Submitted:
14 July 2023
Posted:
17 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Drugs and treatment
2.3. In vivo experiments
2.4. Quantitative real-time PCR
2.5. Western blot analysis
2.6. In vitro experiments
2.6.1. Cell culture
2.6.2. Cell differentiation and viability
2.7. Statistical analysis
3. Results
3.1. Assessment of chronic OLE treatment in neural plasticity indices in PFC
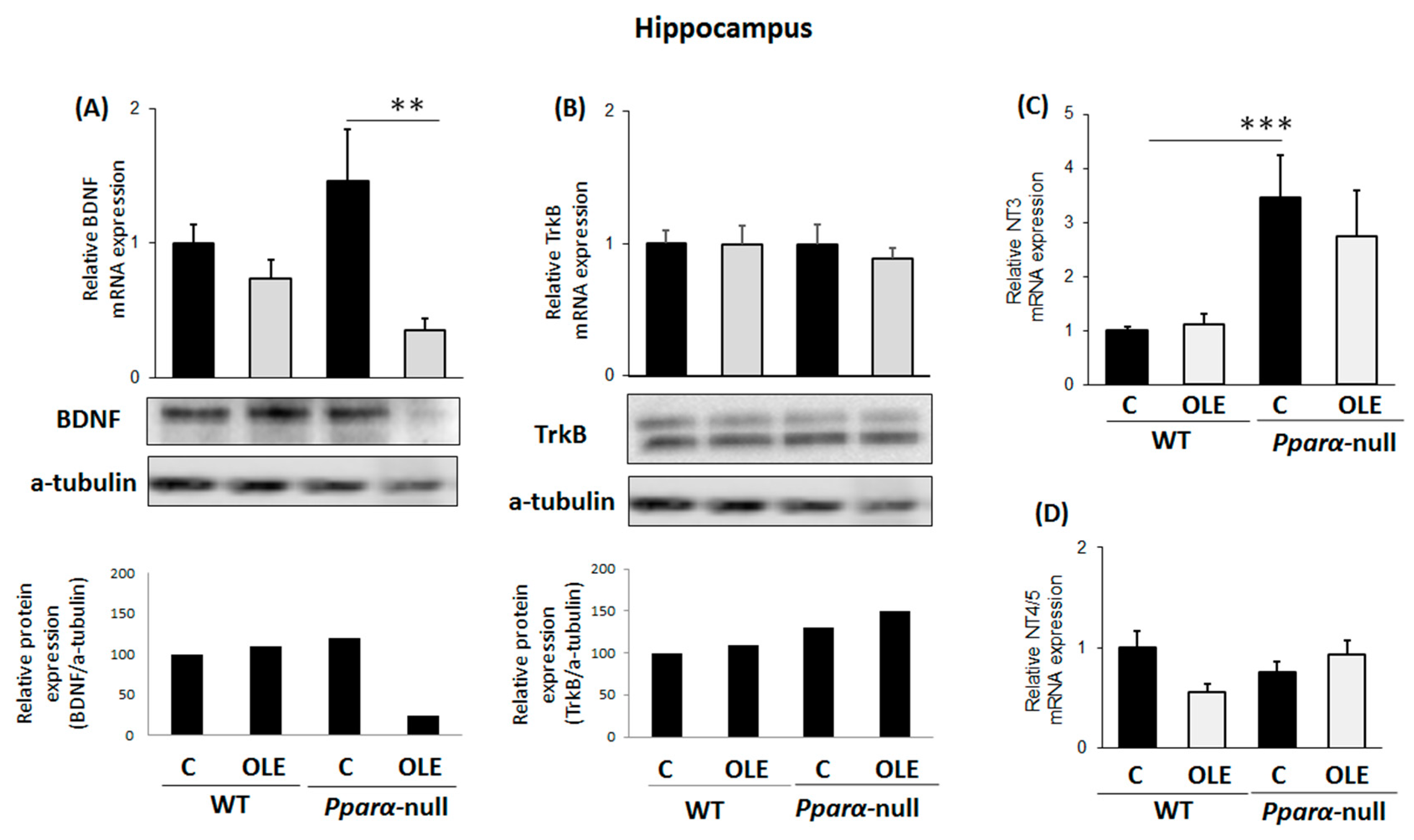
3.2. Assessment of chronic OLE treatment on neural plasticity indices in the hippocampus
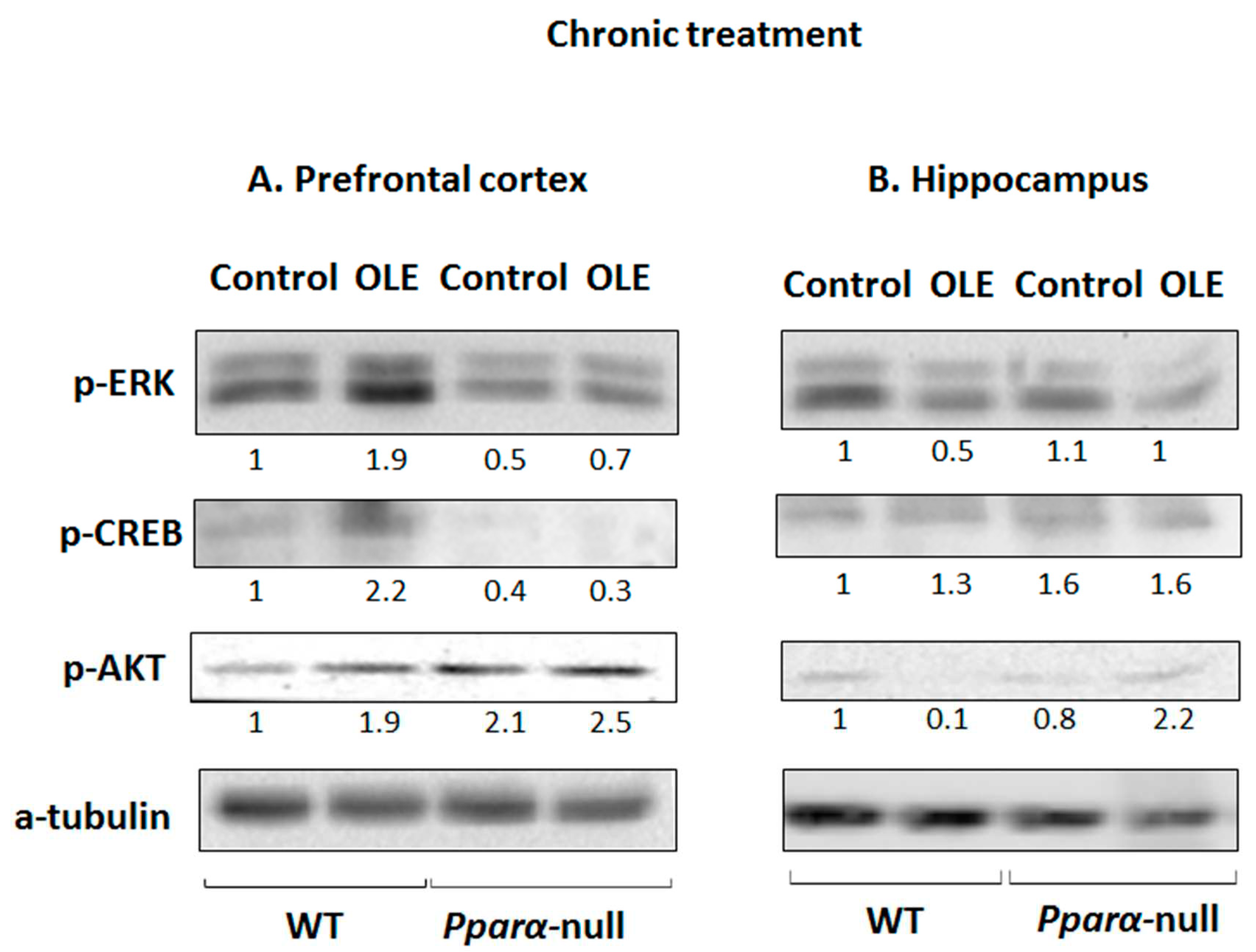
