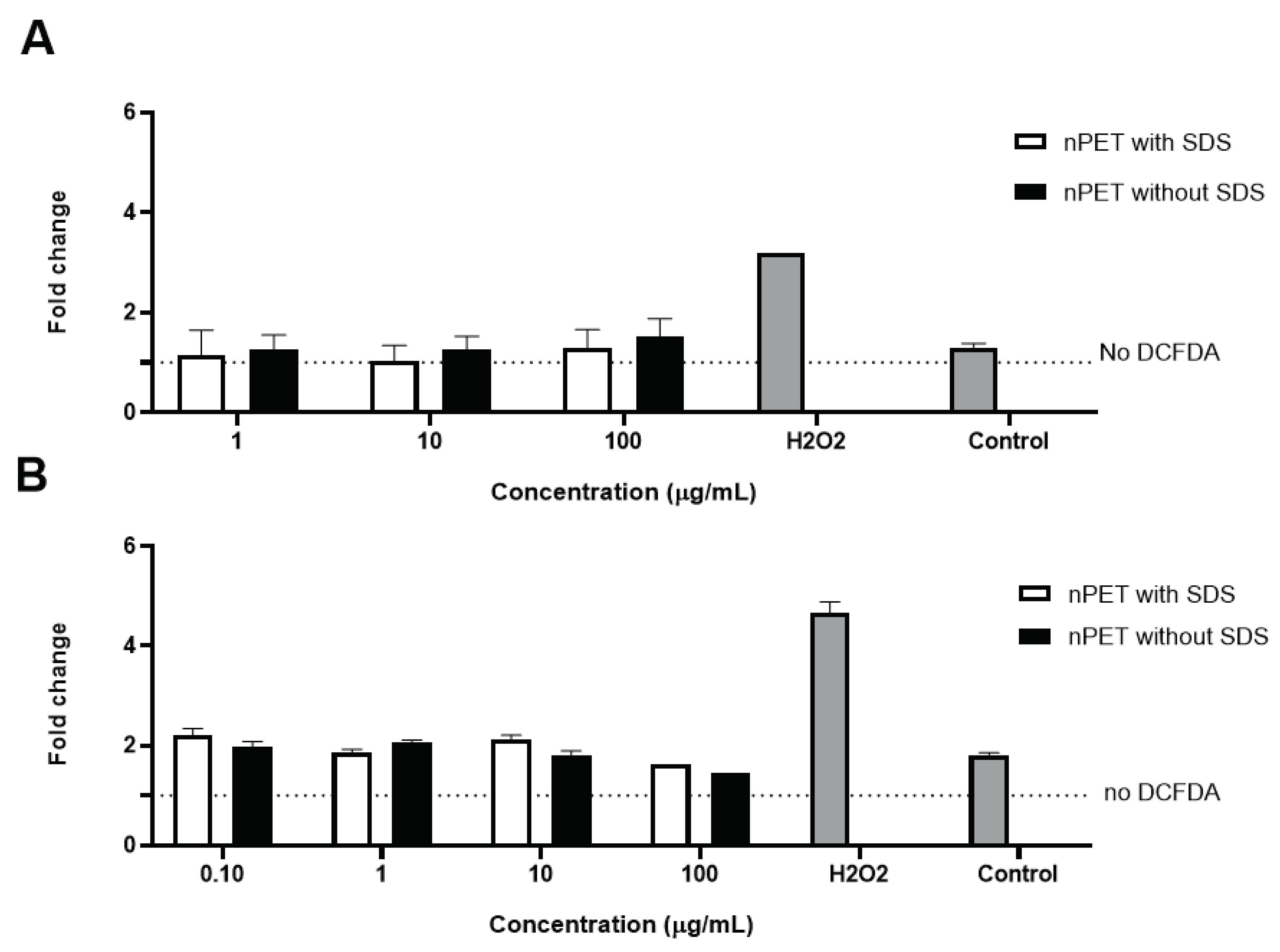
2.3.4. DCFDA Cellular ROS Assay
2.3.5. Uptake of NPs by Monocytes
2.3.6. Hemolysis of Red Blood Cells
2.4. Statistics and Graph Generation
2.5. Ethics
3. Results and Discussion
3.1. PET NPs Characterization
3.1.1. Yield and Concentration Determination of Synthetized NPs
3.1.2. PET NPs Size Distribution
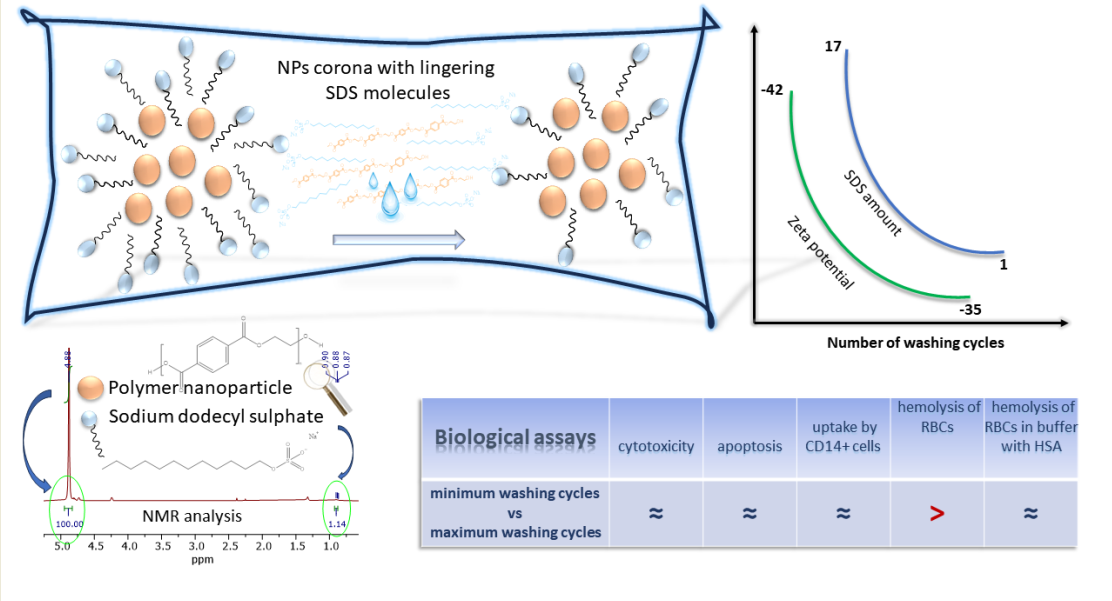
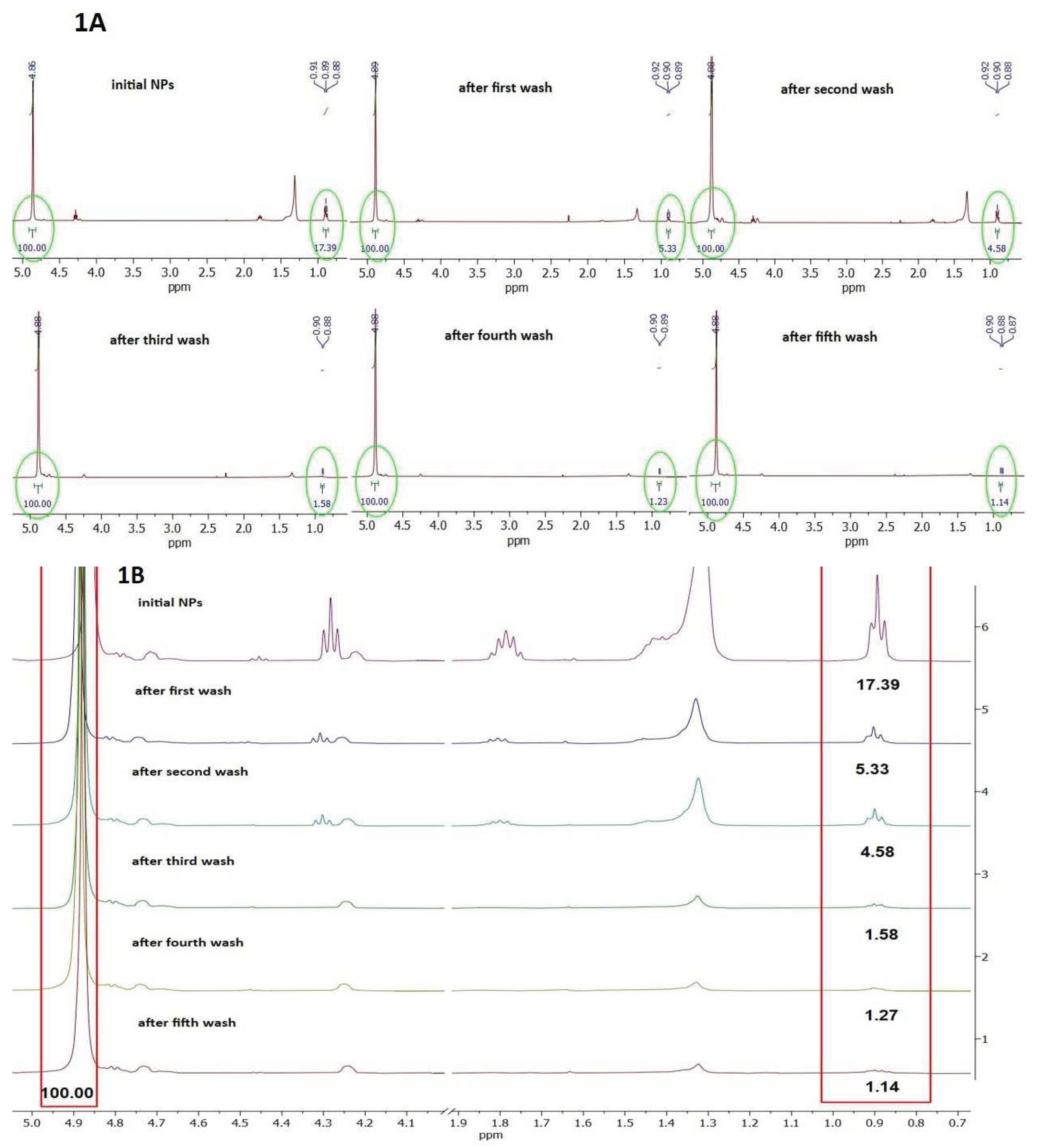
3.1.3. Determination of SDS Level in Corona of PET NPs by 1H NMR
3.1.4. Zeta Potential Determination of PET NPs
3.2. Effects of Residual Ionic Surfactant in NPs Preparations in Cellular Assays
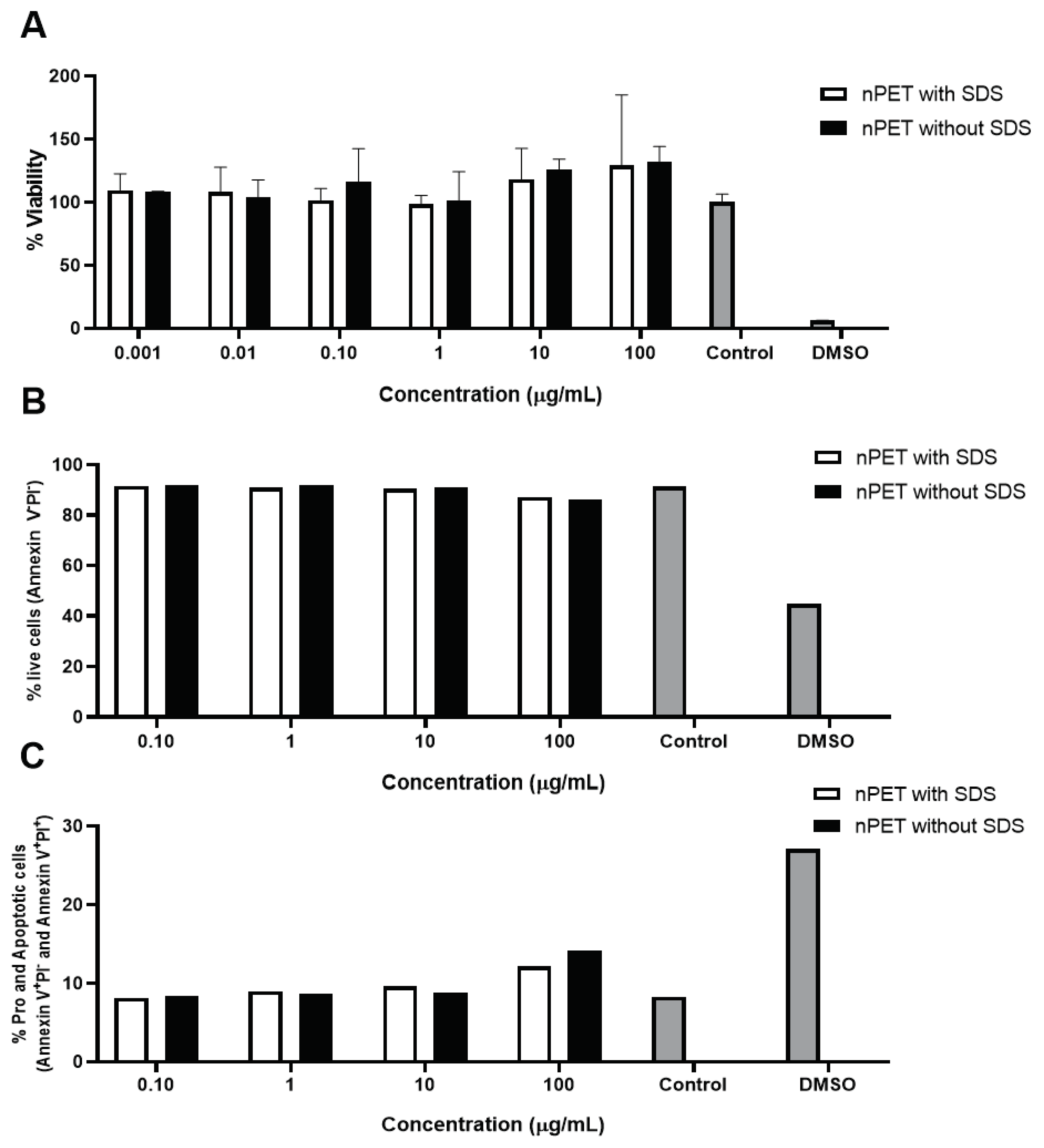
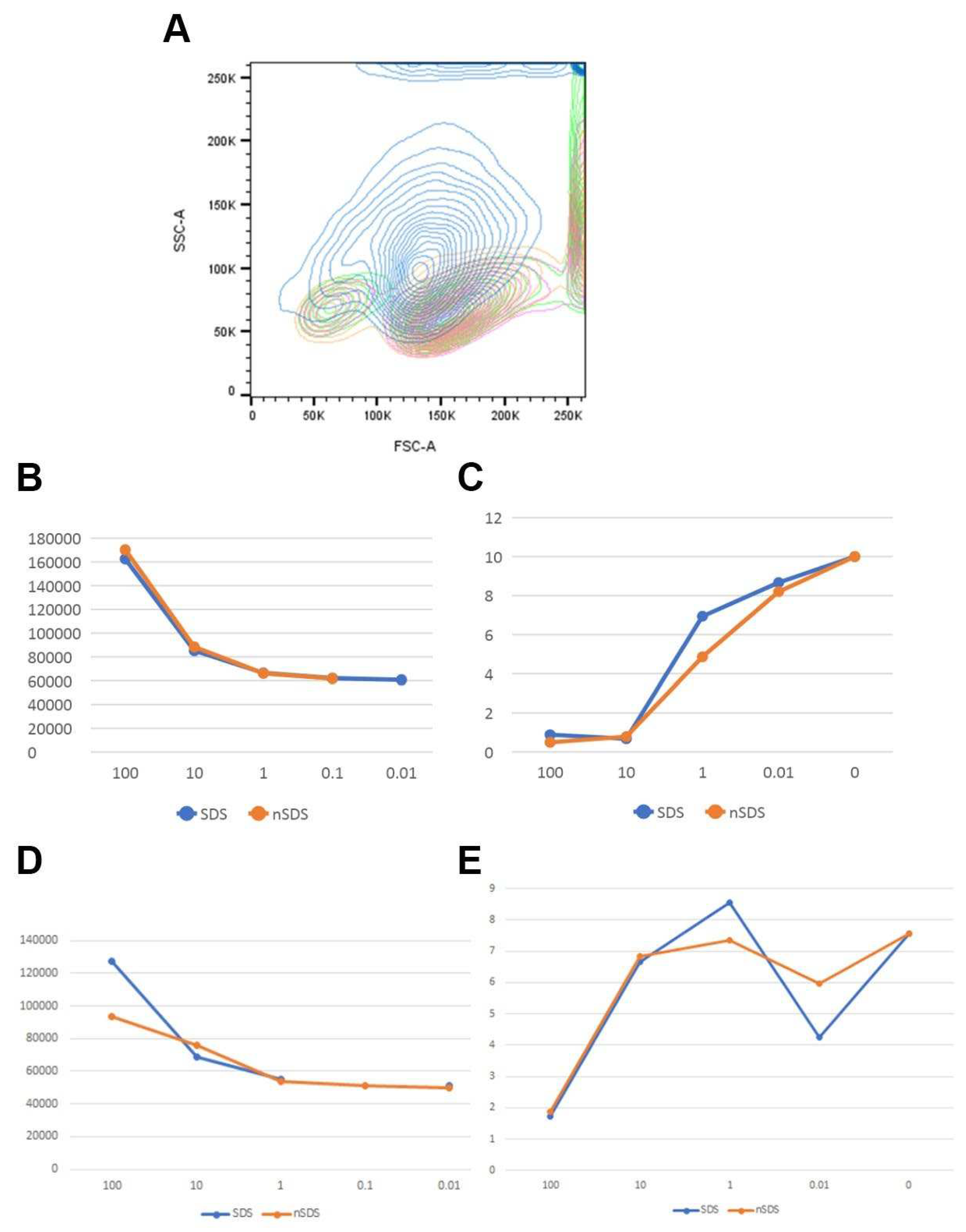
3.2.1. Cytotoxic Effects of PET NPs on Human Peripheral Blood Mononuclear Cells (PBMCs)
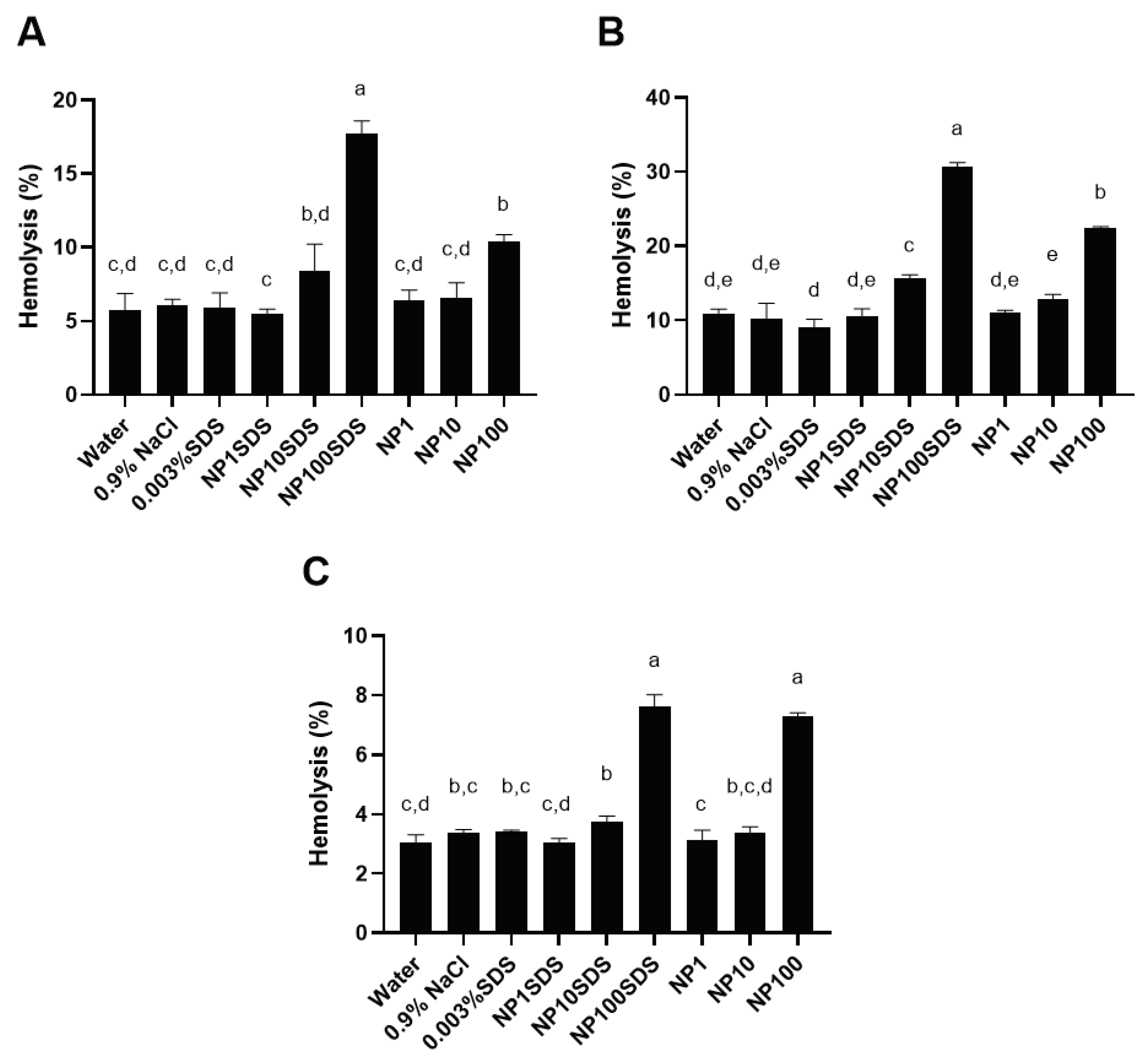
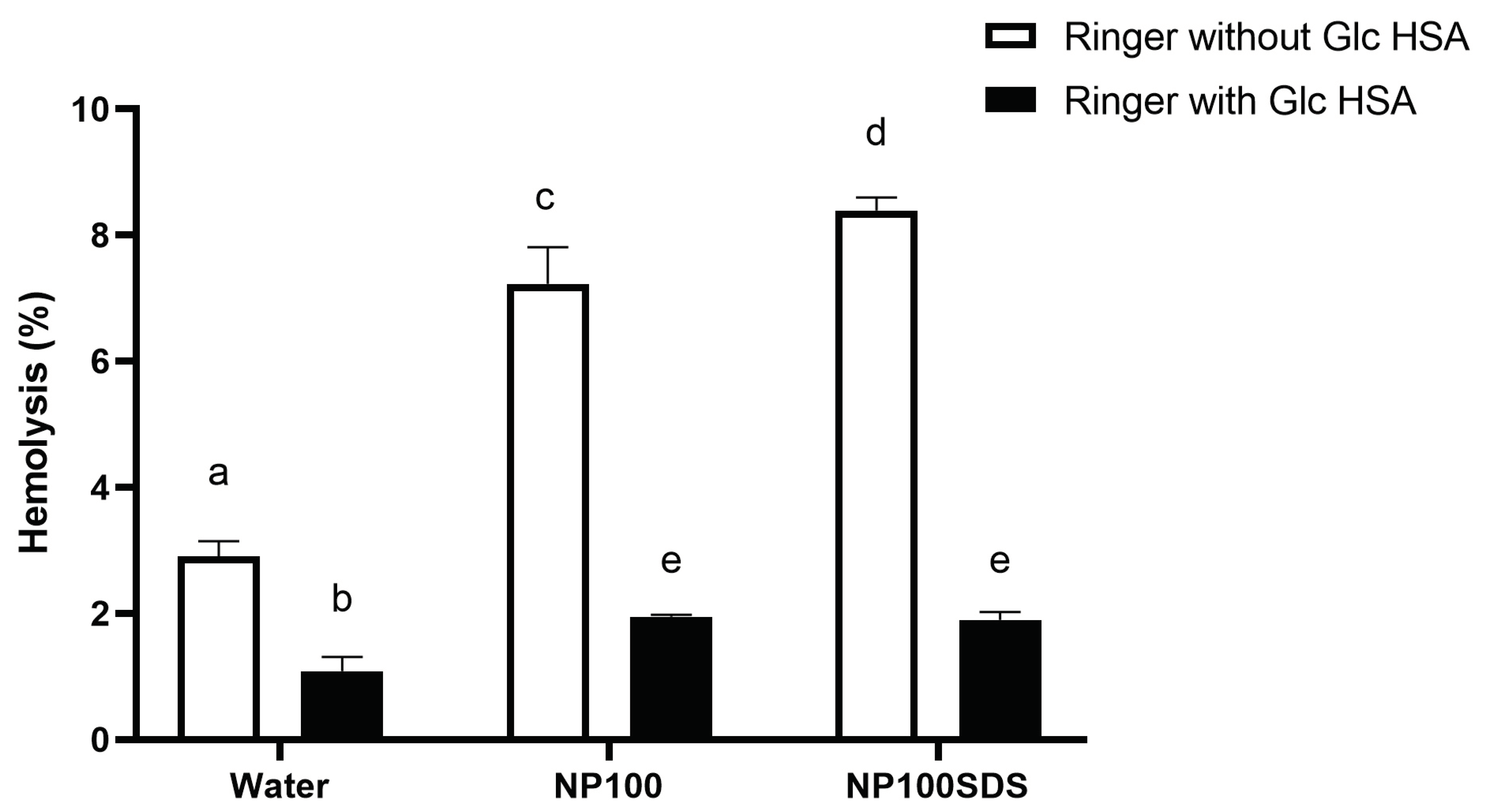
3.2.2. Hemolytic Effects of PET NPs on Human Red Blood Cells
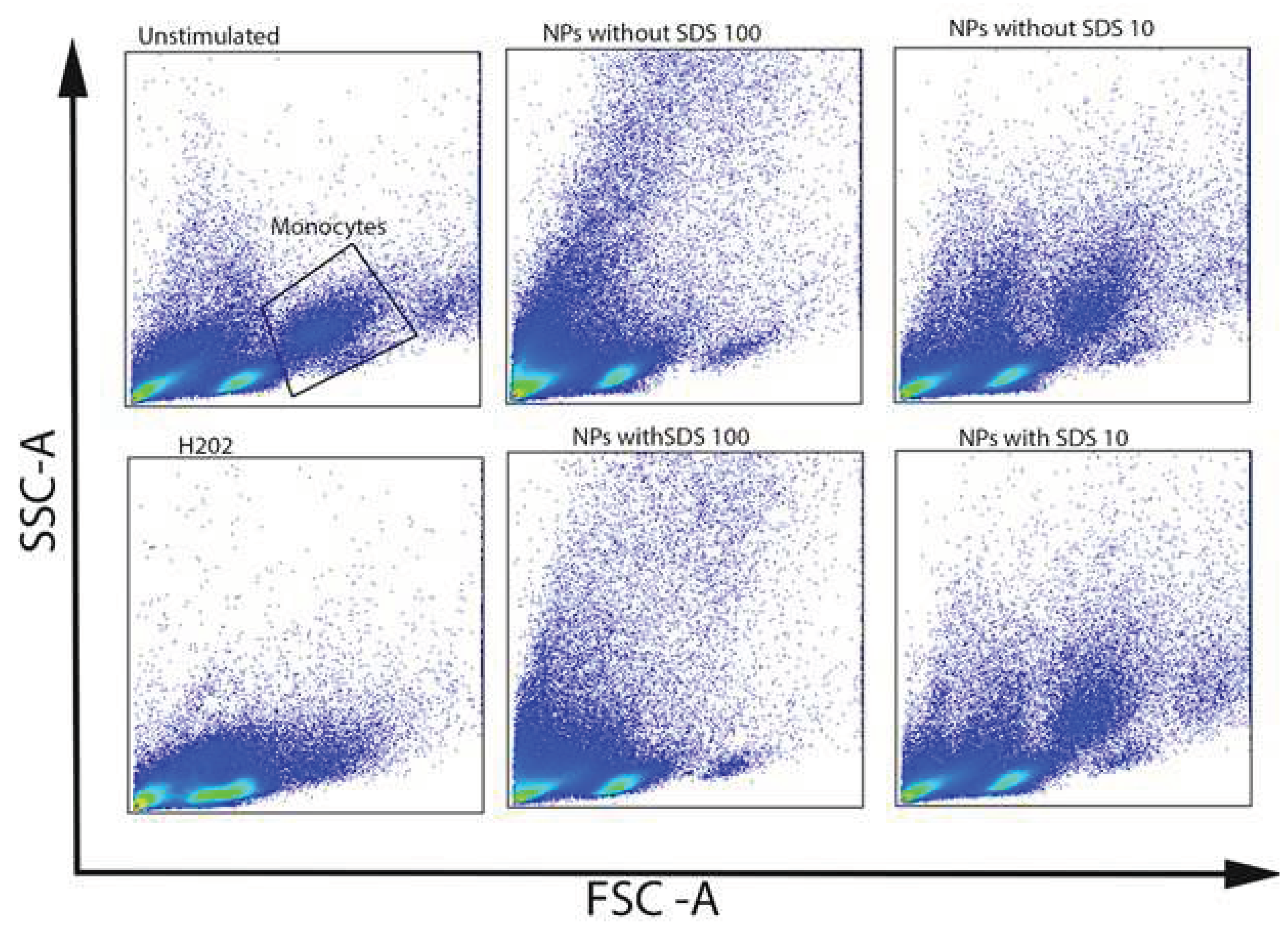
3.2.3. Uptake of NPs by CD14+ Monocytes
4. Conclusions
Supplementary Materials
Funding
References
- Cai, H.; Xu, E.G.; Du, F.; Li, R.; Liu, J.; Shi, H. Analysis of environmental nanoplastics: Progress and challenges. Chem. Eng. J. 2021, 410, 128208. [Google Scholar] [CrossRef]
