Introduction
Glioblastoma multiforme (GBM), the most prevalent primary malignant brain tumour in people, has an estimated overall survival of less than two years. It is still an incurable disease despite the fact that there are many treatments available including surgery, chemotherapy, and radiation, as it is made up of a highly heterogeneous assortment of neoplasms (Lee et al., 2015). Therefore, identifying new targets depends on elucidating the intrinsic molecular processes underlying the abnormal features of GBM. Overexpression of Forkhead box M1 (FOXM1) promotes oncogenesis and increases invasion, angiogenesis, epithelial-mesenchymal transition, self-renewal, and chemotherapy tolerance (Halasi and Gartel, 2013; Meng et al., 2015). Due in part to the pleotropic nature of the FoxM1 pathway, the roles of FoxM1 in GBM remain poorly known.
FOXM1, a transcription factor belonging to the Forkhead box (FOX) family, is well-known for playing a crucial part in many metabolic processes, such as cell cycle control, DNA damage repair, and apoptosis (Su et al 2021). There are three distinct isoforms of the FOXM1 protein. The transcriptional activity of the FOXM1b and FOXM1c splice forms is higher than that of the FOXM1a splice variant. Through homologous recombination, the active forms of FOXM1, especially the FOXM1b and FOXM1c isoforms, can accelerate DNA double-strand repair (Myatt et al., 2014; Monteiro et al., 2013). There are several cancers, including non-small cell lung cancer, breast cancer, basal cell carcinoma, hepatocellular carcinoma, pancreatic cancer, prostate cancer, colon cancer, medulloblastoma, and GBM that are found to have increased FOXM1 expression. In GBM rodent models, high levels of FOXM1 expression in glioma cells increase tumorigenicity, invasiveness, and angiogenesis (Shimet et al., 2022). It is also reported that FOXM1found to be controlled the Wnt/β-catenin signaling pathway by stimulating the nuclear translocation of β-catenin (Zhang et al., 2011). On the other hand, it has been discovered that suppressing FOXM1 expression and activity significantly lowers the rate of cancer metastasis spread by decreasing the expression of matrix metalloproteinases 2 (MMP-2) is regarded as a crucial enzyme in the breakdown of the extracellular matrix (Dai et al., 2007). Targeting FOXM1 in Glioblastoma stem cells may prove to be a novel approach to cancer therapy and also enhance the efficacy of pharmacological therapies in GBM patients.
The identification of natural compounds that specifically target the DNA binding domain (DBD) of FOXM1 has revealed their ability to combat tumors and transform transcription factors into viable drug targets. One such compound is Silybin B, a flavonolignan that occurs naturally and is extracted from the Silybum marianum plant, commonly referred to as Milk Thistle (Soleimani et al., 2019). From thousands of years, Silybin B has been utilized in Indian Ayurvedic medicine as an effective treatment for various diseases.
According to recent research, Silybin B is extensively used extensively as a neuroprotective, hepatoprotective, cardioprotective, antioxidant, anti-cancer, anti-diabetic, anti-viral, anti-hypertensive, immunomodulator, anti-inflammatory, and photoprotective agent by targeting various cellular and molecular pathways, such as MAPK, mTOR, -catenin, and Akt (Binienda et al., 2020). It has potent pleiotropic anti-neoplastic actions against a variety of tumour cells, including glioblastoma (Baiet et al., 2018), lung (Mateen et al., 2013), renal cell carcinoma (Li et al., 2008), bladder (Zeng et al., 2011), colon (Kumar et al., 2014) and skin (Singh et al., 2005).
The purpose of this study was to better understand the antiproliferative properties of Silybin B by examining how it affects Glioblastoma stem cells' ability to undergo senescence. It was also looked into whether Silybin B's impacts on FOXM1 inhibition and cellular senescence are related. Additionally, molecular dynamics simulations (MD) were run to acquire a better understanding of the interactions between Silybin B and FOXM1. Together, these findings may shed light on how Silybin B inhibits FOXM1's transactivation activity and aid in the development of an innovative, efficient FOXM1 molecular inhibitor for the therapy of glioblastoma stem cells. The rational structure-based design of drug candidates may also benefit from a better comprehension of the binding mechanism between Silybin B and FOXM1.
Materials and Methodology
3-Dimentional structures of all 98 phytochemical compounds based on their anticancer activity and 2 control compounds were downloaded in .sdf format obtained from PubChem server (
https://pubchem.ncbi.nlm.nih.gov/). For FOXM1-BD protein, PDB ID: 3G73 was downloaded which is X-ray diffracted having 2.21 Å resolutions.
Appropriate selection and processing of coordinates for receptors and ligands are essential factors in the successful docking process. These preparation processes require the conversion of the PDB file format to the PDBQT file format. The 3D structure of FOXM1-BD with PDB ID 3G73 was then prepared for molecular docking. Chain A, water molecules, DNA, and magnesium ions were removed from the protein molecule using PyMOL software. Further, protein was minimized using Chimaera Software's AutoDock software using an AMBER ff14SB forcefield. Final protein and ligand preparation was done by assigning missing residues, hydrogen polarities, and Gasteiger and Kollman charges to proteins and ligand structures using AutoDock tools (Lim et al., 2011; Jaghoori et al., 2016).
