Submitted:
30 September 2023
Posted:
10 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
3. Discussion
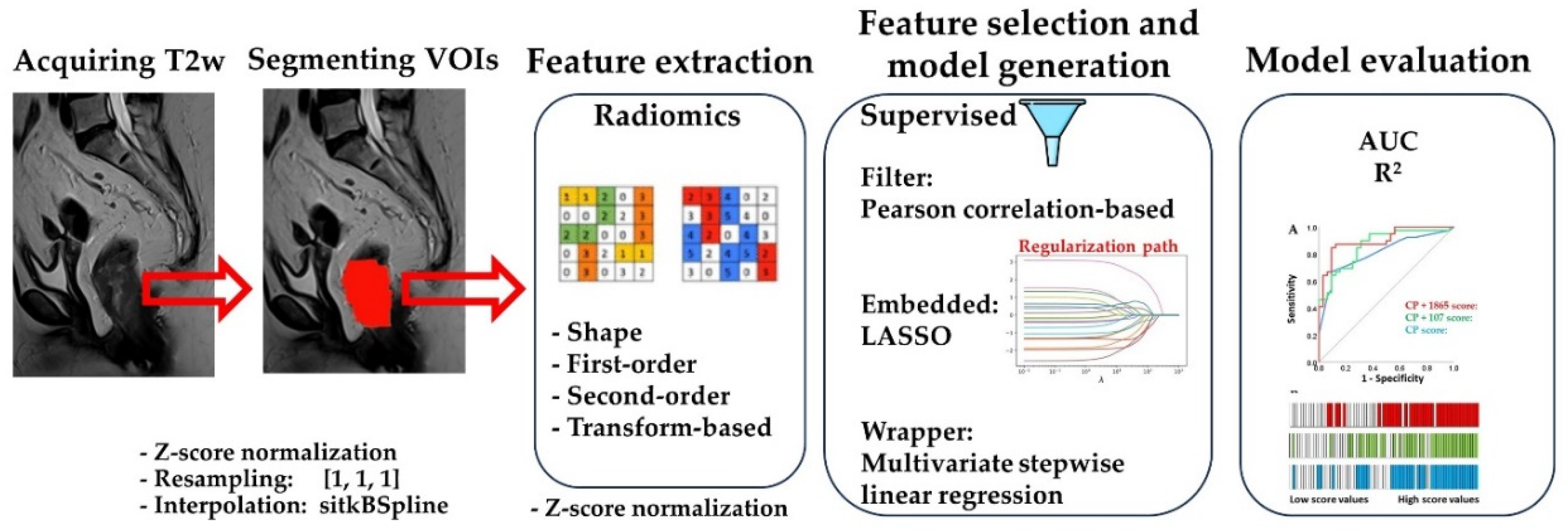
4. Materials and Methods
Ethics Approval Statement
Patients
MRI
Postprocessing
Sample size calculation
MRI normalization
Feature extraction

Normalization of the calculated feature values
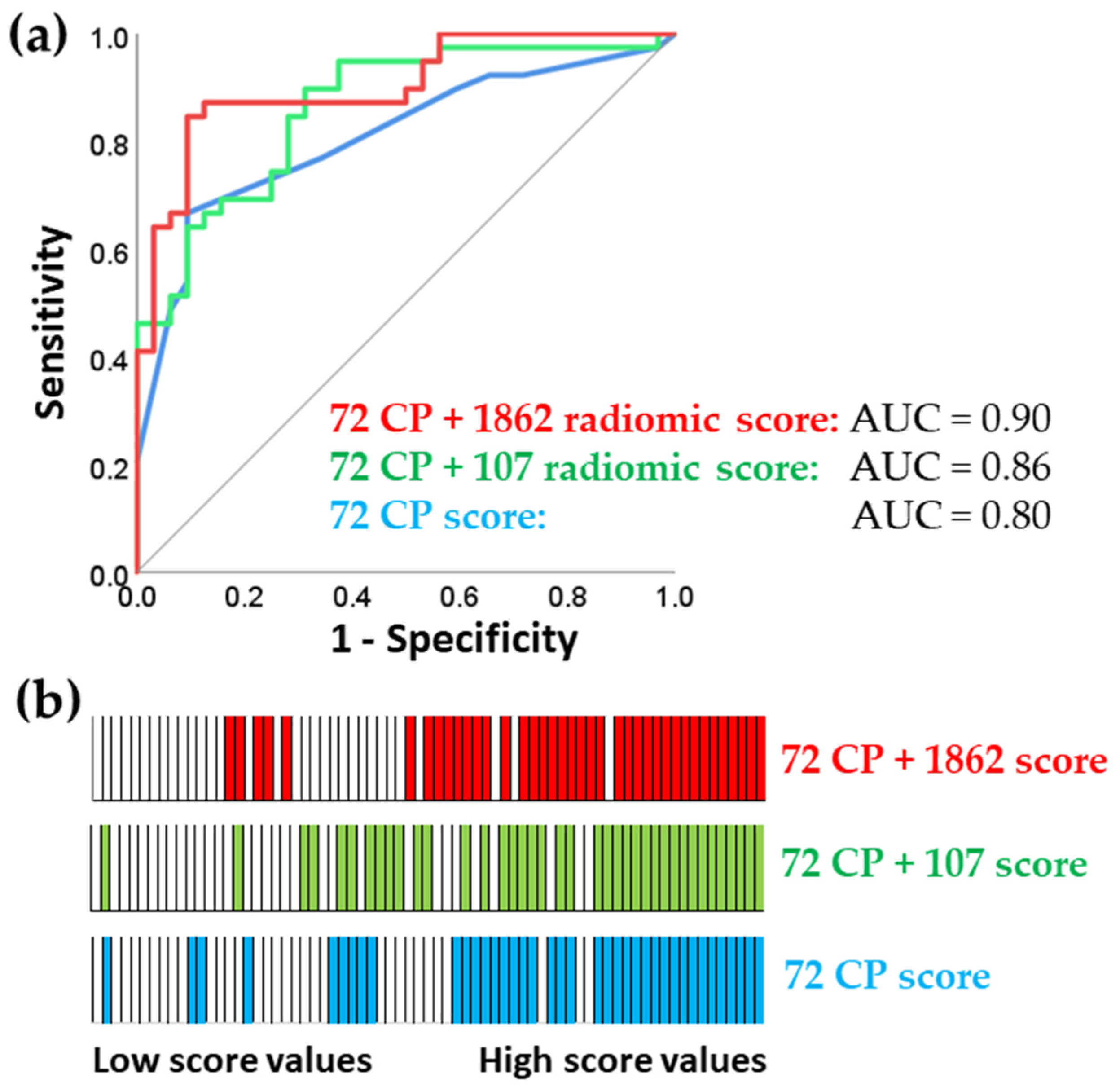
Evaluation of predictive performance
Model selection
Validation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
References
- Sung, H.; Ferlay, J.; Siegel, R. L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. , Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA. Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Saad El Din, K.; Loree, J. M.; Sayre, E. C.; Gill, S.; Brown, C. J.; Dau, H.; De Vera, M. A. , Trends in the epidemiology of young-onset colorectal cancer: a worldwide systematic review. BMC cancer 2020, 20, 288. [Google Scholar] [CrossRef]
- Nikolic, N.; Spasic, J.; Stanic, N.; Nikolic, V.; Radosavljevic, D. , Young-Onset Colorectal Cancer in Serbia: Tertiary Cancer Center Experience. J Adolesc Young Adult Oncol 2023, 12, 207–214. [Google Scholar] [CrossRef]
- Wolf, A. M. D.; Fontham, E. T. H.; Church, T. R.; Flowers, C. R.; Guerra, C. E.; LaMonte, S. J.; Etzioni, R.; McKenna, M. T.; Oeffinger, K. C.; Shih, Y. T.; Walter, L. C.; Andrews, K. S.; Brawley, O. W.; Brooks, D.; Fedewa, S. A.; Manassaram-Baptiste, D.; Siegel, R. L.; Wender, R. C.; Smith, R. A. , Colorectal cancer screening for average-risk adults: 2018 guideline update from the American Cancer Society. CA. Cancer J. Clin. 2018, 68, 250–281. [Google Scholar] [CrossRef]
- Hong, Y.; Kim, J.; Choi, Y. J.; Kang, J. G. , Clinical study of colorectal cancer operation: Survival analysis. Korean J Clin Oncol 2020, 16, 3–8. [Google Scholar] [CrossRef]
- Benson, A. B.; Venook, A. P.; Al-Hawary, M. M.; Azad, N.; Chen, Y. J.; Ciombor, K. K.; Cohen, S.; Cooper, H. S.; Deming, D.; Garrido-Laguna, I.; Grem, J. L.; Gunn, A.; Hecht, J. R.; Hoffe, S.; Hubbard, J.; Hunt, S.; Jeck, W.; Johung, K. L.; Kirilcuk, N.; Krishnamurthi, S.; Maratt, J. K.; Messersmith, W. A.; Meyerhardt, J.; Miller, E. D.; Mulcahy, M. F.; Nurkin, S.; Overman, M. J.; Parikh, A.; Patel, H.; Pedersen, K.; Saltz, L.; Schneider, C.; Shibata, D.; Skibber, J. M.; Sofocleous, C. T.; Stotsky-Himelfarb, E.; Tavakkoli, A.; Willett, C. G.; Gregory, K.; Gurski, L. , Rectal Cancer, Version 2.2022, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 2022, 20, 1139–1167. [Google Scholar] [CrossRef] [PubMed]
