Submitted:
06 September 2023
Posted:
07 September 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
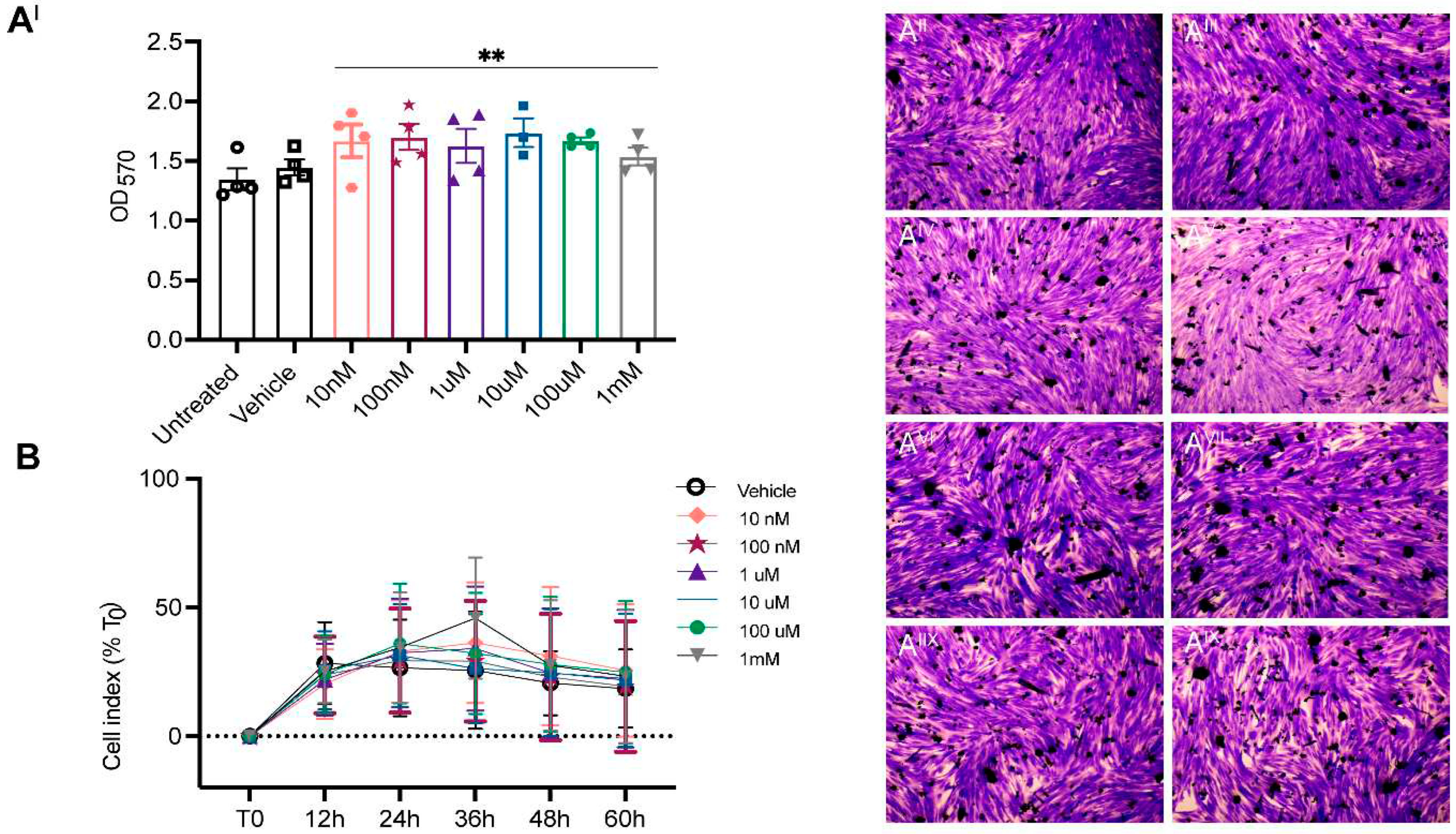
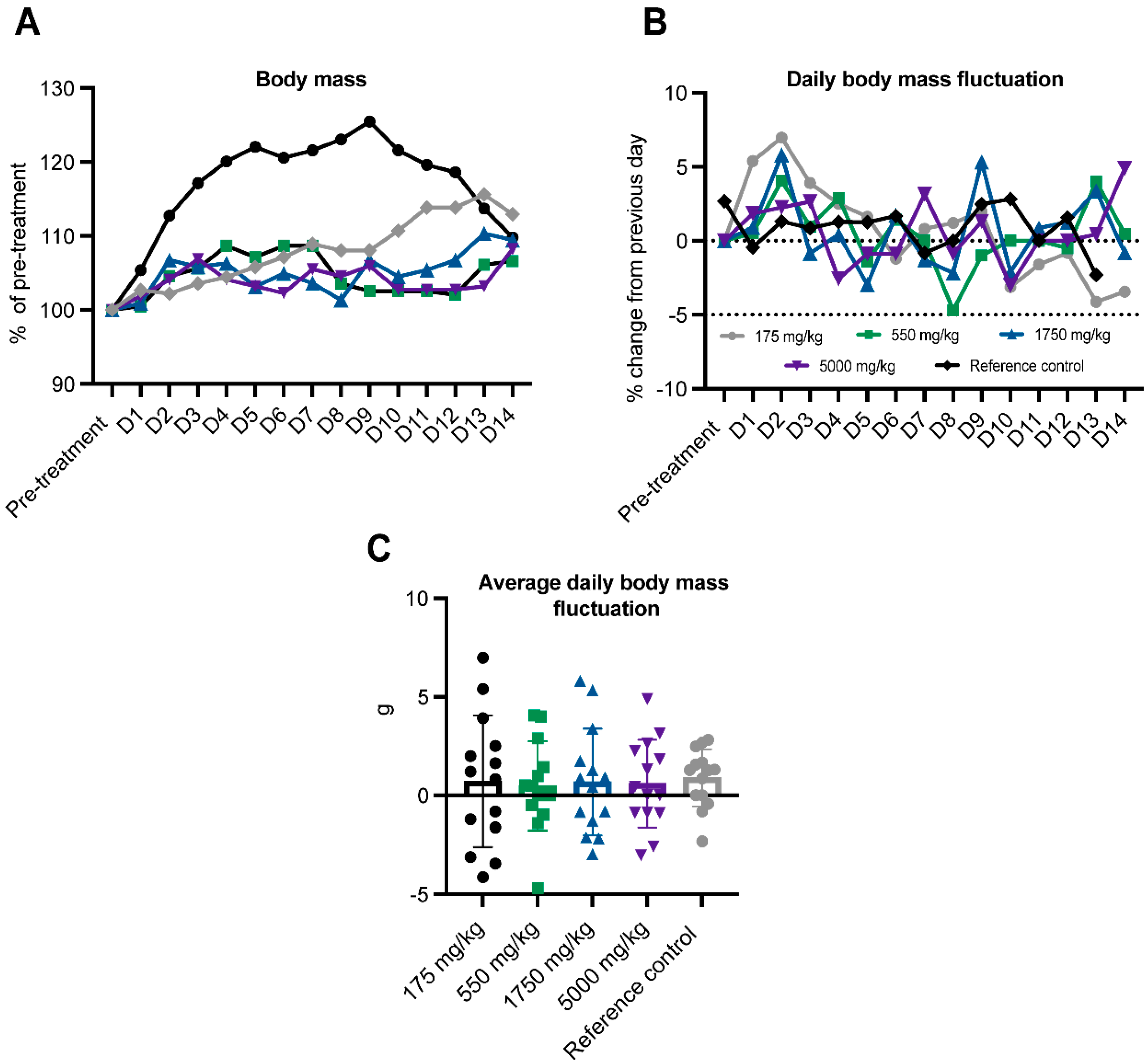
2. Results
| Dose | Post-fasted body weight (g) | ASA mass (mg) | Volume H2O/dose (ml) |
|---|---|---|---|
| 175 mg/kg | 20.4 | 3.57 | 0.204 |
| 550 mg/kg | 19.6 | 10.78 | 0.196 |
| 1750 mg/kg | 22.2 | 38.85 | 0.222 |
| 5000 mg/kg | 21.8 | 109 | 0.218 |
| Observations | ||||||
|---|---|---|---|---|---|---|
| Observations | Symptoms | 175mg/kg ASA n=1 |
550mg/kg ASA n=1 |
1750mg/kg ASA n=1 |
5000mg/kg ASA n=1 |
Untreated |
| Body weight | >5% loss of body weight | - | - | - | - | - |
| Food & water consumption | - | - | - | - | - | - |
| Motor activity | Home-cage activity | - | - | - | + (<30 m) |
- |
| Neurological system | Tremors, limb tone, ataxia | - | - | - | - | - |
| Respiratory system | Gasping, heaving, cyanosis | - | - | - | - | - |
| Gastrointestinal function | Abdominal griping, vomiting, diarrhoea | - | - | - | + (diarrhoea) |
- |
| Skin & mucous membranes | Secretions, excretions | - | - | - | - | - |
| Pain | Grimacing, altered social activity | - | - | - | - | |
| Parameters | Reference Range | ASA Dose | Reference control | |||
|---|---|---|---|---|---|---|
| 175 mg/kg | 550 mg/kg | 1750 mg/kg | 5000 mg/kg |
|||
| Haematological | ||||||
