Submitted:
17 July 2023
Posted:
19 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
Inclusion and Exclusion Criteria
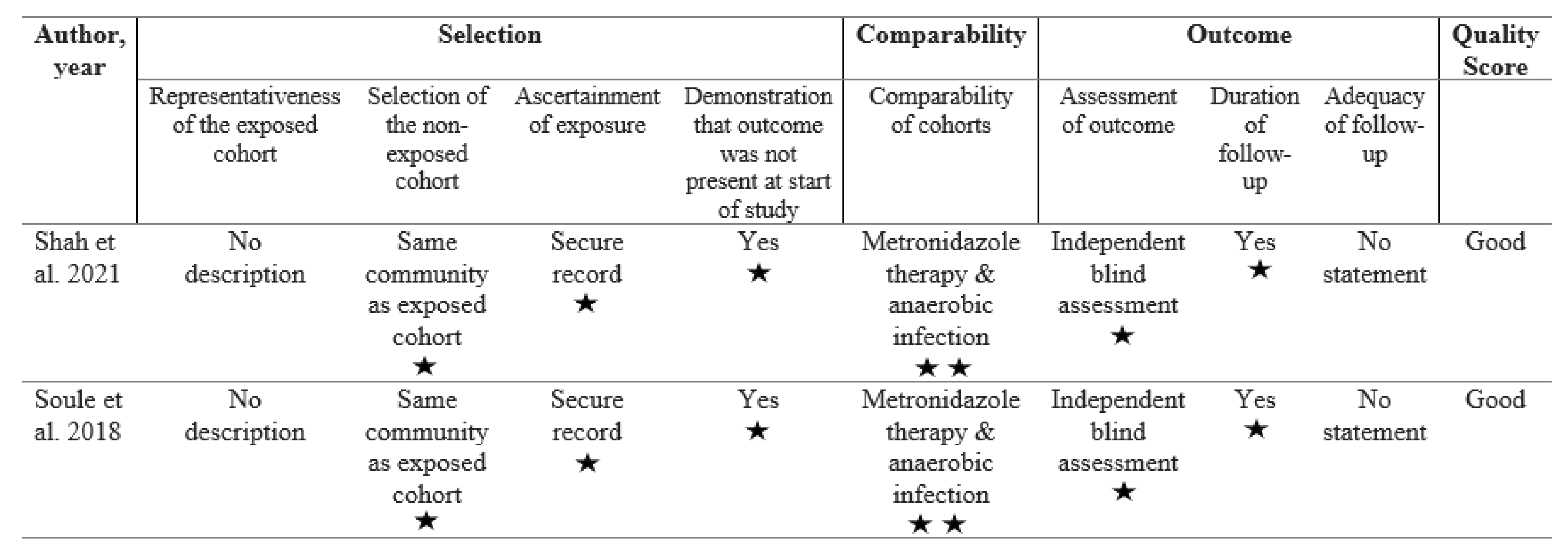
Study Quality
Data Extraction
Statistical Analysis
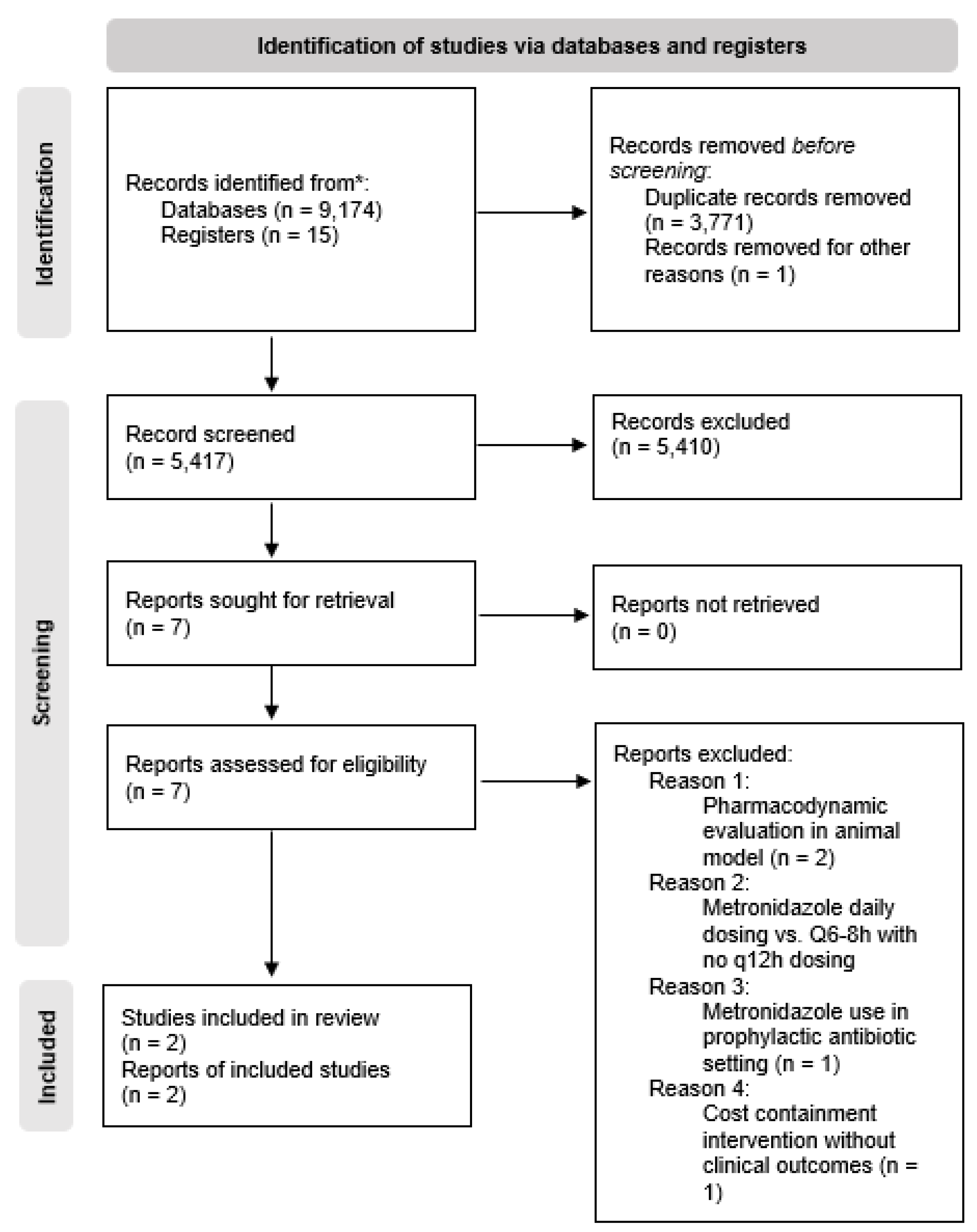
3. Results
Excluded Studies with Reasons
Included Studies and Quality Assessment
Single-Site Study
Multi-Site Study
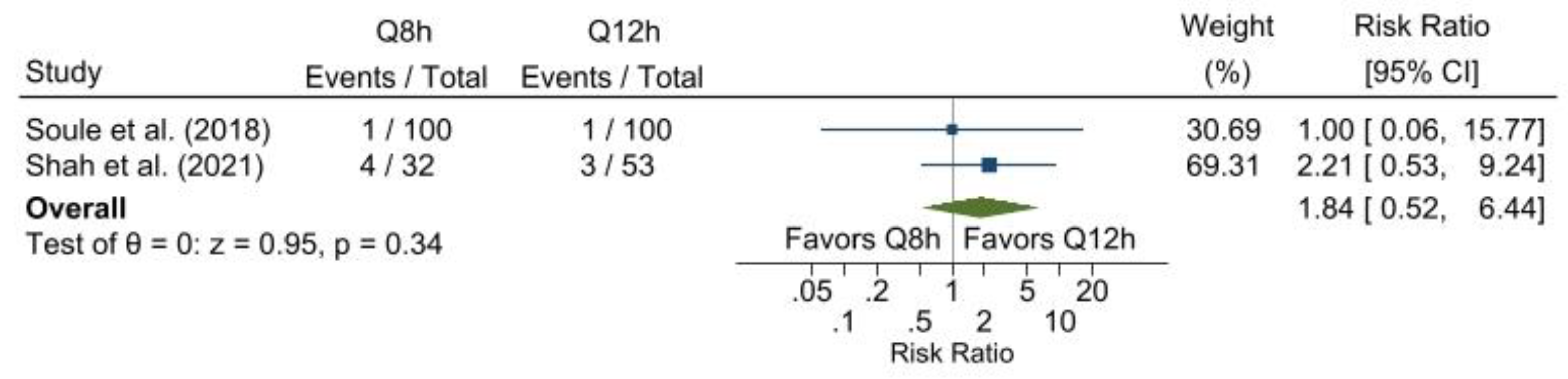
Meta-analysis for Escalation of Antibiotic Therapy
4. Discussion
Limitations
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Placidi, G. F.; Masouka, D.; Alcaraz, A.; Taylor, J. A.; Earle, R. Distribution and metabolism of 14C-metronidazole in mice. Archives Internationales de Pharmacodynamie et de Therapie 1970, 188. [Google Scholar]