- Zorcolo, L.; Rosman, A. S.; Restivo, A.; Pisano, M.; Nigri, G. R.; Fancellu, A.; Melis, M. , Complete pathologic response after combined modality treatment for rectal cancer and long-term survival: a meta-analysis. Ann. Surg. Oncol. 2012, 19, 2822–32. [Google Scholar] [CrossRef]
- Maas, M.; Nelemans, P. J.; Valentini, V.; Das, P.; Rodel, C.; Kuo, L. J.; Calvo, F. A.; Garcia-Aguilar, J.; Glynne-Jones, R.; Haustermans, K.; Mohiuddin, M.; Pucciarelli, S.; Small, W., Jr.; Suarez, J.; Theodoropoulos, G.; Biondo, S.; Beets-Tan, R. G.; Beets, G. L. , Long-term outcome in patients with a pathological complete response after chemoradiation for rectal cancer: a pooled analysis of individual patient data. The lancet oncology 2010, 11, 835–44. [Google Scholar] [CrossRef]
- Vecchio, F. M.; Valentini, V.; Minsky, B. D.; Padula, G. D.; Venkatraman, E. S.; Balducci, M.; Micciche, F.; Ricci, R.; Morganti, A. G.; Gambacorta, M. A.; Maurizi, F.; Coco, C. , The relationship of pathologic tumor regression grade (TRG) and outcomes after preoperative therapy in rectal cancer. Int. J. Radiat. Oncol. Biol. Phys. 2005, 62, 752–60. [Google Scholar] [CrossRef]
- do Canto, L. M.; Barros-Filho, M. C.; Rainho, C. A.; Marinho, D.; Kupper, B. E. C.; Begnami, M.; Scapulatempo-Neto, C.; Havelund, B. M.; Lindebjerg, J.; Marchi, F. A.; Baumbach, J.; Aguiar, S., Jr.; Rogatto, S. R. , Comprehensive Analysis of DNA Methylation and Prediction of Response to NeoadjuvantTherapy in Locally Advanced Rectal Cancer. Cancers 2020, 12. [Google Scholar] [CrossRef]
- Timudom, K.; Akaraviputh, T.; Chinswangwatanakul, V.; Pongpaibul, A.; Korpraphong, P.; Petsuksiri, J.; Ithimakin, S.; Trakarnsanga, A. , Predictive significance of cancer related-inflammatory markers in locally advanced rectal cancer. World J Gastrointest Surg 2020, 12, 390–396. [Google Scholar] [CrossRef]
