Submitted:
14 August 2023
Posted:
15 August 2023
You are already at the latest version
Abstract
Keywords:
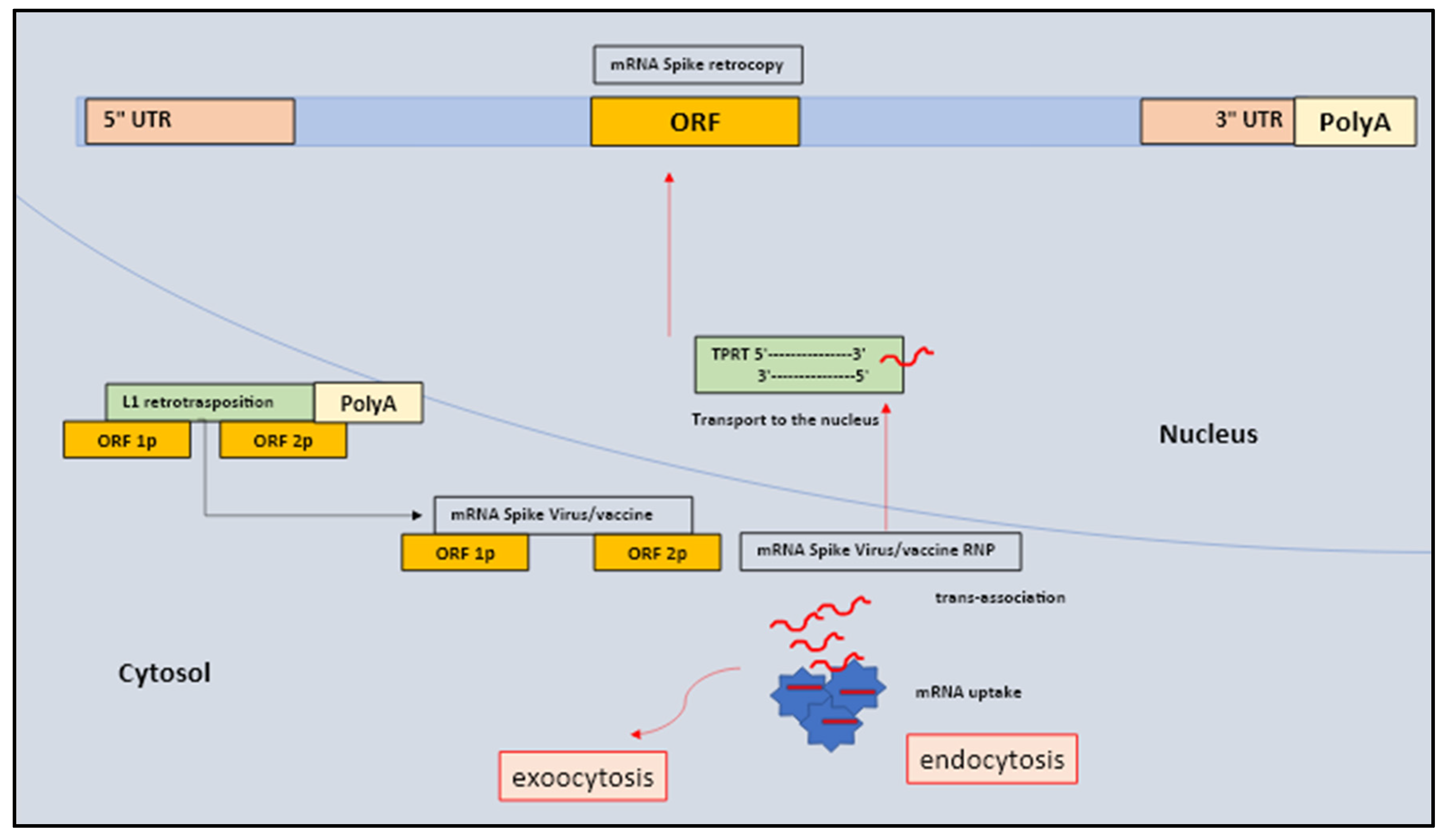
1. Messenger RNA spike protein and host genome alteration the role of retrotransposons
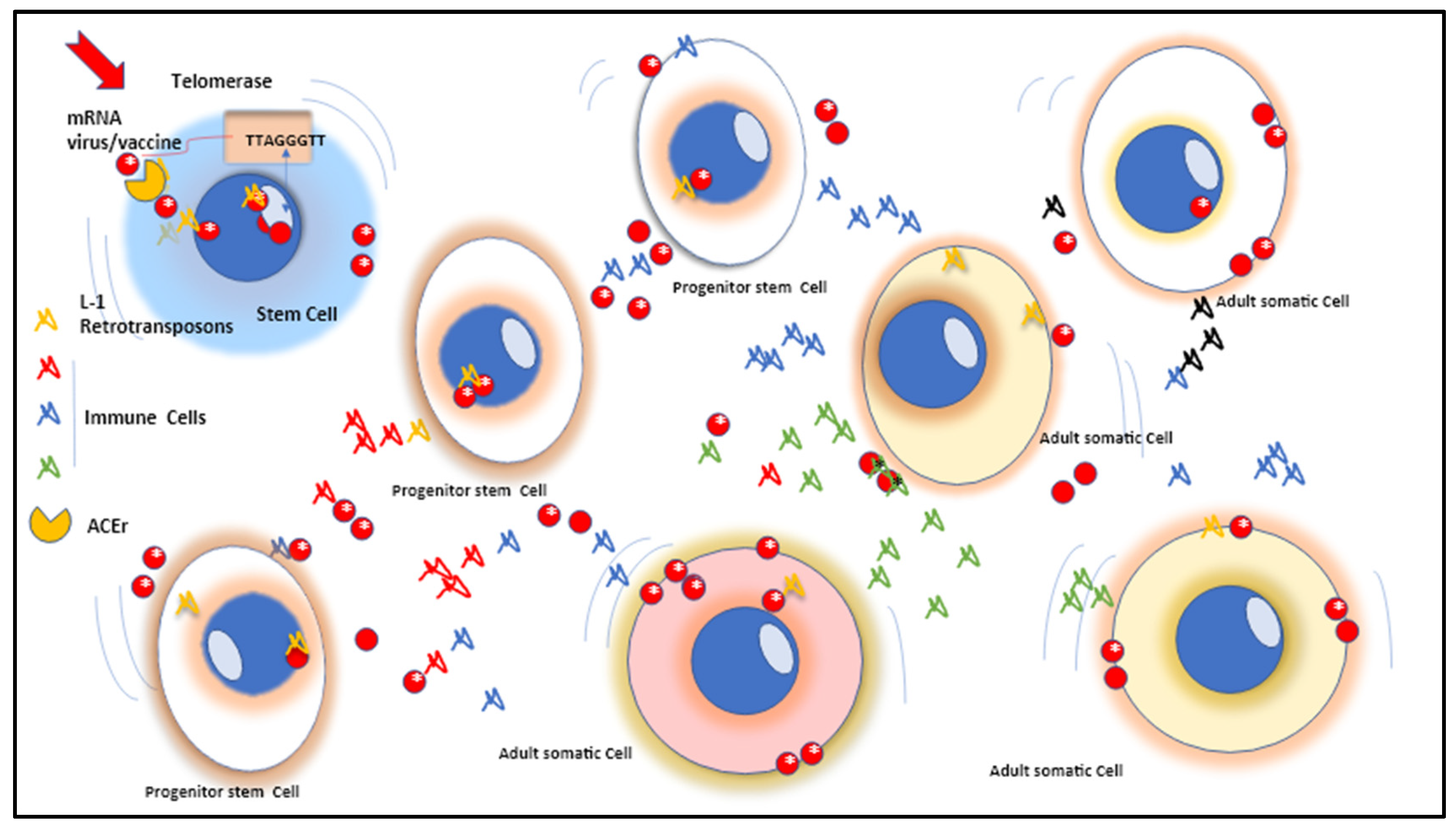
2. Pluripotent, multipotent and progenitor adult stem cells the mRNA of spike protein the telomerasi, the retrotransposition integrated mechanism and the sequential cellular damages
3. Pluripotent, multipotent and progenitor stem cells contamination and further consequences
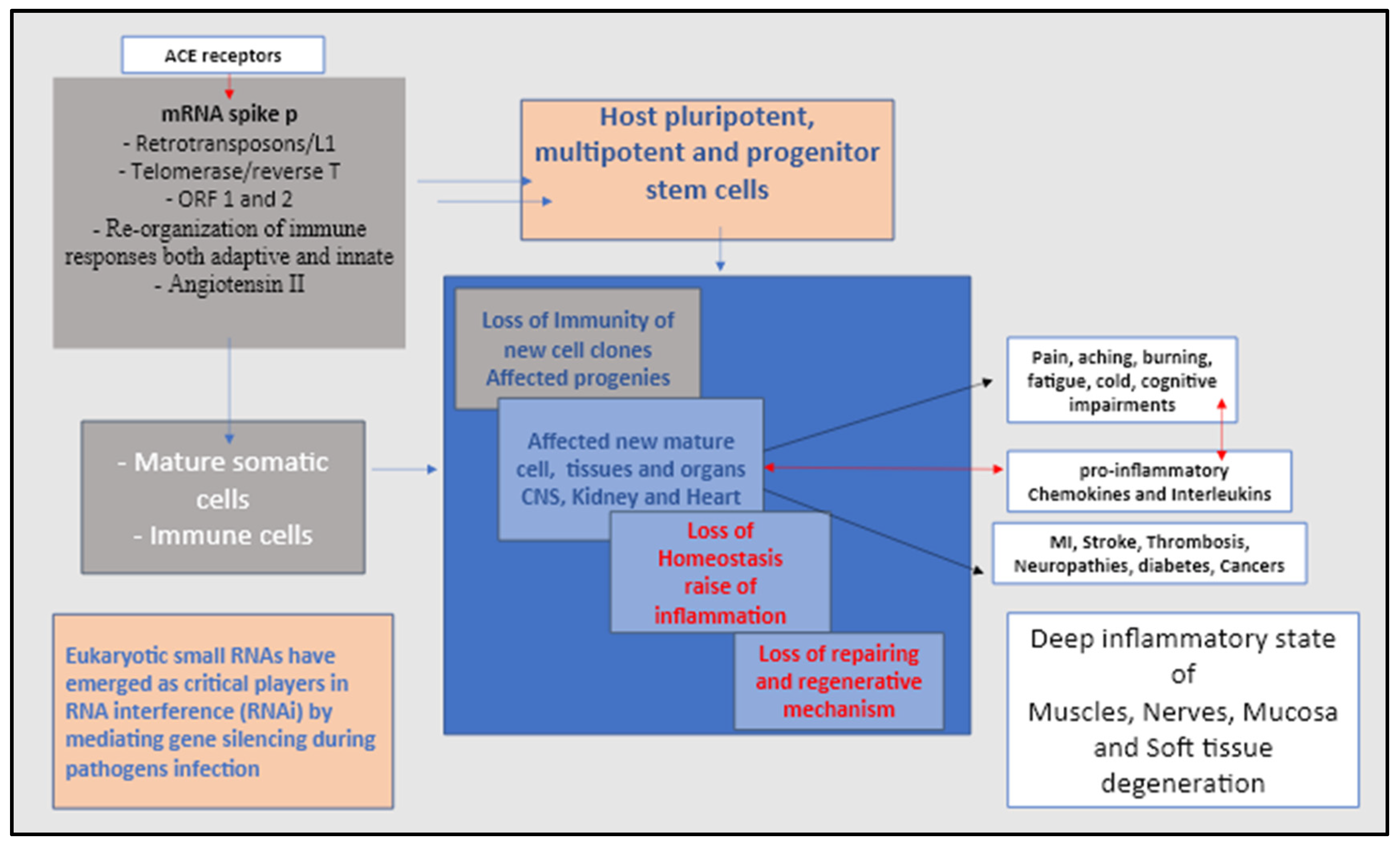
