Submitted:
20 September 2023
Posted:
22 September 2023
You are already at the latest version
Abstract
Keywords:
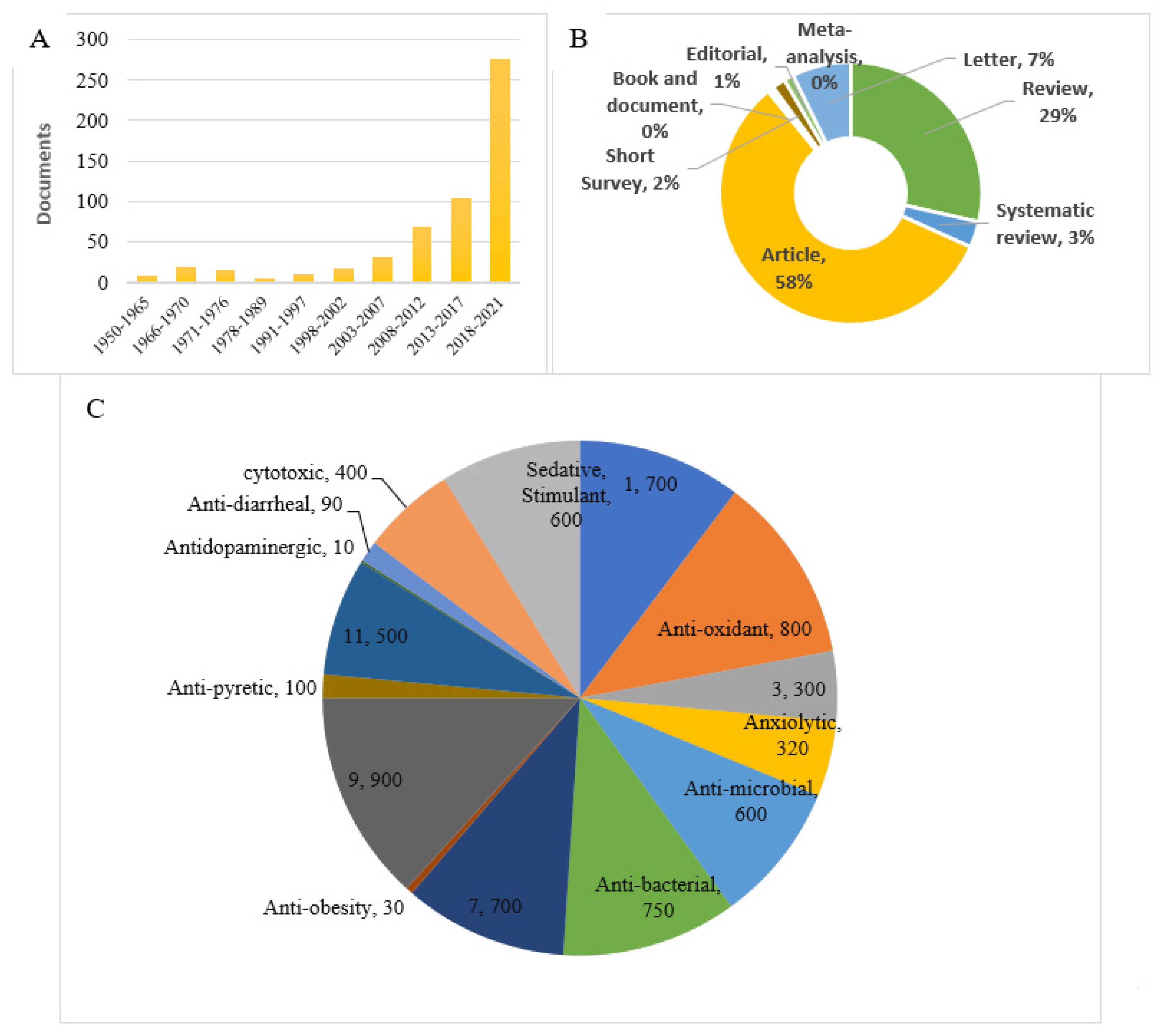
1. Introduction
2. Methodology
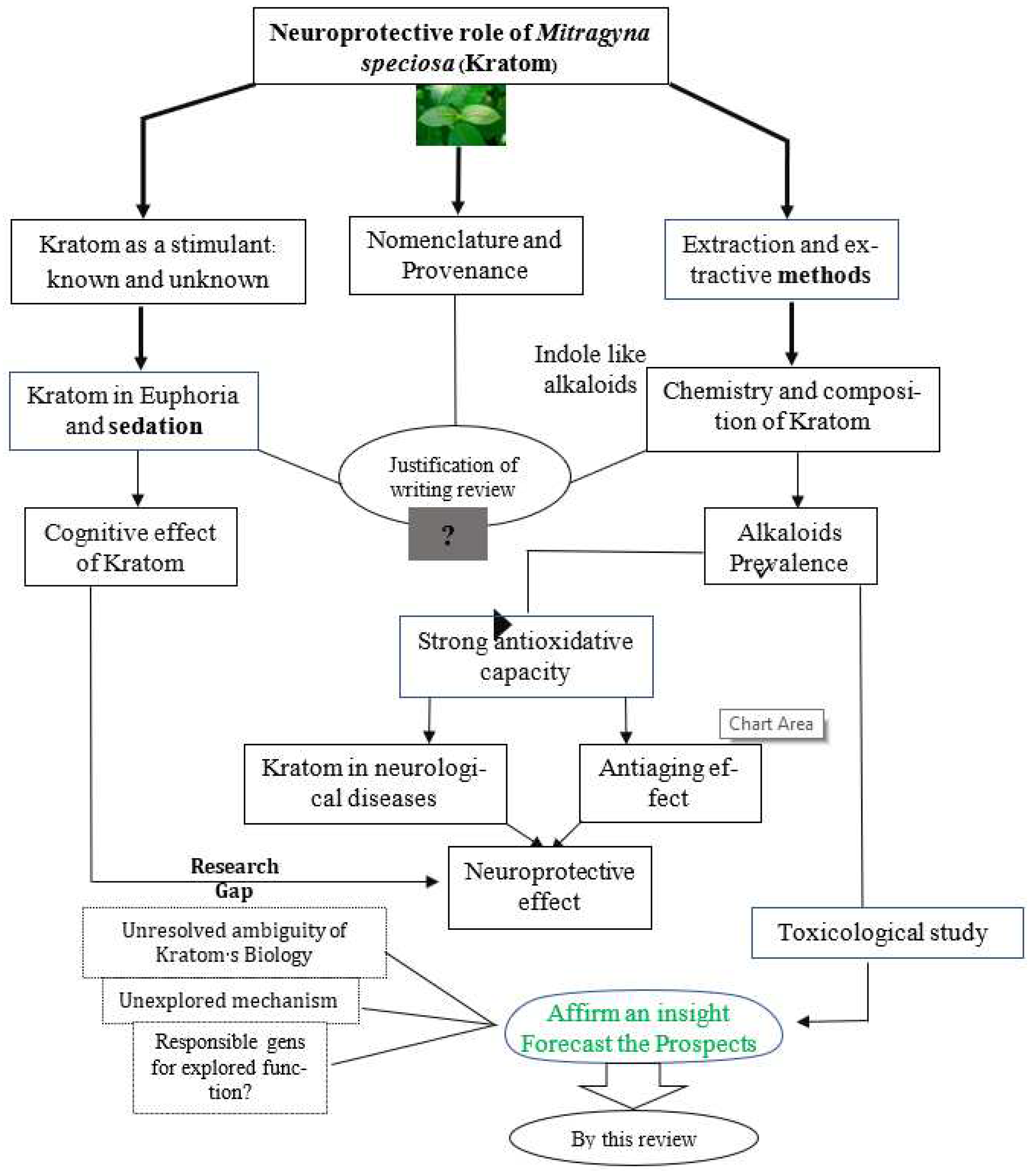
3. Focus of this review
4. Nomenclature and Provenance
5. Extraction and extractive of kratom
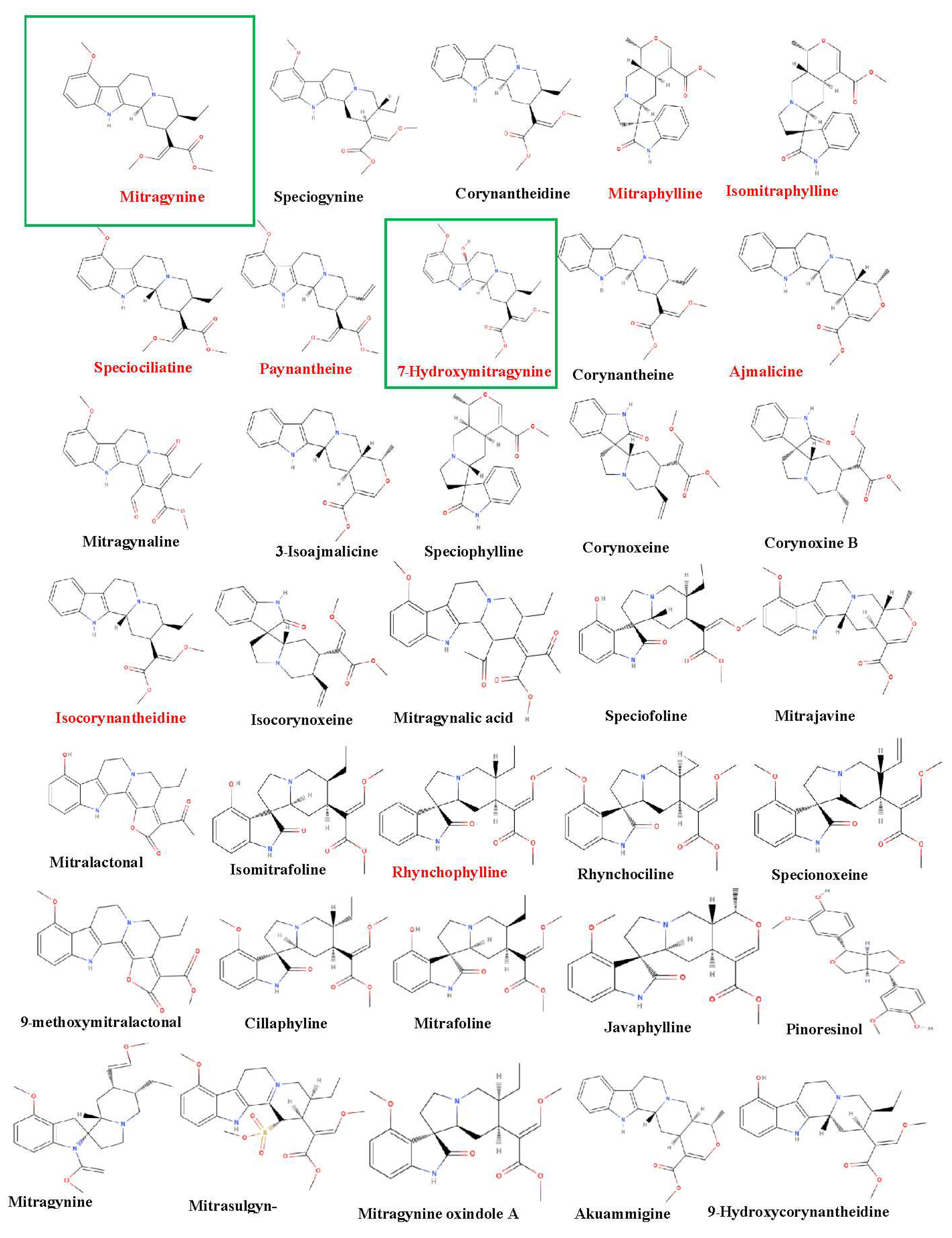
6. Chemistry and Composition-linked actions of kratom
Nature of alkaloids from kratom
7. Toxicology and toxicokinetics of kratom
8. Neurological effects of kratom
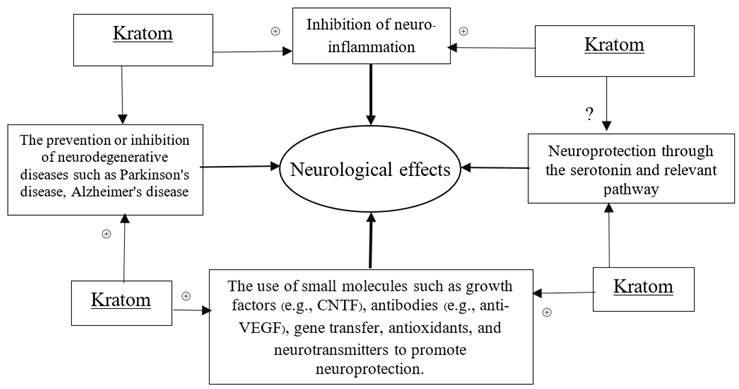
8.1. Kratom, an indole-like alkaloid for neurological effects
8.2. Anti-inflammatory effects leading to neuroprotective effects
8.3. Analgesic and Anti-nociceptive Effects
8.4. Neurological effects by gene regulation
8.5. Antioxidative effects
9. Other pharmacological effects
9.1. Antibacterial effects
9.2. Gastrointestinal effects
10. Adverse effects/ Abuse of kratom
11. Critical Remarks and insights
12. Conclusions and Future prospective
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data availability
Abbreviations
| AChE | Acetylcholinesterase |
| AD | Alzheimer’s disease |
| ALT | Alanine aminotransferase |
| ASE | Accelerated solvent extraction |
| AST | Aspartate aminotransferase |
| BBB | Blood-brain barrier |
| CAT | Catalase |
| COX-2 | Cyclooxygenase-2 |
| CUPRAC | Cupric ion reducing antioxidant capacity |
| CYPs | Cytochromes P50 |
| ED50 | Median effective dose |
| ESI | Electrospray ionization |
| FRAP | Ferric reducing ability of plasma |
| fEPSP | field Excitatory postsynaptic potentials |
| HSP | Heat shock proteins |
| HSF | Heat shock Factors |
| HT2A | Hydroxy-Tryptamine receptor |
| IC50 | Half maximal inhibitory concentration |
| I. P | Intraperitoneal |
| Keap1 | Kelch-like ECH-Associating protein 1 |
| Kg | Kilogram |
| LD50 | Median lethal dose |
| LPS | Lipopolysaccharides |
| LTP | Long-term potentiation |
| MAO | Monoamine oxidase |
| Mg | Milligram |
| µg | Microgram |
| MIC | Minimum inhibitory concentration |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| Nm | Nanometer |
| PD | Parkinson’s disease |
| P. O | Per Oral |
| QTOF-MS | Quadrupole time-of-flight mass spectrometry |
| SOD | Superoxide dismutase |
| TPC | Total phenolic content |
| TFC | Total flavonoid content |
| UDP | Uridine diphosphate |
| UGT | UDP-glucuronosyl transferase |
| UHPLC | Ultra high-performance liquid chromatography |
| w/w | Weight for weight |
References
- Butler, M.S. The role of natural product chemistry in drug discovery. Journal of natural products. 2004, 67, 2141–2153. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. Journal of natural products. 2012, 75, 311–335. [Google Scholar] [CrossRef]
- Koehn, F.E. and Carter, G.T. The evolving role of natural products in drug discovery. Nature reviews Drug discovery. 2005, 4, 206–220. [Google Scholar] [CrossRef]
- Patwardhan, B. and Mashelkar, R.A. Traditional medicine-inspired approaches to drug discovery: can Ayurveda show the way forward? Drug discovery today. 2009, 14, 804–811. [Google Scholar] [CrossRef]
- Arndt, T.; Claussen, U.; Güssregen, B.; Schröfel, S.; Stürzer, B.; Werle, A.; Wolf, G. Kratom alkaloids and O-desmethyltramadol in urine of a “Krypton” herbal mixture consumer. Forensic science international. 2011, 208, 47–52. [Google Scholar] [CrossRef]
- Michael White, C. Pharmacologic and clinical assessment of kratom. The Bulletin of the American Society of Hospital Pharmacists. 2018, 75, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Gong, F.; Gu, H.-P.; Xu, Q.-T.; Kang, W.-Y. Genus Mitragyna: Ethnomedicinal uses and pharmacological studies. Phytopharmacology. 2012, 3, 263–272. [Google Scholar]
- Rech, M.A.; Donahey, E.; Dziedzic, J.M.C.; Oh, L.; Greenhalgh, E. New drugs of abuse. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy. 2015, 35, 189–197. [Google Scholar] [CrossRef]
- Vicknasingam, B.; Narayanan, S.; Beng, G.T.; Mansor, S.M. The informal use of ketum (Mitragyna speciosa) for opioid withdrawal in the northern states of peninsular Malaysia and implications for drug substitution therapy. International Journal of Drug Policy. 2010, 21, 283–288. [Google Scholar] [CrossRef]
- Hassan, Z.; Muzaimi, M.; Navaratnam, V.; Yusoff, N.H.; Suhaimi, F.W.; Vadivelu, R.; Vicknasingam, B.K.; Amato, D.; von Hörsten, S.; Ismail, N.I. From Kratom to mitragynine and its derivatives: physiological and behavioural effects related to use, abuse, and addiction. Neuroscience & Biobehavioral Reviews. 2013, 37, 138–151. [Google Scholar] [CrossRef]
- Eisenman, S.W. The botany of Mitragyna speciosa (Korth.) Havil. and related species. Kratom and other Mitragynines: The chemistry and pharmacology of opioids from a non-opium source. 2014, 57, 57–76. [Google Scholar]
- Warner, M.L.; Kaufman, N.C.; Grundmann, O. The pharmacology and toxicology of kratom: from traditional herb to drug of abuse. International journal of legal medicine. 2016, 130, 127–138. [Google Scholar] [CrossRef]
- Adkins, J.E.; Boyer, E.W.; McCurdy, C.R. Mitragyna speciosa, a psychoactive tree from Southeast Asia with opioid activity. Current topics in medicinal chemistry. 2011, 11, 1165–1175. [Google Scholar] [CrossRef] [PubMed]
- Jaipaew, J.; Padungchareon, T.; Sukrong, S. PCR-reverse dot blot of the nucleotide signature sequences of matK for the identification of Mitragyna speciosa, a narcotic species. Plant Gene. 2018, 14, 46–54. [Google Scholar] [CrossRef]
- Tungphatthong, C.; Urumarudappa, S.K.J.; Awachai, S.; Sooksawate, T.; Sukrong, S. Differentiation of Mitragyna speciosa, a narcotic plant, from allied Mitragyna species using DNA barcoding-high-resolution melting (Bar-HRM) analysis. Scientific reports. 2021, 11, 1–9. [Google Scholar] [CrossRef]
- Parthasarathy, S.; Azizi, J.B.; Ramanathan, S.; Ismail, S.; Sasidharan, S.; Said, M.I.; Mansor, S.M. Evaluation of antioxidant and antibacterial activities of aqueous, methanolic and alkaloid extracts from Mitragyna speciosa (Rubiaceae family) leaves. Molecules. 2009, 14, 3964–3974. [Google Scholar] [CrossRef]
- Meireles, V.; Rosado, T.; Barroso, M.; Soares, S.; Gonçalves, J.; Luís, Â.; Caramelo, D.; Simão, A.Y.; Fernández, N.; Duarte, A.P. Mitragyna speciosa: clinical, toxicological aspects and analysis in biological and non-biological samples. Medicines. 2019, 6, 35. [Google Scholar] [CrossRef]