| Erythrocytes (x1012/L) | 3.47-11.73a | 10.5 | 11.06 | 10.66 | 11.0 | 11.02 |
| Haematocrit (L/L) | 0.16-0.58a | 0.48 | 0.54 | 0.54 | 0.54 | 0.54 |
| Haemoglobin (g/L) | 57.0-170.0a | 156 | 162 | 153 | 157 | 158 |
| MCV (fL) | 25.7-59.7b | 48 | 49 | 50 | 49 | 49 |
| MCH (pg) | 9.0-19.2b | 16 | 15 | 14 | 14 | 14 |
| MCHC (g/L) | 264.6-314.2b | 323* | 300 | 286 | 289 | 290 |
| Platelet (x109/L) | 520-1880b | 942 | 954 | 1040 | 1077 | 1188 |
| WCC (x109/L | 2.2-11.53a | 8.9 | 5.5 | 3.8 | 4.6 | 5.8 |
| Segmented Neutrophils (x109/L) | 0.12-0.30b | 0.3 | 0.7* | 0.2 | 0.4* | 0.5* |
| Lymphocytes (x109/L) | 4.36-5.68b | 8.5* | 4.6 | 3.4* | 3.4* | 4.8 |
| Monocytes (x109/L) | 0-0.3b | 0.18 | 0.06 | 0.23 | 0 | 0 |
| Eosinophils (x109/L) | 0-5.1d | 0 | 2 | 0 | 7* | 0.1 |
| Basophils (x109/L) | 0-0.1d | 0 | 0 | 0 | 0.1 | 0 |
| Biochemical | ||||||
| Total Protein (g/L) | 45-83a | 48 | 42* | 53 | 40* | 53 |
| Albumin (g/L) | 20-47a | 29 | 24 | 30 | 21 | 29 |
| Total globulin (g/L) | 18-21c | 19 | 18 | 23* | 19 | 24 |
| Glucose (mmol/L) | 5.2-12.2a | 18.1* | 13.8* | 15* | IS | IS |
| Sodium (mmol/L) | 149.0-281.4 a | 142* | IS | 145* | IS | IS |
| Potassium (mmol/L) | 4.0-14.0a | 10.8 | IS | 7.5 | IS | IS |
| Chloride (mmol/L) | 110.0-204.4a | 98* | IS | 101* | IS | IS |
| Calcium (mmol/L) | 2.3-3.5a | 2.64 | 2.15* | 2.57 | 1.94* | 2.66 |