- Stanojevic, A.; Samiotaki, M.; Lygirou, V.; Marinkovic, M.; Nikolic, V.; Stojanovic-Rundic, S.; Jankovic, R.; Vlahou, A.; Panayotou, G.; Fijneman, R. J. A.; Castellvi-Bel, S.; Zoidakis, J.; Cavic, M. , Data independent acquisition mass spectrometry (DIA-MS) analysis of FFPE rectal cancer samples offers in depth proteomics characterization of response to neoadjuvant chemoradiotherapy. medRxiv 2023, 2023.05.12.23289671. [Google Scholar]
- Stanojevic, A.; Spasic, J.; Marinkovic, M.; Stojanovic-Rundic, S.; Jankovic, R.; Djuric, A.; Zoidakis, J.; Fijneman, R.; Castellvi-Bel, S.; Cavic, M. , Methylenetetrahydrofolate reductase polymorphic variants in rectal cancer: significance for cancer risk and response to chemoradiotherapy. medRxiv 2023, 2023.09.21.23295916. [Google Scholar]
- Marinkovic, M.; Stojanovic-Rundic, S.; Stanojevic, A.; Ostojic, M.; Gavrilovic, D.; Jankovic, R.; Maksimovic, N.; Stroggilos, R.; Zoidakis, J.; Castellvi-Bel, S.; Fijneman, R. J. A.; Cavic, M. , Exploring novel genetic and hematological predictors of response to neoadjuvant chemoradiotherapy in locally advanced rectal cancer. Front Genet 2023, 14, 1245594. [Google Scholar] [CrossRef]
- Janssen, M. H.; Aerts, H. J.; Buijsen, J.; Lambin, P.; Lammering, G.; Ollers, M. C. , Repeated positron emission tomography-computed tomography and perfusion-computed tomography imaging in rectal cancer: fluorodeoxyglucose uptake corresponds with tumor perfusion. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 849–55. [Google Scholar] [CrossRef]
- van den Bogaard, J.; Janssen, M. H.; Janssens, G.; Buijsen, J.; Reniers, B.; Lambin, P.; Lammering, G.; Ollers, M. C. , Residual metabolic tumor activity after chemo-radiotherapy is mainly located in initially high FDG uptake areas in rectal cancer. Radiother. Oncol. 2011, 99, 137–41. [Google Scholar] [CrossRef]
- Kalisz, K. R.; Enzerra, M. D.; Paspulati, R. M. , MRI Evaluation of the Response of Rectal Cancer to Neoadjuvant Chemoradiation Therapy. Radiographics 2019, 39, 538–556. [Google Scholar] [CrossRef]
