1. Introduction
Bilateral vestibular loss (BVL) causes, amongst other symptoms, postural imbalance during standing and walking, which worsens in darkness and on uneven ground [
1,
2,
3,
4,
5]. These deficits may partially improve as patients adapt behavioral strategies that alter multisensory calibration and locomotor control [
1,
2]. However, postural deficits tend to not dissipate completely over time, which often results in a long-term functional impairment [
8].
Currently available treatment options for BVL are primarily based on physical therapy that may foster behavioral adaptations to loss of vestibular function [
2]. In addition, emerging treatment strategies either aim at restoring peripheral vestibular function (by means of a vestibular implant [
3]), substitute residual vestibular input by other sensory sources (e.g., from proprioception [
4]), or boost vestibular excitability by an imperceptible vestibular noise stimulation using non-invasive noisy galvanic vestibular stimulation (nGVS) [
12,
13]. The latter principle takes advantage of the fact that a majority of patients with BVL retain some degree of vestibular function [
5,
6]. The rationale behind the application of nGVS is stochastic resonance (SR) – a phenomenon according to which (pathologically increased) thresholds for sensory information processing can be lowered by application of an appropriate amount of low-intensity sensory noise [
7,
8]. Beneficial therapeutic effects of nGVS, in particular on static and dynamic postural deficits, are well documented in animal models [
9,
10,
11] and patients with BVL [
12,
13,
14,
15,
16]. In contrast, it is largely unknown 1) which central nervous networks are altered by nGVS, 2) how these alterations translate into improvements in locomotor and postural performance, and 3) if nGVS might interact with naturally occurring mechanisms of BVL-induced adaptive neuroplasticity. The latter implies functional and structural changes in multiple brain networks, including the brainstem, cerebellum, thalamus, striatum, sensorimotor cortices, and limbic areas [
6,
7].
To gain a better understanding of the neurophysiological and neuroanatomical substrates underlying treatment effects of nGVS, the current study investigated regional brain activation patterns and behavioral responses to repeated applications of nGVS in comparison to sham stimulation by serial whole-brain [18F]-FDG-PET measurements and quantitative locomotor assessments from baseline to 9 weeks after chemical bilateral labyrinthectomy (BL) in the rat. We hypothesized that repeated nGVS would alter neuronal activity in (sub-)cortical sensorimotor and brainstem-cerebellar networks, which are also engaged in BVL-induced adaptive neuroplasticity. In terms of behavioral effects, we expected transient effects of nGVS on postural performance following BL.
2. Methods
2.1. Animals and housing
All rodent experiments were approved by the government of Upper Bavaria and performed in accordance with the guidelines for the use of living animals in scientific studies and the German Law for Protection of Animals (ROB-55.2-2532.Vet_02-21-32). Male Sprague-Dawley rats (N=17, weight 400±20 g, age 9–10 weeks at the time of surgery, Charles River Sulzfeld) were housed two animals per cage in a temperature- and humidity-controlled room with a 12h light/dark cycle and free access to food and water. All rodents were placed in double decker cages (GR1800, Tecniplast, Germany).
2.2. Experimental procedures
In all rodents, a chemical BL was performed by transtympanic injection of bupivacaine and p-arsenic acid. On days 1, 3, 6, 7, 15, 21, 30, 45, and 60 post-BL, low-intensity vestibular noise stimulation (nGVS at 0.2mA; N=8 animals) or sham stimulation (nGVS at 0mA; N=9 animals) was administered. Regional brain activation patterns and locomotion performance were assessed before BL and repetitively until day 60 post-BL. Immediate nGVS treatment (after-)effects on regional brain activations were assessed by [
18F]-FDG-PET analysis that was conducted subsequent to stimulation on days 1, 3, 7, 15, 30, and 60 post-BL. During tracer uptake, animals were allowed to move freely for a period of 25min. PET imaging started 30min p.i. On each of these days, a concomitant instrumented analysis of locomotion was performed before nGVS treatment to assess potential long-term effects of previous stimulations on locomotion. Immediate effects of nGVS on locomotion were assessed on days 6, 21, and 45 post-BL by locomotion analysis directly before and after nGVS/sham intervention (
Figure 1A).
2.3. Chemical bilateral labyrinthectomy
Chemical BL was performed as described previously [
17,
18]: Animals were anesthetized with 1.5% isoflurane in O
2 delivered up to 1.2l/min via a mask. For surgical analgesia, 1.5mg/kg meloxicam was injected s.c. before and 3 days after surgery. An additional 5ml saline was injected s.c. as a bolus. After local anesthesia with 1% bupivacaine hydrochloride, a left paramedian incision was made to expose the lamboidal ridge and the external ear canal. The external ear canal was opened just anterior to the exit point of the facial nerve. Then the tympanic membrane was perforated caudally to the hammer shaft with a 26-gauge needle. About 0.150ml of a 20% bupivacaine solution was instilled into the tympanic cavity. For about 2min the bupivacaine solution was aspirated and instilled slowly again multiple times. The same procedure was followed to instill 0.150ml of a 10% solution of p-arsanilic acid, which irreversibly desensitized the primary sensory cells of the inner ear [
19]. The wound closure was followed by skin suture and, for preventive antibiosis, 2mg/kg marbofloxacin was injected s.c. for three days. This procedure was carried out on both sides, starting on the left side.
2.4. Galvanic vestibular stimulation
Before stimulation, rodents were anesthetized with 2% isoflurane in O
2 (1-2l/min) via a mask and were kept warm with a heating pad. Two cannulas (26-gauge) were placed bilaterally s.c. in contact with the mastoid process and connected to the electrodes of a constant current stimulator (neuroConn, Illmenau, Germany). The electrical signal consisted of band-limited zero-mean white noise between 0 to 30Hz [
14,
15]. Initially, the signal intensity was gradually increased until an amplitude, at which small involuntary head movements of the animal occurred. The final nGVS intensity was set to 20% of this amplitude, which yielded 0.2mA across animals. For each animal and time point of intervention, stimulation was applied for a duration of 30min.
2.5. PET imaging and analysis
Rodents were anesthetized with 2% isoflurane in O2 (1–2l/min) via a mask and a cannula was placed in a lateral tail vein for [18F]-FDG bolus injection (in 0.5ml saline, 40MBq). Subsequently, animals were awakened and allowed to move freely for an uptake period of 25min until anesthesia was induced again with isoflurane (1.8%) for the PET-scan (start 30min p.i.). Two animals per scan were positioned in an Inveon PET scanner (Siemens Healthineers, Erlangen, Germany) and were kept warm with a heating pad. The head was fixed using a custom-made head-holder to prevent any passive head movements. Emission data were recorded for 30min followed by a 7min transmission scan using a rotating [57Co] point source. Upon recovery from anesthesia, animals were returned to their home cages.
Image processing was performed as described previously [
18,
20,
21]. The obtained emission measurements were reconstructed using an Ordered Subsets Expectation Maximization (OSEM-3D) algorithm with decay, scatter, attenuation, and dead time correction as well as sensitivity normalization. The resulting images had 212x212x235 voxels of 0.4x0.4x0.4mm
3. Activity distributions of [
18F]-FDG scans were used as a surrogate for regional cerebral glucose metabolism (rGCM). The volume containing the brains in the images was cropped and rigidly registered into PX Rat atlas space (W. Schiffer [
22]) using PMOD medical image analysis software (PMOD Technologies LLC, RRID: SCR_016547, v4.004). To achieve comparability, normalization of the whole brain mean activity was performed after applying a 0.4mm isotropic Gaussian filter, using a brain mask in atlas space. Subsequently, images were segmented into brain regions using Px Rat (W. Schiffer) atlas and in addition regions of interest for the left and right vestibular nucleus were defined. rGCM levels of symmetric brain regions were pooled after preclusion of any statistical side asymmetries. Finally, mean brain normalized rGCM levels for each of the 26 brain regions were obtained and further statistically processed. Additionally, voxel-wise analysis based on t-tests was performed in SPM 8 software (Wellcome Department of Cognitive Neurology, Great Britain) between measurements in the nGVS and sham stimulation group at days 1, 7, 15, 30, 60 post-BL for the sake of visualization. For PET data, p-values <0.001 were considered significant.