- Yuniarti, R.; Nadia, S.; Alamanda, A.; Zubir, M.; Syahputra, R.; Nizam, M. Characterization, phytochemical screenings and antioxidant activity test of kratom leaf ethanol extract (Mitragyna speciosa Korth) using DPPH method. Journal of Physics: Conference Series, IOP Publishing 2020. [Google Scholar] [CrossRef]
- Srichana, K.; Janchawee, B.; Prutipanlai, S.; Raungrut, P.; Keawpradub, N. Effects of mitragynine and a crude alkaloid extract derived from Mitragyna speciosa Korth. on permethrin elimination in rats. Pharmaceutics. 2015, 7, 10–26. [Google Scholar] [CrossRef]
- Goh, Y.S.; Karunakaran, T.; Murugaiyah, V.; Santhanam, R.; Bakar, M.H.A.; Ramanathan, S. Accelerated solvent extractions (ASE) of Mitragyna speciosa Korth.(Kratom) leaves: Evaluation of its cytotoxicity and antinociceptive activity. Molecules. 2021, 26, 3704. [Google Scholar] [CrossRef]
- Mossadeq, W.S.; Sulaiman, M.; Mohamad, T.T.; Chiong, H.; Zakaria, Z.; Jabit, M.; Baharuldin, M.; Israf, D. Anti-inflammatory and antinociceptive effects of Mitragyna speciosa Korth methanolic extract. Medical Principles and Practice. 2009, 18, 378–384. [Google Scholar] [CrossRef]
- Kumarnsit, E.; Keawpradub, N.; Nuankaew, W. Acute and long-term effects of alkaloid extract of Mitragyna speciosa on food and water intake and body weight in rats. Fitoterapia. 2006, 77, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Yano, S.; Horie, S.; Yamamoto, L.T. Inhibitory effect of mitragynine, an alkaloid with analgesic effect from Thai medicinal plant Mitragyna speciosa, on electrically stimulated contraction of isolated guinea-pig ileum through the opioid receptor. Life sciences. 1997, 60, 933–942. [Google Scholar] [CrossRef]
- Pathak, L.; Agrawal, Y.; Dhir, A. Natural polyphenols in the management of major depression. Expert opinion on investigational drugs. 2013, 22, 863–880. [Google Scholar] [CrossRef]
- Carpenter, J.M.; Criddle, C.A.; Craig, H.K.; Ali, Z.; Zhang, Z.; Khan, I.A.; Sufka, K.J. Comparative effects of Mitragyna speciosa extract, mitragynine, and opioid agonists on thermal nociception in rats. Fitoterapia 2016, 109, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Kruegel, A.C.; Uprety, R.; Grinnell, S.G.; Langreck, C.; Pekarskaya, E.A.; Le Rouzic, V.; Ansonoff, M.; Gassaway, M.M.; Pintar, J.E.; Pasternak, G.W. 7-Hydroxymitragynine is an active metabolite of mitragynine and a key mediator of its analgesic effects. ACS central science. 2019, 5, 992–1001. [Google Scholar] [CrossRef]
- Kumarnsit, E.; Keawpradub, N.; Nuankaew, W. Effect of Mitragyna speciosa aqueous extract on ethanol withdrawal symptoms in mice. Fitoterapia. 2007, 78, 182–185. [Google Scholar] [CrossRef]
- Apryani, E.; Hidayat, M.T.; Moklas, M.; Fakurazi, S.; Idayu, N.F. Effects of mitragynine from Mitragyna speciosa Korth leaves on working memory. Journal of Ethnopharmacology. 2010, 129, 357–360. [Google Scholar] [CrossRef]
- Idayu, N.F.; Hidayat, M.T.; Moklas, M.; Sharida, F.; Raudzah, A.N.; Shamima, A.; Apryani, E. Antidepressant-like effect of mitragynine isolated from Mitragyna speciosa Korth in mice model of depression. Phytomedicine. 2011, 18, 402–407. [Google Scholar] [CrossRef] [PubMed]
- Obeng, S.; Kamble, S.H.; Reeves, M.E.; Restrepo, L.F.; Patel, A.; Behnke, M.; Chear, N.J.-Y.; Ramanathan, S.; Sharma, A.; León, F. Investigation of the adrenergic and opioid binding affinities, metabolic stability, plasma protein binding properties, and functional effects of selected indole-based kratom alkaloids. Journal of medicinal chemistry. 2019, 63, 433–439. [Google Scholar] [CrossRef]
- Johnson, L.E.; Balyan, L.; Magdalany, A.; Saeed, F.; Salinas, R.; Wallace, S.; Veltri, C.A.; Swogger, M.T.; Walsh, Z.; Grundmann, O. Focus: Plant-based Medicine and Pharmacology: The Potential for Kratom as an Antidepressant and Antipsychotic. The Yale Journal of Biology and Medicine. 2020, 93, 283. [Google Scholar]
- Ahmad, I.; Prabowo, W.C.; Arifuddin, M.; Fadraersada, J.; Indriyanti, N.; Herman, H.; Purwoko, R.Y.; Nainu, F.; Rahmadi, A.; Paramita, S. Mitragyna species as pharmacological agents: From abuse to promising pharmaceutical products. Life. 2022, 12, 193. [Google Scholar] [CrossRef]
- Vijeepallam, K.; Pandy, V.; Murugan, D.D.; Naidu, M. Methanolic extract of Mitragyna speciosa Korth leaf inhibits ethanol seeking behaviour in mice: Involvement of antidopaminergic mechanism. Metabolic brain disease. 2019, 34, 1713–1722. [Google Scholar] [CrossRef] [PubMed]
- Salleh, N.A.M.; Halim, S.; Ridzuan, P.M.; Uzid, M.M.; Ramli, M.D. The Potential Role of Neuroprotective Effects of Kratom (Mitragyna Speciosa) On Brain Aging. Journal of Cellular & Molecular Anesthesia. 2021, 6, 352–353. [Google Scholar]
- Zul Aznal, A.N.; Mohamad Nor Hazalin, N.A.; Hassan, Z.; Mat, N.H.; Chear, N.J.; Teh, L.K.; Salleh, M.Z.; Suhaimi, F.W. Adolescent kratom exposure affects cognitive behaviours and brain metabolite profiles in Sprague-Dawley rats. Front Pharmacol. 2022, 28, 1057423. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.P.; Narayanan, S.P.; Müller, C.P.P.; Vicknasingam, B.P.; Yücel, M.P.; Ho, E.T.W.P.; Hassan, Z.P.; Mansor, S.M.P. Long-Term Cognitive Effects of Kratom (Mitragyna speciosa Korth.) Use. J Psychoactive Drugs. 2019, 51, 19–27. [Google Scholar] [CrossRef]
- Flores-Bocanegra, L.; Raja, H.A.; Graf, T.N.; Augustinović, M.; Wallace, E.D.; Hematian, S.; Kellogg, J.J.; Todd, D.A.; Cech, N.B.; Oberlies, N.H. The Chemistry of Kratom [Mitragyna speciosa]: Updated Characterization Data and Methods to Elucidate Indole and Oxindole Alkaloids. Journal of Natural Products. 2020, 83, 2165–2177. [Google Scholar] [CrossRef]
- Suwanlert, S. A study of kratom eaters in Thailand. Bull Narc. 1975, 27, 21–27. [Google Scholar]
- Ahmad, K.; Aziz, Z. Mitragyna speciosa use in the northern states of Malaysia: a cross-sectional study. Journal of Ethnopharmacology 2012, 141, 446–450. [Google Scholar] [CrossRef]
- Cinosi, E.; Martinotti, G.; Simonato, P.; Singh, D.; Demetrovics, Z.; Roman-Urrestarazu, A.; Bersani, F.S.; Vicknasingam, B.; Piazzon, G.; Li, J.-H. Following “the roots” of Kratom (Mitragyna speciosa): the evolution of an enhancer from a traditional use to increase work and productivity in Southeast Asia to a recreational psychoactive drug in western countries. BioMed research international 2015. [Google Scholar] [CrossRef]
- Ruck, C. Mushrooms, Myth and Mithras: The drug cult that civilized Europe, City Lights Books. 2021.