4. The Spike protein syndrome in searching for details
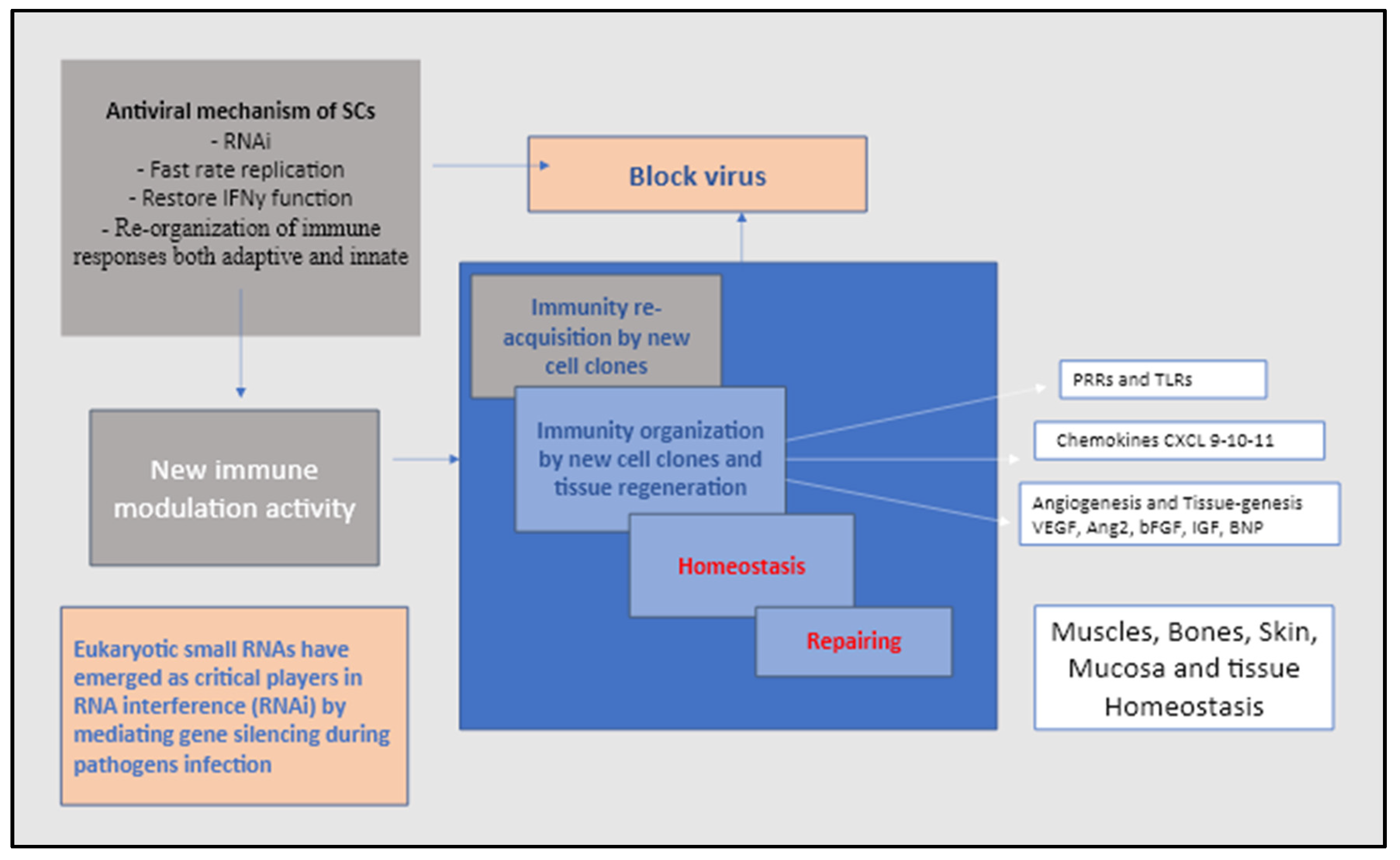
6. The virus non-codingRNA the mechanism to avoid immune surveillance and deactivating stem cells
7. Final considerations
8. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Xu, S.; Yang, K.; Li, R.; Zhang, L. mRNA Vaccine Era—Mechanisms, Drug Platform and Clinical Prospection. Int. J. Mol. Sci. 2020, 21, 6582. [Google Scholar] [CrossRef] [PubMed]
- Domazet-Lošo, T. mRNA Vaccines: Why Is the Biology of Retroposition Ignored? Genes 2022, 13, 719. [Google Scholar] [CrossRef]
- Zhang, L.; Bisht, P.; Flamier, A.; Barrasa, M.I.; Friesen, M.; Richards, A.; Hughes, S.H.; Jaenisch, R. LINE1-Mediated Reverse Transcription and Genomic Integration of SARS-CoV-2 mRNA Detected in Virus-Infected but Not in Viral mRNA-Transfected Cells. Viruses 2023, 15, 629. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Casillas, A.; Redwan, E.M.; Uversky, V.N. SARS-CoV-2: A Master of Immune Evasion. Biomedicines 2022, 10, 1339. [Google Scholar] [CrossRef] [PubMed]
- Milicic Stanic B, Maddox S, de Souza AMA, Wu X et al. Male bias in ACE2 basic science research: Missed opportunity for discovery in the time of COVID-19. Am. J. Physiol. Regul. 2021, 320, R925–R937.
