Submitted:
18 August 2023
Posted:
21 August 2023
You are already at the latest version
Abstract
Keywords:
Introduction
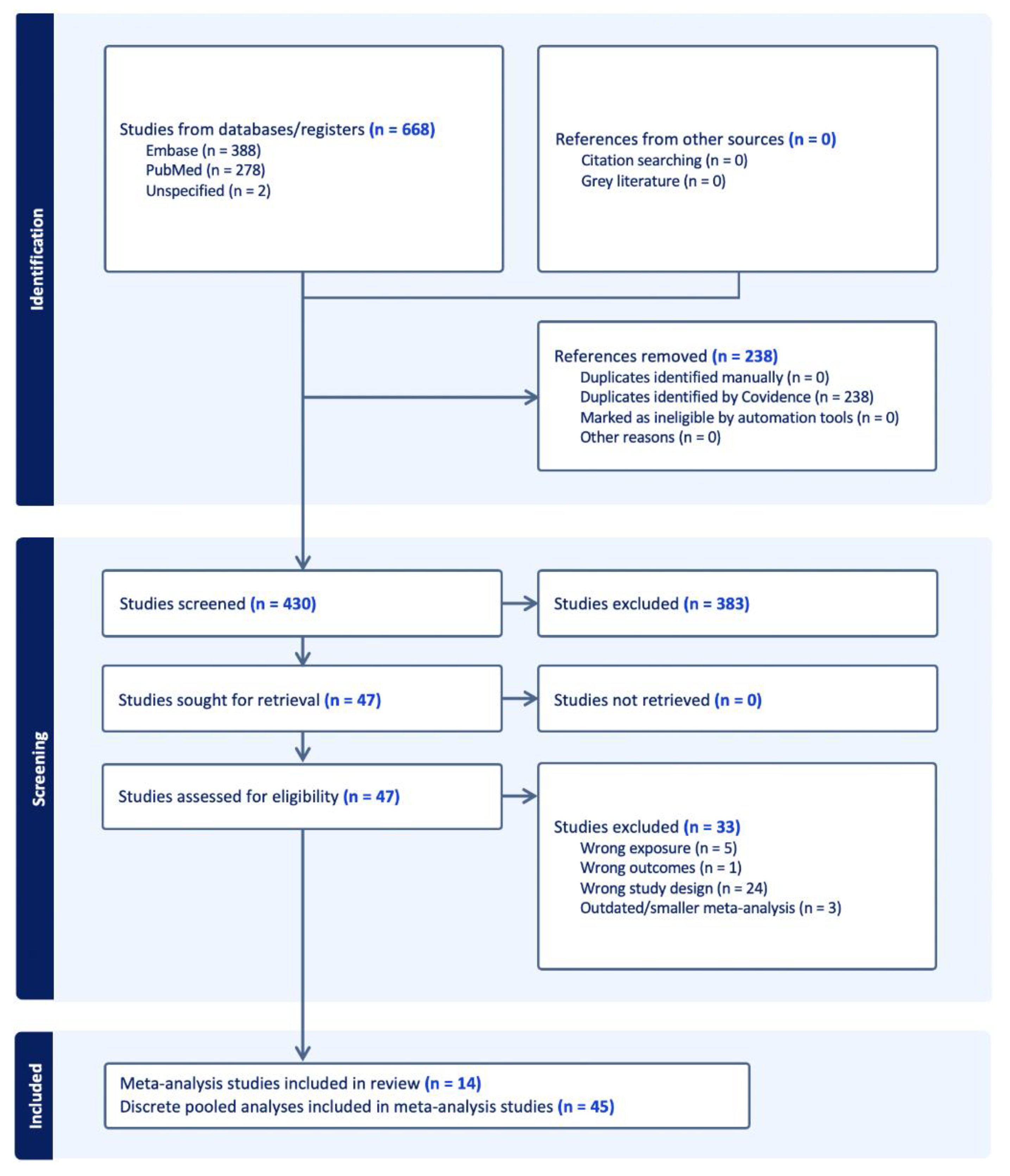
Methods
Inclusion Criteria and Searches
Data Extraction, Outcomes, and Data Synthesis
Data Analysis
Credibility Assessment of Each Pooled Analysis Assessing Associations between Ultra-Processed Food Exposure and Adverse Health Outcomes
Quality Assessment of Each Pooled Analysis Assessing Associations between Ultra-Processed Food Exposure and Adverse Health Outcomes
Quality Assessment of Individual Meta-Analysis Studies
Patient and Public Involvement
Results
Study Characteristics
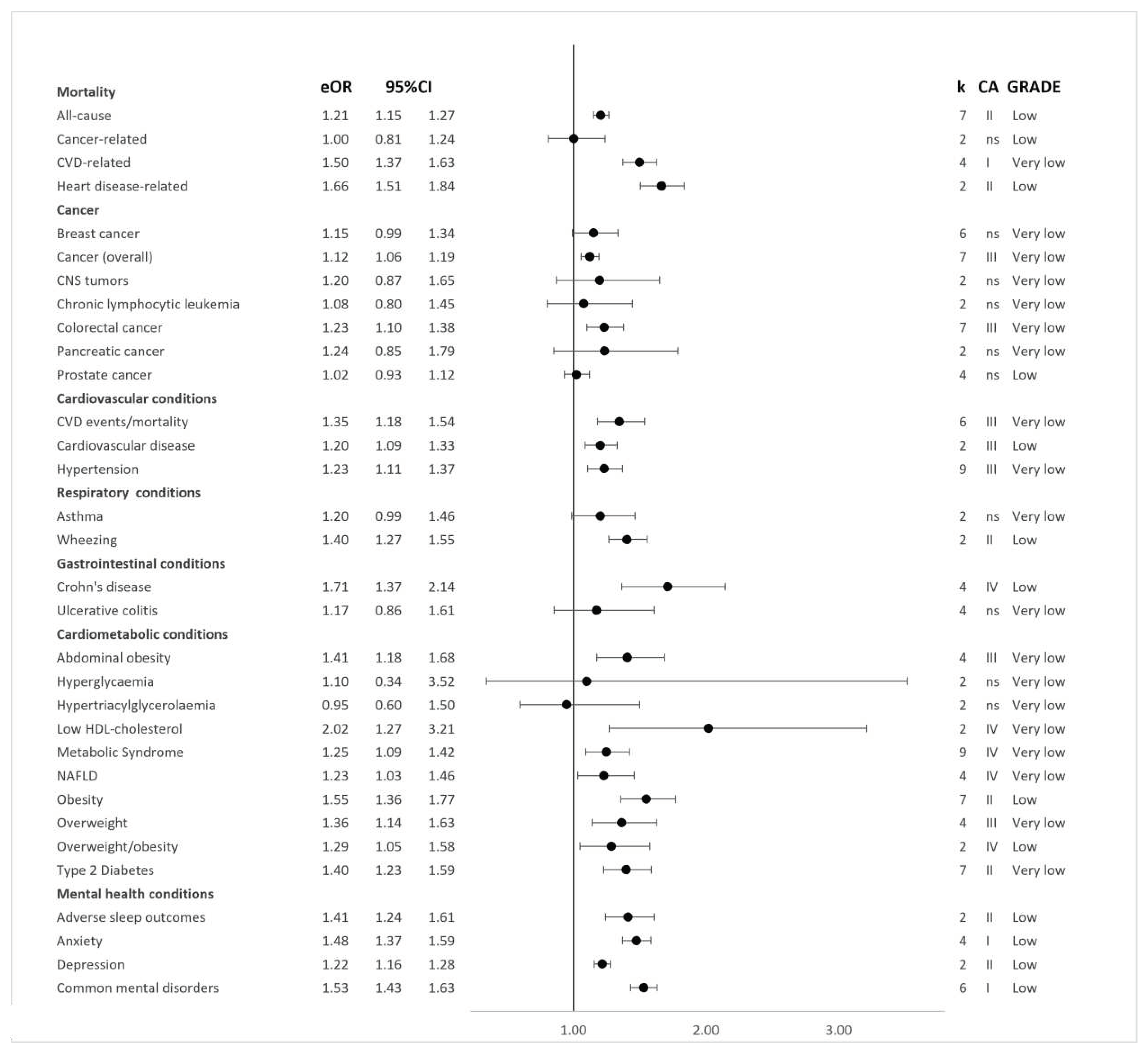
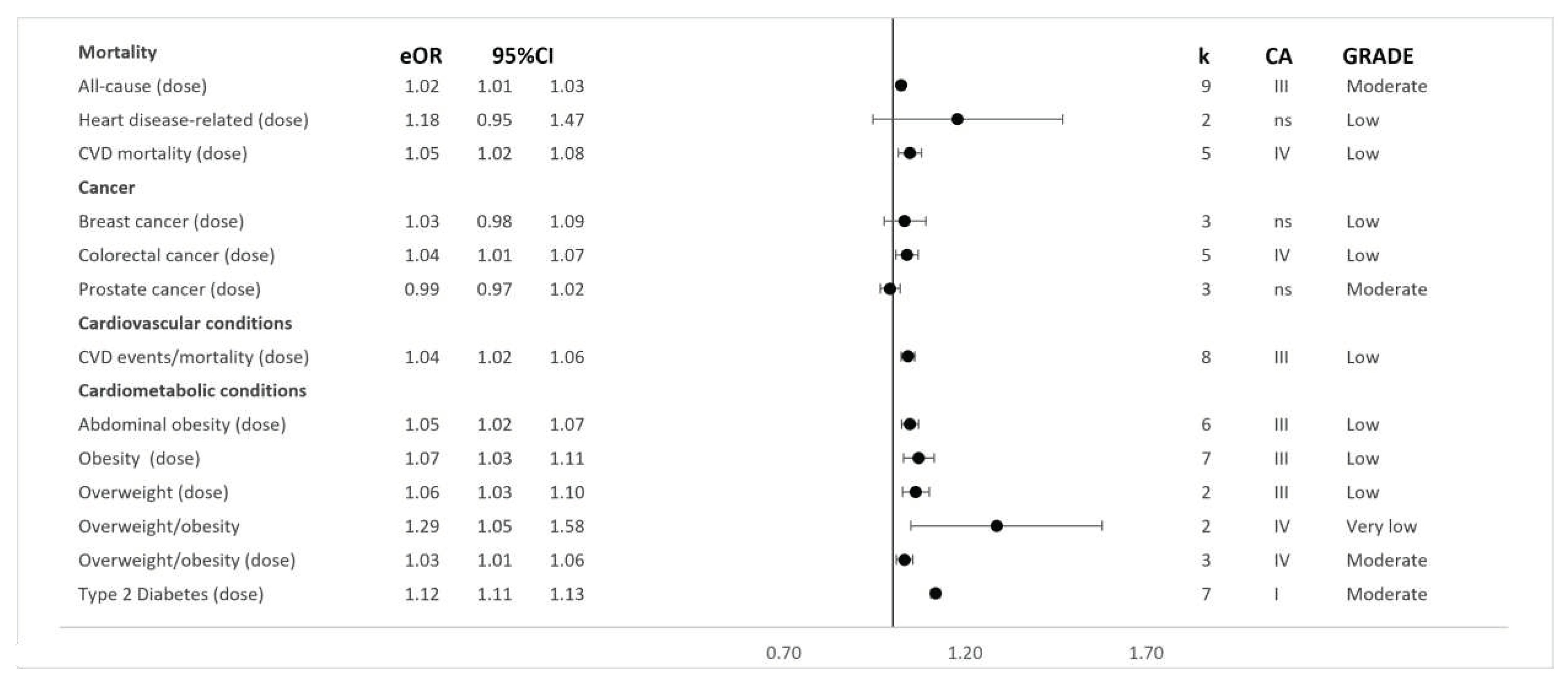
Results of syntheses
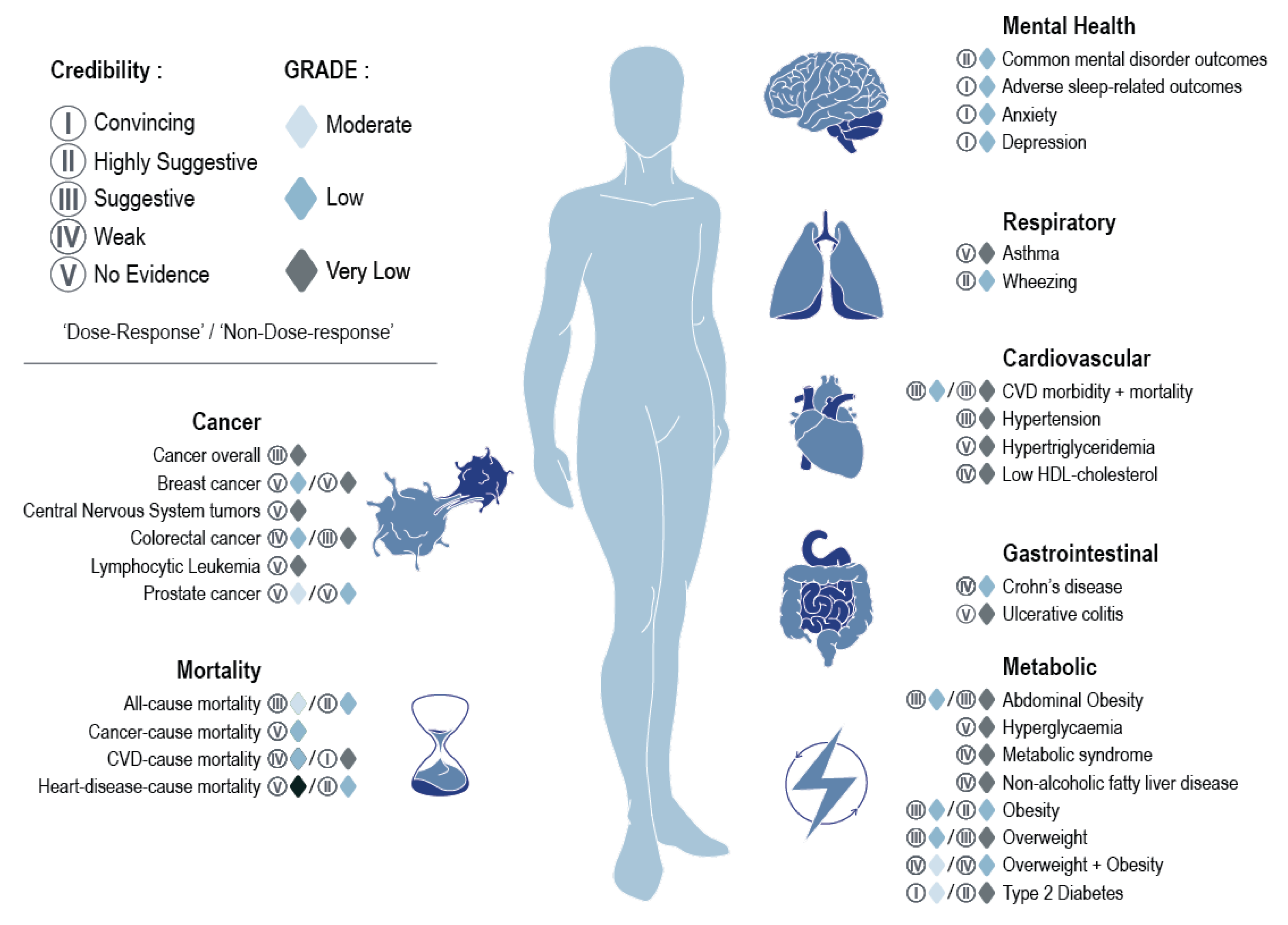
Credibility and Quality Assessments
Cancer
Cardiometabolic Conditions
Gastrointestinal Conditions
Mental Health
Mortality
Respiratory Conditions
Quality Assessment of Individual Meta-Analysis Studies Using the AMSTAR 2 Tool
Discussion
Statement of Principal Findings
Potential Mechanisms of Action
Strengths and Weaknesses in Relation to other Studies
Meaning of the Study: Possible Explanations and Implications for Clinicians and Policymakers
Conclusions and Recommendations
Supplementary Materials
Author Contributions
Funding
Patient and Public Involvement
Conflicts of Interest
References
- Monteiro, C.A.; Cannon, G.; Lawrence, M.; Costa Louzada, M.L.; Machado, P.P. Ultra-processed foods, diet quality, and health using the NOVA classification system; Food and Agriculture Organization of the United Nations (FAO): Rome, 2019. [Google Scholar]
- Monteiro, C.A.; Cannon, G.; Levy, R.B.; Moubarac, J.C.; Louzada, M.L.; Rauber, F.; Khandpur, N.; Cediel, G.; Neri, D.; Martinez-Steele, E.; et al. Ultra-processed foods: What they are and how to identify them. Public Health Nutr 2019, 22, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Baker, P.; Machado, P.; Santos, T.; Sievert, K.; Backholer, K.; Hadjikakou, M.; Russell, C.; Huse, O.; Bell, C.; Scrinis, G.; et al. Ultra-processed foods and the nutrition transition: Global, regional and national trends, food systems transformations and political economy drivers. Obesity Reviews 2020, 21, e13126. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, C.A.; Moubarac, J.C.; Cannon, G.; Ng, S.W.; Popkin, B. Ultra-processed products are becoming dominant in the global food system. Obes Rev 2013, 14 Suppl 2, 21–28. [Google Scholar] [CrossRef]