| Phosphorous (mmol/L) | 2.0-3.1a | 3.32* | 2.23 | 3.95* | 2.83 | 3.69 |
| Urea (mmol/L) | 14.5-21.4a | 4.1* | 5.3* | 5.5* | 4.9* | 5.4* |
| Total bilirubin (µmol/L) | 3.4-14.3a | 9 | 1* | 4 | IS | IS |
| Alanine aminotransferase (ALT; U/L) | 42-73a | 39* | 50 | 75* | 25* | 42 |
| Aspartate aminotransferase (AST; U/L) | 51-122a | 97 | 76 | 220* | 62 | 86 |
| Alkaline phosphatase (ALP; U/L) | 103-217a | 140 | 155 | 193 | 137 | 178 |
| GammaGT (U/L) | 6.0-8.0a | 7 | <5* | <5* | IS | IS |
| Creatine kinase (U/L) | 105-649a | 100* | 43* | 290 | IS | IS |
| Total cholesterol (mmol/L) | 1.3-3.4a | 2.02 | 1.77 | 2.29 | 2.02 | 2.27 |
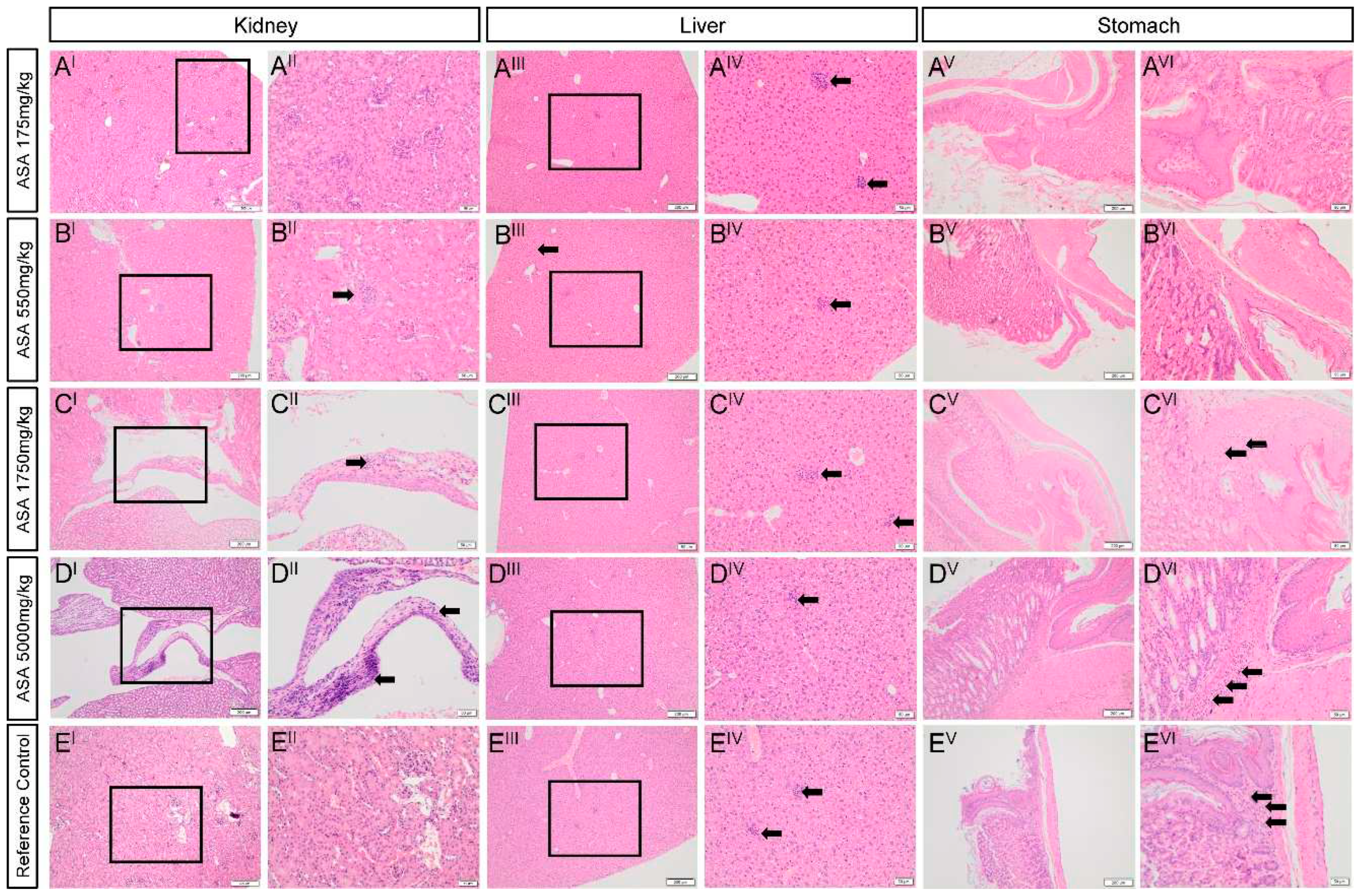
| Pathology | Symptoms | ASA dose | ||||
|---|---|---|---|---|---|---|
| 175 mg/kg | 550 mg/kg |
1750 mg/kg |
5000 mg/kg | Reference control | ||
| Macropathology | Splenic melanosis (background lesion) | - | + | + | - | - |
| Kidney | Rare interstitial lymphoplasmacytic infiltrate with fibrosis | - | + | + | + | - |
| Liver | Multiple random foci of necrosis with neutrophil aggregates | +++ | +++ | + | + | +++ |
| Mild anisocytosis with scant megalocytosis | + | + | + | ++ | - | |
| Stomach | Focal neutrophilic infiltrate into gastric mucosa; | - | - | + | ++ | + |
| Low-grade focal granulocytic gastritis | - | - | + | +++ | +++ | |
3. Discussion
4. Materials and Methods
Animals
Drug details