- Tanguay, P. Kratom in Thailand: Decriminalisation and community control? Series on legislative reform of drug policies, Vol. 13. Transnational Institute (TNI). International Drug Policy Consortium (IDPC). 2011. International Drug Policy Consortium (IDPC).
- Saingam, D.; Assanangkornchai, S.; Geater, A.F.; Balthip, Q. Pattern and consequences of krathom (Mitragyna speciosa Korth.) use among male villagers in southern Thailand: a qualitative study. International Journal of Drug Policy. 2013, 24, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Narayanan, S.; Vicknasingam, B. Traditional and non-traditional uses of Mitragynine (Kratom): A survey of the literature. Brain Research Bulletin 2016, 126, 41–46. [Google Scholar] [CrossRef]
- Khalil, S.; Abdullah, S.A.J.; Ahmad, R. Enforcement status of the poison act 1952 against offences related to kratom (Mitragyna speciosa korth) misuse in Malaysia. UUM Journal of Legal Studies (UUMJLS). 2020, 11, 75–93. [Google Scholar] [CrossRef]
- Swogger, M.T.; Hart, E.; Erowid, F.; Erowid, E.; Trabold, N.; Yee, K.; Parkhurst, K.A.; Priddy, B.M.; Walsh, Z. Experiences of kratom users: a qualitative analysis. Journal of psychoactive Drugs. 2015, 47, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Assanangkornchai, S.; Muekthong, A.; Sam-Angsri, N.; Pattanasattayawong, U. The use of Mitragynine speciosa (“Krathom”), an addictive plant, in Thailand. Substance use & misuse. 2007, 42, 2145–2157. [Google Scholar] [CrossRef]
- Grundmann, O. Patterns of Kratom use and health impact in the US-Results from an online survey. Drug Alcohol Depend. 2017, 176, 63–70. [Google Scholar] [CrossRef]
- Hillebrand, J.; Olszewski, D.; Sedefov, R. Legal highs on the Internet. Subst Use Misuse. 2010, 45, 330–340. [Google Scholar] [CrossRef]
- Schmidt, M.M.; Sharma, A.; Schifano, F.; Feinmann, C. "Legal highs" on the net-Evaluation of UK-based Websites, products and product information. Forensic Sci Int. 2011, 206, 92–97. [Google Scholar] [CrossRef]
- Feng, L.Y.; Battulga, A.; Han, E.; Chung, H.; Li, J.H. New psychoactive substances of natural origin: A brief review. J Food Drug Anal. 2017, 25, 461–471. [Google Scholar] [CrossRef]
- Mustafa, A.; Trevino, L.M.; Turner, C. Pressurized hot ethanol extraction of carotenoids from carrot by-products. Molecules. 2012, 17, 1809–1818. [Google Scholar] [CrossRef]
- Mohamed, H.M. Green, environment-friendly, analytical tools give insights in pharmaceuticals and cosmetics analysis. TrAC Trends in Analytical Chemistry. 2015, 66, 176–192. [Google Scholar] [CrossRef]
- Chimnoi, N.; Khunnawutmanotham, N.; Feungfuloy, P.; Chatrewongwan, K.; Techasakul, S. Facile isolation and purification of thailandine, a biologically active oxoaporphine alkaloid, from Stephania venosa leaves using ion-pair liquid-liquid extraction. Research Journal of Medicinal Plant. 2013, 7, 68–76. [Google Scholar]
- Fatima, N.; Tapondjou, L.A.; Lontsi, D.; Sondengam, B.L.; Ur, R.A.; Choudhary, M.I. Quinovic acid glycosides from Mitragyna stipulosa-first examples of natural inhibitors of snake venom phosphodiesterase I. Nat Prod Lett. 2002, 16, 389–393. [Google Scholar] [CrossRef]
- Asase, A.; Kokubun, T.; Grayer, R.J.; Kite, G.; Simmonds, M.S.; Oteng-Yeboah, A.A.; Odamtten, G.T. Chemical constituents and antimicrobial activity of medicinal plants from Ghana: Cassia sieberiana, Haematostaphis barteri, Mitragyna inermis and Pseudocedrela kotschyi. Phytother Res. 2008, 22, 1013–1016. [Google Scholar] [CrossRef] [PubMed]
- Phongprueksapattana, S.; Putalun, W.; Keawpradub, N.; Wungsintaweekul, J. Mitragyna speciosa: hairy root culture for triterpenoid production and high yield of mitragynine by regenerated plants. Z Naturforsch C J Biosci. 2008, 63, 691–698. [Google Scholar] [CrossRef]
- Ponglux, D.; Wongseripipatana, S.; Takayama, H.; Kikuchi, M.; Kurihara, M.; Kitajima, M.; Aimi, N.; Sakai, S. A New Indole Alkaloid, 7 alpha-Hydroxy-7H-mitragynine, from Mitragyna speciosa in Thailand. Planta Med. 1994, 60, 580–581. [Google Scholar] [CrossRef] [PubMed]
- León, F.; Habib, E.; Adkins, J.E.; Furr, E.B.; McCurdy, C.R.; Cutler, S.J. Phytochemical characterization of the leaves of Mitragyna speciosa grown in U.S.A. Nat Prod Commun. 2009, 4, 907–910. [Google Scholar] [CrossRef]
- Jaleel, C.A.; Gopi, R.; Kishorekumar, A.; Manivannan, P.; Sankar, B.; Panneerselvam, R. Interactive effects of triadimefon and salt stress on antioxidative status and ajmalicine accumulation in Catharanthus roseus. Acta Physiologiae Plantarum. 2008, 30, 287–292. [Google Scholar] [CrossRef]
- Gajalakshmi, S.; Vijayalakshmi, S.; Devi, R.V. Pharmacological activities of Catharanthus roseus: a perspective review. International Journal of Pharma and Bio Sciences. 2013, 4, 431–439. [Google Scholar]
- Duwiejua, M.; Woode, E.; Obiri, D.D. Pseudo-akuammigine, an alkaloid from Picralima nitida seeds, has anti-inflammatory and analgesic actions in rats. J Ethnopharmacol. 2002, 81, 73–79. [Google Scholar] [CrossRef]
- Trager, W.F.; Lee, C.M.; Phillipson, J.D.; Haddock, R.E.; Dwuma-Badu, D.; Beckett, A.H. Configurational analysis of rhynchophylline-type oxindole alkaloids. The absolute configuration of ciliaphylline, rhynchociline, specionoxeine, isospecionoxeine, rotundifoline and isorotundifoline. Tetrahedron. 1968, 24, 523–543. [Google Scholar] [CrossRef]
- Roquebert, J. and Demichel, P. Inhibition of the alpha 1 and alpha 2-adrenoceptor-mediated pressor response in pithed rats by raubasine, tetrahydroalstonine and akuammigine. Eur J Pharmacol. 1984, 106, 203–205. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.L.; Song, J.X.; Lu, J.H.; Yuan, Z.W.; Liu, L.F.; Durairajan, S.S.; Li, M. Corynoxine, a Natural Autophagy Enhancer, Promotes the Clearance of Alpha-Synuclein via Akt/mTOR Pathway. J Neuroimmune Pharmacol. 2014, 9, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Salmean, G.; Ciaraldi, T.P.; Nogueira, L.; Barboza, J.; Taub, P.R.; Hogan, M.C.; Henry, R.R.; Meaney, E.; Villarreal, F.; Ceballos, G.; Ramirez-Sanchez, I. Effects of (-)-epicatechin on molecular modulators of skeletal muscle growth and differentiation. J Nutr Biochem. 2014, 25, 91–94. [Google Scholar] [CrossRef] [PubMed]
- Escandón, R.A.; del Campo, M.; López-Solis, R.; Obreque-Slier, E.; Toledo, H. Antibacterial effect of kaempferol and (−)-epicatechin on Helicobacter pylori. European Food Research and Technology. 2016, 242, 1495–1502. [Google Scholar] [CrossRef]
- Matsumoto, K.; Hatori, Y.; Murayama, T.; Tashima, K.; Wongseripipatana, S.; Misawa, K.; Kitajima, M.; Takayama, H.; Horie, S. Involvement of μ-opioid receptors in antinociception and inhibition of gastrointestinal transit induced by 7-hydroxymitragynine, isolated from Thai herbal medicine Mitragyna speciosa. European journal of pharmacology. 2006, 549, 63–70. [Google Scholar] [CrossRef]
- Shukla, A.; Srinivasan, B.P. 16,17-Dihydro-17b-hydroxy isomitraphylline alkaloid as an inhibitor of DPP-IV, and its effect on incretin hormone and β-cell proliferation in diabetic rat. Eur J Pharm Sci. 2012, 47, 512–519. [Google Scholar] [CrossRef]
- García, R.; Cayunao, C.; Bocic, R.; Backhouse, N.; Delporte, C.; Zaldivar, M.; Erazo, S. Antimicrobial activity of isopteropodine. Z Naturforsch C J Biosci. 2005, 60, 385–388. [Google Scholar] [CrossRef]
- Wang, X.L.; Zhang, L.M.; Hua, Z. Blocking effect of rhynchophylline on calcium channels in isolated rat ventricular myocytes. Zhongguo Yao Li Xue Bao. 1994, 15, 115–118. [Google Scholar]
- Kocialski, A.B.; Marozzi, F.J., Jr.; Malone, M.H. Effects of certain nonsteroid anti-inflammatory drugs, tolbutamide, and tetrahydroalstonine on blood glucose and carrageen in-induced pedal edema in rats. J Pharm Sci. 1972, 61, 1202–1205. [Google Scholar] [CrossRef]
- Kang, T.H.; Murakami, Y.; Matsumoto, K.; Takayama, H.; Kitajima, M.; Aimi, N.; Watanabe, H. Rhynchophylline and isorhynchophylline inhibit NMDA receptors expressed in Xenopus oocytes. Eur J Pharmacol. 2002, 455, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Philipp, A.A.; Wissenbach, D.K.; Weber, A.A.; Zapp, J.; Maurer, H.H. Metabolism studies of the Kratom alkaloids mitraciliatine and isopaynantheine, diastereomers of the main alkaloids mitragynine and paynantheine, in rat and human urine using liquid chromatography-linear ion trap-mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2011, 879, 1049–1055. [Google Scholar] [CrossRef]
- Kruegel, A.C.; Gassaway, M.M.; Kapoor, A.; Váradi, A.; Majumdar, S.; Filizola, M.; Javitch, J.A.; Sames, D. Synthetic and Receptor Signaling Explorations of the Mitragyna Alkaloids: Mitragynine as an Atypical Molecular Framework for Opioid Receptor Modulators. J Am Chem Soc. 2016, 138, 6754–6764. [Google Scholar] [CrossRef]
- Joshi, B. and Taylor, W. Structure of mitragynine (9-methoxycorynantheidine). Chemistry and Industry. 1963, 54, 573. [Google Scholar]
- Zacharias, D.; Rosenstein, R.; Jeffrey, G. The structure of mitragynine hydroiodide. Acta Crystallographica. 1965, 18, 1039–1043. [Google Scholar] [CrossRef]
- Takayama, H.; Ishikawa, H.; Kurihara, M.; Kitajima, M.; Aimi, N.; Ponglux, D.; Koyama, F.; Matsumoto, K.; Moriyama, T.; Yamamoto, L.T.; Watanabe, K.; Murayama, T.; Horie, S. Studies on the synthesis and opioid agonistic activities of mitragynine-related indole alkaloids: discovery of opioid agonists structurally different from other opioid ligands. J Med Chem. 2002, 45, 1949–1956. [Google Scholar] [CrossRef]
- Sharma, A.; Kamble, S.H.; León, F.; Chear, N.J.; King, T.I.; Berthold, E.C.; Ramanathan, S.; McCurdy, C.R.; Avery, B.A. Simultaneous quantification of ten key Kratom alkaloids in Mitragyna speciosa leaf extracts and commercial products by ultra-performance liquid chromatography-tandem mass spectrometry. Drug Test Anal. 2019, 11, 1162–1171. [Google Scholar] [CrossRef]
