Submitted:
14 July 2023
Posted:
17 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Material
2.3. Preparation of the Plant Extracts
2.4. Chemical Characterization Tests in Tubes
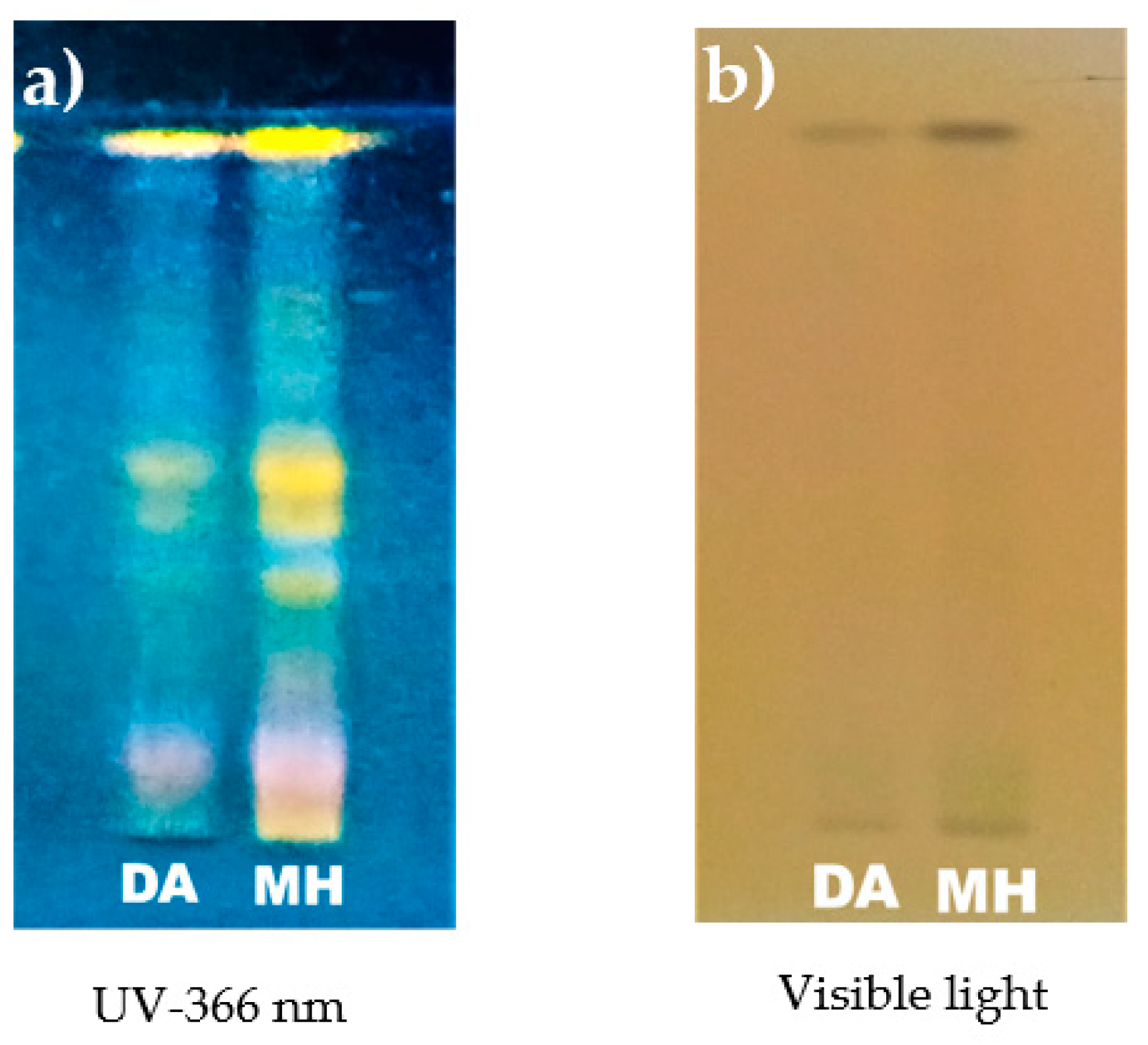
2.5. High-Performance Thin-Layer Chromatography
2.6. Phenolic Compounds Content
2.6.1. Determination of Total Phenolics
2.6.2. Determination of Total Hydrolyzable Tannins
2.6.3. Determination of Total Condensed Tannins
2.6.4. Determination of Total Flavonoids
2.7. Evaluation of Antioxidant Properties
2.7.1. 2,2-DPPH• Radical Scavenging Capacity
2.7.2. ABTS•+ Scavenging Capacity
2.7.3. Ferric-Reducing Power Assay
2.7.4. Lipid Peroxidation Inhibition (LPO) Assay
2.8. Anti-Inflammatory Activity
2.8.1. Lipoxygenase Inhibition Assay
2.8.2. Phospholipase A2 (sPLA2) inhibition assay
2.9. Experimental Animals
2.10. Acute General Toxicity