- Hoffmann M, Kleine-Weber H, Schroeder S, Kruger, N et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280.e278. [CrossRef]
- Devaux, C.A.; Camoin-Jau, L. Molecular Mimicry of the Viral Spike in the SARS-CoV-2 Vaccine Possibly Triggers Transient Dysregulation of ACE2, Leading to Vascular and Coagulation Dysfunction Similar to SARS-CoV-2 Infection. Viruses 2023, 15, 1045. [Google Scholar] [CrossRef]
- Zhang, L.; Bisht, P.; Flamier, A.; Barrasa, M.I.; Friesen, M.; Richards, A.; Hughes, S.H.; Jaenisch, R. LINE1-Mediated Reverse Transcription and Genomic Integration of SARS-CoV-2 mRNA Detected in Virus-Infected but Not in Viral mRNA-Transfected Cells. Viruses 2023, 15, 629. [Google Scholar] [CrossRef]
- Wang S, Yao X, But S, Ping Y et al. A single-cell transcriptomic landscape of the lungs of patients with COVID-19. Nat. Cell Biol. 2021, 23, 1314–1328. [CrossRef]
- Wei, W.; Gilbert, N.; Ooi, S.L.; Lawler, J.F.; Ostertag, E.M.; Kazazian, H.H.; Boeke, J.D.; Moran, J.V. Human L1 Retrotransposition: cisPreference versus trans Complementation. Mol. Cell. Biol. 2001, 21, 1429–1439. [Google Scholar] [CrossRef]
- Dewannieux, M.; Esnault, C.; Heidmann, T. LINE-mediated retrotransposition of marked Alu sequences. Nat. Genet. 2003, 35, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Perez, J.L.; Doucet, A.J.; Bucheton, A.; Moran, J.V.; Gilbert, N. Distinct mechanisms for trans-mediated mobilization of cellular RNAs by the LINE-1 reverse transcriptase. Genome Res. 2007, 17, 602–611. [Google Scholar] [CrossRef] [PubMed]
- Kaessmann, H.; Vinckenbosch, N.; Long, M. RNA-based gene duplication: mechanistic and evolutionary insights. Nat. Rev. Genet. 2009, 10, 19–31. [Google Scholar] [CrossRef]
- Salgado-Albarran M, Navarro-Delgado E. I, Del Moral-Morales A, Alcaraz N et al. Comparative transcriptome analysis reveals key epigenetic targets in SARS-CoV-2 infection. NPJ Syst. Biol. Appl. 2021, 7, 21. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.B.; Song, H.; Xu, Y.; Garrison, K.E.; Buzdin, A.A.; Anwar, N.; Hunter, D.V.; Mujib, S.; Mihajlovic, V.; Martin, E.; et al. LINE-1 Retrotransposable Element DNA Accumulates in HIV-1-Infected Cells. J. Virol. 2013, 87, 13307–13320. [Google Scholar] [CrossRef] [PubMed]
- Sudhindar, P.D.; Wainwright, D.; Saha, S.; Howarth, R.; McCain, M.; Bury, Y.; Saha, S.S.; McPherson, S.; Reeves, H.; Patel, A.H.; et al. HCV Activates Somatic L1 Retrotransposition—A Potential Hepatocarcinogenesis Pathway. Cancers 2021, 13, 5079. [Google Scholar] [CrossRef]
- Schöbel, A.; Nguyen-Dinh, V.; Schumann, G.G.; Herker, E. Hepatitis C virus infection restricts human LINE-1 retrotransposition in hepatoma cells. PLOS Pathog. 2021, 17, e1009496. [Google Scholar] [CrossRef]
- Bonenfant, G.; Meng, R.; Shotwell, C.; Badu, P.; Payne, A.F.; Ciota, A.T.; Sammons, M.A.; Berglund, J.A.; Pager, C.T. Asian Zika Virus Isolate Significantly Changes the Transcriptional Profile and Alternative RNA Splicing Events in a Neuroblastoma Cell Line. Viruses 2020, 12, 510. [Google Scholar] [CrossRef]
- Bellavite, P.; Ferraresi, A.; Isidoro, C. Immune Response and Molecular Mechanisms of Cardiovascular Adverse Effects of Spike Proteins from SARS-CoV-2 and mRNA Vaccines. Biomedicines 2023, 11, 451. [Google Scholar] [CrossRef]
- Carelli, F.N.; Hayakawa, T.; Go, Y.; Imai, H.; Warnefors, M.; Kaessmann, H. The life history of retrocopies illuminates the evolution of new mammalian genes. Genome Res. 2016, 26, 301–314. [Google Scholar] [CrossRef]
- Horie, M.; Honda, T.; Suzuki, Y.; Kobayashi, Y.; Daito, T.; Oshida, T.; Ikuta, K.; Jern, P.; Gojobori, T.; Coffin, J.M.; et al. Endogenous non-retroviral RNA virus elements in mammalian genomes. Nature 2010, 463, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Parrish, N.F.; Fujino, K.; Shiromoto, Y.; Iwasaki, Y.W.; Ha, H.; Xing, J.; Makino, A.; Kuramochi-Miyagawa, S.; Nakano, T.; Siomi, H.; et al. piRNAs derived from ancient viral processed pseudogenes as transgenerational sequence-specific immune memory in mammals. RNA 2015, 21, 1691–1703. [Google Scholar] [CrossRef] [PubMed]
- Son, J.-H.; Do, H.; Han, J. Intragenic L1 Insertion: One Possibility of Brain Disorder. Life 2022, 12, 1425. [Google Scholar] [CrossRef]
- Gundry, SF.; mRNA COVID Vaccines Dramatically Increase Endothelial Inflammatory Markers and ACS Risk as Measured by PULS Cardiac Test: A Warning. Circulation. 2021. Available online: www.ahajournals.org›abs›circ.144.suppl_1.10712 (accessed on 6 April 2023).
- Angels F, Spanevello A, Reboldi G, Visca D, Verdecchia, P. SARS-CoV-2 vaccines: Lights and shadows. Eur. J. Intern. Med. 2021, 88, 1–8. [CrossRef]
- Symer, D.E.; Connelly, C.; Szak, S.T.; Caputo, E.M.; Cost, G.J.; Parmigiani, G.; Boeke, J.D. Human L1 Retrotransposition Is Associated with Genetic Instability In Vivo. Cell 2002, 110, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Beck, C.R.; Garcia-Perez, J.L.; Badge, R.M.; Moran, J.V. LINE-1 Elements in Structural Variation and Disease. Annu. Rev. Genom. Hum. Genet. 2011, 12, 187–215. [Google Scholar] [CrossRef]
- Chen, J.-M.; Férec, C.; Cooper, D.N. LINE-1 Endonuclease-Dependent Retrotranspositional Events Causing Human Genetic Disease: Mutation Detection Bias and Multiple Mechanisms of Target Gene Disruption. J. Biomed. Biotechnol. 2006, 2006, 1–9. [Google Scholar] [CrossRef]
- Burns, K.H. Our Conflict with Transposable Elements and Its Implications for Human Disease. Annu. Rev. Pathol. Mech. Dis. 2020, 15, 51–70. [Google Scholar] [CrossRef]
- Denli, A.M.; Narvaiza, I.; Kerman, B.E.; Pena, M.; Benner, C.; Marchetto, M.C.; Diedrich, J.K.; Aslanian, A.; Ma, J.; Moresco, J.J.; et al. Primate-Specific ORF0 Contributes to Retrotransposon-Mediated Diversity. Cell 2015, 163, 583–593. [Google Scholar] [CrossRef]