- Ellis, C.R.; Racz, R.; Kruhlak, N.L.; Kim, M.T.; Zakharov, A.V.; Southall, N.; Hawkins, E.G.; Burkhart, K.; Strauss, D.G.; Stavitskaya, L. Evaluating kratom alkaloids using PHASE. PLoS One. 2020, 15, e0229646. [Google Scholar] [CrossRef]
- Gutridge, A.M.; Robins, M.T.; Cassell, R.J.; Uprety, R.; Mores, K.L.; Ko, M.J.; Pasternak, G.W.; Majumdar, S.; van Rijn, R.M. G protein-biased kratom-alkaloids and synthetic carfentanil-amide opioids as potential treatments for alcohol use disorder. Br J Pharmacol. 2020, 177, 1497–1513. [Google Scholar] [CrossRef] [PubMed]
- Chear, N.J.; León, F.; Sharma, A.; Kanumuri, S.R.R.; Zwolinski, G.; Abboud, K.A.; Singh, D.; Restrepo, L.F.; Patel, A.; Hiranita, T.; Ramanathan, S.; Hampson, A.J.; McMahon, L.R.; McCurdy, C.R. Exploring the Chemistry of Alkaloids from Malaysian Mitragyna speciosa (Kratom) and the Role of Oxindoles on Human Opioid Receptors. J Nat Prod. 2021, 84, 1034–1043. [Google Scholar] [CrossRef] [PubMed]
- Houghton, P.J. and Said, I.M. 3-dehydromitragynine: An alkaloid from Mitragyna speciosa. Phytochemistry. 1986, 25, 2910–2912. [Google Scholar] [CrossRef]
- Houghton, P.J.; Latiff, A.; Said, I.M. Alkaloids from Mitragyna speciosa. Phytochemistry. 1991, 30, 347–350. [Google Scholar] [CrossRef]
- Limsuwanchote, S.; Wungsintaweekul, J.; Keawpradub, N.; Putalun, W.; Morimoto, S.; Tanaka, H. Development of indirect competitive ELISA for quantification of mitragynine in Kratom (Mitragyna speciosa (Roxb.) Korth.). Forensic Sci Int. 2014, 244, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, S.; León, F.; Chear, N.J.Y.; Yusof, S.R.; Murugaiyah, V.; McMahon, L.R.; McCurdy, C.R. Kratom (Mitragyna speciosa Korth.): A description on the ethnobotany, alkaloid chemistry, and neuropharmacology. 2021. [Google Scholar] [CrossRef]
- Manwill, P.K.; Flores-Bocanegra, L.; Khin, M.; Raja, H.A.; Cech, N.B.; Oberlies, N.H.; Todd, D.A. Kratom (Mitragyna speciosa) Validation: Quantitative Analysis of Indole and Oxindole Alkaloids Reveals Chemotypes of Plants and Products. Planta Medica. 2022, 88, 838–857. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Carrell, E.J.; Ali, Z.; Avula, B.; Avonto, C.; Parcher, J.F.; Khan, I.A. Comparison of three chromatographic techniques for the detection of mitragynine and other indole and oxindole alkaloids in Mitragyna speciosa (kratom) plants. J Sep Sci. 2014, 37, 1411–1418. [Google Scholar] [CrossRef]
- Azizi, J.; Ismail, S.; Mordi, M.N.; Ramanathan, S.; Said, M.I.M.; Mansor, S.M. In vitro and in vivo effects of three different Mitragyna speciosa Korth leaf extracts on phase II drug metabolizing enzymes—glutathione transferases (GSTs). Molecules. 2010, 15, 432–441. [Google Scholar] [CrossRef]
- Raffa, R.B. Kratom and other mitragynines: the chemistry and pharmacology of opioids from a non-opium source, CRC Press. 2014.
- Veeramohan, R.; Azizan, K.A.; Aizat, W.M.; Goh, H.-H.; Mansor, S.M.; Yusof, N.S.M.; Baharum, S.N.; Ng, C.L. Metabolomics data of Mitragyna speciosa leaf using LC-ESI-TOF-MS. Data in brief. 2018, 18, 1212–1216. [Google Scholar] [CrossRef]
- Trakulsrichai, S.; Sathirakul, K.; Auparakkitanon, S.; Krongvorakul, J.; Sueajai, J.; Noumjad, N.; Sukasem, C.; Wananukul, W. Pharmacokinetics of mitragynine in man. Drug design, development and therapy. 2015, 9, 2421. [Google Scholar] [CrossRef]
- Smith, K.E.; Rogers, J.M.; Dunn, K.E.; Grundmann, O.; McCurdy, C.R.; Schriefer, D.; Epstein, D.H. Searching for a Signal: Self-Reported Kratom Dose-Effect Relationships Among a sample of US adults with regular Kratom use histories. Front Pharmacol. 2022, 1, 765917. [Google Scholar] [CrossRef]
- Manda, V.K.; Avula, B.; Ali, Z.; Khan, I.A.; Walker, L.A.; Khan, S.I. Evaluation of in vitro absorption, distribution, metabolism, and excretion (ADME) properties of mitragynine, 7-hydroxymitragynine, and mitraphylline. Planta Med. 2014, 80, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Harizal, S.; Mansor, S.; Hasnan, J.; Tharakan, J.; Abdullah, J. Acute toxicity study of the standardized methanolic extract of Mitragyna speciosa Korth in rodents. Journal of Ethnopharmacology. 2010, 131, 404–409. [Google Scholar] [CrossRef] [PubMed]
- Ilmie, M.U.; Jaafar, H.; Mansor, S.M.; Abdullah, J.M. Subchronic toxicity study of standardized methanolic extract of Mitragyna speciosa Korth in Sprague-Dawley Rats. Frontiers in Neuroscience 2015, 9, 189. [Google Scholar] [CrossRef] [PubMed]
- Saidin, N.A.; Randall, T.; Takayama, H.; Holmes, E.; Gooderham, N.J. Malaysian Kratom, a phyto-pharmaceutical of abuse: studies on the mechanism of its cytotoxicity. Toxicology. 2008, 1, 19–20. [Google Scholar] [CrossRef]
- Rusli, N.; Amanah, A.; Kaur, G.; Adenan, M.I.; Sulaiman, S.F.; Wahab, H.A.; Tan, M.L. The inhibitory effects of mitragynine on P-glycoprotein in vitro. Naunyn-Schmiedeberg's archives of pharmacology. 2019, 392, 481–496. [Google Scholar] [CrossRef]
- Azizi, J.; Ismail, S.; Mansor, S.M. Mitragyna speciosa Korth leaves extracts induced the CYP450 catalyzed aminopyrine-N-demethylase (APND) and UDP-glucuronosyl transferase (UGT) activities in male Sprague-Dawley rat livers. Drug metabolism and drug interactions. 2013, 28, 95–105. [Google Scholar] [CrossRef]
- Kong, W.M.; Chik, Z.; Ramachandra, M.; Subramaniam, U.; Aziddin, R.E.R.; Mohamed, Z. Evaluation of the effects of Mitragyna speciosa alkaloid extract on cytochrome P450 enzymes using a high throughput assay. Molecules. 2011, 16, 7344–7356. [Google Scholar] [CrossRef]
- Kamble, S.H.; Sharma, A.; King, T.I.; León, F.; McCurdy, C.R.; Avery, B.A. Metabolite profiling and identification of enzymes responsible for the metabolism of mitragynine, the major alkaloid of Mitragyna speciosa (kratom). Xenobiotica. 2019, 49, 1279–1288. [Google Scholar] [CrossRef]
- Uno, Y.; Uehara, S.; Murayama, N.; Yamazaki, H. Cytochrome P450 1A1, 2C9, 2C19, and 3A4 polymorphisms account for interindividual variability of toxicological drug metabolism in cynomolgus macaques. Chemical research in toxicology. 2018, 31, 1373–1381. [Google Scholar] [CrossRef]
- Hanapi, N.; Ismail, S.; Mansor, S. Inhibitory effect of mitragynine on human cytochrome P450 enzyme activities. Pharmacognosy research. 2013, 5, 241. [Google Scholar] [CrossRef]
- Showande, S.J.; Fakeye, T.O.; Kajula, M.; Hokkanen, J.; Tolonen, A. Potential inhibition of major human cytochrome P450 isoenzymes by selected tropical medicinal herbs—Implication for herb–drug interactions. Food science & nutrition. 2019, 7, 44–55. [Google Scholar] [CrossRef]
- Ulbricht, C.; Costa, D.; Dao, J.; Isaac, R.; LeBlanc, Y.C.; Rhoades, J.; Windsor, R.C. An evidence-based systematic review of kratom (Mitragyna speciosa) by the Natural Standard Research Collaboration. Journal of Dietary Supplements. 2013, 10, 152–170. [Google Scholar] [CrossRef]
- Hughes, R.L. Fatal combination of mitragynine and quetiapine–a case report with discussion of a potential herb-drug interaction. Forensic Science, Medicine and Pathology. 2019, 15, 110–113. [Google Scholar] [CrossRef]
- Fluyau, D.; Revadigar, N. Biochemical benefits, diagnosis, and clinical risks evaluation of kratom. Frontiers in psychiatry. 2017, 8, 62. [Google Scholar] [CrossRef]
- Grewal, K.S. Observations OX the Pharmacology of Mitragynine. J. Pharmacol. 1932, 46, 251–271. [Google Scholar]
- Mohammad Yusoff, N.H.; Mansor, S.M.; Visweswaran, N.; Muller, C.P.; Hassan, Z. GABAB receptor system modulates mitragynine-induced conditioned place preference in rats. Conference Abstract: 14th Meeting of the Asian-Pacific Society for Neurochemistry 2016. [Google Scholar] [CrossRef]
- Effendy, M.A.; Yunusa, S.; Mat, N.H.; Has, A.T.C.; Müller, C.P.; Hassan, Z. The role of AMPA and NMDA receptors in mitragynine effects on hippocampal synaptic plasticity. Behav Brain Res. 2023, 13, 114169. [Google Scholar] [CrossRef] [PubMed]
- Kruegel, A.C.; Grundmann, O. The medicinal chemistry and neuropharmacology of kratom: A preliminary discussion of a promising medicinal plant and analysis of its potential for abuse. Neuropharmacology 2018, 134 Pt A, 108–108. [Google Scholar] [CrossRef]
- Ammar, I.H.; Muzaimi, M.; Sharif, M.M. The effects on motor behaviour and short-term memory tasks in mice following an acute administration of Mitragyna speciosa alkaloid extract and mitragynine. Journal of Medicinal Plants Research. 2011, 5, 5810–5817. [Google Scholar]
- Ismail, N.I.W.; Jayabalan, N.; Mansor, S.M.; Müller, C.P.; Muzaimi, M. Chronic mitragynine (kratom) enhances punishment resistance in natural reward seeking and impairs place learning in mice. Addiction biology. 2017, 22, 967–976. [Google Scholar] [CrossRef]
- Senik, M.; Mansor, S.; Rammes, G.; Tharakan, J.; Abdullah, J. Mitragyna speciosa Korth standardized methanol extract induced short-term potentiation of CA1 subfield in rat hippocampal slices. Journal of Medicinal Plants Research. 2012, 6, 1234–1243. [Google Scholar] [CrossRef]
- Raymond-Hamet, A. Les alcaloïdes du Mitragyna speciosa Korthals [The alkaloids of Mitragyna speciosa Korthals–French]. Ann Pharm Fr. 1950, 8, 482–490. [Google Scholar]
- Federico, A.; Morgillo, F.; Tuccillo, C.; Ciardiello, F.; Loguercio, C. Chronic inflammation and oxidative stress in human carcinogenesis. International journal of cancer. 2007, 121, 2381–2386. [Google Scholar] [CrossRef]
- Perwez Hussain, S.; Harris, C.C. Inflammation and cancer: an ancient link with novel potentials. International journal of cancer. 2007, 121, 2373–2380. [Google Scholar] [CrossRef] [PubMed]
- Utar, Z.; Majid, M.I.A.; Adenan, M.I.; Jamil, M.F.A.; Lan, T.M. Mitragynine inhibits the COX-2 mRNA expression and prostaglandin E2 production induced by lipopolysaccharide in RAW264.7 macrophage cells. Journal of Ethnopharmacology. 2011, 136, 75–82. [Google Scholar] [CrossRef]