2.6. Locomotion analysis
Quantitative assessment of locomotion was performed with the CatWalk system (CatWalk XT, Noldus Information Technology, Netherlands), which consists of an enclosed walkway (glass plate, 64cm length) that is illuminated by fluorescent light. Locomotion patterns were recorded optically by a high-speed color camera. Rodents were placed individually into the walkway and allowed to move freely in both directions. Only continuous runs across the walkway with a duration <5s and a speed variation <60% were considered for further analysis [
23]. In total, three compliant runs were acquired per animal and assessment time point.
Six spatiotemporal gait parameters linked to balance and coordination were analyzed: locomotion speed (1), and separately for fore and hind limbs base of support (2), mean and variability of stride length (3 & 4) and of stride time (5 & 6). Variability measures were calculated by means of the coefficient of variation [
24].
2.7. Statistical analysis
To assess the impact of BL and the differential effects of post-BL neuroplasticity vs. nGVS on brain activity and locomotion, different univariate and multivariate analyses were performed. Long-term influences of repeated nGVS interventions on the course of post-BL recovery were analyzed by a repeated measures ANOVA with assessment day and stimulation (nGVS vs. sham) separately on each parameter derived from locomotion analysis. Immediate effects of nGVS on locomotor performance on the three intervention days were analyzed by a repeated measures ANOVA with group (nGVS vs. sham), time point (pre vs. post nGVS), and intervention day (6, 21, 45 post BL). Analogous analyses were performed on the PET scan outcomes (i.e., the normalized rGCM levels) using a repeated measures MANOVA that included all analyzed 26 brain regions. Post hoc Bonferroni adjustments were used to control for multiple comparisons within each model. Spearman's rank correlation analysis was performed to assess potential associations between intervention-induced changes in brain activity and locomotor performance. Results were considered significant at p<0.05. Statistical analysis was performed using SPSS (Version 26.0, IBM Corp., USA).
3. Results
3.1. Regional brain activation patterns
Chemical BL had an instantaneous impact on regional brain activation patterns (
Figure 2). Compared to baseline assessment pre-BL, rGCM levels at day 1 post-BL revealed an increased activity in 42% of all analyzed brain regions, particularly within hemispheric sensory and motor networks (e.g., insular, somatosensory, motor cortex, and striatum). A concomitant decrease of activity was found in 19% of regions, mainly within brainstem-cerebellar networks (e.g., colliculus inferior and vestibular nuclei).
During the time period from day 1 to 60 post-BL, a steady decrease of rGCM was found in 31% of brain regions encompassing hemispheric sensory networks (e.g., auditory and somatosensory cortex), motor networks (e.g., medial prefrontal and motor cortex, as well as striatum), and limbic networks (cingulate and retrosplenial cortex). In contrast, rGCM levels in brainstem-cerebellar networks (e.g., cerebellar grey matter and inferior colliculus) and the lateral thalamus exhibited an increase during the course of recovery post-BL.
Repeated treatment with nGVS at days 1, 3, 6, 7, 15, 21, 30, 45, and 60 post-BL resulted in differential rGCM responses that on the whole reversed rGCM modulations occurring in the natural course post-BL. Accordingly, rodents receiving repeated nGVS treatment (compared to sham stimulation) exhibited increased brain activity (
Figure 3), in particular in hemispheric motor networks (e.g., motor cortex and striatum), limbic networks (cingulate and retrosplenial cortex), and a concomitant decrease in brainstem-cerebellar networks (e.g., cerebellar grey and white matter, colliculus inferior, and vestibular nuclei). These stimulation-induced brain activity modulations were present throughout the entire analyzed 60-day period post-BL.
3.2. Locomotor performance
Analogous to effects on regional brain activity, chemical BL also directly affected the locomotor capacity of rodents (
Figure 4). Compared to baseline performance, catwalk assessment at day 1 post-BL revealed a marked slowdown of locomotion linked to a decrease of stride length and a concurrent increase of stride time that was found equally in fore- and hindlimb locomotion. Acute locomotor changes further exhibited typical features of a sensory-ataxic gait pattern with a broadened base of support and increased variability of the spatiotemporal stepping pattern (i.e., stride length CV and stride time CV) that were more pronounced for hindlimb locomotion.
Impaired locomotion steadily improved within the analyzed 60-day period post-BL. Catwalk assessment at day 60 post-BL revealed a close-to-normal locomotion speed, base of support, and spatiotemporal gait variability levels. Repeated nGVS interventions at days 1, 3, 6, 7, 15, 21, 30, 45, and 60 post-BL only marginally affected locomotor recovery in terms of a decreased forelimb stride length CV at day 30 post-BL (
Figure 1D). As a result, the overall course and extent of locomotor recovery following BL was largely comparable between animals receiving nGVS treatment versus animals receiving sham stimulation.
Immediate effects of nGVS stimulation on locomotor performance were assessed at days 6, 21, and 45 post-BL (
Figure 2A,
Figure 5). Compared to sham stimulation, nGVS improved sensory-ataxic gait alterations of treated animals in terms of a reduced base of support (
Figure 5B) and lowered levels of stride length and time variability (
Figure 5DF). These beneficial treatment effects were only observed at day 6 post-BL, when animals still exhibited marked gait ataxia, but not at days 21 and 45 post-BL, when animal locomotion had already largely recovered.
Correlation analysis between stimulation-induced changes in locomotor performance (on day 6 post-BL) and stimulation-induced regional brain activity (on day 7 post-BL) revealed associations with brainstem, hemispheric motor and limbic networks (not corrected for multiple comparisons). Decreased spatial stepping variability correlated with increased rGCM within the vestibular nucleus (R=0.857; p=0.007) and decreased temporal stepping variability with elevated rGCM levels in the cingulate (R=0.821; p=0.012) and retrosplenial cortex (R=0.714; p=0.036). Moreover, a narrower base of support was associated with increased activity within the motor cortex (R=0.821; p=0.012).
4. Discussion
In recent years, low intensity vestibular noise stimulation (i.e., nGVS) has shown promising therapeutic effects on sensorimotor deficits in patients with BVL ([
12,
13,
14,
25]) and other disorders associated with central vestibular deficits (e.g., Parkinson's disease; [
26,
27,
28,
29]). In this study, we applied whole-brain imaging of glucose metabolism and instrumented locomotor analysis to study the potential neurophysiological and neuroanatomical substrates underlying the aforementioned treatment effects of nGVS in a rodent model of BVL. [
18F]-FDG-PET revealed a broad impact of nGVS stimulation on regional brain activation patterns encompassing biologically plausible brain networks in the brainstem, cerebellum, multisensory cortex and motor-basal ganglia circuits. Immediate stimulation-induced modulations of brain activity were consistently present for the entire studied period of 60 days post-BL and broadly reversed brain activity adaptions occurring in the natural course post-BL. Parallel behavioral locomotor assessment, revealed a positive impact of nGVS on sensory-ataxic gait alterations in animals' locomotor pattern that was particularly found during the early stage post-BL. Finally, stimulation-induced locomotor improvements were linked to nGVS brain activity responses in the brainstem, hemispheric motor and limbic networks. In the following, we will discuss these observations and contrast the general impact of BL and post-BL recovery with stimulation-induced effects on regional brain activation patterns and locomotor performance.
4.1. Impact of bilateral labyrinthectomy and post-surgery recovery on brain activation patterns and locomotor performance
As reported previously [
30], chemical BL resulted in an acute decrease of neuronal activity within brainstem-cerebellar networks encompassing primary and secondary processing hubs (e.g., vestibular nuclei, inferior colliculus) of sensory cues from peripheral vestibular and auditory afferents. Decreased regional neuronal activity in brainstem-cerebellar networks only partially recovered during the studied period of 60 days post-BL.
In contrast, we observed a predominant acute increase of regional brain activation in hemispheric sensory, motor, and limbic networks in response to BL. Specifically, neuronal activity in the thalamus – a prominent hub for multisensory integration and vestibular-motor action [
31] – further increased during the course of post-BL recovery, which likely reflects processes of adaptive neuroplasticity that facilitate multisensory substitution and recalibration [
30].