- Chesnokova, E.; Beletskiy, A.; Kolosov, P. The Role of Transposable Elements of the Human Genome in Neuronal Function and Pathology. Int. J. Mol. Sci. 2022, 23, 5847. [Google Scholar] [CrossRef]
- Denli, A.M.; Narvaiza, I.; Kerman, B.E.; Pena, M.; Benner, C.; Marchetto, M.C.; Diedrich, J.K.; Aslanian, A.; Ma, J.; Moresco, J.J.; et al. Primate-Specific ORF0 Contributes to Retrotransposon-Mediated Diversity. Cell 2015, 163, 583–593. [Google Scholar] [CrossRef] [PubMed]
- Esnault, C.; Maestre, J.; Heidmann, T. Human LINE retrotransposons generate processed pseudogenes. Nat. Genet. 2000, 24, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Sultana, T.; van Essen, D.; Siol, O.; Bailly-Bechet, M.; Philippe, C.; El Aabidine, A.Z.; Pioger, L.; Nigumann, P.; Saccani, S.; Andrau, J.-C.; et al. The Landscape of L1 Retrotransposons in the Human Genome Is Shaped by Pre-insertion Sequence Biases and Post-insertion Selection. Mol. Cell 2019, 74, 555–570. [Google Scholar] [CrossRef] [PubMed]
- A Erwin, J.; Paquola, A.C.M.; Singer, T.; Gallina, I.; Novotny, M.; Quayle, C.; A Bedrosian, T.; I A Alves, F.; Butcher, C.R.; Herdy, J.R.; et al. L1-associated genomic regions are deleted in somatic cells of the healthy human brain. Nat. Neurosci. 2016, 19, 1583–1591. [Google Scholar] [CrossRef] [PubMed]
- Zhang L, Richards A, Barrasa MI, Stephen HH et al. Reverse-transcribed SARS-CoV-2 RNA can integrate into the genome of cultured human cells and can be expressed in patient-derived tissues. Proc. Natl. Acad. Sci. USA 2021, 118, e2105968118. [CrossRef] [PubMed]
- Hessien, M.; Donia, T.; Tabll, A.A.; Adly, E.; Abdelhafez, T.H.; Attia, A.; Alkafaas, S.S.; Kuna, L.; Glasnovic, M.; Cosic, V.; et al. Mechanistic-Based Classification of Endocytosis-Related Inhibitors: Does It Aid in Assigning Drugs against SARS-CoV-2? Viruses 2023, 15, 1040. [Google Scholar] [CrossRef]
- Wang, J.; Huang, J.; Shi, G. Retrotransposons in pluripotent stem cells. Cell Regen. 2020, 9, 1–10. [Google Scholar] [CrossRef]
- Patlar, B. On the Role of Seminal Fluid Protein and Nucleic Acid Content in Paternal Epigenetic Inheritance. Int. J. Mol. Sci. 2022, 23, 14533. [Google Scholar] [CrossRef]
- Poiani, A. Complexity of seminal fluid: a review. Behav. Ecol. Sociobiol. 2006, 60, 289–310. [Google Scholar] [CrossRef]
- Rodríguez-Martínez, H.; Kvist, U.; Ernerudh, J.; Sanz, L.; Calvete, J.J. Seminal Plasma Proteins: What Role Do They Play? Am. J. Reprod. Immunol. 2011, 66 (Suppl. 1), 11–22. [Google Scholar] [CrossRef]
- Lupatov, A.Y.; Yarygin, K.N. Telomeres and Telomerase in the Control of Stem Cells. Biomedicines 2022, 10, 2335. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Passos, J.F.; Birket, M.J.; Beckmann, T.; Brings, S.; Peters, H.; Birch-Machin, M.A.; von Zglinicki, T.; Saretzki, G. Telomerase does not counteract telomere shortening but protects mitochondrial function under oxidative stress. J. Cell Sci. 2008, 121, 1046–1053. [Google Scholar] [CrossRef] [PubMed]
- Kaul, Z.; Cesare, A.J.; I Huschtscha, L.; A Neumann, A.; Reddel, R.R. Five dysfunctional telomeres predict onset of senescence in human cells. EMBO Rep. 2011, 13, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Leem, S.-H.; Londoño-Vallejo, J.A.; Kim, J.-H.; Bui, H.; Tubacher, E.; Solomon, G.; Park, J.-E.; Horikawa, I.; Kouprina, N.; Barrett, J.C.; et al. The human telomerase gene: complete genomic sequence and analysis of tandem repeat polymorphisms in intronic regions. Oncogene 2002, 21, 769–777. [Google Scholar] [CrossRef]
- Casacuberta, E. Drosophila: Retrotransposons Making up Telomeres. Viruses 2017, 9, 192. [Google Scholar] [CrossRef]
- Danilevskaya ON, Arkhipova IR, Traverse KL, Pardue ML. Promoting in tandem: The promoter for telomere transposon HeT-A and implications for the evolution of retroviral LTRs. Cell 1997, 88, 647–655. [CrossRef]
- Jenner, L.P.; Peska, V.; Fulnečková, J.; Sýkorová, E. Telomeres and Their Neighbors. Genes 2022, 13, 1663. [Google Scholar] [CrossRef]
- Vaquero-Sedas, M.I.; Vega-Palas, M.A. Assessing the Epigenetic Status of Human Telomeres. Cells 2019, 8, 1050. [Google Scholar] [CrossRef]
- Cubiles, M.D.; Barroso, S.; I Vaquero-Sedas, M.; Enguix, A.; Aguilera, A.; A Vega-Palas, M. Epigenetic features of human telomeres. Nucleic Acids Res. 2018, 46, 2347–2355. [Google Scholar] [CrossRef]
- Gámez-Arjona, F.M.; López-López, C.; Vaquero-Sedas, M.I.; Vega-Palas, M.A. On the organization of the nucleosomes associated with telomeric sequences. Biochim. et Biophys. Acta (BBA) - Mol. Cell Res. 2010, 1803, 1058–1061. [Google Scholar] [CrossRef]
- Vaquero-Sedas, M.I.; Vega-Palas, M.A. On the chromatin structure of eukaryotic telomeres. Epigenetics 2011, 6, 1055–1058. [Google Scholar] [CrossRef] [PubMed]
- Hughes, S.H.; Chandler, M.; Gellert, M.; Lambowitz, A.M.; Rice, P.A.; Sandmeyer, S.B. Reverse Transcription of Retroviruses and LTR Retrotransposons. Microbiol Spectr. 2015, MDNA3-0027-2014. [Google Scholar] [CrossRef]
- Beck, C.R.; Garcia-Perez, J.L.; Badge, R.M.; Moran, J.V. LINE-1 Elements in Structural Variation and Disease. Annu. Rev. Genom. Hum. Genet. 2011, 12, 187–215. [Google Scholar] [CrossRef]
- Kaer, K.; Speek, M. Retroelements in human disease. Gene 2013, 518, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Żyżyńska-Galeńska, K.; Bernat, A.; Piliszek, A.; Karasiewicz, J.; Szablisty, E.; Sacharczuk, M.; Brewińska-Olchowik, M.; Bochenek, M.; Grabarek, J.; Modliński, J.A. Embryonic Environmental Niche Reprograms Somatic Cells to Express Pluripotency Markers and Participate in Adult Chimaeras. Cells 2021, 10, 490. [Google Scholar] [CrossRef] [PubMed]