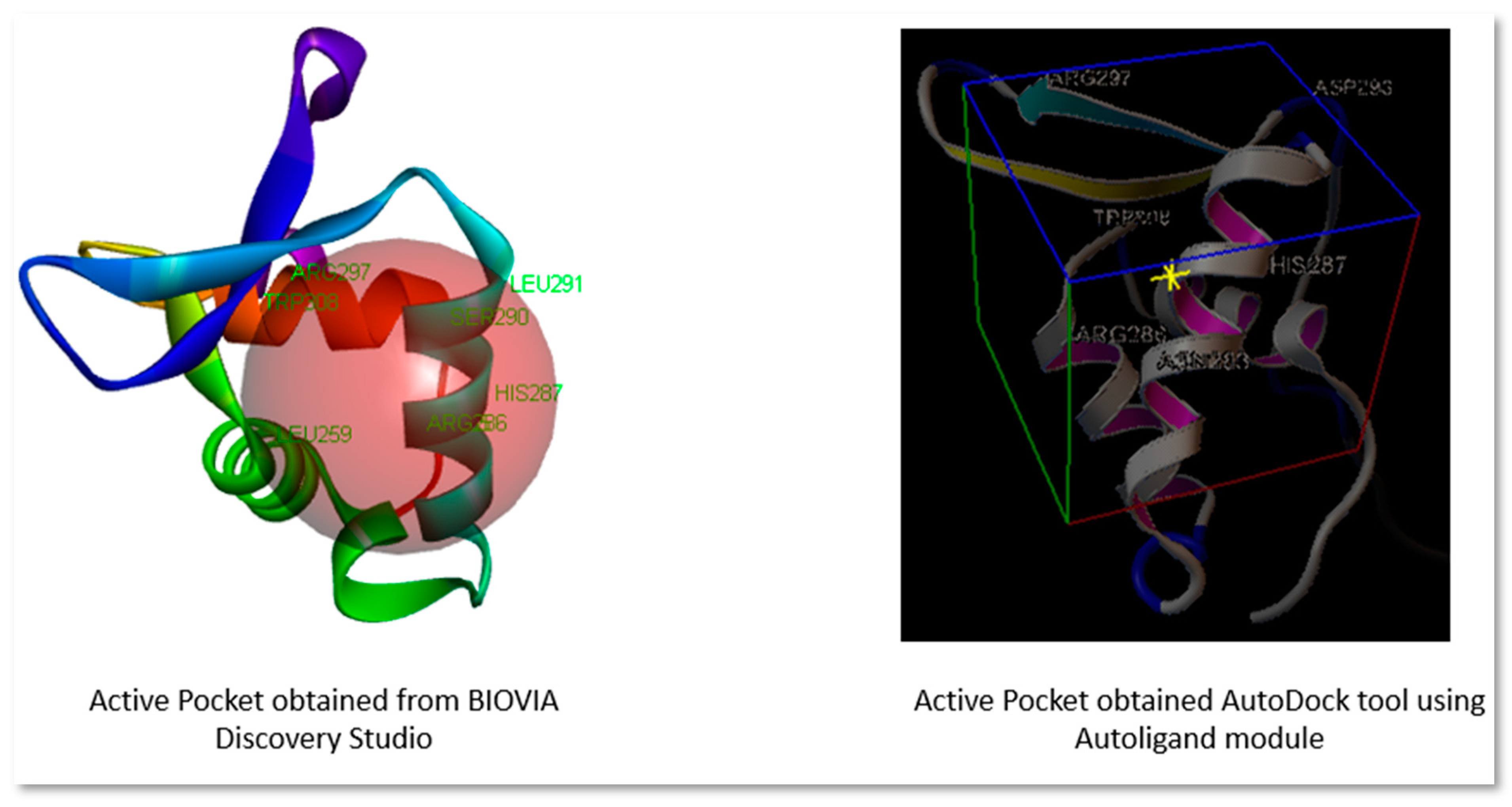
In modern drug design, molecular docking provides valuable information about drug-receptor interactions. It is frequently used to predict the binding orientation of small molecule drug candidates to their protein targets to predict the small molecule's affinity and activity. A grid box was generated, keeping the active site in consideration. The Active site was obtained using BIOVIA Discovery Studio Visualizer 2020 (
https://discover.3ds.com/discovery-studio-visualizer-download) and Autoligand module of AutoDock tools to locate the coordinates for the ligand's possible binding location in the receptor's active site pocket.
Autodock Vina docking tool was used for molecular docking that takes the atomic coordinates of the target protein and selected ligand, thus predicting the most suitable docking conformation of the two. Vina's inability to replicate the docking of more than one ligand to the same target protein at once is one of its drawbacks. This is a barrier for scientists that want to use Vina to do high-throughput virtual screening. A tool called EasyDockVina was developed to make it simple and straightforward to employ numerous ligand docking against a certain protein (Mane et al., 2022; Alshehri et al., 2020). All the 100 ligand compounds were initially docked by using EasyDock Vina. During the entire docking process, the target (protein) was kept rigid while ligands were flexible with the aim to determine the best suitable pose. The selected ligands which possess considerable binding energy and following Lipinski rule were then re-docked using AutoDock Vina. An output log file with all the binding energies between protein and different orientations of ligand was further analysed. On completion of the Docking process, the highest-ranking poses with lesser binding were selected for further protein-ligand interaction analysis.
Understanding drug Absorption, Distribution, Metabolism, Excretion, and Toxicity (ADMET) is crucial for drug research and development. In addition to possessing sufficient activity against the therapeutic target, a high-quality drug candidate should display the appropriate ADMET properties at a therapeutic dosage. Drug-likeness calculation was performed to know the cytotoxicity activity of compounds for human by DruLiTo open-source software (
https://niper.gov.in/pi_dev_tools/DruLiToWeb/DruLiTo_index.html) (Prasanth
et al., 2021; Sharma
et al., 2020). Based on specific physiochemical and structural characteristics, a ligand's drug bioavailability or drug-likeness is used to determine its pharmacological importance (Metwally & Eldaly, 2022). As a result, all ligands were assessed for their druglikeness using Lipinski's five guidelines and Blood Brain Barrier crossing ability by DruLiTo software.