Alterations of brain activation patterns within frontal-basal ganglia loops following BL substantiate the existing evidence for a considerable connection between the vestibular system and the basal ganglia [
32]. Frontal-basal ganglia networks are essentially involved in the supraspinal control of locomotion. In particular they are considered to facilitate or suppress movement via a direct (excitatory) and indirect (inhibitory) pathway [
33]. Altered brain activity within frontal-basal ganglia networks was further associated with a persistent locomotor hyperactivity in the open field in our animals following BL (data not shown), which corresponds to a well-documented behavioral feature in animal models of BVL [
32,
34]. This association fits well with the previously proposed hypothesis that a loss of vestibular input to the striatum may enforce basal ganglia-output via the direct pathway and thereby activate locomotion [
32]. The apparent difference to a previous study that reported decreased instead of increased rGCM levels in frontal-basal ganglia networks following BL might result from methodological differences: While [
18F]-FDG tracer uptake in the previous study was performed in resting rodents, animals in the current study were allowed to move freely during this period. Within the studied period of post-BL recovery, elevated functional activity in frontal-basal ganglia partially decreased but did in general not reach baseline levels of neuronal activity as assessed pre-BL.
Acute concomitant alterations in locomotor performance were observed following chemical BL. A general slowdown of locomotion was accompanied by a pathognomonic increase in the spatiotemporal variability of the stepping pattern and a broadened base of support that are both characteristic for a sensory-ataxic gait disorder [
35]. Increased spatiotemporal stepping variability likely directly reflects the acute loss of vestibular feedback, which is thought to fine-tune and adapt the spatiotemporal stepping pattern to unintended irregularities occurring during locomotion [
36,
37]. In contrast, the broadening of the stepping pattern likely represents a secondary consequence of impaired regulation of balance when the body is moving forward. Both gait impairments were considerably more pronounced in the hindlimb stepping pattern, indicating that vestibulo-spinal regulation of balance during quadruped locomotion predominantly governs hindlimb behavior [
38,
39]. While locomotor impairments steadily improved during the studied recovery period following surgery, moderate signs of gait ataxia at day 60 post-BL indicate a persistent and not fully compensated locomotor deficit.
4.2. Vestibular noise stimulation effects on brain activity and locomotor performance
Low-intensity vestibular noise stimulation is thought to augment residual vestibular function in BVL via SR. We studied the effects of low-intensity nGVS on brain activation patterns on 6 consecutive days spanning a 60-day period post-BL. Although our experimental setup did not allow for a parallel vestibular stimulation and [
18F]-FDG-PET acquisition, consistent stimulation-induced brain activation patterns were still observed during the 30 min period of tracer uptake following the cessation of stimulation. Prolonged 30 min treatment with nGVS yielded a broad impact on neuronal activity in brainstem-cerebellar, limbic, hemispheric motor and sensory networks that was more or less consistent throughout the studied period of post-BL recovery and largely resembled GVS-induced whole-brain activation patterns previously studied in rats using fMRI [
40]. Interestingly, stimulation-induced alterations of brain activity predominantly reversed those observed during the natural course of post-BL adaption. Most prominently, low-intensity nGVS yielded an increase of rGCM within the motor cortex and frontal-basal ganglia loops. This observation is in line with previous reports demonstrating that peripheral vestibular stimulation induces broad neuro-chemical changes at different sites within the basal ganglia [
9,
10,
41,
42]. It may further provide a neurophysiological substrate for the observation that treatment with low-intensity nGVS can ameliorate postural symptoms in basal ganglia disorders like Parkinson's disease [
26,
27].
Attenuating effects of nGVS on brain activity were observed within brainstem-cerebellar networks. In particular, in the cerebellum, which is known to contribute to vestibular compensation [
21] and exhibited increased activity levels during post-BL recovery, stimulation resulted in lowered levels of activity. This observation could indicate that partial restoration of vestibular input by nGVS might interfere with and interrupt compensatory cerebellar-mediated processes linked to recovery post-BL. Surprisingly, we further observed an nGVS-induced attenuation of neuronal activity at primary and secondary hubs of central vestibular and auditory processing (i.e., vestibular nucleus and colliculus inferior) that are known to become considerably modulated by GVS [
43]. However, GVS-induced responses within brainstem-cerebellar networks have been shown to rapidly habituate and become inhibited over time, which could explain the relative deactivation following prolonged 30 min treatment with nGVS [
43,
44].
In parallel to [
18F]-FDG-PET analysis of nGVS effects on brain activity, we studied the instantaneous impact of stimulation on sensory gait ataxia on 3 consecutive days spanning a 45-day period of post-BL recovery. Treatment with nGVS in particular improved sensory-ataxic gait alterations, in terms of a narrower base of support and reduced levels of spatiotemporal stepping variability. These stimulation effects on animals' locomotor pattern broadly resemble previously reported treatment effects of nGVS on locomotor impairments in patients with BVL [
13,
14,
15]. In accordance with these reports, nGVS-induced improvements of locomotor function were only present for a short interval following cessation of stimulation and did not persist over time. Furthermore, nGVS only impacted locomotion at early stages of post-BL recovery but not at later stages where sensory-ataxic gait deficits had to a large extent receded.
nGVS has been shown to facilitate vestibulo-spinal balance responses in healthy humans and patients with BVL [
45,
46], which may result in the observed stabilization of locomotor performance in ataxic animals post-BL. In line with this assumption, nGVS-induced responses in the vestibular nuclei, which relay motor commands through the vestibulo-spinal tract, were found to be correlated with decreased spatial stepping variability under treatment with nGVS. Beyond improvements on the vestibulo-spinal reflex level, nGVS might also contribute to an attenuation of sensory-ataxic gait deficits via modulation of higher brain network activity. Such a link is indicated by correlations found between decreased temporal stepping variability and narrower base of support with nGVS-induced responses in the cingulate, retrosplenial, and motor cortex – areas that have been broadly associated with general motor and locomotor control [
28,
47,
48].
4.3. Conclusions
The findings of this study should be interpreted in light of certain preconditions. First, our experimental setup did not allow for a parallel vestibular stimulation and PET acquisition or locomotor assessment that were performed immediately after a prolonged treatment with nGVS. Hence, we were only able to study immediate after-effects of prolonged nGVS treatment on brain activity and gait ataxia. Our current data suggest that nGVS induces consistent short-term (i.e., <30 min) but not long-term (across intervention days) stimulation after-effects on brain activity and locomotor behavior. Evidence for the presence of such stimulation-induced after-effects on vestibular function is still controversial, with studies reporting prolonged (i.e., lasting for several hours) after-effects on postural stability [
25,
49] and studies that, in contrast, demonstrate a rapid disappearance of any stimulation effects after cessation of nGVS [
50,
51]. Nevertheless, the observed stimulation after-effects on brain activation and on locomotion closely resemble those observed previously in rodents or humans during active GVS [
14,
15,
40]. Furthermore, in order to robustly test for the presence of nGVS-induced stochastic resonance, previous studies suggested that nGVS effects should be examined across a broad range of stimulation intensities [
27,
52,
53], which was not achievable with our current experimental setup. However, the fixed nGVS intensity applied here was close to the average reported optimal intensity from trans-mastoidal nGVS in humans [
12,
27,
52,
54] and the efficacy of stimulation at this intensity is suggested by the close resemblance to stimulation effects previously reported in humans [
14,
15].
In conclusion, using a rodent model of BVL, we observed that low-intensity vestibular noise stimulation modulates brain activity encompassing networks of lower and higher vestibular information processing. This stimulation-induced modulation broadly reverses brain activity adaptions that occur during the natural course of post-BL recovery. Furthermore, stimulation effects on hubs of lower and higher central vestibular information processing are linked to improvements in sensory-ataxic locomotor deficits that closely mimic those previously reported in patients with BVL. Hence, the current findings shed light on the potential neurophysiological and neuroanatomical substrates that mediate previously reported treatment effects of low-intensity vestibular noise stimulation on vestibular perceptual and sensorimotor function in patients with chronic vestibular loss.
Supplementary Materials
Supplementary Table S1.
Author Contributions
Conceptualization, A.Z.; methodology, M.W., E.E., M.G., R.B., S.Z. and A.Z.; software, M.W., E.E., R.B. and M.G.; validation, E.E., R.B., S.Z.; formal analysis, M.W. E.E., M.L. and M.G.; investigation, E.E., M.L. and R.B.; resources, M.W., S.Z. and A.Z.; data curation, M.W., E.E., R.B. and M.G.; writing – original draft preparation, M.W. and A.Z.; writing – review and editing, E.E., M.L., M.G., R.B. and S.Z.; visualization, M.W. and M.G.; supervision, S.Z. and A.Z.; projection administration A.Z.; funding acquisition, A.Z.; All authors have read and agreed to the published version of the manuscript.