- Merdy, P.; Delpy, F.; Bonneau, A.; Villain, S.; Iordachescu, L.; Vollertsen, J.; Lucas, Y. Nanoplastic Production Procedure for Scientific Purposes: PP, PVC, PE-LD, PE-HD, and PS. Heliyon 2023, 9. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Cizdziel, J.V.; Wontor, K.; Olubusoye, B.S. Adsorption/Desorption of Mercury (II) by Artificially Weathered Microplastics: Kinetics, Isotherms, and Influencing Factors. Environ. Pollut. 2023, 337, 122621. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, J.; Thünemann, A.F. Aqueous Dispersions of Polypropylene: Toward Reference Materials for Characterizing Nanoplastics. Macromol. Rapid Commun. 2023, 44, 2200874. [Google Scholar] [CrossRef] [PubMed]
- Petersen, Elijah.J.; Barrios, A.C.; Henry, T.B.; Johnson, M.E.; Koelmans, A.A.; Montoro Bustos, A.R.; Matheson, J.; Roesslein, M.; Zhao, J.; Xing, B. Potential Artifacts and Control Experiments in Toxicity Tests of Nanoplastic and Microplastic Particles. Environ. Sci. Technol. 2022, 56, 15192–15206. [Google Scholar] [CrossRef] [PubMed]
- Pikuda, O.; Xu, E.G.; Berk, D.; Tufenkji, N. Toxicity Assessments of Micro- and Nanoplastics Can Be Confounded by Preservatives in Commercial Formulations. Environ. Sci. Technol. Lett. 2019, 6, 21–25. [Google Scholar] [CrossRef]