Only those ligands were studied for interaction analysis which passes Lipinski's five guidelines and having better binding energy. The interaction was visualized on BIOVIA Discovery Studio Visualizer 2020 (Srivastava
et al., 2021). This program helps to project 3D structure to a 2D image, thus facilitating close inspection of 2D interactions of the protein-ligand complex. Moreover, to estimate final selected molecule toxicity a freely available authenticated and tested software programs ProTox II was used (
https://tox-new.charite.de/protox_II/) (Mehta
et al., 2022; Rachmale
et al., 2022; Rolta
et al., 2022).
The molecular dynamics (MD) simulations method helps follow the atomic motions of molecular systems. Therefore, it's a suitable method to evaluate all the dynamic aspects of proteins, apart from the available structural information from crystallography studies (Limon et al., 2022). Although Molecular docking is a quick method to determine the binding mode of any ligand in the protein's active site, it still possesses several inaccuracies in the outcomes. Therefore, simulation is followed by docking as simulations in the system are simulated under thermal fluctuations. This technique allows studying biological and chemical systems at the atomistic level on timescales from femtoseconds to milliseconds.
MD simulations utilising Schrödinger's Desmond module were used to investigate the binding stability of top-ranked compounds at the atomic level and to comprehend the molecular interaction. The complexes of FoxM1 protein with Bryophyllin A, Silybin B, Withaferin A, Sanguinarine, and the control drugs Troglitazone were solvated using the explicit SPC water model under orthorhombic periodic boundary conditions with a buffer area of 10 between protein atoms and box edges (Zala et al., 2023; Kumar et al., 2023; Desai et al., 2023). To neutralise the charges, sodium and chloride ions were introduced into the system builder, followed by 0.15 M NaCl salt concentrations in accordance with human physiological circumstances. The construction system was then minimised using a fixed value of the OPLS3e force field to reduce electronic conflicts between protein structures and to properly align the protein structure inside the simulation bounds. Long-range electrostatic interactions were evaluated using the smooth particle mesh Ewald technique with a tolerance of 1e-09, whereas short-range Van der Waals and Coulomb interactions were calculated with a cut-off radius of 9.0 (Sudevan et al., 2022; Kikiowo et al., 2022). The simulation was ran for 100 ns under a 'isothermal-isobaric ensemble' (NPT) at a temperature of 300 K and a pressure of 1 bar after importing the built minimised system (.cms file) into the molecular dynamics module. For isothermal-isobaric conditions, the 'Nose-Hoover chain thermostat' and 'Martyna-Tobias-Klein barostat' approaches were ensembled at 100 and 200ps, respectively. Simulation snapshots were obtained at 100ps intervals, and the resultant trajectories were assessed (Aljuhani et al., 2022).
Molecular Docking Analysis
Prior to docking analysis we have minimized protein containing only B chain. After minimization final energy come down to -6241.25 kJ/mol from -3684.35 kJ/mol with RMSD value of 0.12459. Further, we have selected two compounds namely FDI-6 and Troglitazone as our control compounds which are widely reported to be active against the selected protein (Dakhili
et al., 2018; Zhang at el., 2022). Firstly, we have done docking using EasyDock Vina (GUI of AutoDock Vina) and found that. Docked results with binding energy for 100 ligands are shown in
Table 2. Only 6 ligands which follows Lipinski’s rule were found to have binding energy less than -6.6 kcal/mol. These 6 ligands were subjected to re-docking using AutoDock Vina. However, the FDI-6 compound taken as control were having only -6.0 kcal/mol so it was not taken for re-docking using AutoDock Vina. Same active site and configuration file were used to do re-docking of selected 6 ligands with target protein.
Table 3 shows docked results of selected 6 ligands done by using AutoDock Vina. It is found from the docked results that Withaferin A, Bryophyllin A, Silybin B, and Sanguinarine have better binding energy in comparison to control compound
Troglitazone. These 6 ligands were studied for protein-ligand interaction analysis. Out of all the selected phytochemical compounds only Withaferin A is a compound which obeys Lipinski’s rule and have binding energy -7.0 kcal/mol. Thus, this ligand is selected for further simulation studies.
The poses with the highest rankings following the docking procedure were chosen for further protein-ligand interaction research. All six ligands subjected to re-docking using AutoDock Vina were studied for interaction analysis. The interaction was made apparent using BIOVIA Discovery Studio Visualizer 2020. The overall interacting residues with the nature of interaction obtained from Biovia Discovery studio are presented in
Table 3.