- Otero-Losada, M.; Capani, F.; Perez Lloret, S. Neuroprotection - New Approaches and Prospects. IntechOpen. 2020, 77918. [Google Scholar]
- Vermaire, D.J.; Skaer, D.; Tippets, W. Kratom and general anesthesia: a case report and review of the literature. A&A Practice. 2019, 12, 103–105. [Google Scholar] [CrossRef]
- Horie, S.; Koyama, F.; Takayama, H.; Ishikawa, H.; Aimi, N.; Ponglux, D.; Matsumoto, K.; Murayama, T. Indole alkaloids of a Thai medicinal herb, Mitragyna speciosa, that has opioid agonistic effect in guinea-pig ileum. Planta medica. 2005, 71, 231–236. [Google Scholar] [CrossRef]
- Matsumoto, K.; Takayama, H.; Ishikawa, H.; Aimi, N.; Ponglux, D.; Watanabe, K.; Horie, S. Partial agonistic effect of 9-hydroxycorynantheidine on mu-opioid receptor in the guinea-pig ileum. Life Sci. 2005, 78, 2265–2271. [Google Scholar] [CrossRef] [PubMed]
- Reanmongkol, W.; Keawpradub, N.; Sawangjaroen, K. Effects of the extracts from Mitragyna speciosa Korth. leaves on analgesic and behavioral activities in experimental animals. Songklanakarin J. Sci. Technol. 2007, 29 (Suppl. 1), 39–48. [Google Scholar]
- Sabetghadam, A.; Ramanathan, S.; Mansor, S.M. The evaluation of antinociceptive activity of alkaloid, methanolic, and aqueous extracts of Malaysian Mitragyna speciosa Korth leaves in rats. Pharmacognosy research. 2010, 2, 181. [Google Scholar] [CrossRef] [PubMed]
- Idid, S.; Saad, L.; Yaacob, H.; Shahimi, M. Evaluation of analgesia induced by mitragynine, morphine and paracetamol on mice. ASEAN Review of Biodiversity and Environmental Conservation (ARBEC). 1998, 4, 1–7. [Google Scholar]
- Botpiboon, O. Effects of Caffeine and Codeine on Pharmacokinetics and Antinociceptive Activity of Alkaloid Extract from Leaves of Kratom (Mitragyna speciosa Korth.), Prince of Songkla University. 2010.
- Matsumoto, K.; Mizowaki, M.; Suchitra, T.; Takayama, H.; Sakai, S.-I.; Aimi, N.; Watanabe, H. Antinociceptive action of mitragynine in mice: evidence for the involvement of supraspinal opioid receptors. Life Sciences. 1996, 59, 1149–1155. [Google Scholar] [CrossRef]
- Boyer, E.W.; Babu, K.M.; Adkins, J.E.; McCurdy, C.R.; Halpern, J.H. Self-treatment of opioid withdrawal using kratom (Mitragynia speciosa korth). Addiction. 2008, 103, 1048–1050. [Google Scholar] [CrossRef]
- Takayama, H. Chemistry and pharmacology of analgesic indole alkaloids from the rubiaceous plant, Mitragyna speciosa. Chemical and Pharmaceutical Bulletin. 2004, 52, 916–928. [Google Scholar] [CrossRef]
- Thongpradichote, S.; Matsumoto, K.; Tohda, M.; Takayama, H.; Aimi, N.; Sakai, S.-I.; Watanabe, H. Identification of opioid receptor subtypes in antinociceptive actions of supraspinally-admintstered mitragynine in mice. Life Sciences. 1998, 62, 1371–1378. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, L.T.; Horie, S.; Takayama, H.; Aimi, N.; Sakai, S.-I.; Yano, S.; Shan, J.; Pang, P.K.; Ponglux, D.; Watanabe, K. Opioid receptor agonistic characteristics of mitragynine pseudoindoxyl in comparison with mitragynine derived from Thai medicinal plant Mitragyna speciosa. General Pharmacology: The Vascular System. 1999, 33, 73–81. [Google Scholar] [CrossRef]
- Stolt, A.-C.; Schröder, H.; Neurath, H.; Grecksch, G.; Höllt, V.; Meyer, M.R.; Maurer, H.H.; Ziebolz, N.; Havemann-Reinecke, U.; Becker, A. Behavioral and neurochemical characterization of kratom (Mitragyna speciosa) extract. Psychopharmacology. 2014, 231, 13–25. [Google Scholar] [CrossRef]
- Hemby, S.E.; McIntosh, S.; Leon, F.; Cutler, S.J.; McCurdy, C.R. Abuse liability and therapeutic potential of the Mitragyna speciosa (kratom) alkaloids mitragynine and 7-hydroxymitragynine. Addiction biology. 2019, 24, 874–885. [Google Scholar] [CrossRef]
- Havemann-Reinecke, U. P01-50-Kratom and alcohol dependence: Clinical symptoms, withdrawal treatment and pharmacological mechanisms-A case report. European Psychiatry. 2011, 26, 50–50. [Google Scholar] [CrossRef]
- Obeng, S.; Leon, F.; Patel, A.; Gonzalez, J.D.Z.; Da Silva, L.C.; Restrepo, L.F.; Gamez-Jimenez, L.R.; Ho, N.P.; Calvache, M.P.G.; Pallares, V.L.C.; Helmes, J.A.; Shiomitsu, S.K.; Soto, P.L.; McCurdy, C.R.; McMahon, L.R.; Wilkerson, J.L.; Hiranita, T. Interactive Effects of µ-Opioid and Adrenergic-α (2) Receptor Agonists in Rats: Pharmacological Investigation of the Primary Kratom Alkaloid Mitragynine and Its Metabolite 7-Hydroxymitragynine. J Pharmacol Exp Ther. 2022, 383, 182–198. [Google Scholar] [CrossRef]
- Vijeepallam, K.; Pandy, V.; Kunasegaran, T.; Murugan, D.D.; Naidu, M. Mitragyna speciosa leaf extract exhibits antipsychotic-like effect with the potential to alleviate positive and negative symptoms of psychosis in mice. Frontiers in pharmacology. 2016, 7, 464. [Google Scholar] [CrossRef]
- Lu, J.; Wei, H.; Wu, J.; Jamil, M.F.A.; Tan, M.L.; Adenan, M.I.; Wong, P.; Shim, W. Evaluation of the cardiotoxicity of mitragynine and its analogues using human induced pluripotent stem cell-derived cardiomyocytes. PloS one. 2014, 9, e115648. [Google Scholar] [CrossRef]
- Sandager, M.; Nielsen, N.D.; Stafford, G.I.; van Staden, J.; Jäger, A.K. Alkaloids from Boophane disticha with affinity to the serotonin transporter in rat brain. Journal of Ethnopharmacology. 2005, 98, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Elgorashi, E.E.; Stafford, G.I.; Jäger, A.K.; van Staden, J. Inhibition of [3H] citalopram binding to the rat brain serotonin transporter by Amaryllidaceae alkaloids. Planta medica. 2006, 72, 470–473. [Google Scholar] [CrossRef] [PubMed]
- Neergaard, J.; Andersen, J.; Pedersen, M.; Stafford, G.; Van Staden, J.; Jäger, A. Alkaloids from Boophone disticha with affinity to the serotonin transporter. South African Journal of Botany. 2009, 75, 371–374. [Google Scholar] [CrossRef]
- Kong, L.; Cheng, C.H.; Tan, R. Inhibition of MAO A and B by some plant-derived alkaloids, phenols and anthraquinones. Journal of Ethnopharmacology. 2004, 91, 351–355. [Google Scholar] [CrossRef]
- Oboh, G.; Oyeleye, S.; Ademiluyi, A. The food and medicinal values of indigenous leafy vegetables. African Vegetables Forum. 2017, 1238. [Google Scholar] [CrossRef]
- Nwanna, E.; Oyeleye, S.; Ogunsuyi, O.; Oboh, G.; Boligon, A.; Athayde, M. In vitro neuroprotective properties of some commonly consumed green leafy vegetables in Southern Nigeria. NFS journal. 2016, 2, 19–24. [Google Scholar] [CrossRef]
- Oboh, G.; Akinyemi, A.J.; Adeleye, B.; Oyeleye, S.I.; Ogunsuyi, O.B.; Ademosun, A.O.; Ademiluyi, A.O.; Boligon, A.A. Polyphenolic compositions and in vitro angiotensin-I-converting enzyme inhibitory properties of common green leafy vegetables: a comparative study. Food Science and Biotechnology. 2016, 25, 1243–1249. [Google Scholar] [CrossRef] [PubMed]
- Oboh, G.; Ogunruku, O.O.; Oyeleye, S.I.; Olasehinde, T.A.; Ademosun, A.O.; Boligon, A.A. Phenolic extracts from Clerodendrum volubile leaves inhibit cholinergic and monoaminergic enzymes relevant to the management of some neurodegenerative diseases. Journal of Dietary Supplements. 2017, 14, 358–371. [Google Scholar] [CrossRef]
- Lühr, S.; Vilches-Herrera, M.; Fierro, A.; Ramsay, R.R.; Edmondson, D.E.; Reyes-Parada, M.; Cassels, B.K.; Iturriaga-Vásquez, P. 2-Arylthiomorpholine derivatives as potent and selective monoamine oxidase B inhibitors. Bioorganic & medicinal chemistry. 2010, 18, 1388–1395. [Google Scholar] [CrossRef]
- Chen, L.; Fei, S.; Olatunji, O.J. LC/ESI/TOF-MS Characterization, Anxiolytic and Antidepressant-like Effects of Mitragyna speciosa Korth Extract in Diabetic Rats. Molecules. 2022, 27, 2208. [Google Scholar] [CrossRef]
- Innok, W.; Hiranrat, A.; Chana, N.; Rungrotmongkol, T.; Kongsune, P. In silico and in vitro anti-AChE activity investigations of constituents from Mytragyna speciosa for Alzheimer’s disease treatment. J Comput Aided Mol Des. 2021, 35, 325–336. [Google Scholar] [CrossRef]
- Tsuji, M.; Takeuchi, T.; Miyagawa, K.; Ishii, D.; Imai, T.; Takeda, K.; Kitajima, M.; Takeda, H. Yokukansan, a traditional Japanese herbal medicine, alleviates the emotional abnormality induced by maladaptation to stress in mice. Phytomedicine. 2014, 21, 363–371. [Google Scholar] [CrossRef]
- Neef, D.W.; Jaeger, A.M.; Thiele, D.J. Heat shock transcription factor 1 as a therapeutic target in neurodegenerative diseases. Nature reviews Drug discovery. 2011, 10, 930–944. [Google Scholar] [CrossRef]
- Gomez-Pastor, R.; Burchfiel, E.T.; Thiele, D.J. Regulation of heat shock transcription factors and their roles in physiology and disease. Nature reviews Molecular cell biology. 2018, 19, 4–19. [Google Scholar] [CrossRef]
- Steinkraus, K.A.; Smith, E.D.; Davis, C.; Carr, D.; Pendergrass, W.R.; Sutphin, G.L.; Kennedy, B.K.; Kaeberlein, M. Dietary restriction suppresses proteotoxicity and enhances longevity by an hsf-1-dependent mechanism in Caenorhabditis elegans. Aging cell. 2008, 7, 394–404. [Google Scholar] [CrossRef]
- Gülçin, İ.; Elmastaş, M.; Aboul-Enein, H.Y. Antioxidant activity of clove oil–A powerful antioxidant source. Arabian Journal of Chemistry. 2012, 5, 489–499. [Google Scholar] [CrossRef]
- Sadeli, R.A. Uji Aktivitas Antioksidan Dengan Metode DPPH (1, 1-Diphenyl-2-Picrylhydrazyl) Ekstrak Bromelain Buah Nanas (Ananas Comosus (L.) Merr.). Fakultas Farmasi, Universitas Sanata Dharma, Yogyakarta 2016. [Google Scholar]
- Rusmarilin, H.; Lubis, Z.; Lubis, L.; Barutu, Y. Potential of natural antioxidants of black cumin seed (Nigella sativa) and sesame seed (Sesamum indicum) extract by microencapsulation methods. IOP Conference Series: Earth and Environmental Science, IOP Publishing 2019. [Google Scholar] [CrossRef]
- Suhaling, S. Uji Aktivitas Antioksidan Ekstrak Metanol Kacang Merah (Phaseolus vulgaris L.) Dengan Metode DPPH, Universitas Islam Negeri Alauddin Makassar. 2010.