- Pang, X.; Wang, F.; Zhang, Q.; Li, Y.; Huang, R.; Yin, X.; Fan, X. , A Pipeline for Predicting the Treatment Response of Neoadjuvant Chemoradiotherapy for Locally Advanced Rectal Cancer Using Single MRI Modality: Combining Deep Segmentation Network and Radiomics Analysis Based on "Suspicious Region". Frontiers in oncology 2021, 11, 711747. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, X. Y.; Shi, Y. J.; Wang, L.; Zhu, H. T.; Tang, Z.; Wang, S.; Li, X. T.; Tian, J.; Sun, Y. S. , Radiomics Analysis for Evaluation of Pathological Complete Response to Neoadjuvant Chemoradiotherapy in Locally Advanced Rectal Cancer. Clin. Cancer Res. 2017, 23, 7253–7262. [Google Scholar] [CrossRef]
- Petkovska, I.; Tixier, F.; Ortiz, E. J.; Golia Pernicka, J. S.; Paroder, V.; Bates, D. D.; Horvat, N.; Fuqua, J.; Schilsky, J.; Gollub, M. J.; Garcia-Aguilar, J.; Veeraraghavan, H. , Clinical utility of radiomics at baseline rectal MRI to predict complete response of rectal cancer after chemoradiation therapy. Abdom Radiol (NY) 2020, 45, 3608–3617. [Google Scholar] [CrossRef]
- Jiang, H.; Guo, W.; Yu, Z.; Lin, X.; Zhang, M.; Jiang, H.; Zhang, H.; Sun, Z.; Li, J.; Yu, Y.; Zhao, S.; Hu, H. , A Comprehensive Prediction Model Based on MRI Radiomics and Clinical Factors to Predict Tumor Response After Neoadjuvant Chemoradiotherapy in Rectal Cancer. Acad. Radiol. 2023, 30 Suppl 1, S185–S198. [Google Scholar] [CrossRef]
- Shin, J.; Seo, N.; Baek, S. E.; Son, N. H.; Lim, J. S.; Kim, N. K.; Koom, W. S.; Kim, S. , MRI Radiomics Model Predicts Pathologic Complete Response of Rectal Cancer Following Chemoradiotherapy. Radiology 2022, 303, 351–358. [Google Scholar] [CrossRef]
- Dinapoli, N.; Barbaro, B.; Gatta, R.; Chiloiro, G.; Casa, C.; Masciocchi, C.; Damiani, A.; Boldrini, L.; Gambacorta, M. A.; Dezio, M.; Mattiucci, G. C.; Balducci, M.; van Soest, J.; Dekker, A.; Lambin, P.; Fiorino, C.; Sini, C.; De Cobelli, F.; Di Muzio, N.; Gumina, C.; Passoni, P.; Manfredi, R.; Valentini, V. , Magnetic Resonance, Vendor-independent, Intensity Histogram Analysis Predicting Pathologic Complete Response After Radiochemotherapy of Rectal Cancer. Int J Radiat Oncol Biol Phys 2018, 102, 765–774. [Google Scholar] [CrossRef]
- De Cecco, C. N.; Ganeshan, B.; Ciolina, M.; Rengo, M.; Meinel, F. G.; Musio, D.; De Felice, F.; Raffetto, N.; Tombolini, V.; Laghi, A. , Texture analysis as imaging biomarker of tumoral response to neoadjuvant chemoradiotherapy in rectal cancer patients studied with 3-T magnetic resonance. Invest. Radiol. 2015, 50, 239–45. [Google Scholar] [CrossRef]