- Ings, R.M.J.; McFadzean, J.A.; Ormerod, W.E. The Fate of Metronidazole and its Implications in Chemotherapy. Xenobiotica 1975, 5, 223–235. [Google Scholar] [CrossRef]
- Manthei, R.W.; Feo, L.G.; E Stambaugh, J. Identification of the metabolites of metronidazole in the human vagina. Wiadomosci Parazytol. 1969, 15. [Google Scholar]
- Lamp, K.C.; Freeman, C.D.; Klutman, N.E.; Lacy, M.K. Pharmacokinetics and Pharmacodynamics of the Nitroimidazole Antimicrobials. Clin. Pharmacokinet. 1999, 36, 353–373. [Google Scholar] [CrossRef] [PubMed]
- Bunz, D.; Gupta, S.; Jewesson, P. Metronidazole cost containment: a two-stage intervention. Hosp. Formul. 1990, 25. [Google Scholar]
- Richardson, W.S.; Wilson, M.C.; Nishikawa, J.; Hayward, R.S. The well-built clinical question: a key to evidence-based decisions. ACP J. club 1995, 123, A12–A13. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021, 372, 71. [Google Scholar] [CrossRef] [PubMed]
- Wells, G. A.; Shea, B.; O'Connell, D. Wells, G. A.; Shea, B.; O'Connell, D., et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomized studies in meta-analyses. (accessed May 9, 2023). 9 May.
- Agency for Healthcare Research and Quality Assessing the risk of bias of individual studies in systematic review of health care interventions. (accessed , 2023). 15 May.
- StataCorp Stata Statistical Software. 2021, 17. StataCorp Stata Statistical Software. 2021, 17.