Funding
The study was supported by the German Federal Ministry for Education and Science (BMBF, IFB 01EO1401).
Institutional Review Board Statement
All rodent experiments were approved by the government of Upper Bavaria and performed in accordance with the guidelines for the use of living animals in scientific studies and the German Law for Protection of Animals (ROB-55.2-2532.Vet_02-21-32).
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets used and/or analysed during the current study will be available from the corresponding author on reasonable request.
Acknowledgments
We thank Katie Göttlinger for copyediting.
Conflicts of Interest
The authors declare no conflict of interest related to the current study.
References
- Gimmon, Y.; Migliaccio, A.A.; Kim, K.J.; Schubert, M.C. VOR adaptation training and retention in a patient with profound bilateral vestibular hypofunction. Laryngoscope 2019, 129, 2568–2573. [Google Scholar] [CrossRef] [PubMed]
- Sulway, S.; Whitney, S.L. Advances in Vestibular Rehabilitation. Adv Otorhinolaryngol 2019, 82, 164–169. [Google Scholar] [PubMed]
- van de Berg, R.; Ramos, A.; van Rompaey, V.; et al. The vestibular implant: Opinion statement on implantation criteria for research. J Vestib Res 2020, 30, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Kingma, H.; Felipe, L.; Gerards, M.C.; et al. Vibrotactile feedback improves balance and mobility in patients with severe bilateral vestibular loss. J Neurol 2019, 266, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Zingler, V.C.; Weintz, E.; Jahn, K.; et al. Causative factors, epidemiology, and follow-up of bilateral vestibulopathy. Ann N Y Acad Sci 2009, 1164, 505–508. [Google Scholar] [CrossRef]
- van Stiphout, L.; Pleshkov, M.; Lucieer, F.; et al. Patterns of Vestibular Impairment in Bilateral Vestibulopathy and Its Relation to Etiology. Front Neurol 2022, 13. [Google Scholar] [CrossRef]
- Collins, J.; Chow, C.C.; Imhoff, T.T. Stochastic resonance without tuning. Nature 1995, 376, 236–238. [Google Scholar] [CrossRef]
- McDonnell, M.D.; Ward, L.M. The benefits of noise in neural systems: Bridging theory and experiment. Nat Rev Neurosci 2011, 12, 415–426. [Google Scholar] [CrossRef] [PubMed]
- Samoudi, G.; Nissbrandt, H.; Dutia, M.B.; Bergquist, F. Noisy galvanic vestibular stimulation promotes GABA release in the substantia nigra and improves locomotion in hemiparkinsonian rats. PLoS ONE 2012, 7, e29308. [Google Scholar] [CrossRef]
- Samoudi, G.; Nilsson, A.; Carlsson, T.; Bergquist, F. c-Fos Expression after Stochastic Vestibular Stimulation and Levodopa in 6-OHDA Hemilesioned Rats. Neuroscience 2020, 424, 146–154. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Nam, G.S.; Han, G.C.; Le, C.; Oh, S.Y. The Effect of Galvanic Vestibular Stimulation on Visuospatial Cognition in an Incomplete Bilateral Vestibular Deafferentation Mouse Model. Front Neurol 2022, 13, 857736. [Google Scholar] [CrossRef]
- Iwasaki, S.; Yamamoto, Y.; Togo, F.; et al. Noisy vestibular stimulation improves body balance in bilateral vestibulopathy. Neurology 2014, 82, 969–975. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, S.; Fujimoto, C.; Egami, N.; et al. Noisy vestibular stimulation increases gait speed in normals and in bilateral vestibulopathy. Brain Stimul 2018, 11, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Wuehr, M.; Nusser, E.; Decker, J.; et al. Noisy vestibular stimulation improves dynamic walking stability in bilateral vestibulopathy. Neurology 2016, 86, 2196–2202. [Google Scholar] [CrossRef]
- Wuehr, M.; Nusser, E.; Krafczyk, S.; et al. Noise-Enhanced Vestibular Input Improves Dynamic Walking Stability in Healthy Subjects. Brain Stimul 2016, 9, 109–116. [Google Scholar] [CrossRef]
- Eder, J.; Kellerer, S.; Amberger, T.; et al. Combining vestibular rehabilitation with noisy galvanic vestibular stimulation for treatment of bilateral vestibulopathy. J Neurol 2022, 269, 5731–5737. [Google Scholar] [CrossRef]
- Anniko, M.; Wersäll, J. Experimentally (atoxyl) induced ampullar degeneration and damage to the maculae utriculi. Acta Otolaryngol 1977, 83, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Beck, R.; Günther, L.; Xiong, G.; et al. The mixed blessing of treating symptoms in acute vestibular failure--evidence from a 4-aminopyridine experiment. Exp Neurol 2014, 261, 638–645. [Google Scholar] [CrossRef] [PubMed]
- Vignaux, G.; Chabbert, C.; Gaboyard-Niay, S.; et al. Evaluation of the chemical model of vestibular lesions induced by arsanilate in rats. Toxicol Appl Pharmacol 2012, 258, 61–71. [Google Scholar] [CrossRef]
- Grosch, M.; Lindner, M.; Bartenstein, P.; et al. Dynamic whole-brain metabolic connectivity during vestibular compensation in the rat. Neuroimage 2021, 226, 117588. [Google Scholar] [CrossRef] [PubMed]
- Zwergal, A.; Schlichtiger, J.; Xiong, G.; et al. Sequential [(18)F]FDG microPET whole-brain imaging of central vestibular compensation: A model of deafferentation-induced brain plasticity. Brain Struct Funct 2016, 221, 159–170. [Google Scholar] [CrossRef]
- Schiffer, W.K.; Mirrione, M.M.; Biegon, A.; Alexoff, D.L.; Patel, V.; Dewey, S.L. Serial microPET measures of the metabolic reaction to a microdialysis probe implant. J Neurosci Methods 2006, 155, 272–284. [Google Scholar] [CrossRef] [PubMed]
- Herold, S.; Kumar, P.; Jung, K.; et al. CatWalk gait analysis in a rat model of multiple sclerosis. BMC Neurosci 2016, 17, 78. [Google Scholar] [CrossRef] [PubMed]
- Schniepp, R.; Schlick, C.; Schenkel, F.; et al. Clinical and neurophysiological risk factors for falls in patients with bilateral vestibulopathy. J Neurol 2017, 264, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, C.; Egami, N.; Kawahara, T.; et al. Noisy Galvanic Vestibular Stimulation Sustainably Improves Posture in Bilateral Vestibulopathy. Front Neurol 2018, 9, 900. [Google Scholar] [CrossRef] [PubMed]
- Samoudi, G.; Jivegard, M.; Mulavara, A.P.; Bergquist, F. Effects of Stochastic Vestibular Galvanic Stimulation and LDOPA on Balance and Motor Symptoms in Patients With Parkinson's Disease. Brain Stimul 2015, 8, 474–480. [Google Scholar] [CrossRef]
- Wuehr, M.; Schmidmeier, F.; Katzdobler, S.; Fietzek, U.M.; Levin, J.; Zwergal, A. Effects of Low-Intensity Vestibular Noise Stimulation on Postural Instability in Patients with Parkinson’s Disease. J Parkinsons Dis 2022, 12, 1611–1618. [Google Scholar] [CrossRef]
- Lee, S.; Kim, D.J.; Svenkeson, D.; Parras, G.; Oishi, M.M.; McKeown, M.J. Multifaceted effects of noisy galvanic vestibular stimulation on manual tracking behavior in Parkinson's disease. Front Syst Neurosci 2015, 9, 5. [Google Scholar] [CrossRef] [PubMed]
- Tran, S.; Shafiee, M.; Jones, C.B.; et al. Subthreshold stochastic vestibular stimulation induces complex multi-planar effects during standing in Parkinson's disease. Brain Stimul 2018, 11, 1180–1182. [Google Scholar] [CrossRef]
- Antons, M.; Lindner, M.; Grosch, M.; et al. Longitudinal [(18)]UCB-H/[(18)F]FDG imaging depicts complex patterns of structural and functional neuroplasticity following bilateral vestibular loss in the rat. Sci Rep 2022, 12, 6049. [Google Scholar] [CrossRef] [PubMed]