Acute oral toxicity procedure
Haematology
Biochemistry
Histopathology
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bonsett, C. and A. Rudman, The dystrophin connection—ATP? Medical Hypotheses, 1992. 38(2): p. 139-154.
- Timpani, C.A., A. Hayes, and E. Rybalka, Revisiting the dystrophin-ATP connection: How half a century of research still implicates mitochondrial dysfunction in Duchenne Muscular Dystrophy aetiology. Medical hypotheses, 2015. 85(6): p. 1021-1033. [CrossRef]
- Budzinska, M., A. Zimna, and M. Kurpisz, The role of mitochondria in Duchenne muscular dystrophy. J Physiol Pharmacol, 2021. 72(2). [CrossRef]
- Bonsett, C. and A. Rudman, Duchenne’s muscular dystrophy: a tissue culture perspective. Indiana medicine: the journal of the Indiana State Medical Association, 1984. 77(6): p. 446.
- Lowenstein, J.M. and M.N. Goodman, The purine nucleotide cycle in skeletal muscle. Fed Proc, 1978. 37(9): p. 2308-12.
- Arinze, I.J., Facilitating understanding of the purine nucleotide cycle and the one-carbon pool: Part I: The purine nucleotide cycle. Biochemistry and Molecular Biology Education, 2005. 33(3): p. 165-168. [CrossRef]
- Rybalka, E., et al., Adenylosuccinic Acid: An Orphan Drug with Untapped Potential. Pharmaceuticals, 2023. 16(6): p. 822. [CrossRef]