3.3. OLE-induced ERK, AKT and PKA/CREB activation
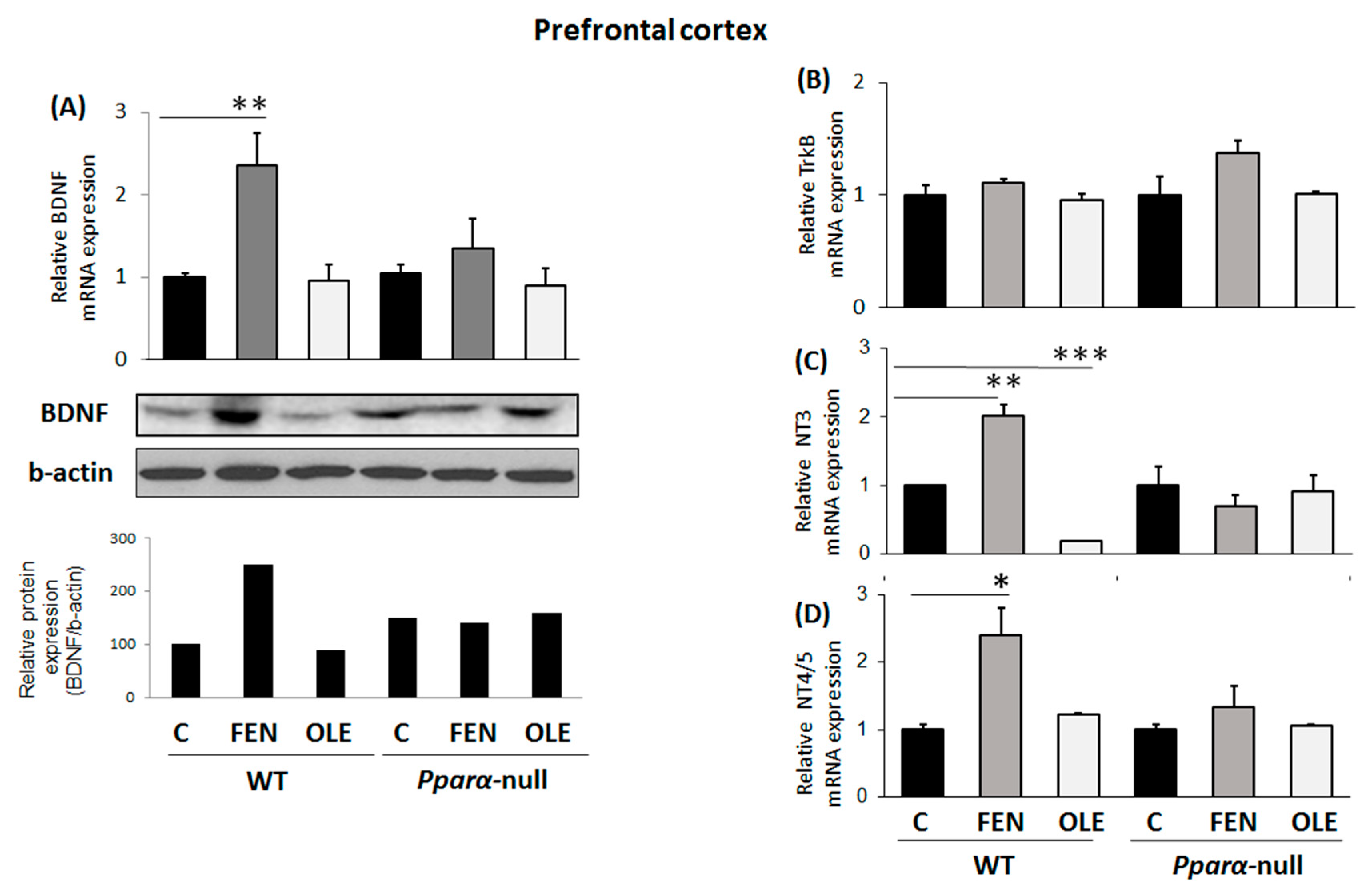
3.4. Assessment of subacute OLE treatment in neural plasticity indices in PFC
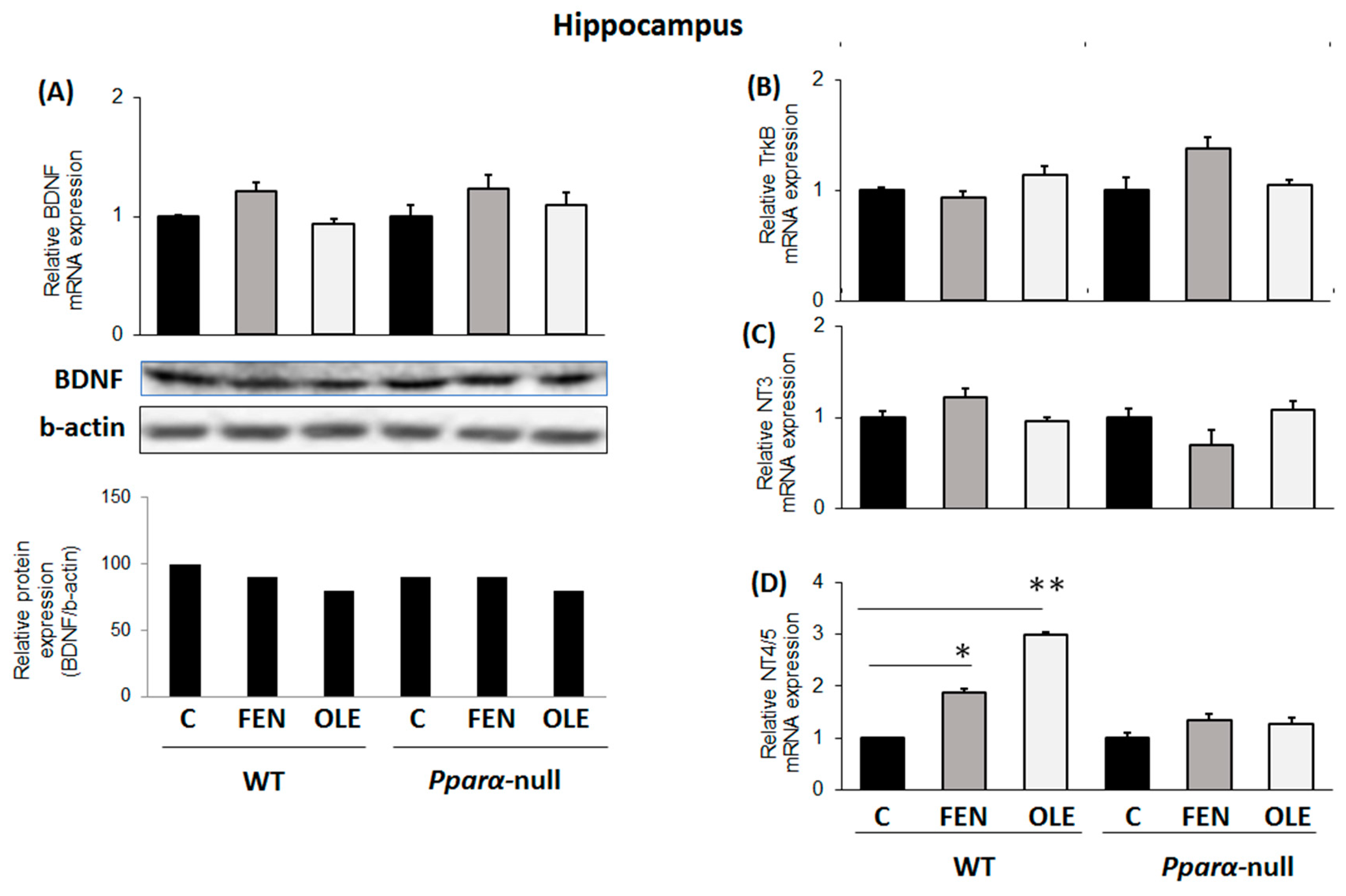
3.5. Assessment of subacute OLE treatment in neural plasticity indices in hippocampus
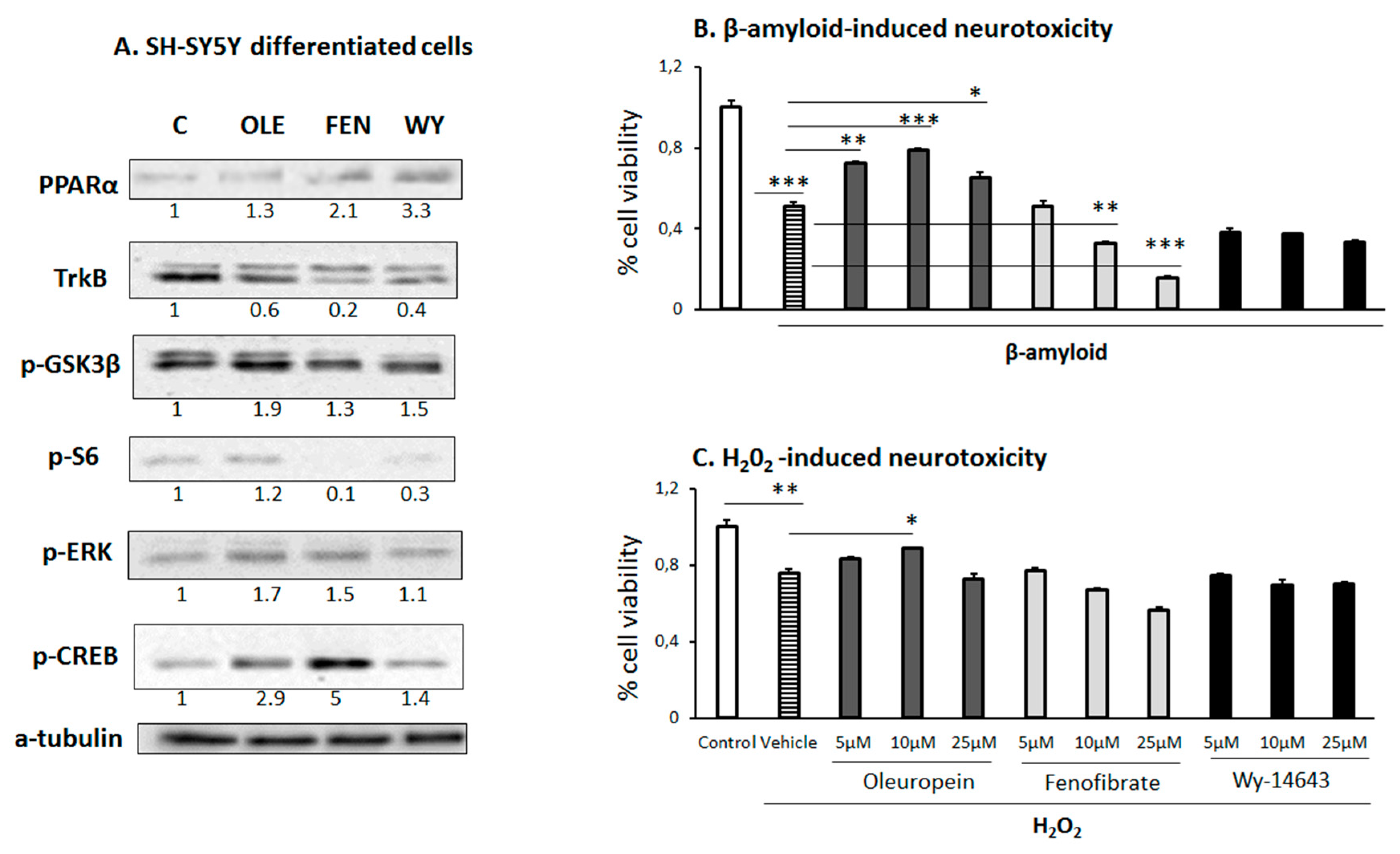
3.6. Effect of OLE on differentiated human SH-SY5Y neuroblastoma cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PPARα | peroxisome proliferator activated receptor α |
| BDNF | brain derived neurotrophic factor |
| TrkB | tyrosine kinase receptor B |
| NT3 | neurotrophic factor 3 |