- Marino, M.; Puppo, F.; Del Bo’, C.; Vinelli, V.; Riso, P.; Porrini, M.; Martini, D. A Systematic Review of Worldwide Consumption of Ultra-Processed Foods: Findings and Criticisms. Nutrients 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Martini, D.; Godos, J.; Bonaccio, M.; Vitaglione, P.; Grosso, G. Ultra-Processed Foods and Nutritional Dietary Profile: A Meta-Analysis of Nationally Representative Samples. Nutrients 2021, 13. [Google Scholar] [CrossRef]
- Swinburn, B.A.; Sacks, G.; Hall, K.D.; McPherson, K.; Finegood, D.T.; Moodie, M.L.; Gortmaker, S.L. The global obesity pandemic: Shaped by global drivers and local environments. Lancet 2011, 378, 804–814. [Google Scholar] [CrossRef] [PubMed]
- Mendes, C.; Miranda, L.; Claro, R.; Horta, P. Food marketing in supermarket circulars in Brazil: An obstacle to healthy eating. Prev Med Rep 2021, 21, 101304. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Hawk, T.; Aggarwal, A.; Drewnowski, A. Characterizing Ultra-Processed Foods by Energy Density, Nutrient Density, and Cost. Front Nutr 2019, 6, 70. [Google Scholar] [CrossRef]
- Luiten, C.M.; Steenhuis, I.H.M.; Eyles, H.; Ni Mhurchu, C.; Waterlander, W.E. Ultra-processed foods have the worst nutrient profile, yet they are the most available packaged products in a sample of New Zealand supermarkets. Public Health Nutrition 2016, 19, 530–538. [Google Scholar] [CrossRef]
- Poti, J.M.; Braga, B.; Qin, B. Ultra-processed Food Intake and Obesity: What Really Matters for Health-Processing or Nutrient Content? Curr Obes Rep 2017, 6, 420–431. [Google Scholar] [CrossRef]
- Srour, B.; Kordahi, M.C.; Bonazzi, E.; Deschasaux-Tanguy, M.; Touvier, M.; Chassaing, B. Ultra-processed foods and human health: From epidemiological evidence to mechanistic insights. The Lancet Gastroenterology & Hepatology 2022. [Google Scholar] [CrossRef]
- Fardet, A. Characterization of the Degree of Food Processing in Relation With Its Health Potential and Effects. Adv Food Nutr Res 2018, 85, 79–129. [Google Scholar] [CrossRef] [PubMed]