- Reynaud, S.; Aynard, A.; Grassl, B.; Gigault, J. Nanoplastics: From Model Materials to Colloidal Fate. Curr. Opin. Colloid Interface Sci. 2022, 57, 101528. [Google Scholar] [CrossRef]
- Heinlaan, M.; Kasemets, K.; Aruoja, V.; Blinova, I.; Bondarenko, O.; Lukjanova, A.; Khosrovyan, A.; Kurvet, I.; Pullerits, M.; Sihtmäe, M.; et al. Hazard Evaluation of Polystyrene Nanoplastic with Nine Bioassays Did Not Show Particle-Specific Acute Toxicity. Sci. Total Environ. 2020, 707, 136073. [Google Scholar] [CrossRef]
- Kelpsiene, E.; Ekvall, M.T.; Lundqvist, M.; Torstensson, O.; Hua, J.; Cedervall, T. Review of Ecotoxicological Studies of Widely Used Polystyrene Nanoparticles. Environ. Sci. Process. Impacts 2022, 24, 8–16. [Google Scholar] [CrossRef]
- Rodríguez-Hernández, A.G.; Muñoz-Tabares, J.A.; Aguilar-Guzmán, J.C.; Vazquez-Duhalt, R. A Novel and Simple Method for Polyethylene Terephthalate (PET) Nanoparticle Production. Environ. Sci. Nano 2019, 6, 2031–2036. [Google Scholar] [CrossRef]