| NT4/5 | neurotrophic factor 4/5 |
| PGC-1α | peroxisome proliferator-activated receptor-gamma coactivator-1 alpha |
| OLE | oleuropein |
| FEN | fenofibrate |
| WY | Wy-14643 |
| PFC | prefrontal cortex |
| AD | Alzheimer’s disease |
| WT | wild type |
References
- Mateos-Aparicio, P. and A. Rodríguez-Moreno, The Impact of Studying Brain Plasticity. Frontiers in Cellular Neuroscience, 2019. 13(66).
- Cramer, S.C. and E.P. Bastings, Mapping clinically relevant plasticity after stroke. Neuropharmacology, 2000. 39(5): p. 842-51.
- Chen, R., L.G. Cohen, and M. Hallett, Nervous system reorganization following injury. Neuroscience, 2002. 111(4): p. 761-773.
- Reichardt, L.F., Neurotrophin-regulated signalling pathways. Philos Trans R Soc Lond B Biol Sci, 2006. 361(1473): p. 1545-64.
- Huang, E.J. and L.F. Reichardt, Neurotrophins: Roles in Neuronal Development and Function. Annual Review of Neuroscience, 2001. 24(1): p. 677-736.
- Bessis, A., et al., Microglial control of neuronal death and synaptic properties. Glia, 2007. 55(3): p. 233-238.
- Cao, L. , et al., Olfactory ensheathing cells promote migration of Schwann cells by secreted nerve growth factor. Glia, 2007. 55(9): p. 897-904.
- Fukuoka, T. , et al., Brain-Derived Neurotrophic Factor Increases in the Uninjured Dorsal Root Ganglion Neurons in Selective Spinal Nerve Ligation Model. The Journal of Neuroscience, 2001. 21(13): p. 4891-4900.
- Bagayogo, I.P. and C.F. Dreyfus, Regulated release of BDNF by cortical oligodendrocytes is mediated through metabotropic glutamate receptors and the PLC pathway. ASN Neuro, 2009. 1(1).
- Lessmann, V., K. Gottmann, and M. Malcangio, Neurotrophin secretion: current facts and future prospects. Progress in Neurobiology, 2003. 69(5): p. 341-374.