- Martínez Leo, E.E.; Peñafiel, A.M.; Hernández Escalante, V.M.; Cabrera Araujo, Z.M. Ultra-processed diet, systemic oxidative stress, and breach of immunologic tolerance. Nutrition 2021, 91-92, 111419. [Google Scholar] [CrossRef] [PubMed]
- Tristan Asensi, M.; Napoletano, A.; Sofi, F.; Dinu, M. Low-Grade Inflammation and Ultra-Processed Foods Consumption: A Review. Nutrients 2023, 15. [Google Scholar] [CrossRef]
- Marx, W.; Lane, M.; Hockey, M.; Aslam, H.; Berk, M.; Walder, K.; Borsini, A.; Firth, J.; Pariante, C.M.; Berding, K.; et al. Diet and depression: Exploring the biological mechanisms of action. Molecular Psychiatry 2021, 26, 134–150. [Google Scholar] [CrossRef]
- Pagliai, G.; Dinu, M.; Madarena, M.P.; Bonaccio, M.; Iacoviello, L.; Sofi, F. Consumption of ultra-processed foods and health status: A systematic review and meta-analysis. British Journal of Nutrition 2020. [Google Scholar] [CrossRef]
- Lane, M.M.; Davis, J.A.; Beattie, S.; Gómez-Donoso, C.; Loughman, A.; O'Neil, A.; Jacka, F.; Berk, M.; Page, R.; Marx, W.; et al. Ultraprocessed food and chronic noncommunicable diseases: A systematic review and meta-analysis of 43 observational studies. Obes Rev 2021, 22, e13146. [Google Scholar] [CrossRef]
- Monteiro, C.A.; Astrup, A. Does the concept of “ultra-processed foods” help inform dietary guidelines, beyond conventional classification systems? YES. The American Journal of Clinical Nutrition 2022. [Google Scholar] [CrossRef]
- Astrup, A.; Monteiro, C.A. Does the concept of “ultra-processed foods” help inform dietary guidelines, beyond conventional classification systems? NO. The American Journal of Clinical Nutrition 2022. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Morgan, R.L.; Whaley, P.; Thayer, K.A.; Schünemann, H.J. Identifying the PECO: A framework for formulating good questions to explore the association of environmental and other exposures with health outcomes. Environ Int 2018, 121, 1027–1031. [Google Scholar] [CrossRef]
- Monteiro, C.A. Nutrition and health. The issue is not food, nor nutrients, so much as processing. Public Health Nutr 2009, 12, 729–731. [Google Scholar] [CrossRef] [PubMed]
- Veritas Health Innovation. Covidence systematic review software, Melbourne, Australia. Available online: www.covidence.org.