- Bonsett, C.A. and A. Rudman, ’Oil globules’ in Duchenne muscular dystrophy--history, demonstration, and metabolic significance. Med Hypotheses, 1994. 43(5): p. 327-38.
- OECD, Test No. 425: Acute Oral Toxicity: Up-and-Down Procedure. 2022.
- McInnes, E.F., Background Lesions in Laboratory Animals: A Color Atlas. 2012: Saunders/Elsevier.
- Mazzaccara, C., et al., Age-Related Reference Intervals of the Main Biochemical and Hematological Parameters in C57BL/6J, 129SV/EV and C3H/HeJ Mouse Strains. PLoS One, 2008. 3(11): p. e3772. [CrossRef]
- Zaias, J., et al., Reference values for serum proteins of common laboratory rodent strains. J Am Assoc Lab Anim Sci, 2009. 48(4): p. 387-90.
- Silva-Santana, G., et al., Clinical hematological and biochemical parameters in Swiss, BALB/c, C57BL/6 and B6D2F1 Mus musculus. Animal Models and Experimental Medicine, 2020. 3(4): p. 304-315. [CrossRef]
- Nemzek, J.A., et al., Differences in normal values for murine white blood cell counts and other hematological parameters based on sampling site. Inflamm Res, 2001. 50(10): p. 523-7. [CrossRef]
- Rybalka, E., et al., Adenylosuccinic acid: a novel inducer of the cytoprotectant Nrf2 with efficacy in Duchenne muscular dystrophy. Curr Med Res Opin, 2021. 37(3): p. 465-467. [CrossRef]
- Zou, G.L., et al., The role of Nrf2/PIWIL2/purine metabolism axis in controlling radiation-induced lung fibrosis. Am J Cancer Res, 2020. 10(9): p. 2752-2767.