- Ji, Y.; Wang, C.; Wang, Y.; Fu, L.; Man, M.; Chen, L. Realistic Polyethylene Terephthalate Nanoplastics and the Size- and Surface Coating-Dependent Toxicological Impacts on Zebrafish Embryos. Environ. Sci. Nano 2020, 7, 2313–2324. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, S.; Duan, Z.; Wang, L. Pulmonary Toxicology Assessment of Polyethylene Terephthalate Nanoplastic Particles in Vitro. Environ. Int. 2022, 162, 107177. [Google Scholar] [CrossRef]
- Johnson, L.M.; Mecham, J.B.; Krovi, S.A.; Caffaro, M.M.M.; Aravamudhan, S.; Kovach, A.L.; Fennell, T.R.; Mortensen, N.P. Fabrication of Polyethylene Terephthalate (PET) Nanoparticles with Fluorescent Tracers for Studies in Mammalian Cells. Nanoscale Adv. 2021, 3, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Villacorta, A.; Rubio, L.; Alaraby, M.; López-Mesas, M.; Fuentes-Cebrian, V.; Moriones, O.H.; Marcos, R.; Hernández, A. A New Source of Representative Secondary PET Nanoplastics. Obtention, Characterization, and Hazard Evaluation. J. Hazard. Mater. 2022, 439, 129593. [Google Scholar] [CrossRef] [PubMed]
- Mahadevan, G.; Valiyaveettil, S. Understanding the Interactions of Poly(Methyl Methacrylate) and Poly(Vinyl Chloride) Nanoparticles with BHK-21 Cell Line. Sci. Rep. 2021, 11, 2089. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Guzmán, J.C.; Bejtka, K.; Fontana, M.; Valsami-Jones, E.; Villezcas, A.M.; Vazquez-Duhalt, R.; Rodríguez-Hernández, A.G. Polyethylene Terephthalate Nanoparticles Effect on RAW 264. 