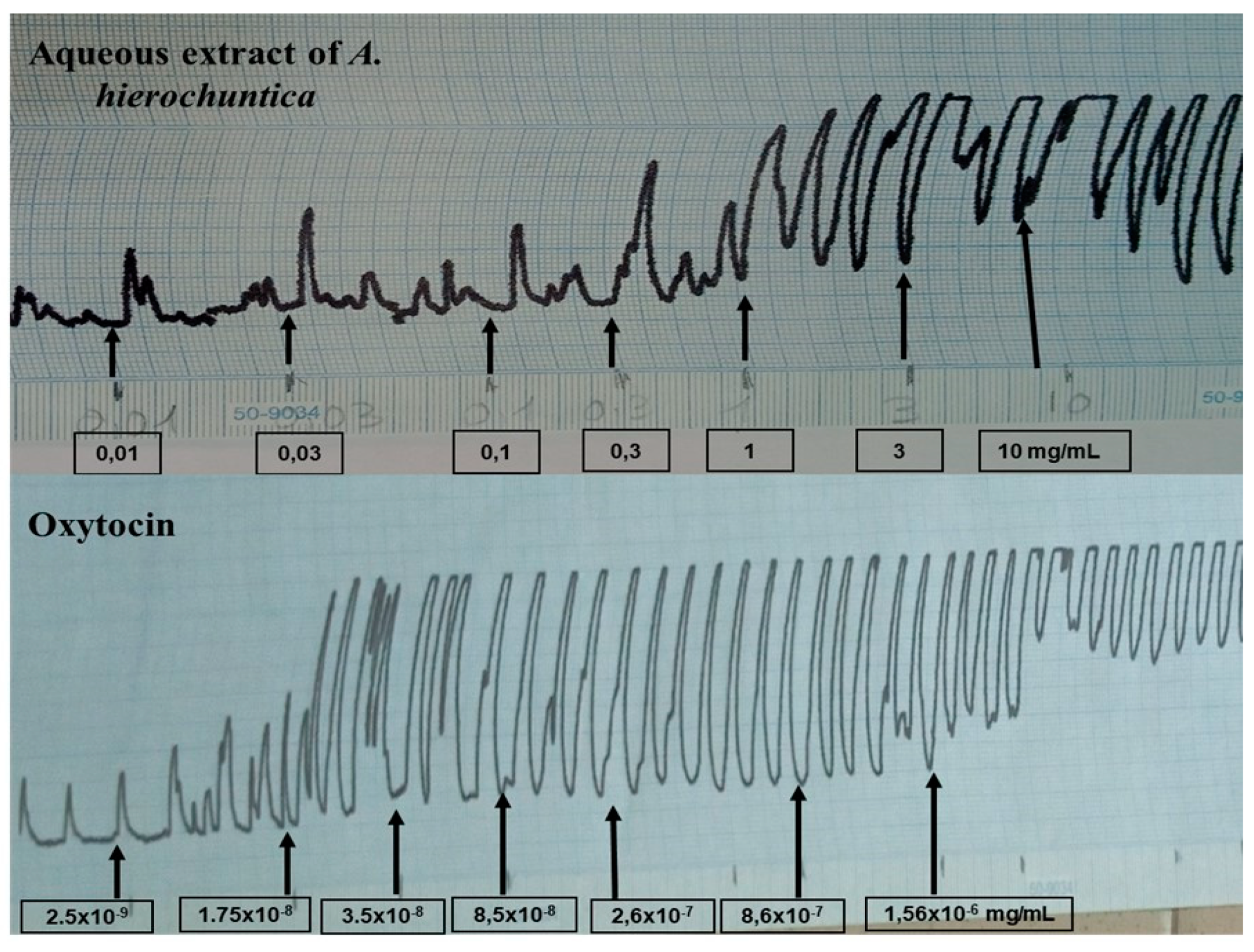
2.11. Evaluation of the Contractile Effect
2.11.1. Isolated Mouse Uterus Preparation
2.11.2. Drug Challenges
2.12. Statistical Analysis
3. Results
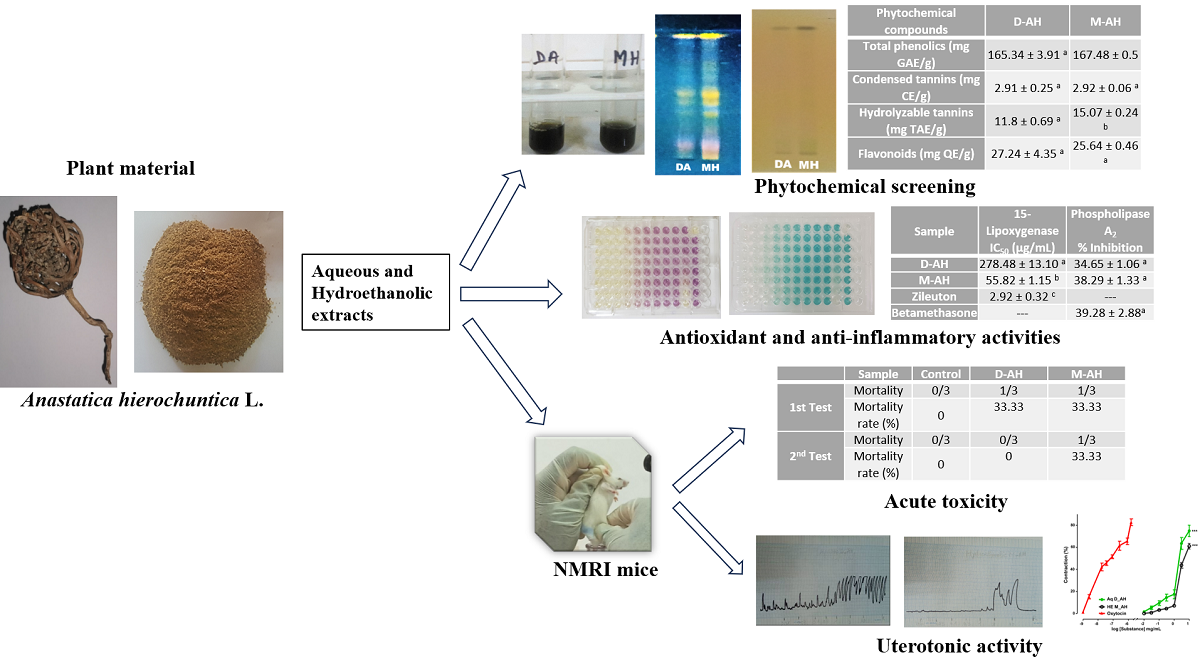
3.1. Preliminary Phytochemical Investigation
3.1.1. Phytochemical Screening
3.1.2. Contents of Total Phenolics, Tannins, and Flavonoids
3.2. Biological Activities
3.2.1. Antioxidant Activity
3.2.2. Anti-Inflammatory Activity
3.2.3. Acute Toxicity
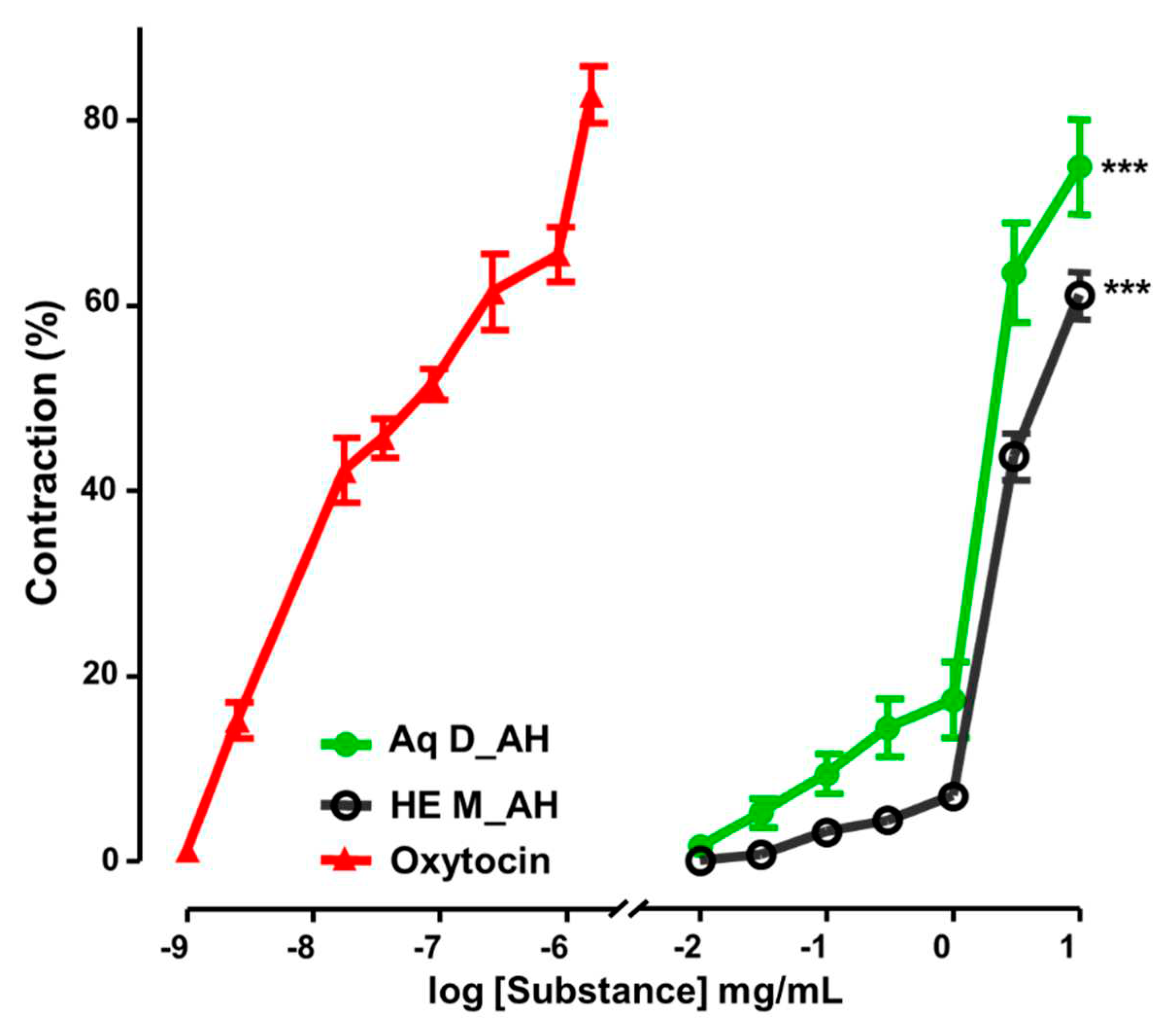
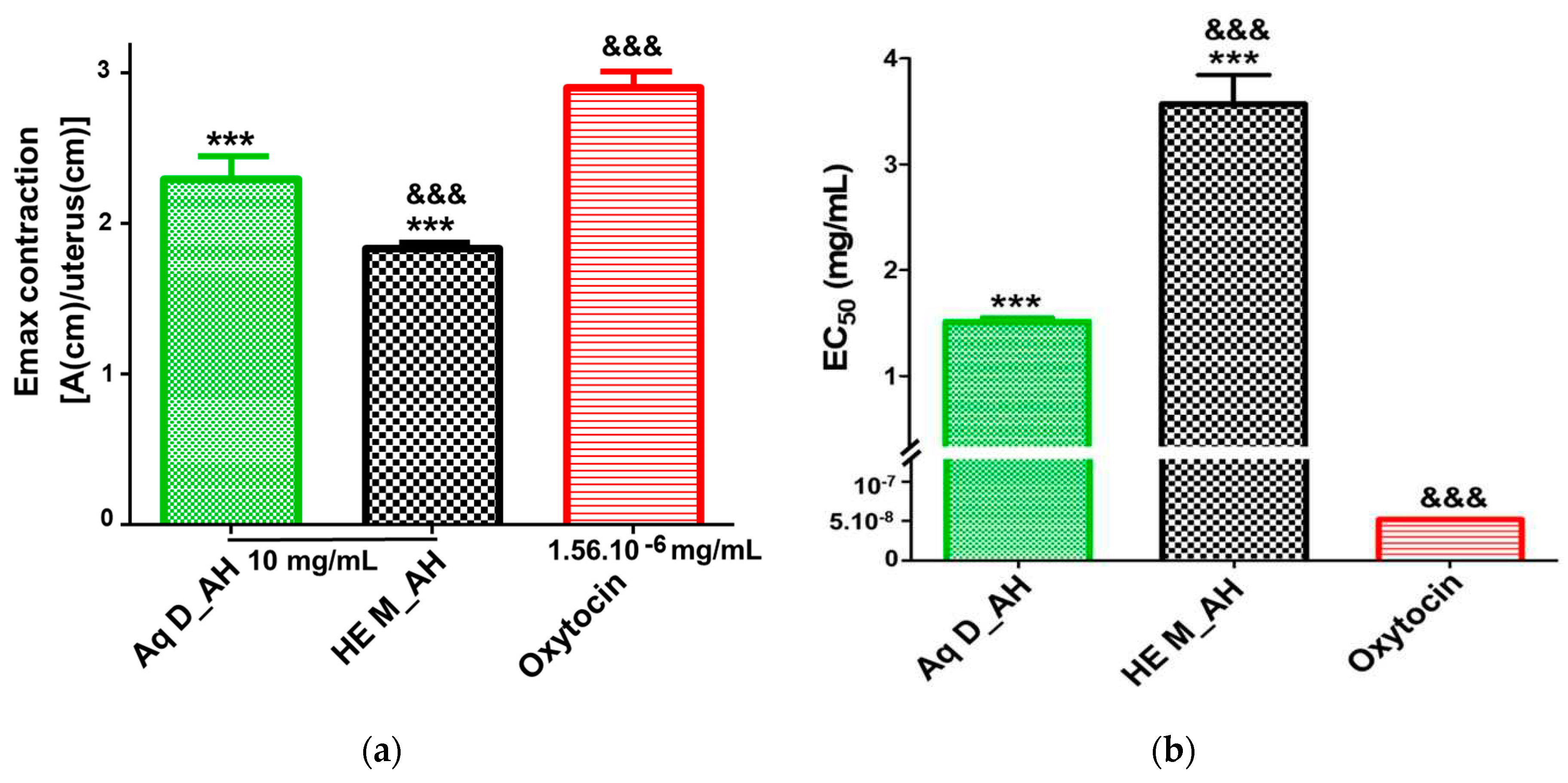
3.2.4. Uterine Contractility Effects of Extracts
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Creanga, A.A.; Syverson, C.; Seed, K.; Callaghan, W.M. Pregnancy-related mortality in the United States, 2011–2013. Obstet Gynecol. 2017, 130, 366–373. [Google Scholar] [CrossRef]