- Ikhlas, N. Uji Aktivitas Antioksidan Ekstrak Herba Kemangi (Ocimum americanum Linn) dengan Metode DPPH (2, 2-Difenil-1-Pikrilhidrazil). 2013.
- Chae, H.S.; Park, H.J.; Hwang, H.R.; Kwon, A.; Lim, W.H.; Yi, W.J.; Han, D.H.; Kim, Y.H.; Baek, J.H. The effect of antioxidants on the production of pro-inflammatory cytokines and orthodontic tooth movement. Mol Cells. 2011, 32, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Nau, R.; Eiffert, H. Modulation of release of proinflammatory bacterial compounds by antibacterials: potential impact on course of inflammation and outcome in sepsis and meningitis. Clinical microbiology reviews. 2002, 15, 95–110. [Google Scholar] [CrossRef] [PubMed]
- Lüderitz, O.; Tanamoto, K.; Galanos, C.; McKenzie, G.R.; Brade, H.; Zähringer, U.; Shiba, T. Lipopolysaccharides: structural principles and biologic activities. Clinical Infectious Diseases. 1984, 6, 428–431. [Google Scholar] [CrossRef] [PubMed]
- Sejvar, J.; Lutterloh, E.; Naiene, J.; Likaka, A.; Manda, R.; Nygren, B.; Monroe, S.; Khaila, T.; Lowther, S.A.; Capewell, L.; Date, K.; Townes, D.; Redwood, Y.; Schier, J.; Barr, B.T.; Demby, A.; Mallewa, M.; Kampondeni, S.; Blount, B.; Humphrys, M.; Talkington, D.; Armstrong, G.L.; Mintz, E. Neurologic manifestations associated with an outbreak of typhoid fever, Malawi—Mozambique, 2009: an epidemiologic investigation. PLoS One. 2012, 7, e46099. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Arellano, L.; Maldonado-Bernal, C. Helicobacter pylori and neurological diseases: Married by the laws of inflammation. World J Gastrointest Pathophysiol. 2014, 15, 5400–5404. [Google Scholar] [CrossRef]
- Arneth, B.M. Gut–brain axis biochemical signalling from the gastrointestinal tract to the central nervous system: gut dysbiosis and altered brain function. Postgraduate medical journal. 2018, 94, 446–452. [Google Scholar] [CrossRef]
- Sabetghadam, A.; Ramanathan, S.; Sasidharan, S.; Mansor, S.M. Subchronic exposure to mitragynine, the principal alkaloid of Mitragyna speciosa, in rats. " Journal of Ethnopharmacology. 2013, 146, 815–823. [Google Scholar] [CrossRef]
- Chittrakarn, S.; Sawangjaroen, K.; Prasettho, S.; Janchawee, B.; Keawpradub, N. Inhibitory effects of kratom leaf extract (Mitragyna speciosa Korth.) on the rat gastrointestinal tract. Journal of ethnopharmacology. 2008, 116, 173–178. [Google Scholar] [CrossRef]
- Tsuchiya, S.; Miyashita, S.; Yamamoto, M.; Horie, S.; Sakai, S.-I.; Aimi, N.; Takayama, H.; Watanabe, K. Effect of mitragynine, derived from Thai folk medicine, on gastric acid secretion through opioid receptor in anesthetized rats. European journal of pharmacology. 2002, 443, 185–188. [Google Scholar] [CrossRef]
- Anwar, M.; Law, R.; Schier, J. Notes from the field: kratom (Mitragyna speciosa) exposures reported to poison centers—United States, 2010–2015. " MMWR Morb Mortal Wkly Rep. 2016, 65, 748–749. [Google Scholar] [CrossRef]
- Scott Gottlieb, M.D. Agency’s Scientific Evidence on the Presence of Opioid Compounds in Kratom, Underscoring Its Potential for Abuse, Food and Drug Administration Statement from FDA Commissioner.
- Sethi, R.; Hoang, N.; Ravishankar, D.A.; McCracken, M.; Manzardo, A.M. Kratom (Mitragyna speciosa): friend or foe? " The Primary Care Companion for CNS Disorders. 2020, 22, 27410. [Google Scholar] [CrossRef]
- Griffiths, Carrie L.; Nidhi Gandhi, and Jacqueline L. Olin. Possible kratom-induced hepatomegaly: a case report. Journal of the American Pharmacists Association. 2018, 58, 561–563. [CrossRef]
- Tayabali, K.; Bolzon, C.; Foster, P.; Patel, J.; Kalim, M.O. Kratom: a dangerous player in the opioid crisis. J Community Hosp Intern Med Perspect. 2018, 8, 107–110. [Google Scholar] [CrossRef] [PubMed]
- Chittrakarn, S.; Keawpradub, N.; Sawangjaroen, K.; Kansenalak, S.; Janchawee, B. The neuromuscular blockade produced by pure alkaloid, mitragynine and methanol extract of kratom leaves (Mitragyna speciosa Korth.). Journal of Ethnopharmacology. 2010, 129, 344–349. [Google Scholar] [CrossRef]
- Hanapi, N.; et al. Evaluation of Selected Malaysian Medicinal Plants on Phase I Drug Metabolizing Enzymes, CYP 2 C 9, CYP 2 D 6 and CYP 3 A 4 Activities in vitro. International Journal of Pharmacology. 2010, 6, 494–499. [Google Scholar] [CrossRef]
- Purintrapiban, J.; Keawpradub, N.; Kansenalak, S.; Chittrakarn, S.; Janchawee, B.; Sawangjaroen, K. Study on glucose transport in muscle cells by extracts from Mitragyna speciosa (Korth) and mitragynine. Nat Prod Res. 2011, 25, 1379–1387. [Google Scholar] [CrossRef]
- Nelsen, J.L.; Lapoint, J.; Hodgman, M.J.; Aldous, K.M. Seizure and coma following Kratom (Mitragynina speciosa Korth) exposure. J Med Toxicol. 2010, 6, 424–426. [Google Scholar] [CrossRef]
- Kapp, F.G.; Maurer, H.H.; Auwärter, V.; Winkelmann, M.; Hermanns-Clausen, M. Intrahepatic cholestasis following abuse of powdered kratom (Mitragyna speciosa). J Med Toxicol. 2011, 7, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Dorman, C.; Wong, M.; Khan, A. Cholestatic hepatitis from prolonged kratom use: a case report. Hepatology. 2015, 61, 1086–1087. [Google Scholar] [CrossRef] [PubMed]
- Riverso, M.; Chang, M.; Soldevila-Pico, C.; Lai, J.; Liu, X. Histologic Characterization of Kratom Use-Associated Liver Injury. Gastroenterology Res. 2018, 11, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, C.T.; Iqbal, U.; Tighe, S.P.; Ahmed, A. Kratom-Induced Cholestatic Liver Injury and Its Conservative Management. J Investig Med High Impact Case Rep. 2019. [CrossRef] [PubMed]