7 Macrophage Cells. Microplastics Nanoplastics 2022, 2, 9. [Google Scholar] [CrossRef]
- Bashirova, N.; Poppitz, D.; Klüver, N.; Scholz, S.; Matysik, J.; Alia, A. A Mechanistic Understanding of the Effects of Polyethylene Terephthalate Nanoplastics in the Zebrafish (Danio Rerio) Embryo. Sci. Rep. 2023, 13, 1891. [Google Scholar] [CrossRef]
- Magrì, D.; Veronesi, M.; Sánchez-Moreno, P.; Tolardo, V.; Bandiera, T.; Pompa, P.P.; Athanassiou, A.; Fragouli, D. PET Nanoplastics Interactions with Water Contaminants and Their Impact on Human Cells. Environ. Pollut. 2021, 271, 116262. [Google Scholar] [CrossRef]
- Chen, L.; Liu, J.; Zhang, Y.; Zhang, G.; Kang, Y.; Chen, A.; Feng, X.; Shao, L. The Toxicity of Silica Nanoparticles to the Immune System. Nanomed. 2018, 13, 1939–1962. [Google Scholar] [CrossRef]
- Forte, M.; Iachetta, G.; Tussellino, M.; Carotenuto, R.; Prisco, M.; De Falco, M.; Laforgia, V.; Valiante, S. Polystyrene Nanoparticles Internalization in Human Gastric Adenocarcinoma Cells. Toxicol. In Vitro 2016, 31, 126–136. [Google Scholar] [CrossRef]