- Say, L.; Chou D Gemmill, A.; Tunçalp, Ö.; Moller, A.; Daniels, J.; Gülmezoglu, A.M.; Temmerman, M.; Alkema, L. Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health. 2014, 2, e323–e333. [Google Scholar] [CrossRef] [PubMed]
- Malik, M.; Roh, M.; England, S.K. Uterine contractions in rodent models and humans. Acta Physiol. 2021, 231, e13607.
- Page, K.; McCool, W.; Guidera, M. Examination of the pharmacology of oxytocin and clinical guidelines for use in labor. J Midwifery Womens Health. 2017, 62, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Kothari, B.; DeGolier, T. The contractile effects of Matricaria chamomilla on Mus musculus isolated uterine tissue. J. pharmacogn. Phytochem. 2022, 11, 303–308. [Google Scholar] [CrossRef]
- Zamawe, C.; King, C.; Jennings, H.; Mandiwa, C.; Fottrell, E. Effectiveness and safety of herbal medicines for induction of labour: A systematic review and metaanalysis. BMJ Open. 2018, 8, e022499. [Google Scholar] [CrossRef]
- Siricilla, S., Iwueke, C.C.; Herington, J.L. Drug discovery strategies for the identification of novel regulators of uterine contractility. Curr Opin Physiol. 2020, 13, 71–86. [CrossRef]
- Ngadjui, E.; Kouam, J.Y.; Fozin, G.R.B.; Momo, A.C.T.; Deeh, P.B.D.; Wankeu-Nya, M.; Nguelefack, T.B.; Watcho, P. Uterotonic Effects of Aqueous and Methanolic Extracts of Lannea acida in Wistar Rats: An In Vitro Study. Reprod Sci. 2021, 28, 2448–2457. [Google Scholar] [CrossRef]
- Monji, F.; Adaikan, P.G.; Lau, L.C.; Bin-Said, B.; Gong, Y.; Tan, H.M.; Choolani, M. Investigation of uterotonic properties of Ananas comosus extracts. J Ethnopharmacol. 2016, 193, 21–9. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, I.; Ghosal, S.; Pradhan, N. Jussiaea repens (L) acts as an uterotonic agent - an in vitro study. Int J Pharm Sci Rev Res. 2015, 27, 368–72. [Google Scholar]
- Abou-Elella, F.; Hanafy, E.A.; Gavamukulya, Y. Determination of antioxidant and antiinflammatory activities, as well as in vitro cytotoxic activities of extracts of Anastatica hierochuntica (Kaff Maryam) against HeLa cell lines. J Med Plants Res. 2016, 10, 77–87. [Google Scholar]
- Alatshan, A.; Esam, Q.; Wedyan, M.; Bseiso, Y.; Alzyoud, E.; Banat, R.; Alkhateeb, H. Antinociceptive and Antiinflammatory Activities of Anastatica hierochuntica and Possible Mechanism of Action. Indian J. Pharm. Sci. 2018, 80, 637–646. [Google Scholar] [CrossRef]
- Mohammd, T.U.; Baker, R.K.; Al-Ameri; K.A.H.; Abd-Ulrazzaq, S.S. Cytotoxic Effect of Aqueous Extract of Anastatica hierochuntica L. on AMN-3 Cell Line in vitro. Adv. life sci. technol. 2015, 31, 59–63.