- Matsuoka, I., M. Meyer, and H. Thoenen, Cell-type-specific regulation of nerve growth factor (NGF) synthesis in non-neuronal cells: comparison of Schwann cells with other cell types. The Journal of Neuroscience, 1991. 11(10): p. 3165.
- Ohta, K. , et al., The effect of dopamine agonists: The expression of GDNF, NGF, and BDNF in cultured mouse astrocytes. Journal of the Neurological Sciences, 2010. 291(1): p. 12-16.
- Schinder, A.F. and M. Poo, The neurotrophin hypothesis for synaptic plasticity. Trends Neurosci, 2000. 23(12): p. 639-45.
- Verderio, C. , et al., Cross talk between vestibular neurons and Schwann cells mediates BDNF release and neuronal regeneration. Brain Cell Biology, 2006. 35(2): p. 187-201.
- Tae, Y.Y. , et al., Minocycline Alleviates Death of Oligodendrocytes by Inhibiting Pro-Nerve Growth Factor Production in Microglia after Spinal Cord Injury. The Journal of Neuroscience, 2007. 27(29): p. 7751.
- Becker, E. , et al., Development of survival responsiveness to brain-derived neurotrophic factor, neurotrophin 3 and neurotrophin 4/5, but not to nerve growth factor, in cultured motoneurons from chick embryo spinal cord. J Neurosci, 1998. 18(19): p. 7903-11.
- Lu, B., P. T. Pang, and N.H. Woo, The yin and yang of neurotrophin action. Nature Reviews Neuroscience, 2005. 6(8): p. 603-614.
- Bramham, C.R. and E. Messaoudi, BDNF function in adult synaptic plasticity: the synaptic consolidation hypothesis. Prog Neurobiol, 2005. 76(2): p. 99-125.
- Michael, E.G. , et al., New Insights in the Biology of BDNF Synthesis and Release: Implications in CNS Function. The Journal of Neuroscience, 2009. 29(41): p. 12764.
- Lu, B., G. Nagappan, and Y. Lu, BDNF and synaptic plasticity, cognitive function, and dysfunction. Handb Exp Pharmacol, 2014. 220: p. 223-50.
- Heldt, S.A. , et al., Hippocampus-specific deletion of BDNF in adult mice impairs spatial memory and extinction of aversive memories. Molecular Psychiatry, 2007. 12(7): p. 656-670.
- Prior, M. , et al., The neurotrophic compound J147 reverses cognitive impairment in aged Alzheimer's disease mice. Alzheimers Res Ther, 2013. 5(3): p. 25.
- Zhang, L. , et al., Brain-Derived Neurotrophic Factor Ameliorates Learning Deficits in a Rat Model of Alzheimer's Disease Induced by Aβ1-42. PLOS ONE, 2015. 10(4): p. e0122415.
- Middlemas, D. , Neurotrophin 3, in xPharm: The Comprehensive Pharmacology Reference, S.J. Enna and D.B. Bylund, Editors. 2007, Elsevier: New York. p. 1-3.
- Warner, S.C. and A.M. Valdes The Genetics of Osteoarthritis: A Review. Journal of Functional Morphology and Kinesiology, 2016. 1. [CrossRef]
- Kang, H. and E.M. Schuman, Long-lasting neurotrophin-induced enhancement of synaptic transmission in the adult hippocampus. Science, 1995. 267(5204): p. 1658-62.
- Vicario-Abejón, C. , et al., Role of neurotrophins in central synapse formation and stabilization. Nat Rev Neurosci, 2002. 3(12): p. 965-74.
- Hock, C. , et al., Region-specific neurotrophin imbalances in Alzheimer disease: decreased levels of brain-derived neurotrophic factor and increased levels of nerve growth factor in hippocampus and cortical areas. Arch Neurol, 2000. 57(6): p. 846-51.
- Wang, M. , et al., Oleuropein promotes hippocampal LTP via intracellular calcium mobilization and Ca(2+)-permeable AMPA receptor surface recruitment. Neuropharmacology, 2020. 176: p. 108196.
- Cheng, K.K. , et al., Highly Stabilized Curcumin Nanoparticles Tested in an In Vitro Blood–Brain Barrier Model and in Alzheimer’s Disease Tg2576 Mice. The AAPS Journal, 2013. 15(2): p. 324-336.
- Maher, P. , et al., A pyrazole derivative of curcumin enhances memory. Neurobiology of Aging, 2010. 31(4): p. 706-709.