- Annangi, B.; Villacorta, A.; Vela, L.; Tavakolpournegari, A.; Marcos, R.; Hernández, A. Effects of True-to-Life PET Nanoplastics Using Primary Human Nasal Epithelial Cells. Environ. Toxicol. Pharmacol. 2023, 100, 104140. [Google Scholar] [CrossRef]
- Sarma, D.K.; Dubey, R.; Samarth, R.M.; Shubham, S.; Chowdhury, P.; Kumawat, M.; Verma, V.; Tiwari, R.R.; Kumar, M. The Biological Effects of Polystyrene Nanoplastics on Human Peripheral Blood Lymphocytes. Nanomaterials 2022, 12, 1632. [Google Scholar] [CrossRef] [PubMed]
- Domenech, J.; de Britto, M.; Velázquez, A.; Pastor, S.; Hernández, A.; Marcos, R.; Cortés, C. Long-Term Effects of Polystyrene Nanoplastics in Human Intestinal Caco-2 Cells. Biomolecules 2021, 11, 1442. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Ma, Y.; Han, X.; Chen, Y. Systematic Toxicity Evaluation of Polystyrene Nanoplastics on Mice and Molecular Mechanism Investigation about Their Internalization into Caco-2 Cells. J. Hazard. Mater. 2021, 417, 126092. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.-Y.; Li, H.; Ren, H.; Zhang, X.; Huang, F.; Zhang, D.; Huang, Q.; Zhang, X. Size-Dependent Effects of Polystyrene Nanoplastics on Autophagy Response in Human Umbilical Vein Endothelial Cells. J. Hazard. Mater. 2022, 421, 126770. [Google Scholar] [CrossRef] [PubMed]