- Baker, R.K.; Mohammd, T.U.; Ali, B.H.; Jameel, N.M. The Effect Of Aqueous Extract Of Anastatica Hierochuntica On Some Hormones In Mouse Females. Ibn al-Haitham j. pure appl. sci. 2017, 26, 198–205. [Google Scholar]
- Rahmy, T.R.; El-Ridi, M.R. Action of Anastatica hierochuntica plant extract on Islets of Langerhans in normal and diabetic rats. Egypt. J. Biol. 2002, 4, 87–94. [Google Scholar]
- Feryal, S.; Hasan, F.A.; Ali, S.M. Effect of alcoholic Anastatica hierochuntica extract on some biochemical and histological parameters in alloxan induced diabetic rats. Iraqi J. Sci. 2011, 52, 445–455. [Google Scholar]
- Yoshikawa, M.; Xu, F.; Morikawa, T.; Ninomiya, K.; Matsuda, H. Anastatin A and B, new skeletal flavonoids with hepatoprotective activities from the desert plant Anastatica hierochuntica. Bioorganic Med. Chem. Lett. 2003, 13, 1045–1049. [Google Scholar] [CrossRef]
- Daur, H. Chemical properties of the medicinal herb Kaff Maryam (Anastatica hierochuntica L.) and its relation to folk medicine use. Afr. J. Microbiol. Res. 2012, 6, 5048–5051. [Google Scholar]
- Boly, R.; Yabre, Z.; Nitiema, M.; Yaro, B.; Yoda, J.; Belemnaba, L.; Ilboudo, S.; Youl, N.H.E.; Guissou, I.P.; Ouedraogo, S. Pharmacological Evaluation of the Bronchorelaxant Effect of Waltheria indica L. (Malvaceae) Extracts on Rat Trachea. Evid Based Complement Alternat Med. 2021, 2021, 5535727. [Google Scholar]
- Parker, J.E.; Chimere, U.Y.; Chinenye, N.P.; Uzoma, I.; Echezona, E.; Innocent, O.O.; Paul, N.A.C. Phytochemical and toxicological studies of methanol and chloroform fractions of Acanthus montanus leaves. J. Biol. Sci. 2021, 21, 52–58. [Google Scholar] [CrossRef]
- Ciulei, I. Methodology for Analysis of Vegetable Drugs. Practical Manual on the Industrial Utilisation of Medicinal and Aromatic Plants, Ed. Ministry of chemical industry, Romania, 1982; pp. 1-62.
- Ouedraogo, W.R.C.; Belemnaba, L.; Nitiema, M.; Kabore, B.; Ouedraogo, N.; Koala, M.; Semde, R.; Ouedraogo, S. Antioxidant and Vasorelaxant Properties of Phaseolus vulgaris Linn (Fabaceae) Immature Pods Extract on the Thoracic Aorta of NMRI Mice. Biomed Pharmacol J. 2023, 16, 533–548. [Google Scholar] [CrossRef]
- Nitthikan, N.; Leelapornpisid, P.; Naksuriya, O.; Intasai, N.; Kiattisin, K. Potential and Alternative Bioactive Compounds from Brown Agaricus bisporus Mushroom Extracts for Xerosis Treatment. Sci. Pharm. 2022, 90, 59. [Google Scholar] [CrossRef]
- Belem-Kabré, W.L.M.E., Kaboré B, Compaoré-Coulibaly A., Traoré T.K., Thiombiano E.A.M., Nebié-Traoré M., Compaoré M., Kini F.B., Ouédraogo S., Kiendrebeogo M., Ouédraogo N., Phytochemical and biological investigations of extracts from the roots of Cocos nucifera L. (Arecaceae) and Carica papaya L. (Caricaceae), two plants used in traditional medicine, Afr. J. Biochem. Res. 2021, 15, 28-35.
- Kaboré, B.; Koala, M.; Ouedraogo, C.W.R.; Belemnaba, L.; Nitiema, M.; Compaoré, S.; Ouedraogo, S.; Ouedraogo, N.; Dabiré, C.M.; Kini, F.B.; Palé, E.; Ouedraogo, S. High-Performance Thin-Layer Chromatography Phytochemical Profiling, Antioxidant Activities, and Acute Toxicity of Leaves Extracts of Lannea velutina A. Rich. J. Med. Chem. Sci., 2023, 6, 410–423. [Google Scholar]
- Arvouet-Grand, A.; Vennat, B.; Pourrat, A.; Legret, P. Standardisation d’un extrait de Propolis et identification des principaux constituants. J Pharm Belg. 1994, 49, 462–468. [Google Scholar] [PubMed]
- Adeyemi, J.O. In Vitro Cytotoxicity and Antioxidant Studies of Dovyallis caffra-Mediated Cassiterite (SnO2) Nanoparticles. Sci. Pharm. 2023, 91, 34. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Poteggent, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–123. [Google Scholar] [CrossRef]
- Moualek, I.; Aiche, G.I.; Guechaoui, N.M.; Lahcene, S.; Houali, K. Antioxidant and anti-inflammatory activities of Arbutus unedo aqueous extract. Asian Pac J Trop Biomed. 2016, 6, 937–944. [Google Scholar] [CrossRef]
- Sombié, E.N.; Traoré, T.K.; Derra, A.N.; N’do, J.Y.P.; Belem-Kabré, W.L.M.E.; Ouédraogo, N.; Hilou, A.; Tibiri, A. Anti-Fibrotic Effects of Calotropis procera (Ait.) R.Br Roots Barks against Diethylnitrosamine-Induced Hepatic Fibrosis in Rats. J. Biosci. Med. 2023, 11, 332–349. [Google Scholar]
- National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals. Guide For the Care and Use of Laboratory Animals, 8th Ed.; National Academies Press, Washington, 2011; 246p.