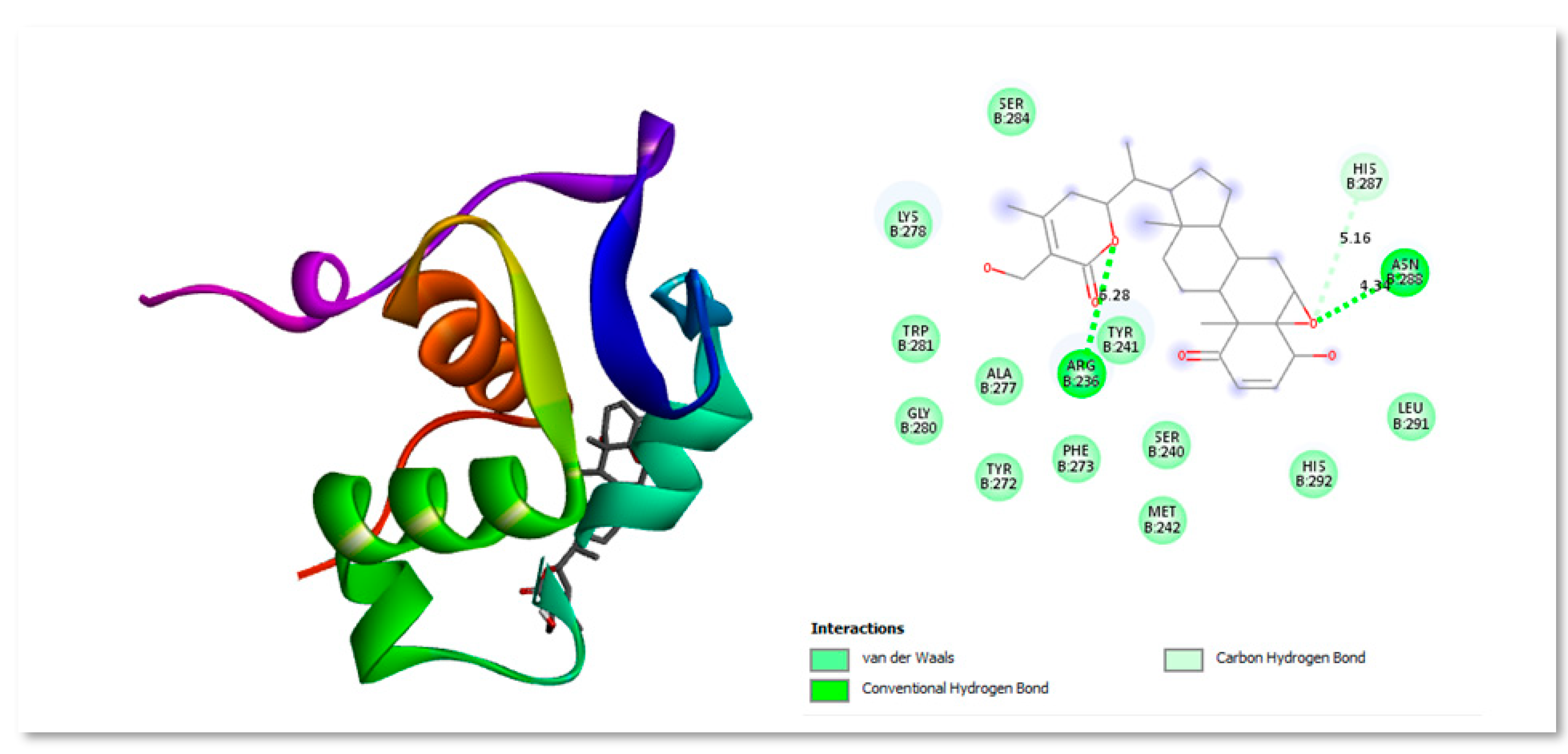
Troglitazone is an antidiabetic drug molecule which is reported to interact with (and inhibit) the FOXM1 DNA binding domain (FOXM1-BD), causing downregulation of protein expression and cancer cell proliferation inhibition. It was taken as control drug for docking analysis. Three structurally distinct FOXM1 inhibitors—troglitazone, thiostrepton, and FDI molecules appear to exert a similar binding pattern inside the FOXM1-DNA binding region (Dakhili
et al., 2018) Using computer-aided drug design, Xie et al. firstly explored the binding site between FDI-6 and FOXM1 and discovered that His287, Arg236 and Ser284 were the important amino acids in the interaction (Xie
et al., 2022).
Figure 2 shows the two- and three-dimensional structure interacting with protein residues present in active site.
Withaferin A is a naturally occurring steroidal lactone compound derived from the
Withania somnifera plant (commonly known as Ashwagandha), which has long been used in Indian Ayurvedic medicine for an effective therapy of a wide range of diseases. By increasing the expression of Bim and Bad, Withaferin A greatly reduced the growth of GBM both in vitro and in vivo and caused the intrinsic death of GBM cells (Tang
et al., 2020). Withaferin A has received a lot of attention as a viable anti-neoplastic agent because of the lactonal steroid's capacity to alter a number of oncogenic pathways (Dhami
et al., 2017).
Figure 3 shows the two- and three-dimensional structure interacting with protein residues present in active site.