- Collin-Faure, V.; Vitipon, M.; Torres, A.; Tanyeres, O.; Dalzon, B.; Rabilloud, T. The Internal Dose Makes the Poison: Higher Internalization of Polystyrene Particles Induce Increased Perturbation of Macrophages. Front. Immunol. 2023, 14. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.; Liu, X.; Qu, M. Nanoplastics and Human Health: Hazard Identification and Biointerface. Nanomaterials 2022, 12, 1298. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.-H.; Choi, S.; Kim, D.; Park, H.J.; Bian, Y.; Choi, S.H.; Chung, H.Y.; Bae, O.-N. Amine-Modified Nanoplastics Promote the Procoagulant Activation of Isolated Human Red Blood Cells and Thrombus Formation in Rats. Part. Fibre Toxicol. 2022, 19, 60. [Google Scholar] [CrossRef] [PubMed]
- Barshtein, G.; Arbell, D.; Yedgar, S. Hemolytic Effect of Polymeric Nanoparticles: Role of Albumin. IEEE Trans. Nanobioscience 2011, 10, 259–261. [Google Scholar] [CrossRef]
- Babonaitė, M.; Čepulis, M.; Kazlauskaitė, J.; Lazutka, J.R. Evaluation of In Vitro Genotoxicity of Polystyrene Nanoparticles in Human Peripheral Blood Mononuclear Cells. Toxics 2023, 11, 627. [Google Scholar] [CrossRef]