- Petkov, S.; Dressel, R.; Rodriguez-Polo, I.; Behr, R. Controlling the Switch from Neurogenesis to Pluripotency during Marmoset Monkey Somatic Cell Reprogramming with Self-Replicating mRNAs and Small Molecules. Cells 2020, 9, 2422. [Google Scholar] [CrossRef] [PubMed]
- Ciomborowska-Basheer, J.; Staszak, K.; Kubiak, M.R.; Makałowska, I. Not So Dead Genes—Retrocopies as Regulators of Their Disease-Related Progenitors and Hosts. Cells 2021, 10, 912. [Google Scholar] [CrossRef]
- Staszak, K.; Makałowska, I. Cancer, Retrogenes, and Evolution. Life 2021, 11, 72. [Google Scholar] [CrossRef]
- Aoyama, K.; Itokawa, N.; Oshima, M.; Iwama, A. Epigenetic Memories in Hematopoietic Stem and Progenitor Cells. Cells 2022, 11, 2187. [Google Scholar] [CrossRef]
- Demongeot, J.; Fougère, C. mRNA COVID-19 Vaccines—Facts and Hypotheses on Fragmentation and Encapsulation. Vaccines 2022, 11, 40. [Google Scholar] [CrossRef]
- Aldén, M.; Olofsson Falla, F.; Yang, D.; Barghouth, M.; Luan, C.; Rasmussen, M.; De Marinis, Y. Intracellular Reverse Transcription of Pfizer BioNTech COVID-19 mRNA Vaccine BNT162b2 In Vitro in Human Liver Cell Line. Curr. Issues Mol. Biol. 2022, 44, 1115–1126. [Google Scholar] [CrossRef] [PubMed]
- Giannotta, G.; Murrone, A.; Giannotta, N. COVID-19 mRNA Vaccines: The Molecular Basis of Some Adverse Events. Vaccines 2023, 11, 747. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.K.; Lavine, K.J.; Lin, C.-Y. Myocarditis after Covid-19 mRNA Vaccination. New Engl. J. Med. 2021, 385, 1332–1334. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.; Kim, M.C.; Kim, K.H.; Jeong, I.-S.; Cho, Y.S.; Choi, Y.D.; Lee, J.E. Case Report: Acute Fulminant Myocarditis and Cardiogenic Shock After Messenger RNA Coronavirus Disease 2019 Vaccination Requiring Extracorporeal Cardiopulmonary Resuscitation. Front. Cardiovasc. Med. 2021, 8, 758996. [Google Scholar] [CrossRef]
- Wilkins, C.; Dishongh, R.; Moore, S.C.; Whitt, M.A.; Chow, M.; Machaca, K. RNA interference is an antiviral defence mechanism in Caenorhabditis elegans. Nature 2005, 436, 1044–1047. [Google Scholar] [CrossRef]
- Balzanelli, M.G.; Distratis, P.; Lazzaro, R.; Pham, V.H.; Tran, T.C.; Dipalma, G.; Bianco, A.; Serlenga, E.M.; Aityan, S.K.; Pierangeli, V.; et al. Analysis of Gene Single Nucleotide Polymorphisms in COVID-19 Disease Highlighting the Susceptibility and the Severity towards the Infection. Diagnostics 2022, 12, 2824. [Google Scholar] [CrossRef]
- Balzanelli, M.G.; Distratis, P.; Lazzaro, R.; D’ettorre, E.; Nico, A.; Inchingolo, F.; Dipalma, G.; Tomassone, D.; Serlenga, E.M.; Dalagni, G.; et al. New Translational Trends in Personalized Medicine: Autologous Peripheral Blood Stem Cells and Plasma for COVID-19 Patient. J. Pers. Med. 2022, 12, 85. [Google Scholar] [CrossRef]
- Sinibaldi-Vallebona, P.; Matteucci, C.; Spadafora, C. Retrotransposon-Encoded Reverse Transcriptase in the Genesis, Progression and Cellular Plasticity of Human Cancer. Cancers 2011, 3, 1141–1157. [Google Scholar] [CrossRef]
- Lavia, P.; Sciamanna, I.; Spadafora, C. An Epigenetic LINE-1-Based Mechanism in Cancer. Int. J. Mol. Sci. 2022, 23, 14610. [Google Scholar] [CrossRef]
- Maupetit-Mehouas, S.; Vaury, C. Transposon Reactivation in the Germline May Be Useful for Both Transposons and Their Host Genomes. Cells 2020, 9, 1172. [Google Scholar] [CrossRef]
- Kravchuk, E.V.; Ashniev, G.A.; Gladkova, M.G.; Orlov, A.V.; Vasileva, A.V.; Boldyreva, A.V.; Burenin, A.G.; Skirda, A.M.; Nikitin, P.I.; Orlova, N.N. Experimental Validation and Prediction of Super-Enhancers: Advances and Challenges. Cells 2023, 12, 1191. [Google Scholar] [CrossRef] [PubMed]
- Seymour, T.; Twigger, A.-J.; Kakulas, F. Pluripotency Genes and Their Functions in the Normal and Aberrant Breast and Brain. Int. J. Mol. Sci. 2015, 16, 27288–27301. [Google Scholar] [CrossRef] [PubMed]
- Balzanelli, M.G.; Distratis, P.; Catucci, O.; Cefalo, A.; Lazzaro, R.; Inchingolo, F.; Tomassone, D.; Aityan, S.K.; Ballini, A.; Nguyen, K.C.D.; et al. Mesenchymal Stem Cells: The Secret Children’s Weapons against the SARS-CoV-2 Lethal Infection. Appl. Sci. 2021, 11, 1696. [Google Scholar] [CrossRef]
- Närvä, E.; Rahkonen, N.; Emani, M.R.; Lund, R.; Pursiheimo, J.; Nästi, J.; Autio, R.; Rasool, O.; Denessiouk, K.; Lähdesmäki, H.; et al. RNA-Binding Protein L1TD1 Interacts with LIN28 via RNA and is Required for Human Embryonic Stem Cell Self-Renewal and Cancer Cell Proliferation. STEM CELLS 2011, 30, 452–460. [Google Scholar] [CrossRef]
- Zhang L, Richards A, Barrasa MI, Stephen HH et al. Reverse-transcribed SARS-CoV-2 RNA can integrate into the genome of cultured human cells and can be expressed in patient-derived tissues. Proc. Natl. Acad. Sci. USA 2021, 118, e2105968118. [CrossRef]
- Dorneles, M.L.d.A.; Cardoso-Lima, R.; Souza, P.F.N.; Rosa, D.S.; Magne, T.M.; Santos-Oliveira, R.; Alencar, L.M.R. Zika Virus (ZIKV): A New Perspective on the Nanomechanical and Structural Properties. Viruses 2022, 14, 1727. [Google Scholar] [CrossRef]
- Barba-Spaeth, G.; Dejnirattisai, W.; Rouvinski, A.; Vaney, M.-C.; Medits, I.; Sharma, A.; Simon-Lorière, E.; Sakuntabhai, A.; Cao-Lormeau, V.-M.; Haouz, A.; et al. Structural basis of potent Zika–dengue virus antibody cross-neutralization. Nature 2016, 536, 48–53. [Google Scholar] [CrossRef]
- Figueiredo, C.P.; Barros-Aragão, F.G.Q.; Neris, R.L.S.; Frost, P.S.; Soares, C.; Souza, I.N.O.; Zeidler, J.D.; Zamberlan, D.C.; de Sousa, V.L.; Souza, A.S.; et al. Zika virus replicates in adult human brain tissue and impairs synapses and memory in mice. Nat. Commun. 2019, 10, 1–16. [Google Scholar] [CrossRef]
- Azevedo RS, de Sousa JR, Araujo MTF, Filho AJM et al. In Situ Immune Response and Mechanisms of Cell Damage in Central Nervous System of Fatal Cases Microcephaly by Zika Virus. Sci. Rep. 2017, 8, 5.