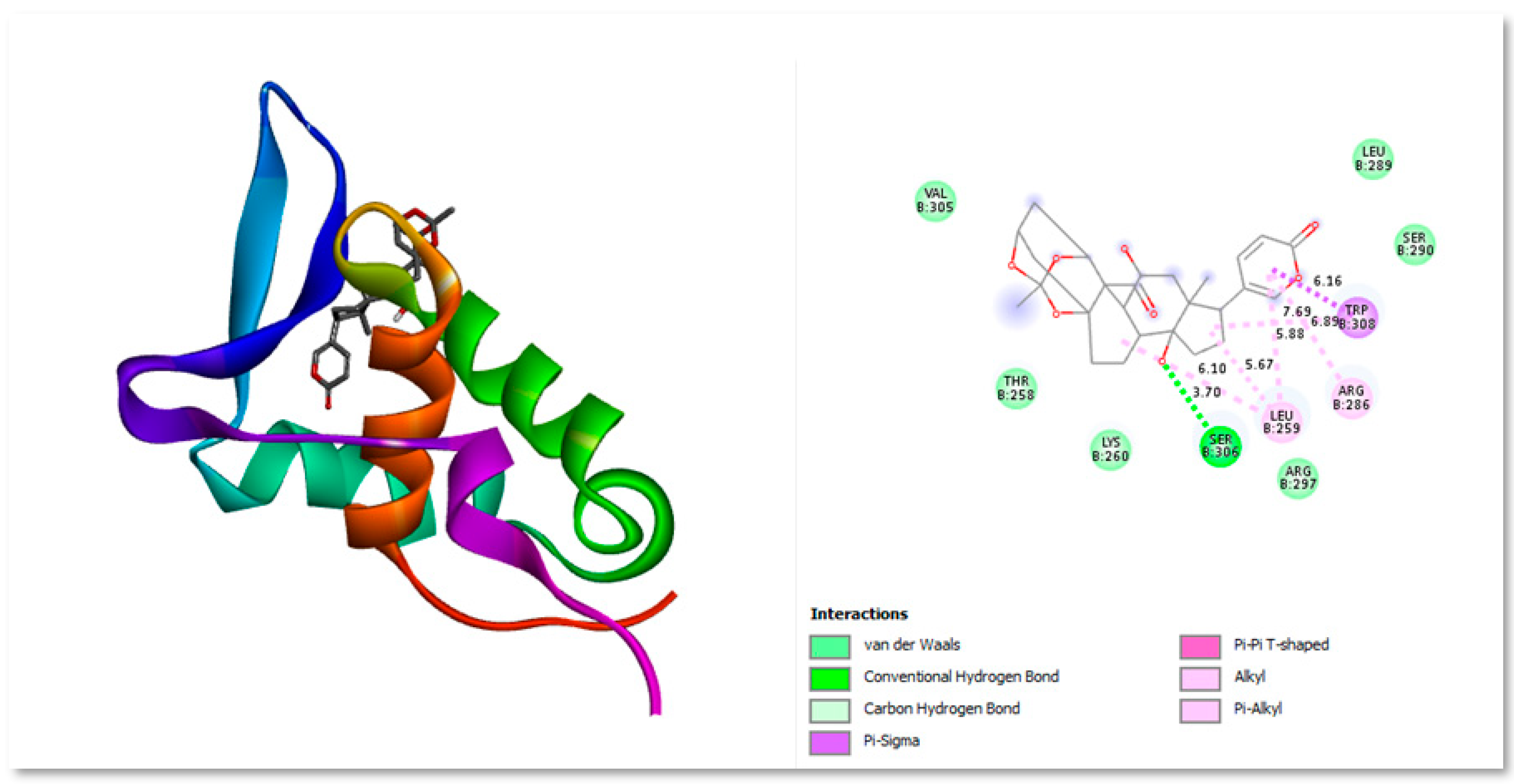
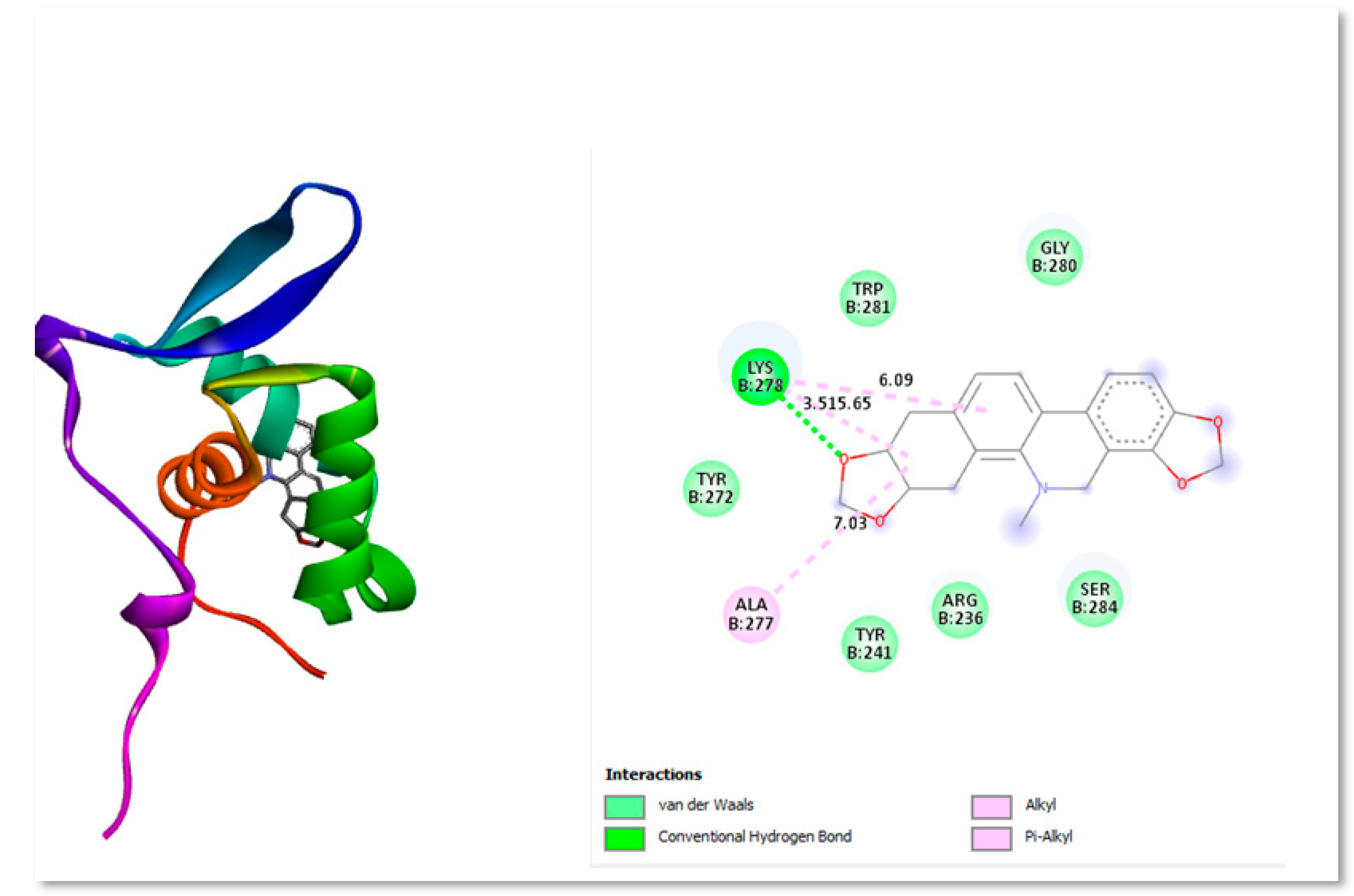
A naturally occurring substance called Bryophyllin A is present in Kalanchoe daigremontiana, Kalanchoe pinnata, and other organisms (Hernández-Caballero et al 2022). People utilise plants from the genus Kalanchoe (Family: Crassulaceae) as decorative plants all over the world in warm regions. Some of the 200 species of Kalanchoe are renowned for their ability to treat illnesses including cancer. Herbal extracts are made from a wide range of plants, and natural treatments are widely utilised throughout the world. Several of the chemical components found in Kalanchoe species, including flavonoids, bufadienolides, fatty acids, triterpenoids, alkaloids, phenolic acids, saponins, tannins, glycosides, and kalanchosides, show significant anticancer potential (Nielsen
et al., 2005; Kamboj
et al., 2009).