- Nitiéma, M.; Ilboudo, S.; Belemnaba, L.; Ouédraogo, G.G.; Ouédraogo, S.; Ouédraogo, N.; Ouedraogo, S.; Guissou, I.P. Acute and sub-acute toxicity studies of aqueous decoction of the trunk barks from Lannea microcarpa Engl. and K. Krause (Anacardiaceae) in rodents. World j. pharm. pharm. Sci. 2018, 7, 30–42. [Google Scholar]
- OECD/OCDE, Guidelines for the testing of chemicals. Guideline 423: Acute oral toxicity-Acute toxic class method. revised method adopted 17th December 2001, Section 4.; 2001; pp. 1-14.
- Omwenga, E.O.; Hensel, A.; Pereira, S.; Shitandi, A.A.; Goycoolea, F.M. Antiquorum sensing, antibiofilm formation and cytotoxicity activity of commonly used medicinal plants by inhabitants of Borabu sub-county, Nyamira County, Kenya. Plos one. 2017, 12, e0185722. [Google Scholar] [CrossRef] [PubMed]
- Md Zin, S.R.; Kassim, N.M.; Mohamed, Z.; Fateh, A.H.; Alshawsh, M.A. Potential toxicity effects of Anastatica hierochuntica aqueous extract on prenatal development of Sprague-Dawley rats. J Ethnopharmacol. 2019, 245, 112180. [Google Scholar] [CrossRef] [PubMed]
- Kausilya, S.R.; Farah, S.T.; Mazidah, M.Z.A.; Mohammad, R.I.S. Effect of pre-treatment and different drying methods on the physicochemical properties of Carica papaya L. leaf powder, J. Saudi Soc. Agric. Sci. 2019, 18, 150–156. [Google Scholar]
- Van Wyk, B.E.; Wink. M. Medicinal Plants of the World: An Illustrated Scientific Guide to Important Medicinal Plants and Their Uses, 1st ed.; Timber Press, Portland, 2004; 480p.
- Hansen, C.J.; Siricilla, S.; Boatwright, N.; Rogers, J.H.; Kumi, M.E.; Herington, J. Effects of Solvents, Emulsions, Cosolvents, and Complexions on Ex Vivo Mouse Myometrial Contractility. Reprod. Sci. 2022, 29, 586–595. [Google Scholar] [CrossRef]
- Plaskova, A.; Mlcek, J. New insights of the application of water or ethanol-water plant extract rich in active compounds in food. Front Nutr. 2023, 28, 10:1118761. [Google Scholar] [CrossRef]
- Corriveau, S.; Rousseau, E.; Berthiaume, M.; Pasquier, J.C. Lipoxygenase and cyclooxygenase inhibitors reveal a complementary role of arachidonic acid derivatives in pregnant human myometrium. Am J Obstet Gynecol. 2010, 203, 266.e1–7. [Google Scholar] [CrossRef] [PubMed]
- Romaniewicz, M.; Całka, J.; Jana, B. Effects of Substance P and Neurokinin A on the Contractile Activity of Inflamed Porcine Uterus. Int. J. Mol. Sci. 2022, 23, 13184. [Google Scholar]
- Blanks, A.M..; Shmygol, A.; Thornton, S. Regulation of oxytocin receptors and oxytocin receptor signaling. Semin. Reprod. Med. 2007, 25, 52–9. [CrossRef]
- Arrowsmith, S.; Wray, S. Oxytocin: its mechanism of action and receptor signalling in the myometrium. J. Neuroendocrinol. 2014, 26, 356–69. [Google Scholar] [CrossRef]
- Porta, M.; Boening, A.; Tiemann, J.; Zack, A.; Patel, A.; Sondgeroth, K. The Contractile Response to Oxytocin in Non-pregnant Rat Uteri Is Modified After the First Pregnancy. Reprod. Sci. 2023, 30, 2152–2165. [Google Scholar] [CrossRef]