- Wijesinghe, R.; Protti, D.A.; Camp, A.J. Vestibular Interactions in the Thalamus. Front Neural Circuits 2015, 9, 79. [Google Scholar] [CrossRef]
- Stiles, L.; Smith, P.F. The vestibular-basal ganglia connection: Balancing motor control. Brain Res 2015, 1597, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Kreitzer, A.C.; Malenka, R.C. Striatal plasticity and basal ganglia circuit function. Neuron 2008, 60, 543–554. [Google Scholar] [CrossRef] [PubMed]
- Goddard, M.; Zheng, Y.; Darlington, C.L.; Smith, P.F. Locomotor and exploratory behavior in the rat following bilateral vestibular deafferentation. Behav Neurosci 2008, 122, 448–459. [Google Scholar] [CrossRef] [PubMed]
- Schniepp, R.; Mohwald, K.; Wuehr, M. Gait ataxia in humans: Vestibular and cerebellar control of dynamic stability. J Neurol 2017, 264, 87–92. [Google Scholar] [CrossRef]
- Dietrich, H.; Heidger, F.; Schniepp, R.; MacNeilage, P.R.; Glasauer, S.; Wuehr, M. Head motion predictability explains activity-dependent suppression of vestibular balance control. Sci Rep 2020, 10, 668. [Google Scholar] [CrossRef]
- Jahn, K.; Wuehr, M. Postural Control Mechanisms in Mammals, Including Humans. In Fritzsch, B.; ed. The Senses: A Comprehensive Reference (Second Edition). Oxford: Elsevier, 2020. [Google Scholar]
- Witts, E.C.; Murray, A.J. Vestibulospinal contributions to mammalian locomotion. Curr Opin Physiol 2019, 8, 56–62. [Google Scholar] [CrossRef]
- Murray, A.J.; Croce, K.; Belton, T.; Akay, T.; Jessell, T.M. Balance Control Mediated by Vestibular Circuits Directing Limb Extension or Antagonist Muscle Co-activation. Cell Rep 2018, 22, 1325–1338. [Google Scholar] [CrossRef] [PubMed]
- Rancz, E.A.; Moya, J.; Drawitsch, F.; Brichta, A.M.; Canals, S.; Margrie, T.W. Widespread vestibular activation of the rodent cortex. J Neurosci 2015, 35, 5926–5934. [Google Scholar] [CrossRef]
- Stiles, L.; Reynolds, J.N.; Napper, R.; Zheng, Y.; Smith, P.F. Single neuron activity and c-Fos expression in the rat striatum following electrical stimulation of the peripheral vestibular system. Physiol Rep 2018, 6, e13791. [Google Scholar] [CrossRef] [PubMed]
- Stiles, L.; Zheng, Y.; Smith, P.F. The effects of electrical stimulation of the peripheral vestibular system on neurochemical release in the rat striatum. PLoS ONE 2018, 13, e0205869. [Google Scholar] [CrossRef]
- Dlugaiczyk, J.; Wuehr, M.; Straka, H. Electrical Stimulation of Vestibular Endorgans. In Fritzsch, B.; ed. The Senses: A Comprehensive Reference (Second Edition). Oxford: Elsevier, 2020. [Google Scholar]
- Courjon, J.H.; Precht, W.; Sirkin, D.W. Vestibular nerve and nuclei unit responses and eye movement responses to repetitive galvanic stimulation of the labyrinth in the rat. Exp Brain Res 1987, 66, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Wuehr, M.; Boerner, J.C.; Pradhan, C.; et al. Stochastic resonance in the human vestibular system - Noise-induced facilitation of vestibulospinal reflexes. Brain Stimul 2018, 11, 261–263. [Google Scholar] [CrossRef]
- Schniepp, R.; Boerner, J.C.; Decker, J.; Jahn, K.; Brandt, T.; Wuehr, M. Noisy vestibular stimulation improves vestibulospinal function in patients with bilateral vestibulopathy. J Neurol 2018, 265, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Devinsky, O.; Morrell, M.J.; Vogt, B.A. Contributions of anterior cingulate cortex to behaviour. Brain 1995, 118, 279–306. [Google Scholar] [CrossRef]
- Cho, J.; Sharp, P.E. Head direction, place, and movement correlates for cells in the rat retrosplenial cortex. Behav Neurosci 2001, 115, 3–25. [Google Scholar] [CrossRef]
- Fujimoto, C.; Yamamoto, Y.; Kamogashira, T.; et al. Noisy galvanic vestibular stimulation induces a sustained improvement in body balance in elderly adults. Sci Rep 2016, 6, 37575. [Google Scholar] [CrossRef] [PubMed]
- Keywan, A.; Badarna, H.; Jahn, K.; Wuehr, M. No evidence for after-effects of noisy galvanic vestibular stimulation on motion perception. Sci Rep 2020, 10, 2545. [Google Scholar] [CrossRef]
- Nooristani, M.; Maheu, M.; Houde, M.-S.; Bacon, B.-A.; Champoux, F. Questioning the lasting effect of galvanic vestibular stimulation on postural control. PLoS ONE 2019, 14, e0224619–e19. [Google Scholar] [CrossRef] [PubMed]
- Galvan-Garza, R.C.; Clark, T.K.; Mulavara, A.P.; Oman, C.M. Exhibition of stochastic resonance in vestibular tilt motion perception. Brain Stimul 2018, 11, 716–722. [Google Scholar] [CrossRef]
- Assländer, L.; Giboin, L.S.; Gruber, M.; Schniepp, R.; Wuehr, M. No evidence for stochastic resonance effects on standing balance when applying noisy galvanic vestibular stimulation in young healthy adults. Sci Rep 2021, 11, 12327. [Google Scholar] [CrossRef] [PubMed]
- Wuehr, M.; Decker, J.; Schniepp, R. Noisy galvanic vestibular stimulation: An emerging treatment option for bilateral vestibulopathy. J Neurol 2017, 264, 81–86. [Google Scholar] [CrossRef] [PubMed]
Figure 1.
Experimental protocol and processing of brain scans: (A) [18F]-FDG-PET and locomotion analysis were performed before (baseline) and post bilateral labyrinthectomy (BL) on 9 sequential days encompassing a total period of 60 days. A 30min long treatment with noisy galvanic vestibular stimulation at 0.2 mA (nGVS; N = 8) vs. sham (N = 9) was applied on each assessment day post-BL. (B) On days 1, 3, 7, 15, 30, and 60 post-BL, immediate stimulation effects on regional brain activation were tested by whole-brain [18F]-FDG-PET imaging directly following nGVS treatment. A concomitant locomotion analysis before nGVS treatment was performed on each of these days to assess potential long-term effects of stimulation. (C) Immediate effects of nGVS on locomotion performance were assessed on days 6, 21, and 45 post-BL. (B – D) Post-processing of PET serial scans: Images were acquired, reconstructed, cropped, registered, filtered, normalized, and segmented into 26 brain regions. Subsequently, activity levels for each brain region normalized to the whole brain were obtained.
Figure 1.
Experimental protocol and processing of brain scans: (A) [18F]-FDG-PET and locomotion analysis were performed before (baseline) and post bilateral labyrinthectomy (BL) on 9 sequential days encompassing a total period of 60 days. A 30min long treatment with noisy galvanic vestibular stimulation at 0.2 mA (nGVS; N = 8) vs. sham (N = 9) was applied on each assessment day post-BL. (B) On days 1, 3, 7, 15, 30, and 60 post-BL, immediate stimulation effects on regional brain activation were tested by whole-brain [18F]-FDG-PET imaging directly following nGVS treatment. A concomitant locomotion analysis before nGVS treatment was performed on each of these days to assess potential long-term effects of stimulation. (C) Immediate effects of nGVS on locomotion performance were assessed on days 6, 21, and 45 post-BL. (B – D) Post-processing of PET serial scans: Images were acquired, reconstructed, cropped, registered, filtered, normalized, and segmented into 26 brain regions. Subsequently, activity levels for each brain region normalized to the whole brain were obtained.