Figure 4 shows the two- and three-dimensional structure interacting with protein residues present in active site.
Sanguinarine (SAG), a naturally occurring benzophenanthridine alkaloid produced from the root of the bloodroot plant Sanguinaria canadensis Linn. havingchemo-preventive properties (Xu
et al., 2022). According to several studies, SANG prevents tumour metastasis and growth by interfering with a variety of cell signalling pathways and its molecular targets, including BCL-2, MAPKs, Akt, NF-B, ROS, and microRNAs (miRNAs) (Ullah
et al., 2023).
Figure 5 shows the two- and three-dimensional structure interacting with protein residues present in active site.
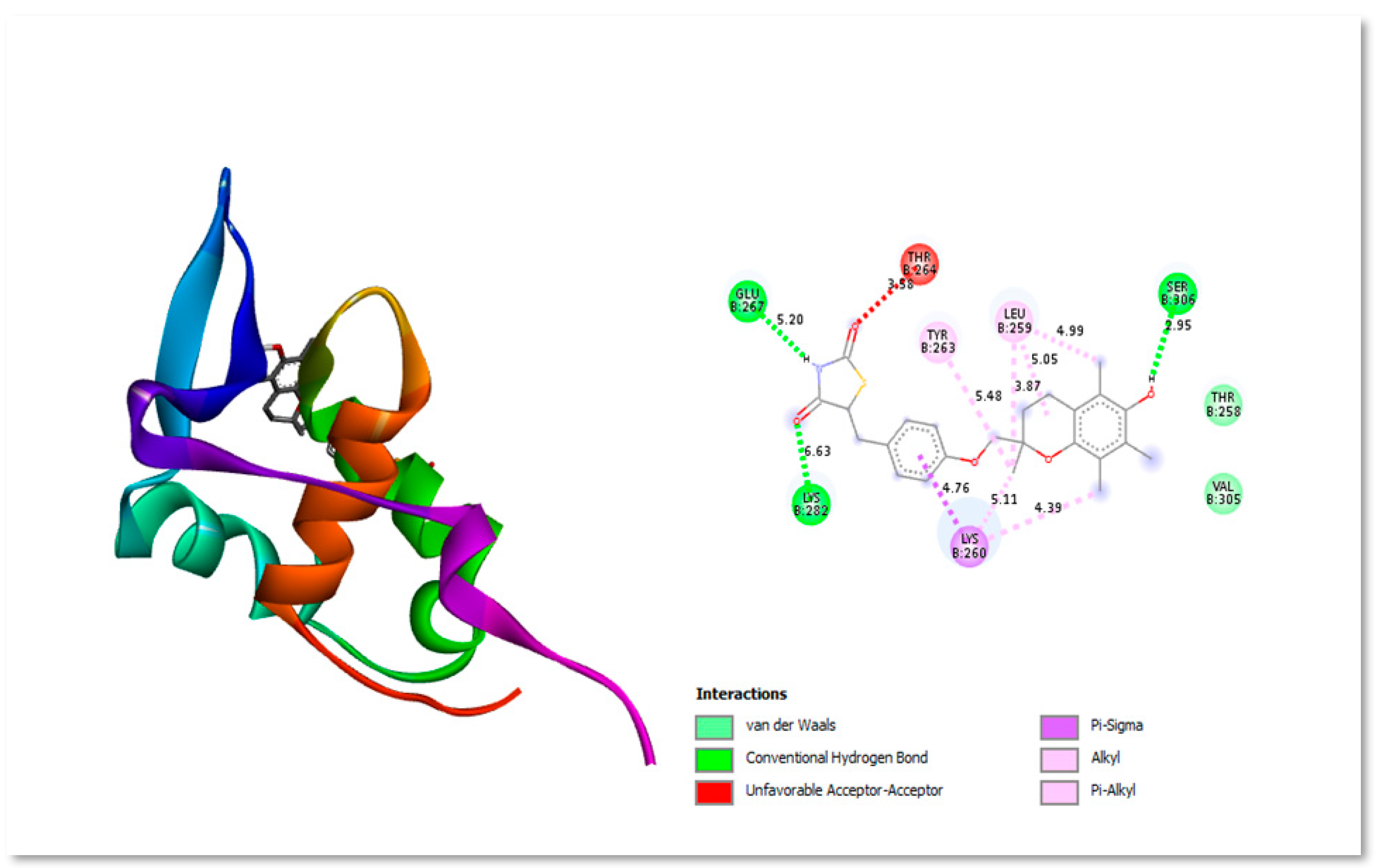
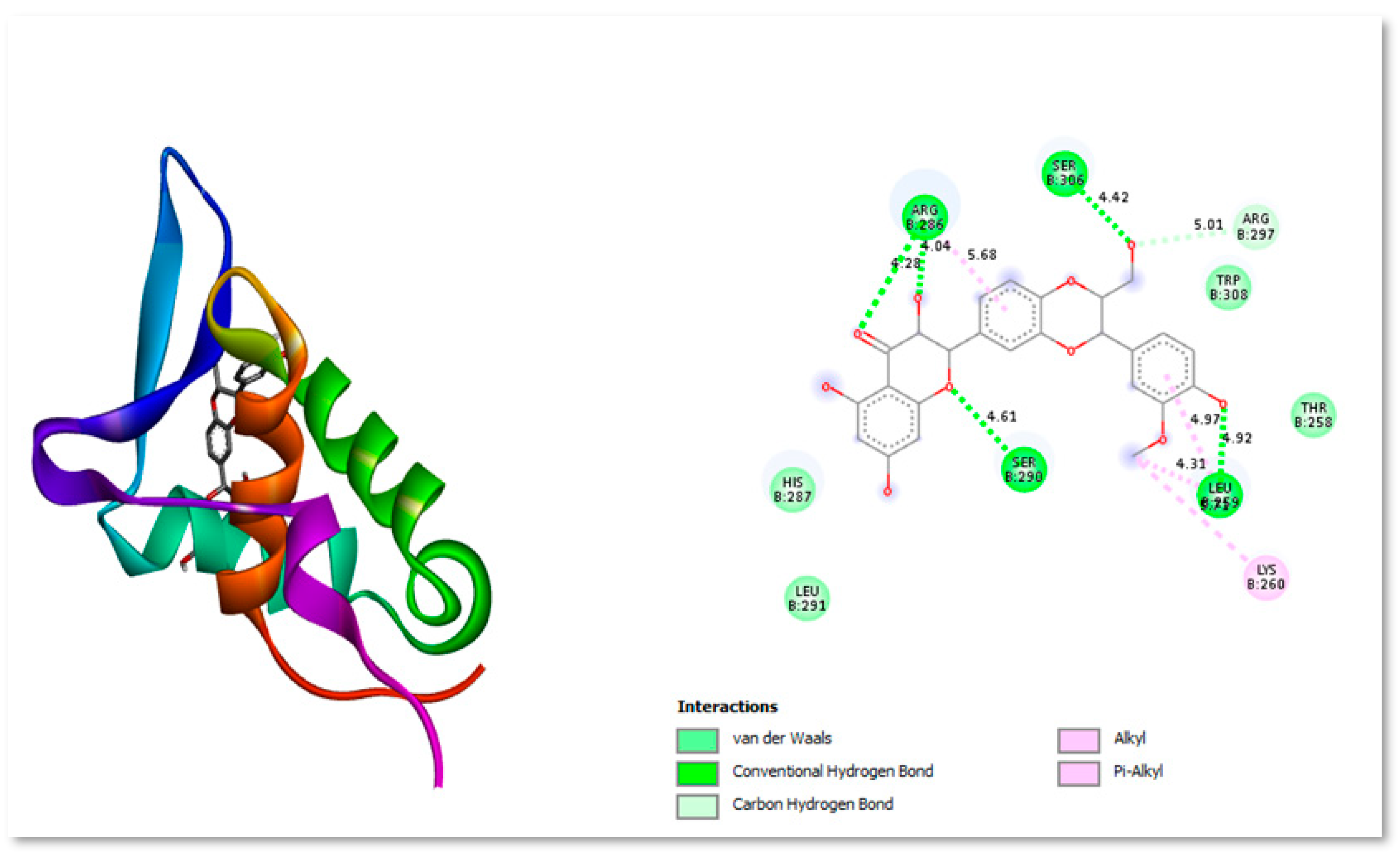
Silybin B is extensively used extensively as a neuroprotective, hepatoprotective, cardioprotective, antioxidant, anti-cancer, anti-diabetic, anti-viral, anti-hypertensive, immunomodulator, anti-inflammatory, and photoprotective agent by targeting various cellular and molecular pathways, such as MAPK, mTOR, -catenin, and Akt (Binienda et al., 2020). Figure 6 shows the two- and three-dimensional structure interacting with protein residues present in active site.
MD simulations can be utilized to evaluate the stability of protein-ligand complexes under physiological conditions. By employing MD simulations, researchers can observe and analyse the conformational changes of proteins over time, gaining valuable insights into the movements of proteins and their impact on the stability of ligands. In this study, a 100 ns MD simulations on the