- Veiga, G.A.; Milazzotto, M.P.; Nichi, M.; Lúcio, C.F.; Silva, L.C.; Angrimani, D.S.; Vannucchi, C.I. Gene expression of estrogen and oxytocin receptors in the uterus of pregnant and parturient bitches. Braz J Med Biol Res. 2015, 48, 339–43. [Google Scholar] [CrossRef] [PubMed]
- Astutik, H.; Santoso, B.; Agil, M. The Effect of Anastatica hierochuntica L. Extract on the Histology of Myometrial Cells and Prostaglandin Levels (PGE2, PGF2α) in Pregnant Mice. Advances in Health Sciences Research. 2019, 22, 81–86. [Google Scholar]
- Adebisi, M.I; Alka, H. Uterotonic effect of aqueous extract of Launaea taraxacifolia Willd on rat isolated uterine horns. Afr. J. Biotechnol. 2019, 18, 399–407. [Google Scholar]

| Phytochemical analysis | Extracts | |||
|---|---|---|---|---|
| D-AH | M-AH | Powder | ||
|
Phytoconstituents Steroidal and triterpene glycosides |
+ |
+ |
--- |
|
| Alkaloids salts | - | - | --- | |
| Flavone glycosides | + | + | --- | |
| Saponins | + | + | --- | |
| Anthracenosides | - | - | --- | |
| Tannins | + | + | --- | |
| Cardenolides | - | - | --- | |
| Reducing compounds | + | + | --- | |
| Coumarins | + | + | --- | |
| Residual moisture content (%) | --- | --- | 6.51 ± 0.08 | |
| Extract yield (%) | 8.79 | 5.91 | --- | |
| Phytochemical compounds | D-AH | M-AH |
|---|---|---|
| Total phenolics (mg GAE/g) | 165.34 ± 3.91 a | 167.48 ± 0.5 a |
| Condensed tannins (mg CE/g) | 2.91 ± 0.25 a | 2.92 ± 0.06 a |
| Hydrolyzable tannins (mg TAE/g) | 11.8 ± 0.69 a | 15.07 ± 0.24 b |
| Flavonoids (mg QE/g) | 27.24 ± 4.35 a | 25.64 ± 0.46 a |
| Sample | DPPH | ABTS | FRAP | LPO | ||
|---|---|---|---|---|---|---|
| IC50 (µg/mL) | ARP | IC50 (µg/mL) | ARP | mmol AAE/100g | % Inhibition | |
| D-AH | 66.20 ± 0.60 a | 0.015 | 16.87 ± 0.49 a | 0.06 | 12.60 ± 0.34 a | 66.97 ± 1.46 a |
| M-AH | 63.56 ± 1.34 b | 0.016 | 33.92 ± 0.6 b | 0.03 | 13.56 ± 0.43 a | 73.97 ± 1.03 b |
| AA | 1.82 ± 0.02 c | 0.55 | 0.94 ± 0.08 c | 1.06 | --- | 95.66 ± 0.27 c |
| Sample | 15-Lipoxygenase IC50 (µg/mL) |
Phospholipase A2 % Inhibition |
|---|---|---|
| D-AH | 278.48 ± 13.10 a | 34.65 ± 1.06 a |
| M-AH | 55.82 ± 1.15 b | 38.29 ± 1.33 a |
| Zileuton | 2.92 ± 0.32 c | --- |
| Betamethasone | --- | 39.28 ± 2.88a |
| Sample | Control | D-AH | M-AH | |
|---|---|---|---|---|
| 1st Test | Mortality | 0/3 | 1/3 | 1/3 |
| Mortality rate (%) | 0 | 33.33 | 33.33 | |
| 2nd Test | Mortality | 0/3 | 0/3 | 1/3 |
| Mortality rate (%) | 0 | 0 | 33.33 |
| Day 0 | Day 1 | Day 2 | Day 3 | Day 7 | Day 14 | |
|---|---|---|---|---|---|---|
| Control | 26 ± 2.8 | 26.5 ± 2.1 | 27.5 ± 3.5 | 28 ± 2.8 | 28 ± 2.8 | 29.5 ± 3.5 |
| D-AH | 27.3 ± 1.2 | 28 ± 1.3 | 29 ± 1 | 29.8 ± 1.3 | 30.4 ± 0.9 | 31.4 ± 0.9 |
| M-AH | 26.2 ± 0.8 | 26.2 ± 0.8 | 27.3 ± 1 | 28 ± 1.4 | 28.5 ± 1.3 | 31.8 ± 1.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