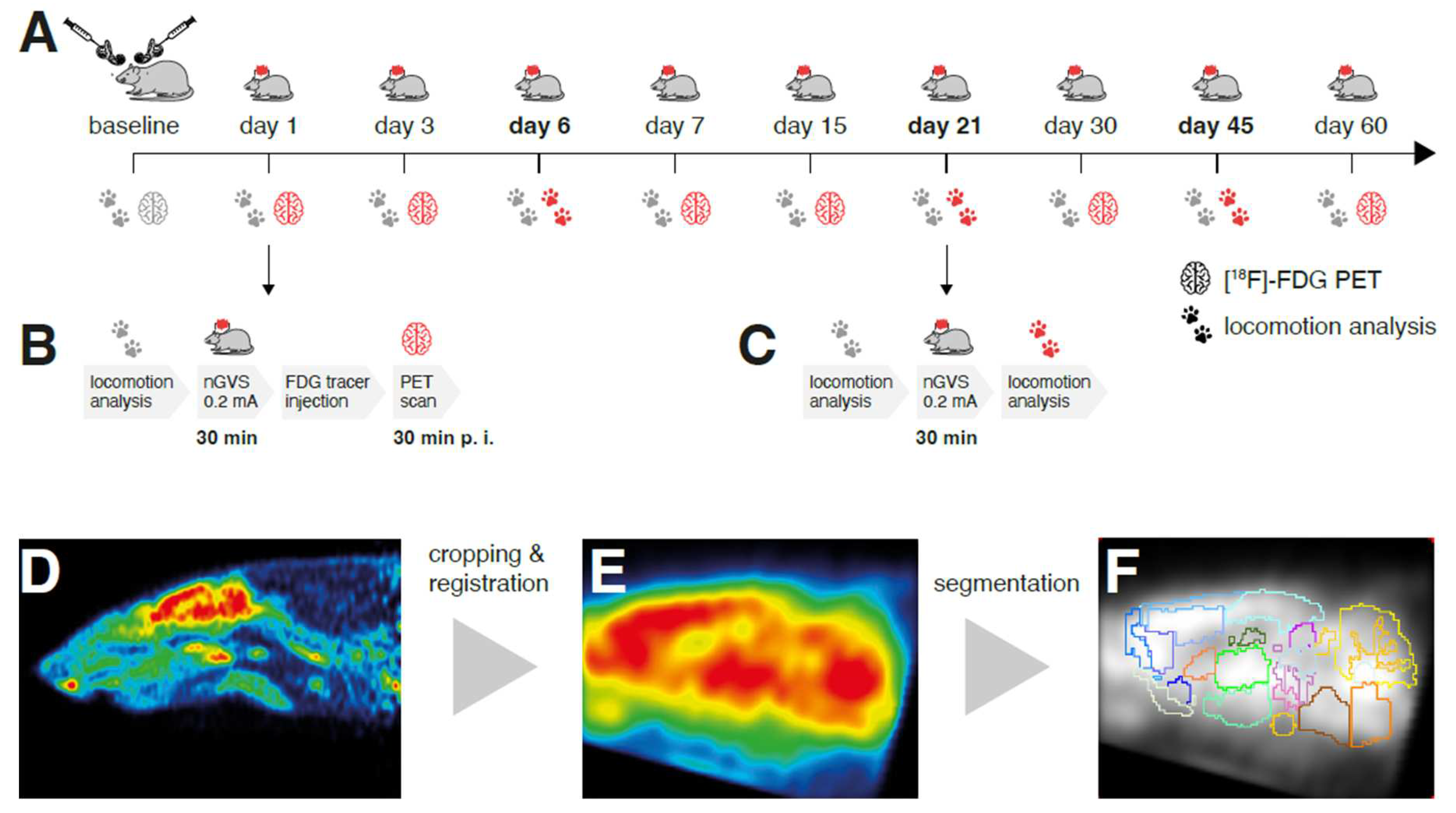
Figure 2.
Changes in regional brain activity in response to bilateral labyrinthectomy and low-intensity vestibular noise stimulation. Overview of changes in mean normalized levels of brain activity in 26 selected brain regions measured by [18F]-FDG-PET. Significant increases in activity are visualized in red (light red: p<0.05; medium red: p<0.01; dark red: p<0.001) and decreases in activity in blue (light blue: p<0.05; medium blue: p<0.01; dark blue: p<0.001). First column: instantaneous changes in response to bilateral labyrinthectomy (baseline vs. day 1); second column: changes within the course of recovery (day 1-60 post bilateral labyrinthectomy); third column: changes in response to low-intensity vestibular noise stimulation (nGVS vs. sham); fourth to ninth columns: time-dependent changes in response to vestibular noise stimulation, i.e., decomposition of significant interaction effects between recovery time and treatment. Abbreviations: nGVS: noisy galvanic vestibular stimulation; vest: vestibular; c: cortex; ad: anterior-dorsal; p: posterior; v: ventral.
Figure 2.
Changes in regional brain activity in response to bilateral labyrinthectomy and low-intensity vestibular noise stimulation. Overview of changes in mean normalized levels of brain activity in 26 selected brain regions measured by [18F]-FDG-PET. Significant increases in activity are visualized in red (light red: p<0.05; medium red: p<0.01; dark red: p<0.001) and decreases in activity in blue (light blue: p<0.05; medium blue: p<0.01; dark blue: p<0.001). First column: instantaneous changes in response to bilateral labyrinthectomy (baseline vs. day 1); second column: changes within the course of recovery (day 1-60 post bilateral labyrinthectomy); third column: changes in response to low-intensity vestibular noise stimulation (nGVS vs. sham); fourth to ninth columns: time-dependent changes in response to vestibular noise stimulation, i.e., decomposition of significant interaction effects between recovery time and treatment. Abbreviations: nGVS: noisy galvanic vestibular stimulation; vest: vestibular; c: cortex; ad: anterior-dorsal; p: posterior; v: ventral.