protein-ligand complex structures were conducted to assess the consistency of proposed phytochemicals, namely Bryophyllin A, Silybin B, Withaferin A, and Sanguinarine, within the binding cavity of the FoxM1 protein's active site. During the MD simulations, various parameters collected from the trajectory of the simulations. These parameters included Root Mean Square Deviation (RMSD), Root Mean Square Fluctuation (RMSF), and Radius of Gyration (RGyr). These metrics served as indicators for evaluating the stability of the protein-ligand complex in dynamic states.
Root-mean-square deviation (RMSD) is a widely used metric in MD simulations to measure the average movement or deviation of atoms between two reference frames. It provides valuable information about the stability and conformational changes within protein-ligand complexes during MD simulations, typically conducted over various nanosecond timescales. Higher RMSD values for the protein backbone indicate a greater degree of unfolding or conformational changes, suggesting decreased stability of the complex. Conversely, lower RMSD values suggest a more compact and stable conformation of the protein-ligand complex (Derya
et al., 2022; Halder
et al., 2022). Graphical analyzing the protein Cα RMSD values, interpret the stability of the docking complex over time (
Figure 7). Patterns and trends in the RMSD plot can provide valuable insights into the dynamic behavior of the protein-ligand complex, indicating regions of stability and potential conformational changes that may impact the ligand's binding within the protein's active site.