- Earl, P.; Sisson, P.R.; Ingham, H.R. Twelve-hourly dosage schedule for oral and intravenous metronidazole. J. Antimicrob. Chemother. 1989, 23, 619–621. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, K.H.; Gunderson, B.W.; Hermsen, E.D.; Hovde, L.B.; Rotschafer, J.C. Pharmacodynamics of Pulse Dosing versus Standard Dosing: In Vitro Metronidazole Activity against Bacteroides fragilis and Bacteroides thetaiotaomicron. Antimicrob. Agents Chemother. 2004, 48, 4195–4199. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, C.; Earl, P. Metronidazole: two or three times daily—a comparative controlled clinical trial of the efficacy of two different dosing schedules of metronidazole for chemoprophylaxis following third molar surgery. Br. J. Oral Maxillofac. Surg. 1994, 32, 165–167. [Google Scholar] [CrossRef]
- Wang, S.; Cunha, B.; Hamid, N.; Amato, B.; Feuerman, M.; Malone, B. Metronidazole Single versus Multiple Daily Dosing in Serious Intraabdominal/Pelvic and Diabetic Foot Infections. J. Chemother. 2007, 19, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Soule, A.F.; Green, S.B.; Blanchette, L.M. Clinical efficacy of 12-h metronidazole dosing regimens in patients with anaerobic or mixed anaerobic infections. Ther. Adv. Infect. Dis. 2018, 5, 57–62. [Google Scholar] [CrossRef]
- Shah, S.; Adams, K.; Merwede, J.; McManus, D.; Topal, J. Three is a crowd: Clinical outcomes of a twice daily versus a thrice daily metronidazole dosing strategy from a multicenter study. Anaerobe 2021, 71, 102378. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention Pelvic inflammatory disease. https://www.cdc.gov/std/treatment-guidelines/pid.htm.
- Chey, W.D.; Leontiadis, G.I.; Howden, C.W.; Moss, S.F. ACG Clinical Guideline: Treatment of Helicobacter pylori Infection. Am. J. Gastroenterol. 2017, 112, 212–239. [Google Scholar] [CrossRef] [PubMed]
- A Dingsdag, S.; Hunter, N. Metronidazole: an update on metabolism, structure–cytotoxicity and resistance mechanisms. J. Antimicrob. Chemother. 2017, 73, 265–279. [Google Scholar] [CrossRef] [PubMed]
| Population | Hospitalized adolescents (≥ 12 years) and adult patients. |
| Intervention | Received metronidazole for a culture-confirmed or suspected anaerobic infection. |
| Comparison | We compared the patients who received intravenous or oral metronidazole on an every 8-hour versus an every 12-hour interval. |
| Outcome | Mortality is defined as death during the hospital encounter. The need to escalate antibiotic therapy is defined as the addition of at least 1 antimicrobial agent. |
| Database | Search Strategy Terms | Number of Retrieved Studies |
|---|---|---|
| Cochrane Central Register of Controlled Trials | Metronidazole; Filter: Title Only | 15 |
| Cochrane Database of Systematic Reviews | Metronidazole; Filter: Title, Abstract, or Keyword | 28 |
| PubMed | Metronidazole AND “extended dose” Metronidazole AND Dose; Filter: Title Metronidazole AND “12-hour” OR “12 hour” Metronidazole AND “twice daily” |
1 2,005 4,991 589 |
| Science Direct | Metronidazole, “dose” Metronidazole, “12 hour” OR “12-hour” Metronidazole AND “twice daily” |
63 3 89 |
| Scopus | Metronidazole AND “extended dose*” Metronidazole AND “extended dose* interval” Metronidazole AND “12-hour” OR “12 hour” Metronidazole AND “dose*” Metronidazole AND “twice daily” |
2 3 172 411 817 |
 |
| Author, year | Design | Country | Subjects With Q8h Dosing | Subjects With Q12h Dosing | Total Subjects |
|---|---|---|---|---|---|
| Shah et al. 2021 | Cohort | USA, 5 hospitals | 32 | 53 | 85 |
| Soule et al. 2018 | Cohort | USA, 1 hospital | 100 | 100 | 200 |
| Author, year |
Clinical Condition |
Prescribed Antibiotics |
Dosing Regimen |
Clinical Outcomes |
| Shah et al. 2021 | Hospitalized with anaerobic bacteremia | Metronidazole for ≥ 3 days | Intravenous or oral formulation dosed every 12 hours or every 8 hours | Mortality (30-day all-cause), post-infection hospital days, escalation of antibiotic therapy, 30-day readmission due to infection, repeat positive blood cultures |
| Soule et al. 2018 | Hospitalized with presumed anaerobic or mixed anaerobic infection | Metronidazole for ≥ 3 days | Intravenous or oral formulation dosed every 12 hours or every 8 hours | Clinical cure, escalation of antibiotic therapy, death |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