- Shi, X.; Wang, X.; Huang, R.; Tang, C.; Hu, C.; Ning, P.; Wang, F. Cytotoxicity and Genotoxicity of Polystyrene Micro- and Nanoplastics with Different Size and Surface Modification in A549 Cells. Int. J. Nanomedicine 2022, 17, 4509. [Google Scholar] [CrossRef] [PubMed]
- Collin-Faure, V.; Dalzon, B.; Devcic, J.; Diemer, H.; Cianférani, S.; Rabilloud, T. Does Size Matter? A Proteomics-Informed Comparison of the Effects of Polystyrene Beads of Different Sizes on Macrophages. Environ. Sci. Nano 2022, 9, 2827–2840. [Google Scholar] [CrossRef]
| Samples | Dispersant | Z- Average (d.nm) |
Polydispersity Index (PdI) |
|---|---|---|---|
| NPs washed | MilliQ water | 311.7 ± 4.4 | 0.375 ± 0.026 |
| BSA (0.05%) | 243.5 ± 1.3 | 0.352 ± 0.037 | |
| SDS (0.1%) | 265.7 ± 5.9 | 0.351 ± 0.041 | |
| NPs unwashed | MilliQ water | 236.4 ± 1.2 | 0.325 ± 0.006 |
| NPs Washed (Dispersed in MilliQ Water) |
NPs Unwashed (Dispersed in MilliQ Water) |
NPs Washed (Dispersed in 0.5% SDS) |
|
|---|---|---|---|
| Zeta potential (mV) | -34.93 ± 1.18 | -42.10 ± 1.30 | -63.53 ± 1.47 |
| Mobility (µmcm/V) | -2.739 ± 0.094 | -3.301 ± 0.100 | -4.980 ± 0.114 |
| Conductivity (mS/cm) | 0.0205 ± 0.0001 | 0.0287 ± 0.0105 | 0.9710 ± 0.0301 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