- Veronese, N.; Demurtas, J.; Thompson, T.; Solmi, M.; Pesolillo, G.; Celotto, S.; Barnini, T.; Stubbs, B.; Maggi, S.; Pilotto, A.; et al. Effect of low-dose aspirin on health outcomes: An umbrella review of systematic reviews and meta-analyses. Br J Clin Pharmacol 2020, 86, 1465–1475. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Chen, Z.; Chen, B.; Li, J.; Yuan, X.; Li, J.; Wang, W.; Dai, T.; Chen, H.; Wang, Y.; et al. Dietary sugar consumption and health: Umbrella review. BMJ 2023, 381, e071609. [Google Scholar] [CrossRef] [PubMed]
- Belbasis, L.; Bellou, V.; Evangelou, E.; Ioannidis, J.P.; Tzoulaki, I. Environmental risk factors and multiple sclerosis: An umbrella review of systematic reviews and meta-analyses. Lancet Neurol 2015, 14, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Mann, C.J. Observational research methods. Research design II: Cohort, cross sectional, and case-control studies. Emergency Medicine Journal 2003, 20, 54–60. [Google Scholar] [CrossRef]
- Dettori, J.R.; Norvell, D.C.; Chapman, J.R. Fixed-Effect vs Random-Effects Models for Meta-Analysis: 3 Points to Consider. Global Spine J 2022, 12, 1624–1626. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G.; Spiegelhalter, D.J. A re-evaluation of random-effects meta-analysis. J R Stat Soc Ser A Stat Soc 2009, 172, 137–159. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ (Clinical research ed.) 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Sterne, J.A.; Egger, M. Funnel plots for detecting bias in meta-analysis: Guidelines on choice of axis. J Clin Epidemiol 2001, 54, 1046–1055. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. Bmj 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Ioannidis, J.P.; Trikalinos, T.A. An exploratory test for an excess of significant findings. Clin Trials 2007, 4, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Markozannes, G.; Aretouli, E.; Rintou, E.; Dragioti, E.; Damigos, D.; Ntzani, E.; Evangelou, E.; Tsilidis, K.K. An umbrella review of the literature on the effectiveness of psychological interventions for pain reduction. BMC Psychol 2017, 5, 31. [Google Scholar] [CrossRef]
- Gosling, C.J.; Solanes, A.; Fusar-Poli, P.; Radua, J. metaumbrella: The first comprehensive suite to perform data analysis in umbrella reviews with stratification of the evidence. BMJ Ment Health 2023, 26. [Google Scholar] [CrossRef]
- Fusar-Poli, P.; Radua, J. Ten simple rules for conducting umbrella reviews. Evid Based Ment Health 2018, 21, 95–100. [Google Scholar] [CrossRef]
- Ioannidis, J.P. Integration of evidence from multiple meta-analyses: A primer on umbrella reviews, treatment networks and multiple treatments meta-analyses. Cmaj 2009, 181, 488–493. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; deBeer, H.; et al. GRADE guidelines: 1. 1. Introduction—GRADE evidence profiles and summary of findings tables. Journal of Clinical Epidemiology 2011, 64, 383–394. [Google Scholar] [CrossRef]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E.; et al. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. Bmj 2017, 358, j4008. [Google Scholar] [CrossRef]
- Lian, Y.; Wang, G.-P.; Chen, G.-Q.; Chen, H.-N.; Zhang, G.-Y. Association between ultra-processed foods and risk of cancer: A systematic review and meta-analysis. Frontiers in Nutrition 2023, 10. [Google Scholar] [CrossRef]
- Isaksen, I.M.; Dankel, S.N. Ultra-processed food consumption and cancer risk: A systematic review and meta-analysis. Clin Nutr 2023, 42, 919–928. [Google Scholar] [CrossRef]
- Moradi, S.; Entezari, M.H.; Mohammadi, H.; Jayedi, A.; Lazaridi, A.V.; Kermani, M.A.H.; Miraghajani, M. Ultra-processed food consumption and adult obesity risk: A systematic review and dose-response meta-analysis. Crit Rev Food Sci Nutr 2023, 63, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Hu, H.; Li, T.; Zhang, J.; Feng, Y.; Yang, X.; Li, Y.; Wu, Y.; Li, X.; Huang, H.; et al. Dose-response meta-analysis of ultra-processed food with the risk of cardiovascular events and all-cause mortality: Evidence from prospective cohort studies. Food Funct 2023, 14, 2586–2596. [Google Scholar] [CrossRef] [PubMed]
- Pagliai, G.; Dinu, M.; Madarena, M.P.; Bonaccio, M.; Iacoviello, L.; Sofi, F. Consumption of ultra-processed foods and health status: A systematic review and meta-analysis. Br J Nutr 2021, 125, 308–318. [Google Scholar] [CrossRef]
- Wang, M.; Du, X.; Huang, W.; Xu, Y. Ultra-processed Foods Consumption Increases the Risk of Hypertension in Adults: A Systematic Review and Meta-analysis. Am J Hypertens 2022, 35, 892–901. [Google Scholar] [CrossRef] [PubMed]
- Shu, L.; Zhang, X.; Zhou, J.; Zhu, Q.; Si, C. Ultra-processed food consumption and increased risk of metabolic syndrome: A systematic review and meta-analysis of observational studies. Frontiers in Nutrition 2023, 10. [Google Scholar] [CrossRef] [PubMed]
- Henney, A.E.; Gillespie, C.S.; Alam, U.; Hydes, T.J.; Cuthbertson, D.J. Ultra-Processed Food Intake Is Associated with Non-Alcoholic Fatty Liver Disease in Adults: A Systematic Review and Meta-Analysis. Nutrients 2023, 15. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Khandpur, N.; Desjardins, C.; Wang, L.; Monteiro, C.A.; Rossato, S.L.; Fung, T.T.; Manson, J.E.; Willett, W.C.; Rimm, E.B.; et al. Ultra-Processed Food Consumption and Risk of Type 2 Diabetes: Three Large Prospective U.S. Cohort Studies. Diabetes Care 2023. [Google Scholar] [CrossRef]