- Furuhashi, M., New insights into purine metabolism in metabolic diseases: role of xanthine oxidoreductase activity. Am J Physiol Endocrinol Metab, 2020. 319(5): p. E827-E834. [CrossRef]
- Gooding, J.R., et al., Adenylosuccinate Is an Insulin Secretagogue Derived from Glucose-Induced Purine Metabolism. Cell Rep, 2015. 13(1): p. 157-167. [CrossRef]
- Cader, M.Z., et al., FAMIN Is a Multifunctional Purine Enzyme Enabling the Purine Nucleotide Cycle. Cell, 2020. 180(2): p. 278-295.e23.
- Kourakis, S., et al., Dimethyl Fumarate and Its Esters: A Drug with Broad Clinical Utility? Pharmaceuticals (Basel), 2020. 13(10): p. 306.
- Ormerod, A.D. and U. Mrowietz, Fumaric acid esters, their place in the treatment of psoriasis. Br J Dermatol, 2004. 150(4): p. 630-2. [CrossRef]
- Harries, M.J., R.J. Chalmers, and C.E. Griffiths, Fumaric acid esters for severe psoriasis: a retrospective review of 58 cases. Br J Dermatol, 2005. 153(3): p. 549-51. [CrossRef]
- Smith, D., Fumaric acid esters for psoriasis: a systematic review. Ir J Med Sci, 2017. 186(1): p. 161-177. [CrossRef]
- Palte, M.J., et al., Improving the Gastrointestinal Tolerability of Fumaric Acid Esters: Early Findings on Gastrointestinal Events with Diroximel Fumarate in Patients with Relapsing-Remitting Multiple Sclerosis from the Phase 3, Open-Label EVOLVE-MS-1 Study. Adv Ther, 2019. 36(11): p. 3154-3165. [CrossRef]
- Colas, C., P.M. Ung, and A. Schlessinger, SLC Transporters: Structure, Function, and Drug Discovery. Medchemcomm, 2016. 7(6): p. 1069-1081. [CrossRef]
- Bonsett, C.A. and A. Rudman, The dystrophin connection--ATP? Med Hypotheses, 1992. 38(2): p. 139-54.
- Alqahtani, M.S., et al., Advances in Oral Drug Delivery. Frontiers in Pharmacology, 2021. 12. [CrossRef]
- Coleman, J.J. and S.K. Pontefract, Adverse drug reactions. Clin Med (Lond), 2016. 16(5): p. 481-485.
- Guengerich, F.P., Mechanisms of drug toxicity and relevance to pharmaceutical development. Drug Metab Pharmacokinet, 2011. 26(1): p. 3-14. [CrossRef]
- Timpani, C.A., et al., Adenylosuccinic acid therapy ameliorates murine Duchenne Muscular Dystrophy. Sci Rep, 2020. 10(1): p. 1125. [CrossRef]
- Longbrake, E.E., et al., Dimethyl fumarate-associated lymphopenia: risk factors and clinical significance. Multiple Sclerosis Journal–Experimental, Translational and Clinical, 2015. 1: p. 2055217315596994. [CrossRef]
- Timpani, C.A. and E. Rybalka, Calming the (Cytokine) Storm: Dimethyl Fumarate as a Therapeutic Candidate for COVID-19. Pharmaceuticals, 2021. 14(1): p. 15. [CrossRef]
- Timpani, C.A., et al., Nitric Oxide (NO) and Duchenne Muscular Dystrophy: NO Way to Go? Antioxidants, 2020. 9(12): p. 1268.
- Rybalka, E., et al., Chemotherapeutic agents induce mitochondrial superoxide production and toxicity but do not alter respiration in skeletal muscle in vitro. Mitochondrion, 2018. 42: p. 33-49. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