- Volpe, S.; Isaksson, L. J.; Zaffaroni, M.; Pepa, M.; Raimondi, S.; Botta, F.; Lo Presti, G.; Vincini, M. G.; Rampinelli, C.; Cremonesi, M.; de Marinis, F.; Spaggiari, L.; Gandini, S.; Guckenberger, M.; Orecchia, R.; Jereczek-Fossa, B. A. , Impact of image filtering and assessment of volume-confounding effects on CT radiomic features and derived survival models in non-small cell lung cancer. Transl Lung Cancer Res 2022, 11, 2452–2463. [Google Scholar] [CrossRef]
- Simha, V.; Kapoor, R.; Gupta, R.; Bahl, A.; Nada, R. , Mucinous adenocarcinoma of the rectum: a poor candidate for neo-adjuvant chemoradiation? J Gastrointest Oncol 2014, 5, 276–9. [Google Scholar]
- Chand, M.; Bhangu, A.; Wotherspoon, A.; Stamp, G. W. H.; Swift, R. I.; Chau, I.; Tekkis, P. P.; Brown, G. , EMVI-positive stage II rectal cancer has similar clinical outcomes as stage III disease following pre-operative chemoradiotherapy. Ann. Oncol. 2014, 25, 858–863. [Google Scholar] [CrossRef]
- Kensen, C. M.; Betgen, A.; Wiersema, L.; Peters, F. P.; Kayembe, M. T.; Marijnen, C. A. M.; van der Heide, U. A.; Janssen, T. M. , Online Adaptive MRI-Guided Radiotherapy for Primary Tumor and Lymph Node Boosting in Rectal Cancer. Cancers 2023, 15. [Google Scholar] [CrossRef]
- Wu, C.; Qiu, Y.; Zhang, R.; Li, X.; Liang, H.; Wang, M.; Li, F.; Zhu, M.; Ye, G.; Liu, H.; Li, G.; Zhao, L. , Association of peripheral basophils with tumor M2 macrophage infiltration and outcomes of the anti-PD-1 inhibitor plus chemotherapy combination in advanced gastric cancer. Journal of translational medicine 2022, 20, 386. [Google Scholar] [CrossRef]
- Miranda, J.; Tan, G. X. V.; Fernandes, M. C.; Yildirim, O.; Sims, J. A.; Araujo-Filho, J. A. B.; de, M. M. F. A.; Assuncao-Jr, A. N.; Nomura, C. H.; Horvat, N. , Rectal MRI radiomics for predicting pathological complete response: Where we are. Clin. Imaging 2022, 82, 141–149. [Google Scholar] [CrossRef]
- Demircioglu, A. , The effect of preprocessing filters on predictive performance in radiomics. Eur Radiol Exp 2022, 6, 40. [Google Scholar] [CrossRef]