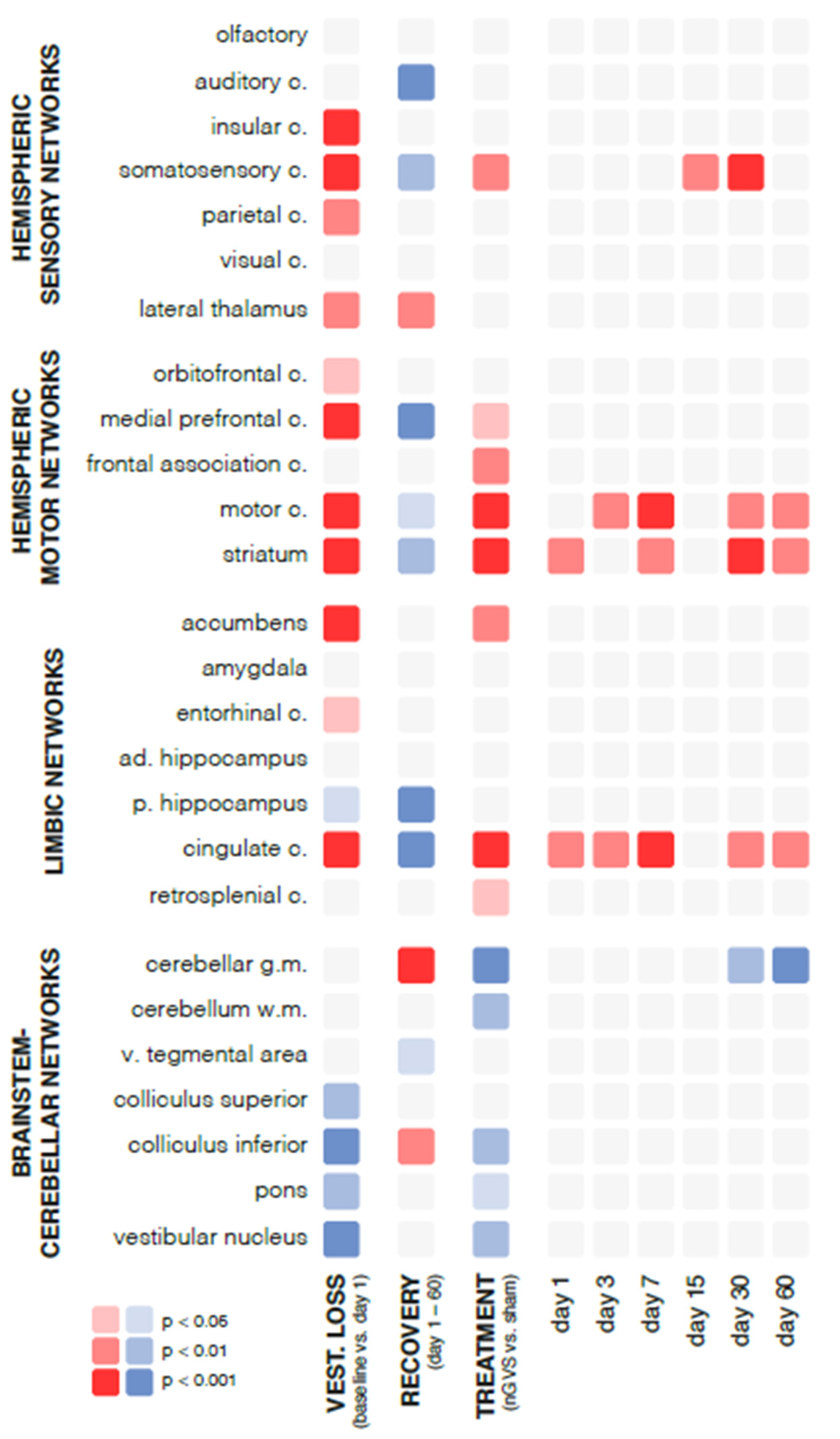
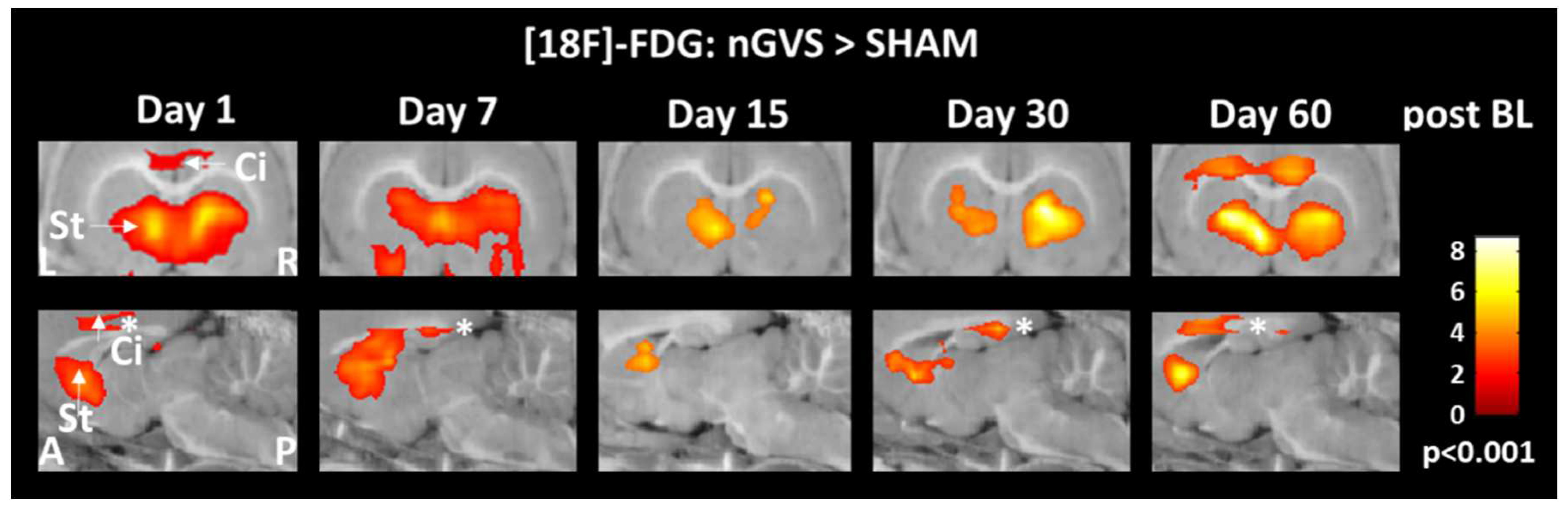
Figure 3.
Brain activity responses to low-intensity vestibular noise stimulation in hemispheric motor and limbic networks. Comparison of regional cerebral glucose metabolism in the cingulate cortex and striatum between animals receiving noisy galvanic vestibular stimulation (nGVS) vs. sham stimulation at days 1, 7, 15, 30, and 60 post BL. nGVS treatment results in increased activity in both brain regions on all intervention days. Abbreviations: BL: bilateral labyrinthectomy; Ci: cingulate cortex; St: striatum; L: left; R: right; A: anterior; P: posterior.
Figure 3.
Brain activity responses to low-intensity vestibular noise stimulation in hemispheric motor and limbic networks. Comparison of regional cerebral glucose metabolism in the cingulate cortex and striatum between animals receiving noisy galvanic vestibular stimulation (nGVS) vs. sham stimulation at days 1, 7, 15, 30, and 60 post BL. nGVS treatment results in increased activity in both brain regions on all intervention days. Abbreviations: BL: bilateral labyrinthectomy; Ci: cingulate cortex; St: striatum; L: left; R: right; A: anterior; P: posterior.
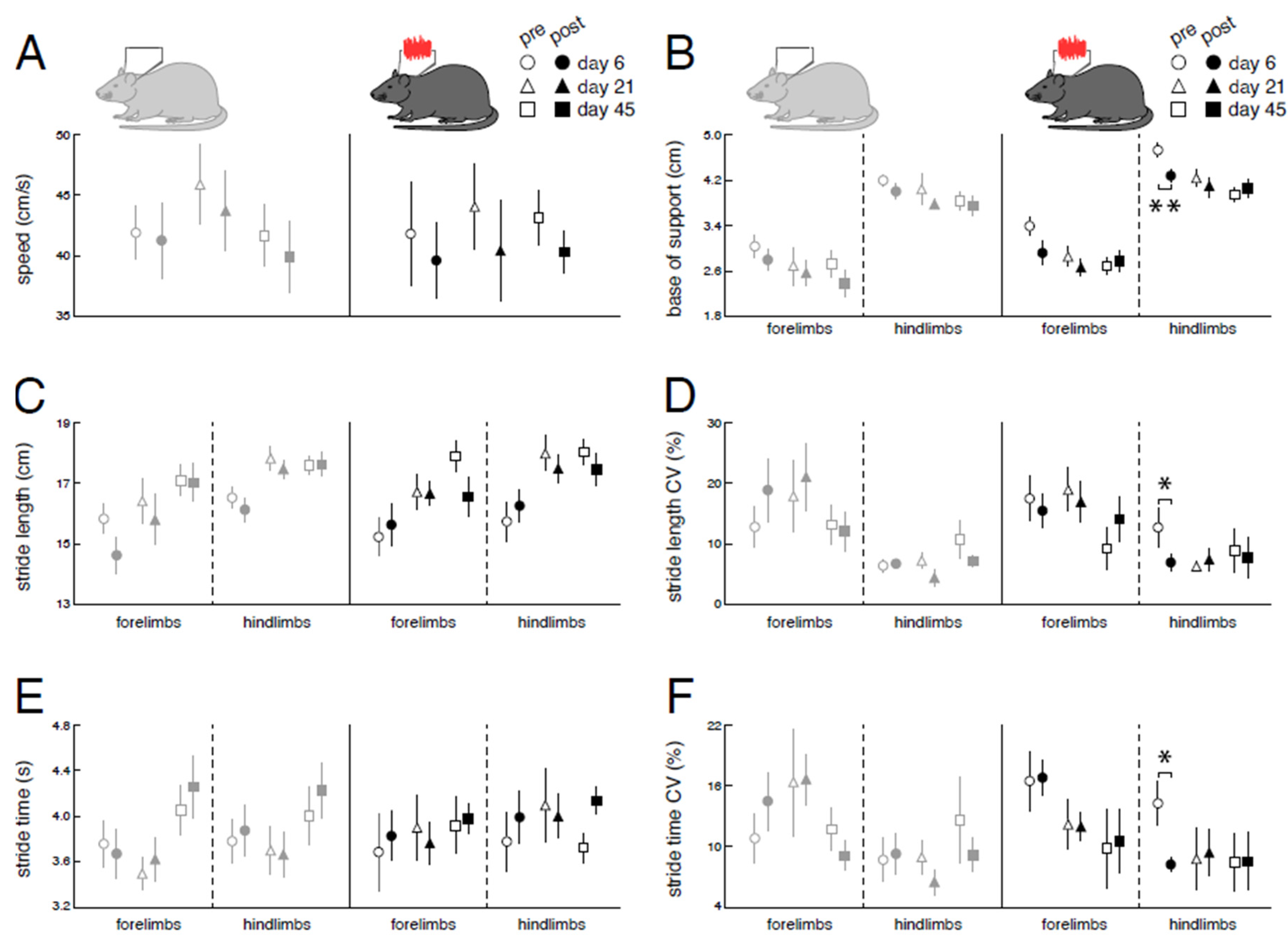
Figure 4.