- Palasamudram Shekar, S.; Rojas, E.E.; D'Angelo, C.C.; Gillenwater, S.R.; Galvis, N.P.M.; C. Legally Lethal Kratom: A Herbal Supplement with Overdose Potential. J Psychoactive Drugs. 2019, 51, 28–30. [Google Scholar] [CrossRef] [PubMed]
- Mousa, M.S.; Sephien, A.; Gutierrez, J.; OʼLeary, C. N-Acetylcysteine for Acute Hepatitis Induced by Kratom Herbal Tea. Am J Ther. 2018, 25, e550–e551. [Google Scholar] [CrossRef]
- Osborne, C.S.; Overstreet, A.N.; Rockey, D.C.; Schreiner, A.D. Drug-Induced Liver Injury Caused by Kratom Use as an Alternative Pain Treatment Amid an Ongoing Opioid Epidemic. J Investig Med High Impact Case Rep. 2019. [CrossRef]

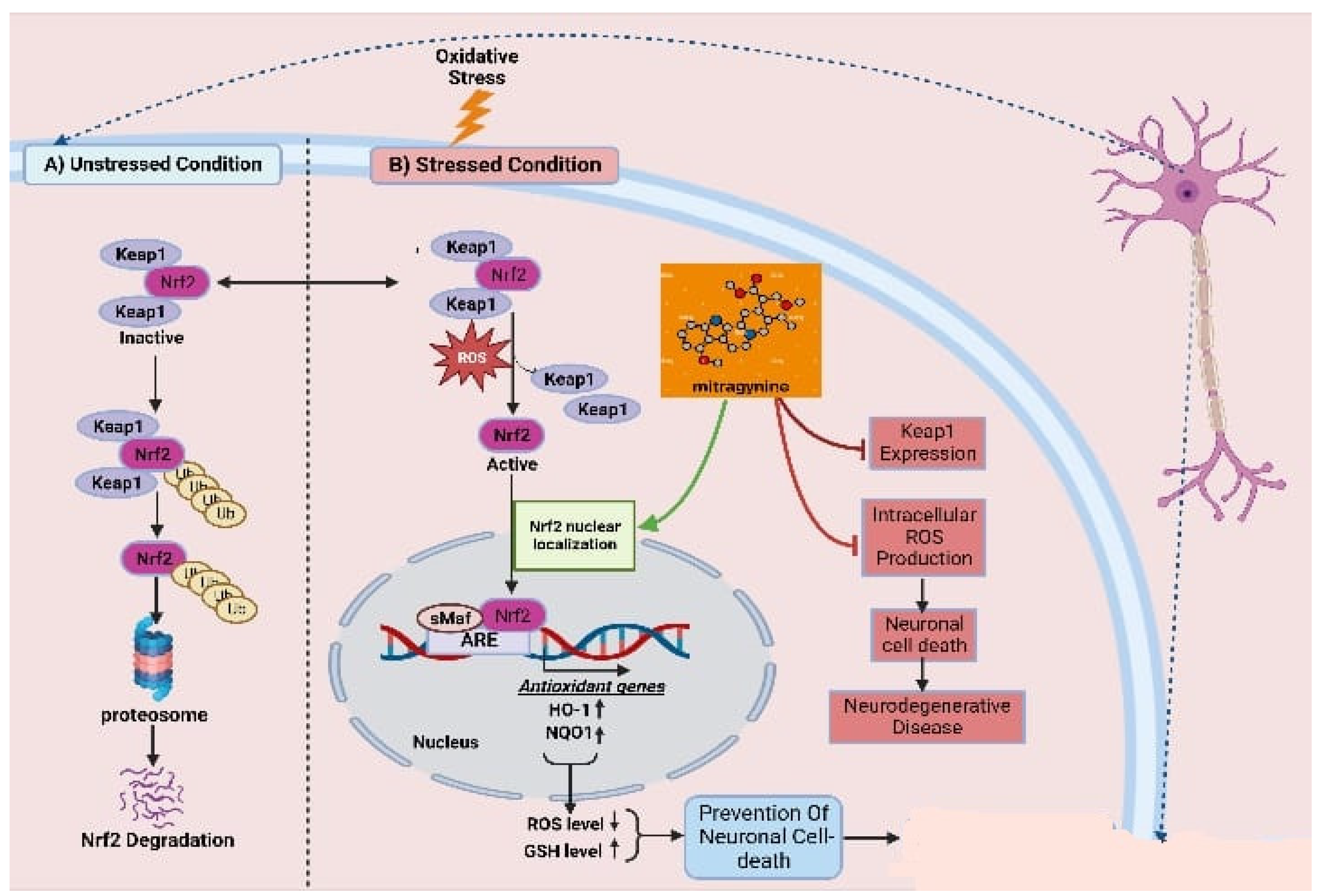
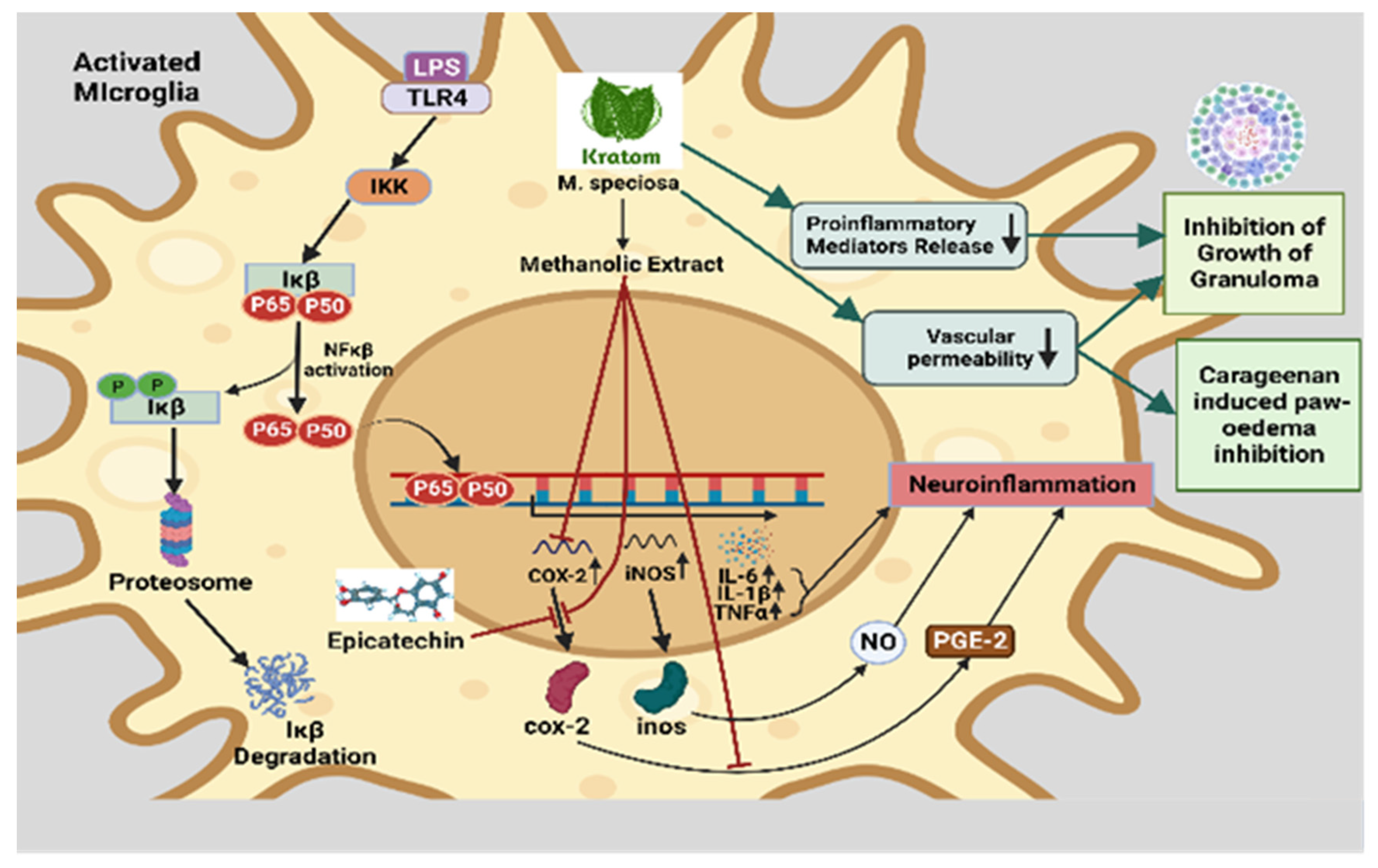
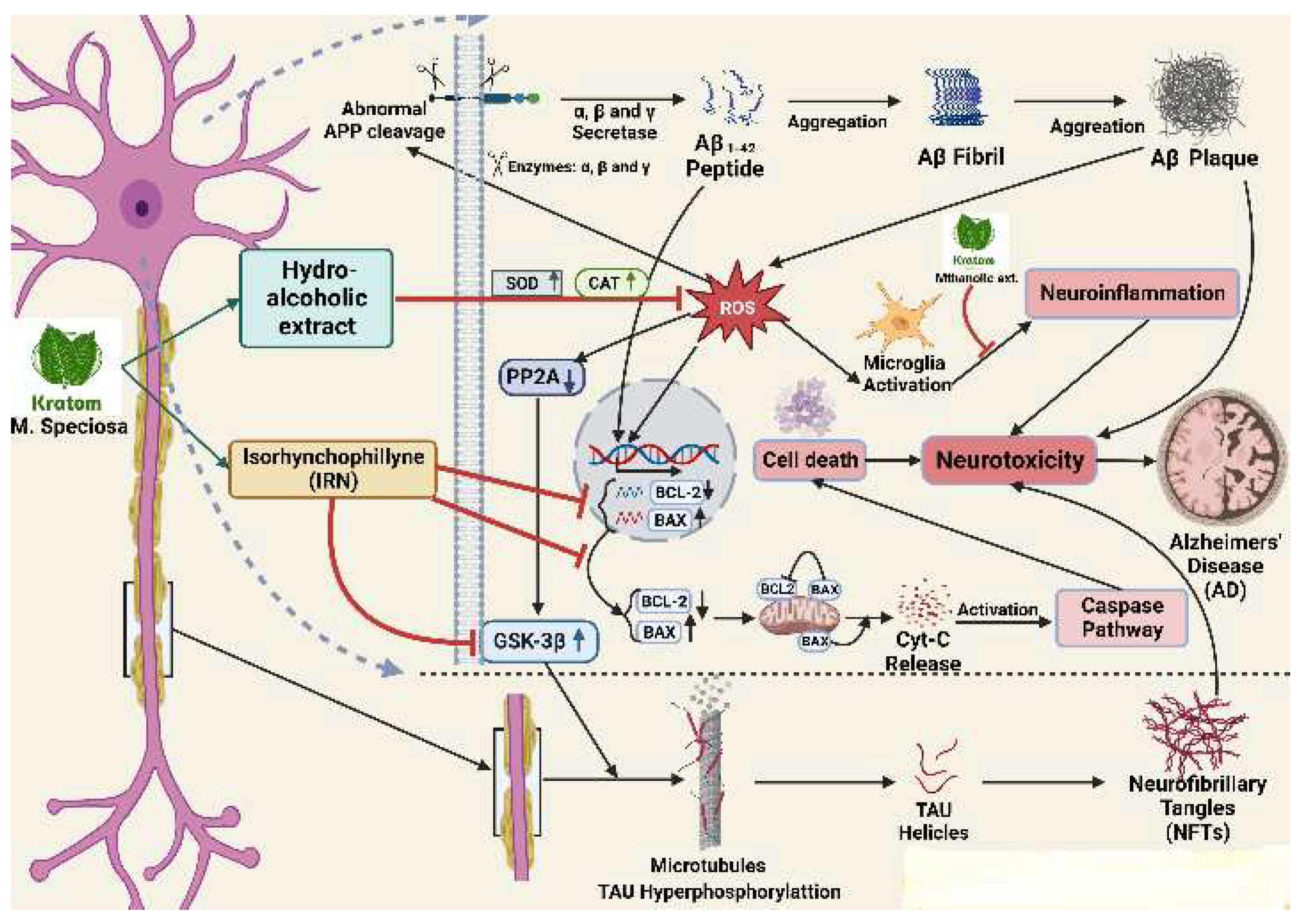
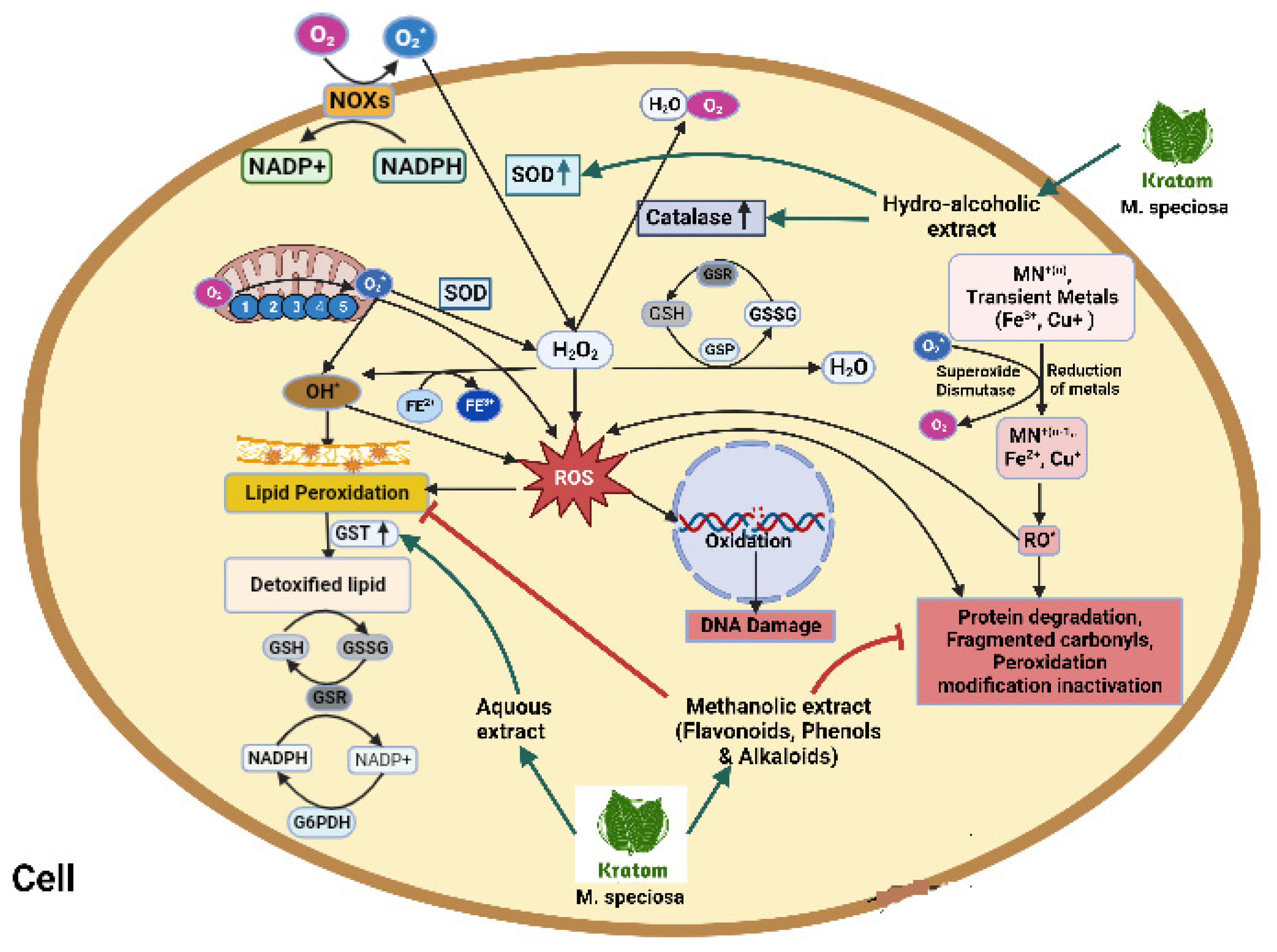
| Antioxidative effect | ||||
|---|---|---|---|---|
| Treatment with doses | Nature of kratom product | Experimental model | Major findings (molecular changes) | Reference |
| Kratom | Methanolic, water, alkaloids | In vitro | The high content of phenolic, flavonoid compounds and the result of DPPH, high antioxidant activity in methanolic extract | (Parthasarathy, Bin Azizi et al. 2009)16 |
| Ethanolic extract | In vitro (DPPH) | The IC50 value of 38.56 μg / mL | (Yuniarti, Nadia et al. 2020)18 | |
| Aqueous extract (100 mg/kg) | Male Sprague Dawley rats | ↑Glutathione transferase (GSTs) activity | (Azizi, Ismail et al. 2010)89 | |
| Neurophysiological | ||||
| Kratom | Mitragynine (5, 10 and 15 mg/kg) | Male ICR mice | Mitragynine neither altered locomotor activity nor its high or low dose | (Apryani, Hidayat et al. 2010)28 |
| Methanolic extract (0.008%) | Male Sprague Dawley rats | ↓ Field excitatory post-synaptic potentials (fEPSP) in the CA1 regionconcentration-dependently, and blocked long-term potentiation (LTP) | (Senik, Mansor et al. 2012)114 | |
| Anti-inflammatory | ||||
| Kratom | Methanolic extract (100-200 mg/kg) | Male Sprague Dawley rats | Dose-dependently suppressedthe development of carrageenan-induced rat paw edema, and ↓ granulomatous tissue formation at 200 mg/kg | (Mossadeq, Sulaiman et al. 2009)21 |
| Methanolic extract (10 and 20 g/mL) | RAW264.7 macrophage cells | ↓mRNA expression of COX-2, ↓PGE2 production, and ↓COX-1 expression | (Utar, Majid et al. 2011)118 | |
| Analgesic/Anti-nociceptive | ||||
| Kratom | 7-hydroxymitragynine (ED50= 0.80 mg/kg, and ED50= 0.93 mg/kg) | Male ddY-strain mice, and male albino guinea-pigs | 4.4–5.7 times morepotent as μ-opioid agonist than morphine in tail-flick and hot-plate test | (Matsumoto, Hatori et al. 2006)68 |
| 7-hydroxymitragynine (100 nM), speciociliatine (30 μM) | Male Albino Dunkin-Hartley guinea-pigs | ↓Twitch contraction and 7-hydroxymitragynine showed most potent opioid effect on the electrically stimulated contraction (pD2 = 8.38 ± 0.12) | (Horie, Koyama et al. 2005)121 | |
| Methanolic and alkaloid extract (100 mg/kg) | Male Swiss mice and Wistar rats | Prolong the latency of nociceptive response in the hot plate test | (Reanmongkol, Keawpradub et al. 2007)123 | |
| Mitragynine alkaloid (10 nM–1 μM) | Male Albino guinea pigs | Block the reversible Ca2+ channel that activates neurotransmitters | (Matsumoto, Yamamoto et al. 2005)122 | |
| Alkaloid (20 mg/kg), methanolic (200 mg/kg), and aqueous extract (400 mg/kg) | Male SpraqueDawley rats | Both hot plate and tail-flick tests showed prolonged nociceptive responses | (Sabetghadam, Ramanathan et al. 2010)124 | |
| Mitragynine alkaloid (100 mg/kg), co-administration of caffeine (25 mg/kg, p.o.) and codeine (3 mg/kg, p.o.) | Male Wistar rats | ↑Latency period in a hot plate test after 30 minutes | (Botpiboon 2010)126 | |
| Mitragynine (2.0 mg/kg) and paynantheine (0.1 mg/kg) | Male wild-type mice (+/+) and μ opioid receptor (MOR) knockout mice (−/−) | Exert analgesic effects predominantly via κ opioid receptors | (Stolt, Schröder et al. 2014)132 | |
| Anti-depressant | ||||
| Kratom | Mitragynine(10 mg/kg and 30 mg/kg) | Male mice from the ICR strain | ↓Corticosterone in forced swim test (FST) and tail suspension test (TST) | (Idayu, Hidayat et al. 2011)29 |
| Aqueous extract (100, 300, and 500 mg/kg) | Male Swiss albino mice | Effects on serotonin or noradrenaline neurotransmissions | (Kumarnsit, Keawpradub et al. 2006)22 | |
| Mitragynine or alkaloid extract (20, 40, and 80 mg/kg) | Male Swiss albino mice | ↑Total number of arm entries, rearing frequency and ↓grooming, and immobility time in the Y-maze test,Alkaloid extract exhibits more potent opioid agonistic effects than mitragynine | (Ammar, Muzaimi et al. 2011)112 | |
| Mitragynine (5, 15, 20, and 25 mg/kg) | Male Swiss albino mice | Chronic mitragynine treatment impaired spatial learning and memory | (Ismail, Jayabalan et al. 2017)113 | |
| Mitragynine (72.5 mg and 74.9 mg) of Kratom tea or >3 glasses daily | Human | Executive function, memory, and attention were not impaired | (Singh, Narayanan et al. 2019)36 | |
| Anti-psychotic/Anti-dopaminergic | ||||
| Kratom | Methanolic extract(75 and 100 mg/kg) | Male Swiss albino mice | Apomorphine-induced cage climbing behavior↓, ↓dopamine-induced contractile response | (Vijeepallam, Pandy et al. 2016)136 |