- Zhou, X.; Yi, Y.; Liu, Z.; Cao, W.; Lai, B.; Sun, K.; Li, L.; Zhou, Z.; Feng, Y.; Tian, J. , Radiomics-Based Pretherapeutic Prediction of Non-response to Neoadjuvant Therapy in Locally Advanced Rectal Cancer. Ann Surg Oncol 2019, 26, 1676–1684. [Google Scholar] [CrossRef]
- Gillies, R. J.; Kinahan, P. E.; Hricak, H. , Radiomics: Images Are More than Pictures, They Are Data. Radiology 2016, 278, 563–77. [Google Scholar] [CrossRef]
- Jiang, Z.; Dong, Y.; Yang, L.; Lv, Y.; Dong, S.; Yuan, S.; Li, D.; Liu, L. , CT-Based Hand-crafted Radiomic Signatures Can Predict PD-L1 Expression Levels in Non-small Cell Lung Cancer: a Two-Center Study. J. Digit. Imaging 2021, 34, 1073–1085. [Google Scholar] [CrossRef]
- Jeon, S. H.; Song, C.; Chie, E. K.; Kim, B.; Kim, Y. H.; Chang, W.; Lee, Y. J.; Chung, J. H.; Chung, J. B.; Lee, K. W.; Kang, S. B.; Kim, J. S. , Combining Radiomics and Blood Test Biomarkers to Predict the Response of Locally Advanced Rectal Cancer to Chemoradiation. In Vivo 2020, 34, 2955–2965. [Google Scholar] [CrossRef] [PubMed]
- Capirci, C.; Valentini, V.; Cionini, L.; De Paoli, A.; Rodel, C.; Glynne-Jones, R.; Coco, C.; Romano, M.; Mantello, G.; Palazzi, S.; Mattia, F. O.; Friso, M. L.; Genovesi, D.; Vidali, C.; Gambacorta, M. A.; Buffoli, A.; Lupattelli, M.; Favretto, M. S.; La Torre, G. , Prognostic value of pathologic complete response after neoadjuvant therapy in locally advanced rectal cancer: long-term analysis of 566 ypCR patients. Int. J. Radiat. Oncol. Biol. Phys. 2008, 72, 99–107. [Google Scholar] [CrossRef]
- Marzi, C.; Marfisi, D.; Barucci, A.; Del Meglio, J.; Lilli, A.; Vignali, C.; Mascalchi, M.; Casolo, G.; Diciotti, S.; Traino, A. C.; Tessa, C.; Giannelli, M. , Collinearity and Dimensionality Reduction in Radiomics: Effect of Preprocessing Parameters in Hypertrophic Cardiomyopathy Magnetic Resonance T1 and T2 Mapping. Bioengineering (Basel) 2023, 10. [Google Scholar] [CrossRef]
- Fedorov, A.; Beichel, R.; Kalpathy-Cramer, J.; Finet, J.; Fillion-Robin, J. C.; Pujol, S.; Bauer, C.; Jennings, D.; Fennessy, F.; Sonka, M.; Buatti, J.; Aylward, S.; Miller, J. V.; Pieper, S.; Kikinis, R. , 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magn Reson Imaging 2012, 30, 1323–41. [Google Scholar] [CrossRef] [PubMed]