- Narula, N.; Chang, N.H.; Mohammad, D.; Wong, E.C.L.; Ananthakrishnan, A.N.; Chan, S.S.M.; Carbonnel, F.; Meyer, A. Food Processing and Risk of Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. Clin Gastroenterol Hepatol 2023. [Google Scholar] [CrossRef]
- Delpino, F.M.; Figueiredo, L.M.; Flores, T.R.; Silveira, E.A.; Silva Dos Santos, F.; Werneck, A.O.; Louzada, M.; Arcêncio, R.A.; Nunes, B.P. Intake of ultra-processed foods and sleep-related outcomes: A systematic review and meta-analysis. Nutrition 2023, 106, 111908. [Google Scholar] [CrossRef]
- Lane, M.M.; Gamage, E.; Travica, N.; Dissanayaka, T.; Ashtree, D.N.; Gauci, S.; Lotfaliany, M.; O’Neil, A.; Jacka, F.N.; Marx, W. Ultra-Processed Food Consumption and Mental Health: A Systematic Review and Meta-Analysis of Observational Studies. Nutrients 2022, 14. [Google Scholar] [CrossRef]
- Suksatan, W.; Moradi, S.; Naeini, F.; Bagheri, R.; Mohammadi, H.; Talebi, S.; Mehrabani, S.; Hojjati Kermani, M.A.; Suzuki, K. Ultra-Processed Food Consumption and Adult Mortality Risk: A Systematic Review and Dose-Response Meta-Analysis of 207,291 Participants. Nutrients 2021, 14. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Li, T.; Deeks, J. Chapter 6: Choosing effect measures and computing estimates of effect. In Cochrane Handbook for Systematic Reviews of Interventions version 6.3 (updated 22); Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; Cochrane, 20 February 2022. [Google Scholar]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.T.; Rothstein, H.R. When Does it Make Sense to Perform a Meta-Analysis? In Introduction to Meta-Analysis; John Wiley & Sons, 2009. [Google Scholar]
- Jones, S.R.; Carley, S.; Harrison, M. An introduction to power and sample size estimation. Emergency Medicine Journal 2003, 20, 453. [Google Scholar] [CrossRef] [PubMed]
- Tobias, D.K.; Hall, K.D. Eliminate or reformulate ultra-processed foods? Biological mechanisms matter. Cell Metabolism 2021. [Google Scholar] [CrossRef] [PubMed]
- Machado, P.; Cediel, G.; Woods, J.; Baker, P.; Dickie, S.; Gomes, F.S.; Scrinis, G.; Lawrence, M. Evaluating intake levels of nutrients linked to non-communicable diseases in Australia using the novel combination of food processing and nutrient profiling metrics of the PAHO Nutrient Profile Model. European Journal of Nutrition 2022, 61, 1801–1812. [Google Scholar] [CrossRef]
- Coletro, H.N.; Bressan, J.; Diniz, A.P.; Hermsdorff, H.H.M.; Pimenta, A.M.; Meireles, A.L.; Mendonça, R.D.; Carraro, J.C.C. Habitual polyphenol intake of foods according to NOVA classification: Implications of ultra-processed foods intake (CUME study). Int J Food Sci Nutr 2023, 74, 338–349. [Google Scholar] [CrossRef]
- Fardet, A. Minimally processed foods are more satiating and less hyperglycemic than ultra-processed foods: A preliminary study with 98 ready-to-eat foods. Food Funct 2016, 7, 2338–2346. [Google Scholar] [CrossRef]
- Debras, C.; Chazelas, E.; Srour, B.; Druesne-Pecollo, N.; Esseddik, Y.; Szabo de Edelenyi, F.; Agaësse, C.; De Sa, A.; Lutchia, R.; Gigandet, S.; et al. Artificial sweeteners and cancer risk: Results from the NutriNet-Santé population-based cohort study. PLOS Medicine 2022, 19, e1003950. [Google Scholar] [CrossRef]
- Debras, C.; Chazelas, E.; Sellem, L.; Porcher, R.; Druesne-Pecollo, N.; Esseddik, Y.; de Edelenyi, F.S.; Agaësse, C.; De Sa, A.; Lutchia, R.; et al. Artificial sweeteners and risk of cardiovascular diseases: Results from the prospective NutriNet-Santé cohort. BMJ 2022, 378, e071204. [Google Scholar] [CrossRef]
- Suez, J.; Cohen, Y.; Valdés-Mas, R.; Mor, U.; Dori-Bachash, M.; Federici, S.; Zmora, N.; Leshem, A.; Heinemann, M.; Linevsky, R.; et al. Personalized microbiome-driven effects of non-nutritive sweeteners on human glucose tolerance. Cell 2022, 185, 3307–3328. [Google Scholar] [CrossRef]
- Bettini, S.; Boutet-Robinet, E.; Cartier, C.; Coméra, C.; Gaultier, E.; Dupuy, J.; Naud, N.; Taché, S.; Grysan, P.; Reguer, S.; et al. Food-grade TiO2 impairs intestinal and systemic immune homeostasis, initiates preneoplastic lesions and promotes aberrant crypt development in the rat colon. Scientific Reports 2017, 7, 40373. [Google Scholar] [CrossRef]
- Srour, B.; Chazelas, E.; Druesne-Pecollo, N.; Esseddik, Y.; de Edelenyi, F.S.; Agaësse, C.; De Sa, A.; Lutchia, R.; Debras, C.; Sellem, L.; et al. Dietary exposure to nitrites and nitrates in association with type 2 diabetes risk: Results from the NutriNet-Santé population-based cohort study. PLoS Med 2023, 20, e1004149. [Google Scholar] [CrossRef] [PubMed]
- Chassaing, B.; Compher, C.; Bonhomme, B.; Liu, Q.; Tian, Y.; Walters, W.; Nessel, L.; Delaroque, C.; Hao, F.; Gershuni, V.; et al. Randomized controlled-feeding study of dietary emulsifier carboxymethylcellulose reveals detrimental impacts on the gut microbiota and metabolome. Gastroenterology 2021. [Google Scholar] [CrossRef] [PubMed]
- Lane, M.; Howland, G.; West, M.; Hockey, M.; Marx, W.; Loughman, A.; O’Hely, M.; Jacka, F.; Rocks, T. The effect of ultra-processed very low-energy diets on gut microbiota and metabolic outcomes in individuals with obesity: A systematic literature review. Obesity Research & Clinical Practice 2020, 14, 197–204. [Google Scholar] [CrossRef]
- World Health Organization (WHO) - Guidelines Review Committee - Nutrition and Food Safety. Use of non-sugar sweeteners: WHO guideline; ISBN: 978-92-4-007361-6. Access date: July 7th, 2023; 2023.