- Praag, H.v. , et al., Plant-Derived Flavanol (−)Epicatechin Enhances Angiogenesis and Retention of Spatial Memory in Mice. The Journal of Neuroscience, 2007. 27(22): p. 5869-5878.
- Tuck, K.L. and P.J. Hayball, Major phenolic compounds in olive oil: metabolism and health effects. The Journal of Nutritional Biochemistry, 2002. 13(11): p. 636-644.
- Visioli, F., A. Poli, and C. Gall, Antioxidant and other biological activities of phenols from olives and olive oil. Medicinal Research Reviews, 2002. 22(1): p. 65-75.
- Visioli, F. and C. Galli, Oleuropein protects low density lipoprotein from oxidation. Life Sciences, 1994. 55(24): p. 1965-1971.
- Briante, R. , et al., Antioxidant activity of the main bioactive derivatives from oleuropein hydrolysis by hyperthermophilic beta-glycosidase. J Agric Food Chem, 2001. 49(7): p. 3198-203.
- Oi-Kano, Y. , et al., Oleuropein, a phenolic compound in extra virgin olive oil, increases uncoupling protein 1 content in brown adipose tissue and enhances noradrenaline and adrenaline secretions in rats. J Nutr Sci Vitaminol (Tokyo), 2008. 54(5): p. 363-70.
- Visioli, F. , et al., Biological activities and metabolic fate of olive oil phenols. European Journal of Lipid Science and Technology, 2002. 104(9-10): p. 677-684.
- Malliou, F. , et al., The olive constituent oleuropein, as a PPARα agonist, markedly reduces serum triglycerides. The Journal of Nutritional Biochemistry, 2018. 59: p. 17-28.
- Andreadou, I. , et al., The olive constituent oleuropein exhibits anti-ischemic, antioxidative, and hypolipidemic effects in anesthetized rabbits. J Nutr, 2006. 136(8): p. 2213-9.
- Qabaha, K. , et al., Oleuropein Is Responsible for the Major Anti-Inflammatory Effects of Olive Leaf Extract. Journal of Medicinal Food, 2017. 21(3): p. 302-305.
- Jemai, H., A. El Feki, and S. Sayadi, Antidiabetic and Antioxidant Effects of Hydroxytyrosol and Oleuropein from Olive Leaves in Alloxan-Diabetic Rats. Journal of Agricultural and Food Chemistry, 2009. 57(19): p. 8798-8804.
- Alirezaei, M. , et al., Antioxidant effects of oleuropein versus oxidative stress induced by ethanol in the rat intestine. Comparative Clinical Pathology, 2014. 23(5): p. 1359-1365.
- Luccarini, I. , et al., Oleuropein aglycone protects against pyroglutamylated-3 amyloid-ß toxicity: biochemical, epigenetic and functional correlates. Neurobiology of Aging, 2015. 36(2): p. 648-663.
- Nardiello, P. , et al., Diet Supplementation with Hydroxytyrosol Ameliorates Brain Pathology and Restores Cognitive Functions in a Mouse Model of Amyloid-β Deposition. Journal of Alzheimer's Disease, 2018. 63: p. 1161-1172.
- Grossi, C. , et al., The Polyphenol Oleuropein Aglycone Protects TgCRND8 Mice against Aß Plaque Pathology. PLoS ONE, 2013. 8(8): p. e71702.
- Pourkhodadad, S. , et al., Neuroprotective effects of oleuropein against cognitive dysfunction induced by colchicine in hippocampal CA1 area in rats. The Journal of Physiological Sciences, 2016. 66(5): p. 397-405.
- Pitozzi, V. , et al., Long-Term Dietary Extra-Virgin Olive Oil Rich in Polyphenols Reverses Age-Related Dysfunctions in Motor Coordination and Contextual Memory in Mice: Role of Oxidative Stress. Rejuvenation Research, 2012. 15(6): p. 601-612.
- Farr, S.A. , et al., Extra Virgin Olive Oil Improves Learning and Memory in SAMP8 Mice. Journal of Alzheimer's Disease, 2012. 28: p. 81-92.
- Rizk, F.H. , et al., Fenofibrate Improves Cognitive Impairment Induced by High-Fat High-Fructose Diet: A Possible Role of Irisin and Heat Shock Proteins. ACS Chem Neurosci, 2022. 13(12): p. 1782-1789.
- Akiyama, T.E. , et al., Peroxisome proliferator-activated receptor-alpha regulates lipid homeostasis, but is not associated with obesity: studies with congenic mouse lines. J Biol Chem, 2001. 276(42): p. 39088-93.