- Roth, H.; Schneider, L.; Eberle, R.; Lausen, J.; Modlich, U.; Blümel, J.; Baylis, S.A. Zika virus infection studies with CD34 + hematopoietic and megakaryocyte-erythroid progenitors, red blood cells and platelets. Transfusion 2020, 60, 561–574. [Google Scholar] [CrossRef]
- Balzanelli, M.G.; Distratis, P.; Dipalma, G.; Vimercati, L.; Inchingolo, A.D.; Lazzaro, R.; Aityan, S.K.; Maggiore, M.E.; Mancini, A.; Laforgia, R.; et al. Sars-CoV-2 Virus Infection May Interfere CD34+ Hematopoietic Stem Cells and Megakaryocyte–Erythroid Progenitors Differentiation Contributing to Platelet Defection towards Insurgence of Thrombocytopenia and Thrombophilia. Microorganisms 2021, 9, 1632. [Google Scholar] [CrossRef]
- Heazlewood, S.Y.; Ahmad, T.; Cao, B.; Cao, H.; Domingues, M.; Sun, X.; Heazlewood, C.K.; Li, S.; Williams, B.; Fulton, M.; et al. High ploidy large cytoplasmic megakaryocytes are hematopoietic stem cells regulators and essential for platelet production. Nat. Commun. 2023, 14, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Balzanelli, M.G.; Distratis, P.; Lazzaro, R.; Pham, V.H.; Tran, T.C.; Dipalma, G.; Bianco, A.; Serlenga, E.M.; Aityan, S.K.; Pierangeli, V.; et al. Analysis of Gene Single Nucleotide Polymorphisms in COVID-19 Disease Highlighting the Susceptibility and the Severity towards the Infection. Diagnostics 2022, 12, 2824. [Google Scholar] [CrossRef] [PubMed]
- Rossi, F.; Josey, B.; Sayitoglu, E.C.; Potens, R.; Sultu, T.; Duru, A.D.; Beljanski, V. Characterization of zika virus infection of human fetal cardiac mesenchymal stromal cells. PLOS ONE 2020, 15, e0239238. [Google Scholar] [CrossRef] [PubMed]
- Strange, D.P.; Jiyarom, B.; Pourhabibi Zarandi, N.; Xie, X.; Baker, C.; Sadri-Ardekani, H.; Shi, P.-Y.; Verma, S. Axl Promotes Zika Virus Entry and Modulates the Antiviral State of Human Sertoli Cells. mBio 2019, 10. [Google Scholar] [CrossRef]
- Hastings, A.K.; Hastings, K.; Uraki, R.; Hwang, J.; Gaitsch, H.; Dhaliwal, K.; Williamson, E.; Fikrig, E. Loss of the TAM Receptor Axl Ameliorates Severe Zika Virus Pathogenesis and Reduces Apoptosis in Microglia. iScience 2019, 13, 339–350. [Google Scholar] [CrossRef]
- Wang, S.; Qiu, Z.; Hou, Y.; Deng, X.; Xu, W.; Zheng, T.; Wu, P.; Xie, S.; Bian, W.; Zhang, C.; et al. AXL is a candidate receptor for SARS-CoV-2 that promotes infection of pulmonary and bronchial epithelial cells. Cell Res. 2021, 31, 126–140. [Google Scholar] [CrossRef]
- Shastri, A.; Al Aiyan, A.; Kishore, U.; Farrugia, M.E. Immune-Mediated Neuropathies: Pathophysiology and Management. Int. J. Mol. Sci. 2023, 24, 7288. [Google Scholar] [CrossRef]
- Tulbă, D.; Popescu, B.O.; Manole, E.; Băicuș, C. Immune Axonal Neuropathies Associated With Systemic Autoimmune Rheumatic Diseases. Front. Pharmacol. 2021, 12. [Google Scholar] [CrossRef]
- Martyniak, A.; Tomasik, P.J. A New Perspective on the Renin-Angiotensin System. Diagnostics 2022, 13, 16. [Google Scholar] [CrossRef]
- Chen L, Li X, Chen M, Feng Y, Xiong C. The ACE2 expression in human heart indicates new potential mechanism of heart injury among patients infected with SARS-CoV-2. Cardiovascular 2020, 116, 1097–1100. [CrossRef] [PubMed]
- Dolhnikoff M, Ferreira Ferranti J, de Almeida Monteiro RA, Duarte-Neto A.N et al. SARS-CoV-2 in cardiac tissue of a child with COVID-19-related multisystem inflammatory syndrome. Lancet Child Adolescent. Health 2020, 4, 790–794. [CrossRef] [PubMed]
- Marchiano, S.; Hsiang, T.-Y.; Khanna, A.; Higashi, T.; Whitmore, L.S.; Bargehr, J.; Davaapil, H.; Chang, J.; Smith, E.; Ong, L.P.; et al. SARS-CoV-2 Infects Human Pluripotent Stem Cell-Derived Cardiomyocytes, Impairing Electrical and Mechanical Function. Stem Cell Rep. 2021, 16, 478–492. [Google Scholar] [CrossRef] [PubMed]
- Jura, M.; Garus, M.; Krakowska, K.; Urban, S.; Błaziak, M.; Iwanek, G.; Zymliński, R.; Biegus, J.; Paleczny, B. A Methodological Perspective on the Function and Assessment of Peripheral Chemoreceptors in Heart Failure: A Review of Data from Clinical Trials. Biomolecules 2022, 12, 1758. [Google Scholar] [CrossRef]