- Riboli, E.; Beland, F.A.; Lachenmeier, D.W.; Marques, M.M.; Phillips, D.H.; Schernhammer, E.; Afghan, A.; Assunção, R.; Caderni, G.; Corton, J.C.; et al. Carcinogenicity of aspartame, methyleugenol, and isoeugenol. The Lancet Oncology. [CrossRef]
- Chazelas, E.; Druesne-Pecollo, N.; Esseddik, Y.; de Edelenyi, F.S.; Agaesse, C.; De Sa, A.; Lutchia, R.; Rebouillat, P.; Srour, B.; Debras, C.; et al. Exposure to food additive mixtures in 106,000 French adults from the NutriNet-Santé cohort. Scientific Reports 2021, 11, 19680. [Google Scholar] [CrossRef]
- Hall, K.D.; Ayuketah, A.; Brychta, R.; Cai, H.; Cassimatis, T.; Chen, K.Y.; Chung, S.T.; Costa, E.; Courville, A.; Darcey, V.; et al. Ultra-Processed Diets Cause Excess Calorie Intake and Weight Gain: An Inpatient Randomized Controlled Trial of Ad Libitum Food Intake. Cell Metab 2019, 30, 67–77. [Google Scholar] [CrossRef]
- Sartorelli, D.S.; Crivellenti, L.C.; Baroni, N.F.; de Andrade Miranda, D.E.G.; da Silva Santos, I.; Carvalho, M.R.; de Lima, M.C.; Carreira, N.P.; Chaves, A.V.L.; Manochio-Pina, M.G.; et al. Effectiveness of a minimally processed food-based nutritional counselling intervention on weight gain in overweight pregnant women: A randomized controlled trial. Eur J Nutr 2023, 62, 443–454. [Google Scholar] [CrossRef]
- Fazzino, T.L.; Courville, A.B.; Guo, J.; Hall, K.D. Ad libitum meal energy intake is positively influenced by energy density, eating rate and hyper-palatable food across four dietary patterns. Nature Food 2023, 4, 144–147. [Google Scholar] [CrossRef]
- Adams, J.; Hofman, K.; Moubarac, J.-C.; Thow, A.M. Public health response to ultra-processed food and drinks. BMJ 2020, 369, m2391. [Google Scholar] [CrossRef]
- Dicken, S.J.; Batterham, R.L. The Role of Diet Quality in Mediating the Association between Ultra-Processed Food Intake, Obesity and Health-Related Outcomes: A Review of Prospective Cohort Studies. Nutrients 2021, 14. [Google Scholar] [CrossRef]
- Kipnis, V.; Midthune, D.; Freedman, L.; Bingham, S.; Day, N.E.; Riboli, E.; Ferrari, P.; Carroll, R.J. Bias in dietary-report instruments and its implications for nutritional epidemiology. Public Health Nutr 2002, 5, 915–923. [Google Scholar] [CrossRef]
- Boyko, E.J. Observational research--opportunities and limitations. J Diabetes Complications 2013, 27, 642–648. [Google Scholar] [CrossRef] [PubMed]
- Vadiveloo, M.K.; Gardner, C.D. Not All Ultra-Processed Foods Are Created Equal: A Case for Advancing Research and Policy That Balances Health and Nutrition Security. Diabetes Care 2023, 46, 1327–1329. [Google Scholar] [CrossRef]
- Scrinis, G.; Monteiro, C. From ultra-processed foods to ultra-processed dietary patterns. Nature Food 2022, 3, 671–673. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, A.H.; Appel, L.J.; Vadiveloo, M.; Hu, F.B.; Kris-Etherton, P.M.; Rebholz, C.M.; Sacks, F.M.; Thorndike, A.N.; Horn, L.V.; Wylie-Rosett, J. 2021 Dietary Guidance to Improve Cardiovascular Health: A Scientific Statement From the American Heart Association. Circulation 2021, 144, e472–e487. [Google Scholar] [CrossRef] [PubMed]
- Slimani, N.; Deharveng, G.; Southgate, D.A.; Biessy, C.; Chajès, V.; van Bakel, M.M.; Boutron-Ruault, M.C.; McTaggart, A.; Grioni, S.; Verkaik-Kloosterman, J.; et al. Contribution of highly industrially processed foods to the nutrient intakes and patterns of middle-aged populations in the European Prospective Investigation into Cancer and Nutrition study. Eur J Clin Nutr 2009, 63 Suppl 4, S206–225. [Google Scholar] [CrossRef]
- Eicher-Miller, H.A.; Fulgoni, V.L., 3rd; Keast, D.R. Contributions of processed foods to dietary intake in the US from 2003-2008: A report of the Food and Nutrition Science Solutions Joint Task Force of the Academy of Nutrition and Dietetics, American Society for Nutrition, Institute of Food Technologists, and International Food Information Council. J Nutr 2012, 142, 2065s–2072s. [Google Scholar] [CrossRef]
- Asfaw, A. Does consumption of processed foods explain disparities in the body weight of individuals? The case of Guatemala. Health Econ 2011, 20, 184–195. [Google Scholar] [CrossRef]
- Poti, J.M.; Mendez, M.A.; Shu Wen, N.; Popkin, B.M. Is the degree of food processing and convenience linked with the nutritional quality of foods purchased by US households? American Journal of Clinical Nutrition 2015, 101, 1251–1262. [Google Scholar] [CrossRef]
- Monteiro, C.A.; Levy, R.B.; Claro, R.M.; Castro, I.R.R.d.; Cannon, G. A new classification of foods based on the extent and purpose of their processing. Cadernos de Saúde Pública 2010, 26, 2039–2049. [Google Scholar] [CrossRef]
- Monteiro, C.A.; Cannon, G.; Moubarac, J.-C.; Levy, R.B.; Louzada, M.L.C.; Jaime, P.C. The UN Decade of Nutrition, the NOVA food classification and the trouble with ultra-processing. Public Health Nutrition 2018, 21, 5–17. [Google Scholar] [CrossRef]
- Food Agriculture Organization (FAO) of the United Nations. Guidelines on the collection of information on food processing through food consumption surveys; Rome, 2015. [Google Scholar]
- Food and Agriculture Organization of the United Nations (FAO). Minimum dietary diversity for women; Rome, 2021. [Google Scholar]
- United Nations Children's Fund (UNICEF); for every child. Policy Brief: Sugar-Sweetened Beverage Taxation; 2021. [Google Scholar]
- AO; IFAD; UNICEF; WFP; and WHO. The State of Food Security and Nutrition in the World 2023. Urbanization, agrifood systems transformation and healthy diets across the rural–urban continuum.; FAO: Rome, 2023. [Google Scholar]
- Moubarac, J.C. Ultra-processed food and drink products in Latin America: Trends, impact on obesity, policy implications; Pan American Health Organization World Health Organization: Washington, DC, USA, 2015. [Google Scholar]
- Scientific Advisory Committee on Nutrition (SACN). SACN statement on processed foods and health, 2023.
- Gibney, M.J. Ultra-Processed Foods: Definitions and Policy Issues. Current Developments in Nutrition 2018, 3. [Google Scholar] [CrossRef] [PubMed]
- Gibney, M.J.; Forde, C.G.; Mullally, D.; Gibney, E.R. Ultra-processed foods in human health: A critical appraisal. American Journal of Clinical Nutrition 2017, 106, 712–724. [Google Scholar] [CrossRef]
- Jones, J.M. Food processing: Criteria for dietary guidance and public health? Proc Nutr Soc 2019, 78, 4–18. [Google Scholar] [CrossRef] [PubMed]
- Braesco, V.; Souchon, I.; Sauvant, P.; Haurogné, T.; Maillot, M.; Féart, C.; Darmon, N. Ultra-processed foods: How functional is the NOVA system? European Journal of Clinical Nutrition 2022. [Google Scholar] [CrossRef]
- Sneed, N.M.; Ukwuani, S.; Sommer, E.C.; Samuels, L.R.; Truesdale, K.P.; Matheson, D.; Noerper, T.E.; Barkin, S.L.; Heerman, W.J. Reliability and validity of assigning ultraprocessed food categories to 24-h dietary recall data. The American Journal of Clinical Nutrition 2023, 117, 182–190. [Google Scholar] [CrossRef]
- McHugh, M.L. Interrater reliability: The kappa statistic. Biochem Med (Zagreb) 2012, 22, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Khandpur, N.; Rossato, S.; Drouin-Chartier, J.-P.; Du, M.; Martinez, E.; Sampson, L.; Monteiro, C.; Zhang, F.F.; Willett, W.; Fung, T.T.; et al. Categorizing ultra-processed food intake in large-scale cohort studies: Evidence from the Nurses’ Health Studies, the Health Professionals Follow-up Study, and the Growing Up Today Study. medRxiv 2021. [Google Scholar] [CrossRef]
- Louzada, M.; Cruz, G.L.D.; Silva, K.; Grassi, A.G.F.; Andrade, G.C.; Rauber, F.; Levy, R.B.; Monteiro, C.A. Consumption of ultra-processed foods in Brazil: Distribution and temporal evolution 2008-2018. Rev Saude Publica 2023, 57, 12. [Google Scholar] [CrossRef]
- Martinez-Steele, E.; Khandpur, N.; Batis, C.; Bes-Rastrollo, M.; Bonaccio, M.; Cediel, G.; Huybrechts, I.; Juul, F.; Levy, R.B.; da Costa Louzada, M.L.; et al. Best practices for applying the Nova food classification system. Nature Food 2023, 4, 445–448. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations (FAO). Food-based dietary guidelines. Available online: https://www.fao.org/nutrition/education/food-dietary-guidelines/regions/en/.