- van Griethuysen, J. J. M.; Fedorov, A.; Parmar, C.; Hosny, A.; Aucoin, N.; Narayan, V.; Beets-Tan, R. G. H.; Fillion-Robin, J. C.; Pieper, S.; Aerts, H. , Computational Radiomics System to Decode the Radiographic Phenotype. Cancer Res. 2017, 77, e104–e107. [Google Scholar] [CrossRef]
- Efron, B. , Bootstrap Methods: Another Look at the Jackknife. The Annals of Statistics 1979, 7, 1–26. [Google Scholar] [CrossRef]
| Characteristics | N (%) |
|---|---|
| Age (years) | |
| Mean (SD) (Range) | 60.8 (10.6) (33.0-81.0) |
| Gender | |
| Female / Male | 25 (33.3%) / 50 (66.7%) |
| T in clinical TNM | |
| T2 | 2 (2.7%) |
| T3 | 64 (85.3%) |
| T4 | 9 (12.0%) |
| N in clinical TNM | |
| N0 | 1 (1.3%) |
| N1a | 3 (4.0%) |
| N1b | 18 (24.0%) |
| N1c | 1 (1.3%) |
| N2a | 22 (29.3%) |
| N2b | 30 (40.0%) |
| Tumor differentiation | |
| well / moderate / poor | 39 (52.0%)/30 (40.0%)/6 (8.0%) |
| Mucinous histological type | |
| Yes / No | 13 (17.3) / 62 (85.7) |
| Absolute basophil count (109/L) | |
| Mean (SD) (Range) | 0.1 (0.1) (0.0-1.0) |
| Tumor length (mm) | |
| Mean (SD) (Range) | 63.2 (18.6) (24-150) |
| CRM status | |
| Uninvolved / Involved | 36 (48.0%) / 39 (52.0%) |
| Type of CRM involvement | |
| By direct tumor spread | 20 (26.7%) |
| By mesorectal TD or metastatic LN | 15 (20.0%) |
| Both categories | 4 (5.3%) |
| Uninvolved | 36 (48.0%) |
| Extramural vascular invasion (EMVI) | |
| Yes / No | 25 (33.3%) / 50 (66.7%) |
| Operative treatment | |
| No (cCR) / Yes | 12 (16.0%) / 63 (84.0%) |
| TRG score (operated patients) | |
| TRG1 | 13/63 (20.6%) |
| TRG2 | 10/63 (15.9%) |
| TRG3 | 30/63 (47.6%) |
| TRG4 | 10/63 (15.9%) |
| Response to the treatment | |
| R (cCR+TRG1+TRG2) | 35/75 (46.7%) |
| NonR (TRG3+TRG4) | 40/75 (53.3%) |
| Feature | Pearson b | P | R2 | P |
|---|---|---|---|---|
| Clinicopathological features | ||||
| Mucinous histological type | 0.396 | <0.001 | 0.157 | <0.001 |
| N stage | 0.302 | <0.001 | 0.091 | 0.010 |
| Extramural vascular invasion | 0.293 | <0.001 | 0.086 | 0.013 |
| Type of CRM involvement | 0.278 | <0.001 | 0.078 | 0.020 |
| Initial basophil count | 0.275 | <0.001 | 0.076 | 0.020 |
| Features calculated in original images | ||||