| Treatment with doses | Nature of kratom product | Experimental model | Major findings (molecular changes) | Reference |
|---|---|---|---|---|
| Anti-bacterial | ||||
| Kratom | Methanolic extract (3.12 to 6.25 mg/mL) | Salmonella typhi and Bacillus subtilis | Minimum inhibitory concentrations (MICs) by the broth dilution method | (Parthasarathy, Bin Azizi et al. 2009)16 |
| Mitragynine (40 mg/kg), alkaloid extract (100 mg/kg) | Adult male Wistar rats | Detoxification and elimination of permethrin | (Srichana, Janchawee et al. 2015)19 | |
| Gastrointestinal effects | ||||
| Kratom | Methanolic extract (50, 100, 200 and 400 mg/kg) | Adult Wistar rats | Protection against castor oil-induced diarrhea, ↓ intestinal transit | (Chittrakarn, Sawangjaroen et al. 2008)165 |
| mitragynine (3–30 μg) | Male Wistar rats | ↓ 2-deoxy-d-glucose-stimulated gastric acid secretion | (Tsuchiya, Miyashita et al. 2002)166 | |
| 7-hydroxymintragynine (ED50 = 1.19 mg/kg) | Male ddY-strain mice | ↓ Gastrointestinal transit and significantly antagonized by β-funaltrexamine hydrochloride (β-FNA) pretreatment, but slightlyantagonized by naloxonazine | (Matsumoto, Hatori et al. 2006)68 | |
| Muscle relaxant | ||||
| Kratom | Methanolic extract (10–40 mg/mL), mitragynine (2 mg/mL) | Wistar rats | Blockade of nerve conduction, amplitude, and duration | (Chittrakarn, Keawpradub et al. 2010)172 |
| Potential to inhibit enzyme activity | ||||
| Kratom | Methanolic extract | Three main CYP450 enzymes CYP2C9, CYP2D6, and CYP3A4 | Most potent effect on CYP2D6 at IC50 (3.6 ± 0.1 μg/mL) | (Hanapi 2010)173 |
| Alkaloid extract | CYP450 enzymes, Quinidine (CYP2D6), ketoconazole (CYP3A4), tranylcypromine (CYP2C19), and furafylline (CYP1A2) | Most potent inhibitory effect on CYP3A4 and CYP2D6 at IC50 values of 0.78 µg/mL and 0.636 µg/mL | (Kong, Chik et al. 2011)100 | |
| Anti-diabetic | ||||
| Kratom | Water extract 0.6 mg mL-1 | L8 muscle cells | ↑ Glucose transporters (GLUT1) | (Purintrapiban, Keawpradub et al. 2011)174 |
| Anti-hypertensive | ||||
| Kratom | Methanolic extract (100, 500, and 1000 mg/kg) | Male Albino rats | Blood pressure (diastolic: 102.7 ± 0.72,98.74 ± 7.95 and 86.85 ± 3.34), and ↑ ALT, AST, albumin, triglycerides, cholesterol, albumin levels | (Harizal, Mansor et al. 2010)95 |
| Weight reduction | ||||
| Kratom | Mitragynine (45 and 50 mg/kg) | Male Wistar rats | ↓Food and water intakes | (Kumarnsit, Keawpradub et al. 2006)22 |
| Mitragynine (100 mg/kg) | Male and female Sprague-Dawley rats | ↓Food intake, ↓Body weight of female rats, and ↑liver weight of both male and female rats | (Sabetghadam, Ramanathan et al. 2013)164 | |
| Uses pattern | Side effects of Kratom | Condition | History | Reference |
|---|---|---|---|---|
| For 1 month, kratom leaf tea is brewed with Datura stramonium | 4-5 mm pupils, minimally reactive, roving conjugate gaze, spasticity of lower extremities with manipulation | Chronic pain after post-colostomy surgery | 64 years male | (Nelsen, Lapoint et al. 2010)175 |
| Powder of leaf 4.6-7 to 8.6-14 g/day for 2 weeks | Loss of appetite, fever and chills, slight abdominal discomfort, concomitant brown discoloration of the urine, jaundice, and pruritus | Intrahepatic cholestasis | 25 years male | (Kapp, Maurer et al. 2011)176 |
| Kratom tea 4 times a day for 3.5 years | A generalized tonic-clonic seizure lasting 5 min, pulse 123 beats per min | Tonic colonic seizure | 43 years male | (Boyer, Babu et al. 2008)128 |
| 1 tablespoon of powder daily for 3 months | Jaundice, dark urine, mild confusion, liver injury | Cholestatic hepatitis | 58 years male | (Dorman, Wong et al. 2015)177 |
| 6 g Kratom capsules daily for 2 weeks | Palpation of the right upper quadrant (RUQ) in the presence of vomiting, fatigue, abdominal pain, and brown urine | Hepatomegaly | 21 years male | (Griffiths, Gandhi et al. 2018)170 |
| Sixty tablets over 1 week | A yellowish appearance to the skin, usually associated with nausea,fatigue, joint pains, night sweats, pale stools anddark urine | Hepatitis | 32 years male | (Tayabali, Bolzon et al. 2018)171 |
| Herbal drug Kratom | Distention, mass, tenderness, rebound, sternal pleuritic chest pain, mild shortness of breath, mild cough, mild coughing, and mild chest pain | Intrahepatic cholestasis | 38 years male | (Riverso, Chang et al. 2018)178 |
| A tablespoon of crushed leaves (-1.5 g/d) | Yellow discoloration of eyes and skin, mild fatigue, jaundice | Intrahepatic cholestasis | 52 years male | (Fernandes, Iqbal et al. 2019)179 |
| Green-colored herbal powder supplement for a few weeks with increasing daily dosage | Pupils were pinpoint and not reactive to light and cool peripheries, the abdomenand pelvis revealed cholestasis without cholecystitis | Intrahepatic cholestasis | 36 years male | (Palasamudram Shekar, Rojas et al. 2019)180 |
| Kratom tea for 2 weeks | Tea-colored urine, malaise, fatigue, and intermittent subjective fever | Acute hepatitis | 31 years male | (Mousa, Sephien et al. 2018)181 |
| Kratom capsules for 3 weeks | Dark urine, pruritus, subjective fevers, fatigue, nonbloody, nonbilious emesis, nonicteric sclera, and sublingual jaundice | Hepatitis | 47 years male | (Osborne, Overstreet et al. 2019)182 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