- Ministry of Health of Brazil. Dietary guidelines for the Brazilian population. Available online: https://bvsms.saude.gov.br/bvs/publicacoes/dietary_guidelines_brazilian_population.pdf.
- Le Haut Conseil de la santé publique (The High Council for Public Health). Relatif Aux Objectifs de Santé Publique Quantifiés Pour La Politique Nutritionnelle de Santé Publique (PNNS) 2018–2022 (Quantified Public Health Objectives for Public Health Nutrition Policy (PNNS) 2018–2022). Available online: https://www.hcsp.fr/explore.cgi/avisrapportsdomaine?clefr=648.
- The Israeli Ministry of Health. Nutritional recommendations. Available online: https://www.health.gov.il/PublicationsFiles/dietary%20guidelines%20EN.pdf.
- Ministry of Health Malaysia. Malaysian dietary guidelines. Available online: https://nutrition.moh.gov.my/wp-content/uploads/2021/07/Web%20MDG.pdf.
- Karlsen, T.H.; Sheron, N.; Zelber-Sagi, S.; Carrieri, P.; Dusheiko, G.; Bugianesi, E.; Pryke, R.; Hutchinson, S.J.; Sangro, B.; Martin, N.K.; et al. The EASL-Lancet Liver Commission: Protecting the next generation of Europeans against liver disease complications and premature mortality. The Lancet 2022, 399, 61–116. [Google Scholar] [CrossRef] [PubMed]
- Childs, R.; Sibson, V. Ultra-processed foods (UPF) in the diets of infants and young children in the UK: What they are, how they harm health, and what needs to be done to reduce intakes; 2023; pp 1-96.
- World Health Organization (WHO). Guideline: Sugars intake for adults and children. Available online: https://www.who.int/publications/i/item/9789241549028.
- Silver, L.D.; Ng, S.W.; Ryan-Ibarra, S.; Taillie, L.S.; Induni, M.; Miles, D.R.; Poti, J.M.; Popkin, B.M. Changes in prices, sales, consumer spending, and beverage consumption one year after a tax on sugar-sweetened beverages in Berkeley, California, US: A before-and-after study. PLoS medicine 2017, 14, e1002283. [Google Scholar] [CrossRef] [PubMed]
- Pell, D.; Mytton, O.; Penney, T.L.; Briggs, A.; Cummins, S.; Penn-Jones, C.; Rayner, M.; Rutter, H.; Scarborough, P.; Sharp, S.J. Changes in soft drinks purchased by British households associated with the UK soft drinks industry levy: Controlled interrupted time series analysis. bmj 2021, 372. [Google Scholar] [CrossRef] [PubMed]
- Colchero, M.A.; Rivera-Dommarco, J.; Popkin, B.M.; Ng, S.W. In Mexico, evidence of sustained consumer response two years after implementing a sugar-sweetened beverage tax. Health Affairs 2017, 36, 564–571. [Google Scholar] [CrossRef]
- Popkin, B.M.; Hawkes, C. Sweetening of the global diet, particularly beverages: Patterns, trends, and policy responses. Lancet Diabetes Endocrinol 2016, 4, 174–186. [Google Scholar] [CrossRef]
- Muth, N.D.; Dietz, W.H.; Magge, S.N.; Johnson, R.K. Public Policies to Reduce Sugary Drink Consumption in Children and Adolescents. Pediatrics 2019, 143. [Google Scholar] [CrossRef]
| Convincing (Class I) |
|
| Highly suggestive (Class II) |
|
| Suggestive (Class III) |
|
| Weak (Class IV) |
|
| No evidence (Class V) |
|
| Outcome | Level of exposure comparison |
Studies, n | Participants, n | Cases, n | Effect size metric | Effect size | 95% CIs | P value | Largest study significant | Small study effect | Excess significance bias | I2 | Evidence Class |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cancer | |||||||||||||
| Breast cancer 41 | Dose-response | 3 | 282684 | 5240 | OR | 1.033 | (0.976, 1.092) | 2.61E-01 | ns | ns | ns | 59.216 | V |
| Breast cancer 41 | Non-dose-response | 6 | 284644 | 6220 | OR | 1.151 | (0.993, 1.335) | 6.28E-02 | ns | sig. | ns | 45.751 | V |
| Cancer overall 42 | Non-dose-response | 7 | 825701 | 7004 | HR | 1.123 | (1.057, 1.193) | 1.77E-04 | ns | ns | ns | 33.301 | III |
| Central nervous system tumours 41 | Non-dose-response | 2 | 197558 | 328 | OR | 1.199 | (0.871, 1.652) | 2.66E-01 | sig. | na | na | 68.228 | V |
| Chronic lymphocytic leukemia 41 | Non-dose-response | 2 | 199086 | 448 | OR | 1.078 | (0.804, 1.445) | 6.16E-01 | ns | na | na | 0 | V |
| Colorectal cancer 41 | Dose-response | 5 | 720143 | 6881 | OR | 1.039 | (1.008, 1.07) | 1.20E-02 | ns | ns | ns | 55.933 | IV |
| Colorectal cancer 41 | Non-dose-response | 7 | 723262 | 8405 | OR | 1.232 | (1.101, 1.378) | 2.63E-04 | ns | ns | ns | 67.103 | III |
| Pancreatic cancer 41 | Non-dose-response | 2 | 295691 | 773 | OR | 1.235 | (0.853, 1.788) | 2.64E-01 | ns | na | na | 59.484 | V |
| Prostate cancer 41 | Dose-response | 3 | 222460 | 4853 | OR | 0.992 | (0.965, 1.021) | 6.02E-01 | ns | ns | ns | 0 | V |
| Prostate cancer 41 | Non-dose-response | 4 | 226370 | 6772 | OR | 1.023 | (0.933, 1.122) | 6.27E-01 | ns | ns | ns | 0 | V |
| Cardiometabolic conditions | |||||||||||||
| Abdominal obesity 43 | Dose-response | 6 | 66235 | 17011 | OR | 1.047 | (1.024, 1.071) | 5.38E-05 | sig. | ns | ns | 76.474 | III |