| Original_shape_Max2DDiameterSlice | 0.387 | <0.001 | 0.150 | <0.001 |
| Original_GLSZM_SizeZoneNonUniformity | 0.382 | <0.001 | 0.146 | <0.001 |
| Original_GLSZM_SmallHighGrayLevelEmphasis | 0.373 | <0.001 | 0.140 | <0.001 |
| Original_GLDM_SmallDepHighGrayLevEmphasis | 0.367 | <0.001 | 0.135 | <0.001 |
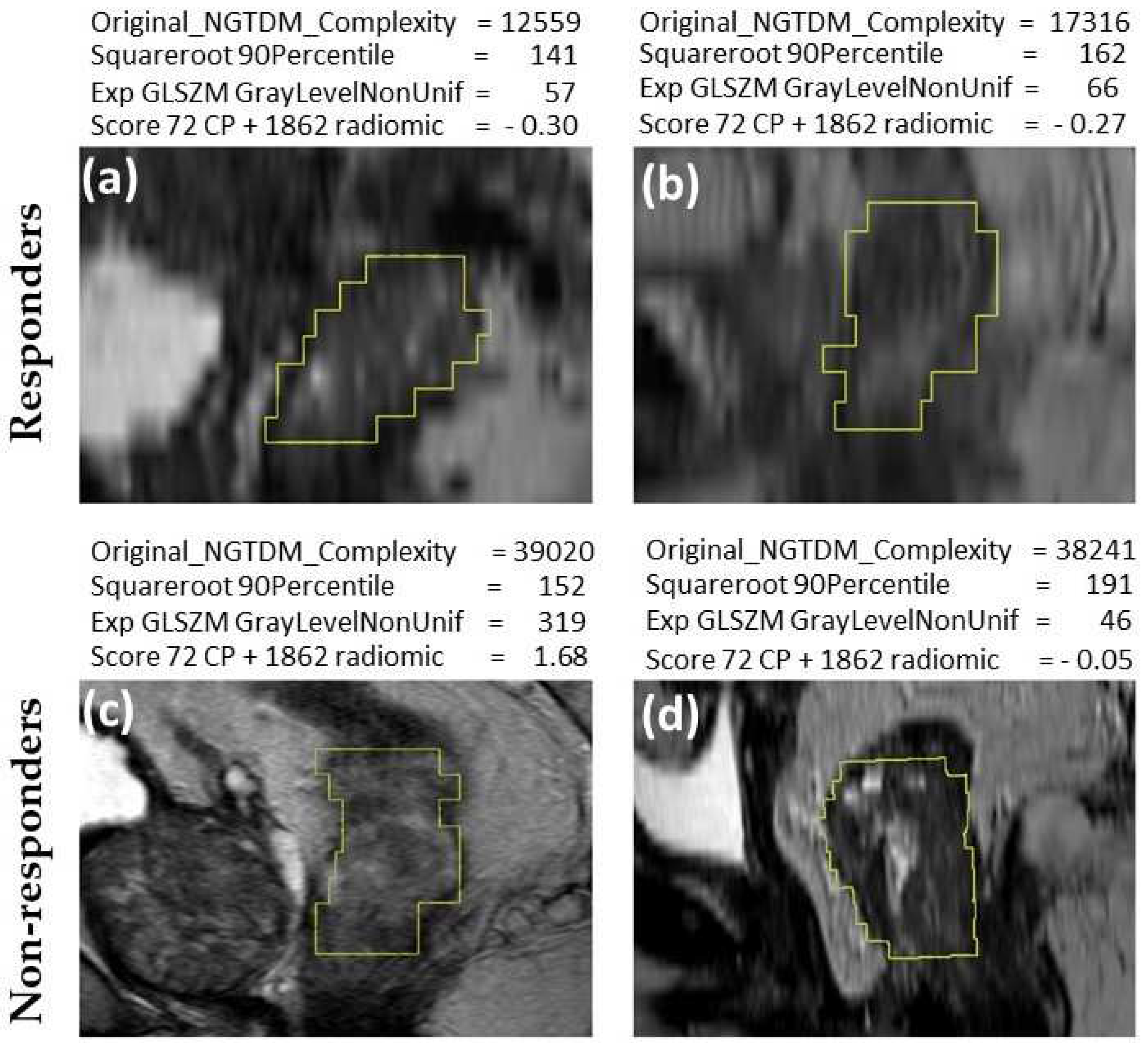
| Original_NGTDM_Complexity | 0.356 | <0.001 | 0.127 | <0.001 |
| Features calculated in filtered images | ||||
| Sqroot_GLDM_SmallDepHighGrayLevEmphasis | 0.401 | <0.001 | 0.161 | <0.001 |
| Squareroot_GLSZM_SmallHighGrayLevelEmphasis | 0.398 | <0.001 | 0.158 | <0.001 |
| Exponential_GLSZM_GrayLevelNonUniformity | 0.377 | <0.001 | 0.143 | <0.001 |
| Logarithm_firstorder_90Percentile | 0.375 | <0.001 | 0.141 | <0.001 |
| Squareroot_firstorder_90Percentile | 0.372 | <0.001 | 0.139 | <0.001 |
|
a Univariate linear regression test b Pearson correlation coefficient Abbreviations: CRM, circumferential resection margin |
||||
| Feature | coefficient c 95%CI |
P-value |
|---|---|---|
| Model: from 72 CP features R2 = 0.30 b | ||
| Mucinous histological type | Coefficient = 0.478 0.277 - 0.697 |
P=0.001 |
| Extramural vascular invasion | Coefficient = 0.432 0.174 - 0.704 |
P=0.001 |
| Type of CRM involvement | Coefficient = 0.322 0.058 - 0.600 |
P=0.027 |
| Model: from 72 CP + 107 radiomic features R2 = 0.45 | ||
| Mucinous histological type | Coefficient = 0.396 0.163 - 0.603 |
P=0.001 |
| Initial basophil count | Coefficient = 0.395 0.249 - 1.301 |
P=0.025 |
| N stage | Coefficient = 0.393 0.110 - 0.653 |
P=0.010 |
| Original_NGTDM_Complexity | Coefficient = 0.385 0.111 - 0.581 |
P=0.002 |
| Original_firstorder_90Percentile | Coefficient = 0.218 -0.006 - 0.482 |
P=0.049 |
| Model: from 72 CP + 1862 radiomics features R2 = 0.47 | ||
| Mucinous histological type | Coefficient = 0.258 0.068- 0.431 |
P=0.009 |
| Initial basophil count | Coefficient = 0.384 0.270 - 1.207 |
P=0.0014 |
| Type of CRM involvement | Coefficient = 0.319 0.071 - 0.542 |
P=0.008 |
| Squareroot_firstorder_90Percentile | Coefficient = 0.483 0.262 - 0.659 |
P=0.001 |
| Exponential_GLSZM_GrayLevelNonUniformity | Coefficient = 0.377 0.178 - 0.528 |
P=0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