- Grassi, G.; Seravalle, G.; Cattaneo, B.M.; Lanfranchi, A.; Vailati, S.; Giannattasio, C.; Del Bo, A.; Sala, C.; Bolla, G.B.; Pozzi, M.; et al. Sympathetic Activation and Loss of Reflex Sympathetic Control in Mild Congestive Heart Failure. Circulation 1995, 92, 3206–3211. [Google Scholar] [CrossRef]
- DeBurgh Daly M, Scott MJ. An Analysis of the Primary Cardiovascular Reflex Effects of Stimulation of the Carotid Body Chemoreceptors in the Dog. J. Physiol. 1962, 162, 555–573. [CrossRef]
- Daly MD, Scott MJ. The Cardiovascular Responses to Stimulation of the Carotid Body Chemoreceptors in the Dog. J. Physiol. 1963, 165, 179–197. [CrossRef]
- Lenka, N.; Krishnan, S.; Board, P.; Rangasamy, D. Exploiting the power of LINE-1 retrotransposon mutagenesis for identification of genes involved in embryonic stem cell differentiation. Stem Cell Rev. Rep. 2014, 10, 408–416. [Google Scholar] [CrossRef]
- Esposito, M.; Gualandi, N.; Spirito, G.; Ansaloni, F.; Gustincich, S.; Sanges, R. Transposons Acting as Competitive Endogenous RNAs: In-Silico Evidence from Datasets Characterised by L1 Overexpression. Biomedicines 2022, 10, 3279. [Google Scholar] [CrossRef]
- Blackburn, E.H. TELOMERASES. Annu. Rev. Biochem. 1992, 61, 113–129. [Google Scholar] [CrossRef]
- Balzanelli M, Distratis P, Lazzaro Rita, Pham VH et al. The Anti-Viral Activity of Stem Cells: A Rational Explanation for their Use in Clinical Application. Bentham Science Publishers 2023, 23, 738–747.
- Blackburn, E.H.; Collins, K. Telomerase: An RNP Enzyme Synthesizes DNA. Cold Spring Harb. Perspect. Biol. 2010, 3, a003558–a003558. [Google Scholar] [CrossRef] [PubMed]
- Gargiulo Isacco C, Pham Hung V, Nguyen Cao DK, Vo Lh, T et al. Autologous Peripheral Blood Stem Cells Increase the Telomere Length in Patients: A Case Report of 13 Patients. Journal of Stem Cell Research & Therapy 2016, 6, 1–6.
- AbdelMassih, A.; Agha, H.; El-Saiedi, S.; El-Sisi, A.; El Shershaby, M.; Gaber, H.; Ismail, H.-A.; El-Husseiny, N.; Amin, A.R.; ElBoraie, A.; et al. The role of miRNAs in viral myocarditis, and its possible implication in induction of mRNA-based COVID-19 vaccines-induced myocarditis. Bull. Natl. Res. Cent. 2022, 46, 1–8. [Google Scholar] [CrossRef]
- Siu, R.W.C.; Fragkoudis, R.; Simmonds, P.; Donald, C.L.; Chase-Topping, M.E.; Barry, G.; Attarzadeh-Yazdi, G.; Rodriguez-Andres, J.; Nash, A.A.; Merits, A.; et al. Antiviral RNA Interference Responses Induced by Semliki Forest Virus Infection of Mosquito Cells: Characterization, Origin, and Frequency-Dependent Functions of Virus-Derived Small Interfering RNAs. J. Virol. 2011, 85, 2907–2917. [Google Scholar] [CrossRef]
- Mishra, R.; Kumar, A.; Ingle, H.; Kumar, H. The Interplay Between Viral-Derived miRNAs and Host Immunity During Infection. Front. Immunol. 2020, 10, 3079. [Google Scholar] [CrossRef]
- Qiu, Y.; Xu, Y.; Zhang, Y.; Zhou, H.; Deng, Y.-Q.; Li, X.-F.; Miao, M.; Zhang, Q.; Zhong, B.; Hu, Y.; et al. Human Virus-Derived Small RNAs Can Confer Antiviral Immunity in Mammals. Immunity 2017, 46, 992–1004. [Google Scholar] [CrossRef]
- Henzinger, H.; Barth, D.A.; Klec, C.; Pichler, M. Non-Coding RNAs and SARS-Related Coronaviruses. Viruses 2020, 12, 1374. [Google Scholar] [CrossRef]
- Zambon, R.A.; Vakharia, V.N.; Wu, L.P. RNAi is an antiviral immune response against a dsRNA virus in Drosophila melanogaster. Cell. Microbiol. 2006, 8, 880–889. [Google Scholar] [CrossRef]
- Ding, S.-W.; Voinnet, O. Antiviral Immunity Directed by Small RNAs. Cell 2007, 130, 413–426. [Google Scholar] [CrossRef]
- Ghildiyal, M.; Seitz, H.; Horwich, M.D.; Li, C.; Du, T.; Lee, S.; Xu, J.; Kittler, E.L.; Zapp, M.L.; Weng, Z.; et al. Endogenous siRNAs Derived from Transposons and mRNAs in Drosophila Somatic Cells. Science 2008, 320, 1077–1081. [Google Scholar] [CrossRef] [PubMed]
- Shiu, P.K.T.; Ilieva, M.; Holm, A.; Uchida, S.; DiStefano, J.K.; Bronisz, A.; Yang, L.; Asahi, Y.; Goel, A.; Yang, L.; et al. The Non-Coding RNA Journal Club: Highlights on Recent Papers—12. Non-Coding RNA 2023, 9, 28. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