| Abdominal obesity 43 | Non-dose-response | 4 | 31749 | 13928 | OR | 1.41 | (1.175, 1.684) | 2.02E-04 | sig. | ns | sig. | 62.29 | III |
| Cardiovascular disease events combined (morbidity and mortality) 44 | Dose-response | 8 | 289077 | 11054 | RR | 1.042 | (1.023, 1.061) | 1.15E-05 | sig. | ns | ns | 75.862 | III |
| Cardiovascular disease events combined (morbidity and mortality) 44 | Non-dose-response | 6 | 269136 | 8235 | RR | 1.347 | (1.182, 1.536) | 8.16E-06 | sig. | ns | ns | 62.12 | III |
| Cardiovascular disease morbidity 44 | Dose-response | 2 | 117298 | 3308 | RR | 1.04 | (1.023, 1.057) | 1.68E-06 | sig. | na | na | 0 | III |
| Cardiovascular disease morbidity 44 | Non-dose-response | 2 | 116357 | 3308 | RR | 1.204 | (1.089, 1.331) | 2.77E-04 | sig. | na | na | 0 | III |
| Hyperglycaemia 45 | Non-dose-response | 2 | 1113 | 511 | OR | 1.1 | (0.344, 3.515) | 8.72E-01 | sig. | na | na | 67.435 | V |
| Hypertension 46 | Non-dose-response | 9 | 111594 | 13386 | OR | 1.232 | (1.107, 1.371) | 1.39E-04 | ns | ns | sig. | 51.994 | III |
| Hypertriglyceridaemia 45 | Non-dose-response | 2 | 1113 | 392 | OR | 0.947 | (0.597, 1.501) | 8.17E-01 | ns | na | na | 0 | V |
| Low high-density lipoprotein concentration 45 | Non-dose-response | 2 | 1113 | 475 | OR | 2.019 | (1.27, 3.21) | 2.97E-03 | sig. | na | na | 0 | IV |
| Metabolic syndrome 47 | Non-dose-response | 9 | 23500 | 8998 | RR | 1.247 | (1.093, 1.422) | 1.05E-03 | sig. | sig. | sig. | 85.01 | IV |
| Non-alcoholic fatty liver disease 48 | Non-dose-response | 4 | 23110 | 9057 | RR | 1.229 | (1.034, 1.46) | 1.90E-02 | sig. | ns | sig. | 89.936 | IV |
| Obesity 43 | Dose-response | 7 | 80064 | 15769 | OR | 1.071 | (1.03, 1.114) | 5.47E-04 | ns | sig. | sig. | 88.4 | III |
| Obesity 43 | Non-dose-response | 7 | 96485 | 21241 | OR | 1.55 | (1.357, 1.771) | 1.18E-10 | sig. | ns | sig. | 54.818 | II |
| Overweight 43 | Dose-response | 2 | 24954 | 14530 | OR | 1.063 | (1.027, 1.101) | 5.37E-04 | sig. | na | na | 54.221 | III |
| Overweight 43 | Non-dose-response | 4 | 44820 | 21927 | OR | 1.362 | (1.139, 1.63) | 7.25E-04 | ns | ns | ns | 72.555 | III |
| Overweight/obesity 43 | Dose-response | 3 | 15152 | 4302 | OR | 1.032 | (1.01, 1.055) | 5.15E-03 | sig. | ns | ns | 38.885 | IV |
| Overweight/obesity 43 | Non-dose-response | 2 | 32417 | 13791 | OR | 1.287 | (1.05, 1.578) | 1.53E-02 | ns | na | na | 0 | IV |
| Type two diabetes 49 | Dose-response | 7 | 415554 | 21932 | RR | 1.118 | (1.105, 1.131) | 2.28E-77 | sig. | ns | ns | 2.189 | I |
| Type two diabetes 49 | Non-dose-response | 7 | 415554 | 21932 | OR | 1.397 | (1.229, 1.588) | 3.13E-07 | sig. | ns | sig. | 88.114 | II |
| Gastrointestinal conditions | |||||||||||||
| Crohn’s disease 50 | Non-dose-response | 4 | 962593 | 889 | HR | 1.709 | (1.365, 2.141) | 3.05E-06 | ns | ns | ns | 0 | IV |
| Ulcerative colitis 50 | Non-dose-response | 4 | 962593 | 1886 | HR | 1.172 | (0.856, 1.606) | 3.22E-01 | ns | ns | ns | 73.901 | V |
| Mental health | |||||||||||||
| Adverse sleep-related outcomes 51 | Non-dose-response | 2 | 102191 | 4804 | OR | 1.412 | (1.241, 1.606) | 1.11E-06 | sig. | na | na | 41.645 | II |
| Anxiety outcomes 52 | Non-dose-response | 4 | 101709 | 11711 | OR | 1.475 | (1.371, 1.586) | 1.35E-25 | sig. | ns | ns | 0 | I |
| Combined common mental disorder outcomes 52 | Non-dose-response | 6 | 185773 | 41948 | OR | 1.529 | (1.432, 1.632) | 4.91E-37 | sig. | ns | ns | 9.08 | I |
| Depressive outcomes 52 | Non-dose-response | 2 | 41637 | 2995 | HR | 1.216 | (1.158, 1.277) | 3.42E-15 | sig. | na | na | 0 | II |
| Mortality | |||||||||||||
| All-cause mortality 44 | Dose-response | 9 | 295651 | 35080 | RR | 1.023 | (1.014, 1.032) | 1.11E-06 | sig. | sig. | sig. | 45.571 | III |
| All-cause mortality 44 | Non-dose-response | 7 | 287969 | 19827 | RR | 1.207 | (1.151, 1.266) | 8.60E-15 | sig. | sig. | sig. | 11.867 | II |
| Cancer mortality 53 | Non-dose-response | 2 | 42203 | 641 | HR | 1.003 | (0.811, 1.24) | 9.79E-01 | ns | na | na | 0 | V |
| Cardiovascular disease mortality 44 | Dose-response | 5 | 147961 | 7135 | RR | 1.047 | (1.015, 1.08) | 4.12E-03 | sig. | ns | ns | 85.405 | IV |
| Cardiovascular disease mortality 44 | Non-dose-response | 4 | 152779 | 4927 | RR | 1.497 | (1.374, 1.63) | 2.07E-20 | sig. | ns | ns | 0 | I |
| Heart disease-related mortality 53 | Dose-response | 2 | 114366 | 4240 | HR | 1.179 | (0.946, 1.47) | 1.42E-01 | sig. | na | na | 0 | II |
| Heart disease-related mortality 53 | Non-dose-response | 2 | 114366 | 4240 | HR | 1.664 | (1.506, 1.838) | 1.36E-23 | sig. | na | na | 75.522 | V |
| Respiratory conditions | |||||||||||||
| Asthma 18 | Non-dose-response | 2 | 111294 | 14037 | RR | 1.203 | (0.988, 1.464) | 6.55E-02 | sig. | na | na | 36.063 | V |
| Wheezing 18 | Non-dose-response | 2 | 111294 | 25590 | RR | 1.403 | (1.266, 1.554) | 9.06E-11 | sig. | na | na | 7.622 | II |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