- Lee, S.S. , et al., Targeted disruption of the alpha isoform of the peroxisome proliferator-activated receptor gene in mice results in abolishment of the pleiotropic effects of peroxisome proliferators. Mol Cell Biol, 1995. 15(6): p. 3012-22.
- Andreadou, I. , et al., Oleuropein prevents doxorubicin-induced cardiomyopathy interfering with signaling molecules and cardiomyocyte metabolism. J Mol Cell Cardiol, 2014. 69: p. 4-16.
- Impellizzeri, D. , et al., The effects of a polyphenol present in olive oil, oleuropein aglycone, in an experimental model of spinal cord injury in mice. Biochem Pharmacol, 2012. 83(10): p. 1413-26.
- Edgecombe, S.C., G. L. Stretch, and P.J. Hayball, Oleuropein, an antioxidant polyphenol from olive oil, is poorly absorbed from isolated perfused rat intestine. J Nutr, 2000. 130(12): p. 2996-3002.
- Pappenheimer, J.R. and K.Z. Reiss, Contribution of solvent drag through intercellular junctions to absorption of nutrients by the small intestine of the rat. J Membr Biol, 1987. 100(2): p. 123-36.
- Del Boccio, P. , et al., Liquid chromatography-tandem mass spectrometry analysis of oleuropein and its metabolite hydroxytyrosol in rat plasma and urine after oral administration. J Chromatogr B Analyt Technol Biomed Life Sci, 2003. 785(1): p. 47-56.
- Xu, A.N. and F. Nie, Brain-derived neurotrophic factor enhances the therapeutic effect of oleuropein in the lipopolysaccharide-induced models of depression. Folia Neuropathol, 2021. 59(3): p. 249-262.
- Impey, S. , et al., Cross talk between ERK and PKA is required for Ca2+ stimulation of CREB-dependent transcription and ERK nuclear translocation. Neuron, 1998. 21(4): p. 869-83.
- Cañón, E. , et al., Rapid effects of retinoic acid on CREB and ERK phosphorylation in neuronal cells. Mol Biol Cell, 2004. 15(12): p. 5583-92.
- Bonni, A. , et al., Cell survival promoted by the Ras-MAPK signaling pathway by transcription-dependent and -independent mechanisms. Science, 1999. 286(5443): p. 1358-62.
- Nakagawa, S. , et al., Localization of phosphorylated cAMP response element-binding protein in immature neurons of adult hippocampus. J Neurosci, 2002. 22(22): p. 9868-76.
- Shaywitz, A.J. and M.E. Greenberg, CREB: a stimulus-induced transcription factor activated by a diverse array of extracellular signals. Annu Rev Biochem, 1999. 68: p. 821-61.
- Cooke, S.F. and T.V. Bliss, Plasticity in the human central nervous system. Brain, 2006. 129(Pt 7): p. 1659-73.
- Dinda, B. , et al., Therapeutic potentials of plant iridoids in Alzheimer's and Parkinson's diseases: A review. Eur J Med Chem, 2019. 169: p. 185-199.
- Balint, L.B. and L. Nagy, Selective Modulators of PPAR Activity as New Therapeutic Tools in Metabolic Diseases. Endocrine, Metabolic & Immune Disorders - Drug Targets, 2006. 6(1): p. 33-43.
- Fei-fei, P. , et al., Oleuropein Improves Long Term Potentiation at Perforant Path-dentate Gyrus Synapses in vivo. Chinese Herbal Medicines (CHM). 7(3): p. 255-260.
- Gelé, P. , et al., Recovery of brain biomarkers following peroxisome proliferator-activated receptor agonist neuroprotective treatment before ischemic stroke. Proteome Sci, 2014. 12: p. 24.
- Butt, M.S. , et al., Neuroprotective effects of oleuropein: Recent developments and contemporary research. Journal of Food Biochemistry, 2021. 45(12): p. e13967.

| Gene | Sequences of primers | |
|---|---|---|
| BDNF | F | 5'-TGAGTCTCCAGGACAGCAAA-3' |
| R | 5'-GACGTTTACTTCTTTCATGGGC-3' | |
| TrkB | F | 5'-TGATGTTGCTCCTGCTCAAG-3' |
| R | 5'-CCCAGCCTTTGTCTTTCCTT-3' | |
| NT3 | F | 5'-CGGATGCCATGGTTACTTCT-3' |
| R | 5'-AGTCTTCCGGCAAACTCCTT-3' | |
| NT4/5 | F | 5'-AGCCGGGGAGCAGAGAAG-3' |
| R | 5'-CACCTCCTCACTCTGGGACT-3' | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