Long-term influences of low-intensity vestibular noise stimulation on locomotor recovery following bilateral labyrinthectomy. Comparison of changes in locomotor performance within a period of 60 days following BL in the nGVS group (dark green and black circles) vs. the sham stimulation group (light green and grey circles). Spatiotemporal gait parameters (A – F) of locomotor performance assessed on days 1, 3, 7, 15, 30, and 60 post-BL. Locomotion analysis on these days was performed ahead of a 30 min long treatment with nGVS or sham stimulation. Compared to baseline, BL results in a slowdown of locomotion (A, C, E) with a broadened base of support and increased gait variability (i.e., CV; D, F). Locomotor alterations are more pronounced for hindlimbs. Following BL, impaired locomotion performance steadily recovers close to baseline levels. Repeated nGVS interventions only marginally influence the long-term course of locomotor recovery post-BL (D, left column). Abbreviations: nGVS: noisy galvanic vestibular stimulation; BL: bilateral labyrinthectomy; CV: coefficient of variation; * p < 0.05; ** p < 0.01; *** p < 0.001.
Figure 4.
Long-term influences of low-intensity vestibular noise stimulation on locomotor recovery following bilateral labyrinthectomy. Comparison of changes in locomotor performance within a period of 60 days following BL in the nGVS group (dark green and black circles) vs. the sham stimulation group (light green and grey circles). Spatiotemporal gait parameters (A – F) of locomotor performance assessed on days 1, 3, 7, 15, 30, and 60 post-BL. Locomotion analysis on these days was performed ahead of a 30 min long treatment with nGVS or sham stimulation. Compared to baseline, BL results in a slowdown of locomotion (A, C, E) with a broadened base of support and increased gait variability (i.e., CV; D, F). Locomotor alterations are more pronounced for hindlimbs. Following BL, impaired locomotion performance steadily recovers close to baseline levels. Repeated nGVS interventions only marginally influence the long-term course of locomotor recovery post-BL (D, left column). Abbreviations: nGVS: noisy galvanic vestibular stimulation; BL: bilateral labyrinthectomy; CV: coefficient of variation; * p < 0.05; ** p < 0.01; *** p < 0.001.
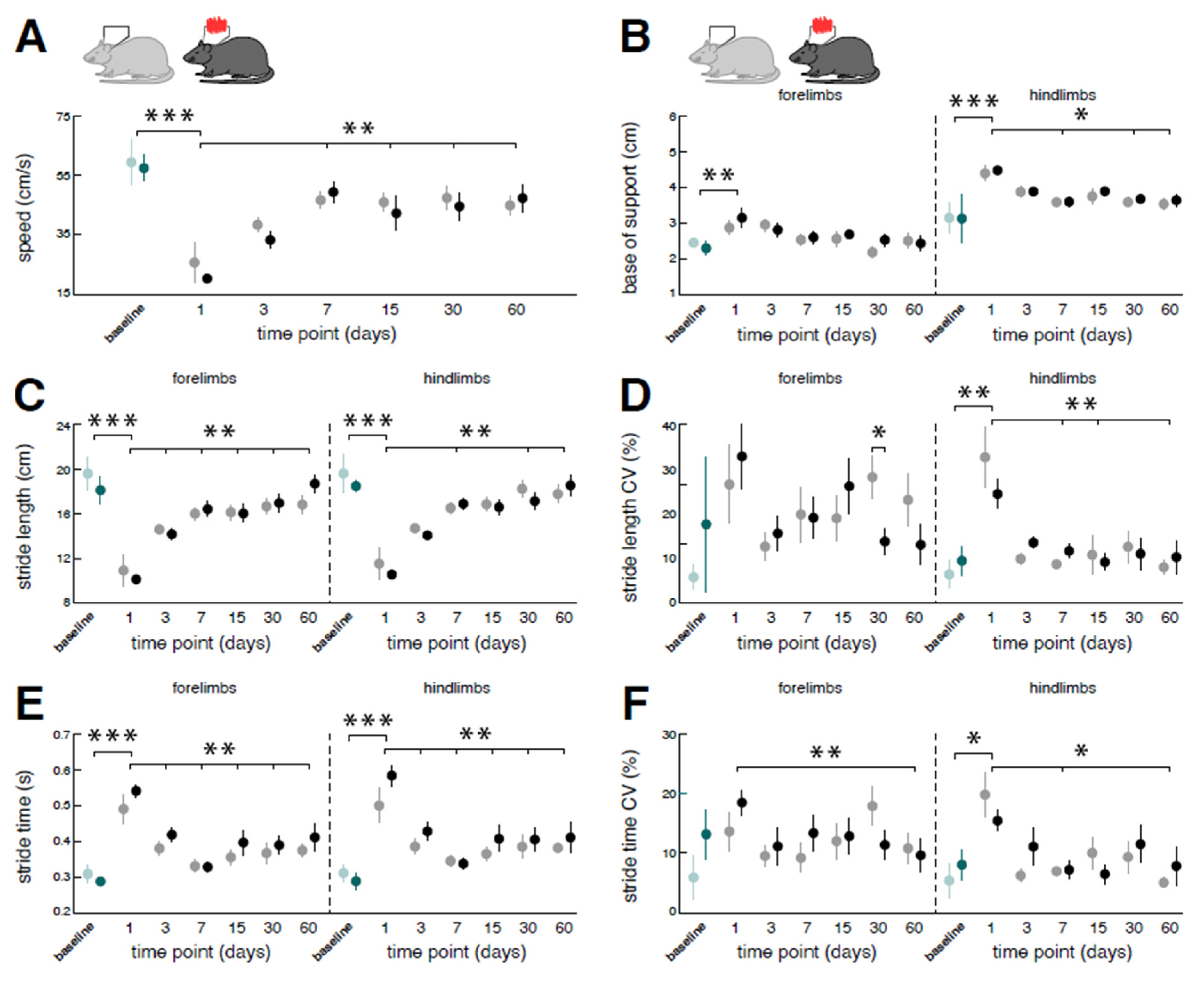
Figure 5.
Immediate effects of low-intensity vestibular noise on locomotor performance. Comparison of pre- (open symbols) and post-stimulation (filled symbols) performance with nGVS (N = 8; right column) vs. sham stimulation (N = 9; left column) on intervention days 6 (circles), 21 (triangles), and 45 (squares) on spatiotemporal gait parameters (A – F). nGVS alters locomotion in terms of a narrower base of support (B) and reduced variability (i.e., CV) of stride length (D) and stride time (F). nGVS effects on locomotion are restricted to hindlimb gait performance and only present during intervention day 6. Abbreviations: nGVS: noisy galvanic vestibular stimulation; CV: coefficient of variation; * p < 0.05; ** p < 0.01; *** p < 0.001.
Figure 5.
Immediate effects of low-intensity vestibular noise on locomotor performance. Comparison of pre- (open symbols) and post-stimulation (filled symbols) performance with nGVS (N = 8; right column) vs. sham stimulation (N = 9; left column) on intervention days 6 (circles), 21 (triangles), and 45 (squares) on spatiotemporal gait parameters (A – F). nGVS alters locomotion in terms of a narrower base of support (B) and reduced variability (i.e., CV) of stride length (D) and stride time (F). nGVS effects on locomotion are restricted to hindlimb gait performance and only present during intervention day 6. Abbreviations: nGVS: noisy galvanic vestibular stimulation; CV: coefficient of variation; * p < 0.05; ** p < 0.01; *** p < 0.001.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).