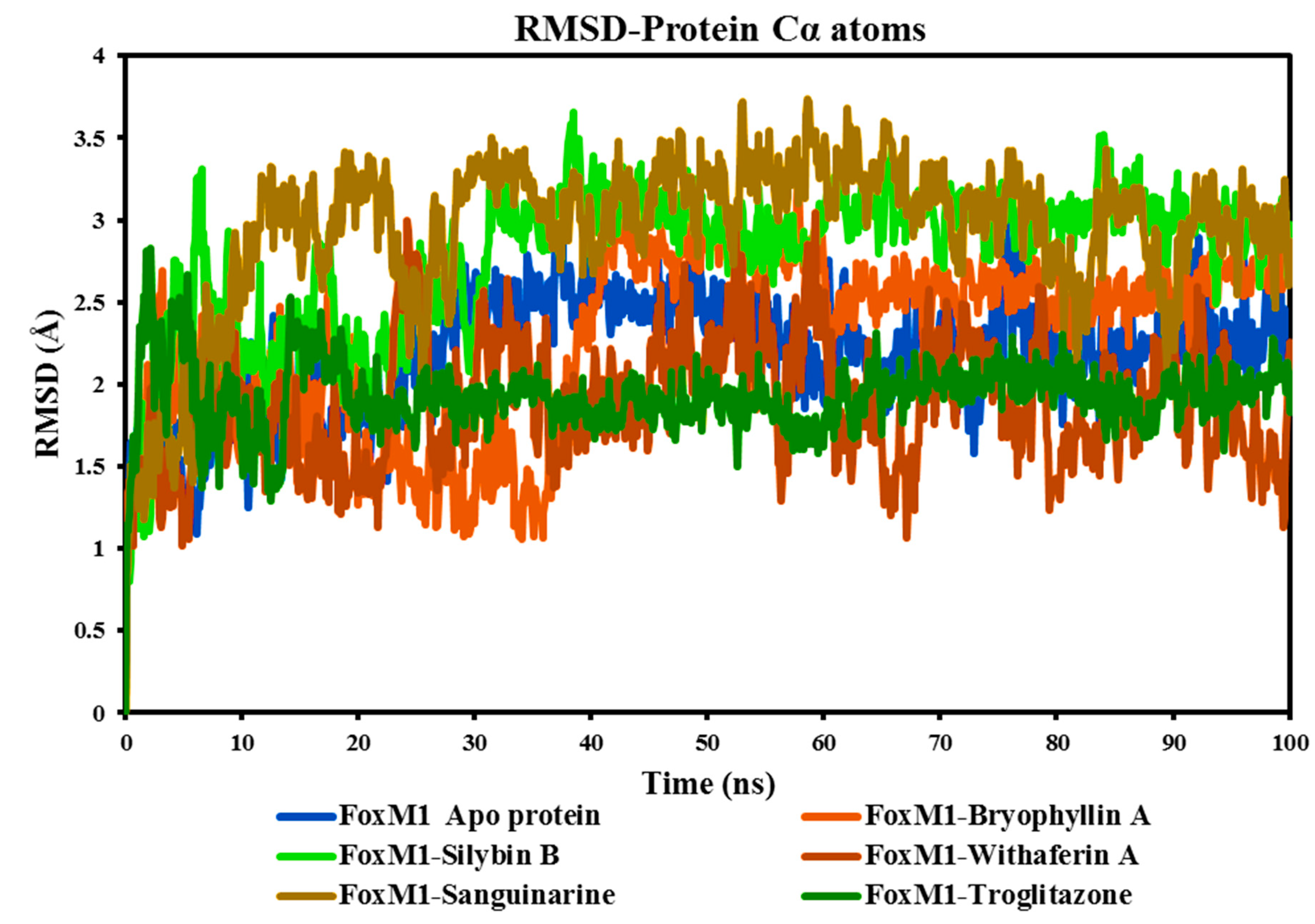
As depicted in
Figure 7, the Cα RMSD demonstrates consistent stability throughout the simulation, with values ranging between 0.80 Å and 3.66 Å. The average RMSD values for the Apo FoxM1 protein and its complexes with Bryophyllin A, Silybin B, Withaferin A, Sanguinarine, and the control drug Troglitazone are 2.18 Å, 2.28 Å, 2.79 Å, 1.88 Å, 2.93 Å, and 1.93 Å, respectively. Among the complexes, notable fluctuations are observed in the FoxM1-Silybin B complex (light green colour) and the FoxM1-Sanguinarine complex (grey colour). The FoxM1-Withaferin A complex exhibits the lowest minimum RMSD value of 0.82 Å, indicating a relatively stable structure. Additionally, it has a comparatively low average RMSD value of 1.88 Å, further supporting its status as the most stable complex among the analyzed complexes. On the contrary, the FoxM1-Sanguinarine complex demonstrates the highest maximum RMSD value of 3.73 Å, indicating significant structural fluctuations. Moreover, it possesses the highest average RMSD value of 2.93 Å among the complexes, suggesting relatively lower stability compared to the others. Based on the minimal fluctuations and modest differences in RMSD values observed in the protein Cα RMSD plot, it can be concluded that the protein-ligand complexes, except for FoxM1-Sanguinarine, demonstrated stability under dynamic conditions. The minor conformational adaptations observed suggest that the complexes were able to accommodate the ligand molecules while maintaining overall stability throughout the MD simulations.
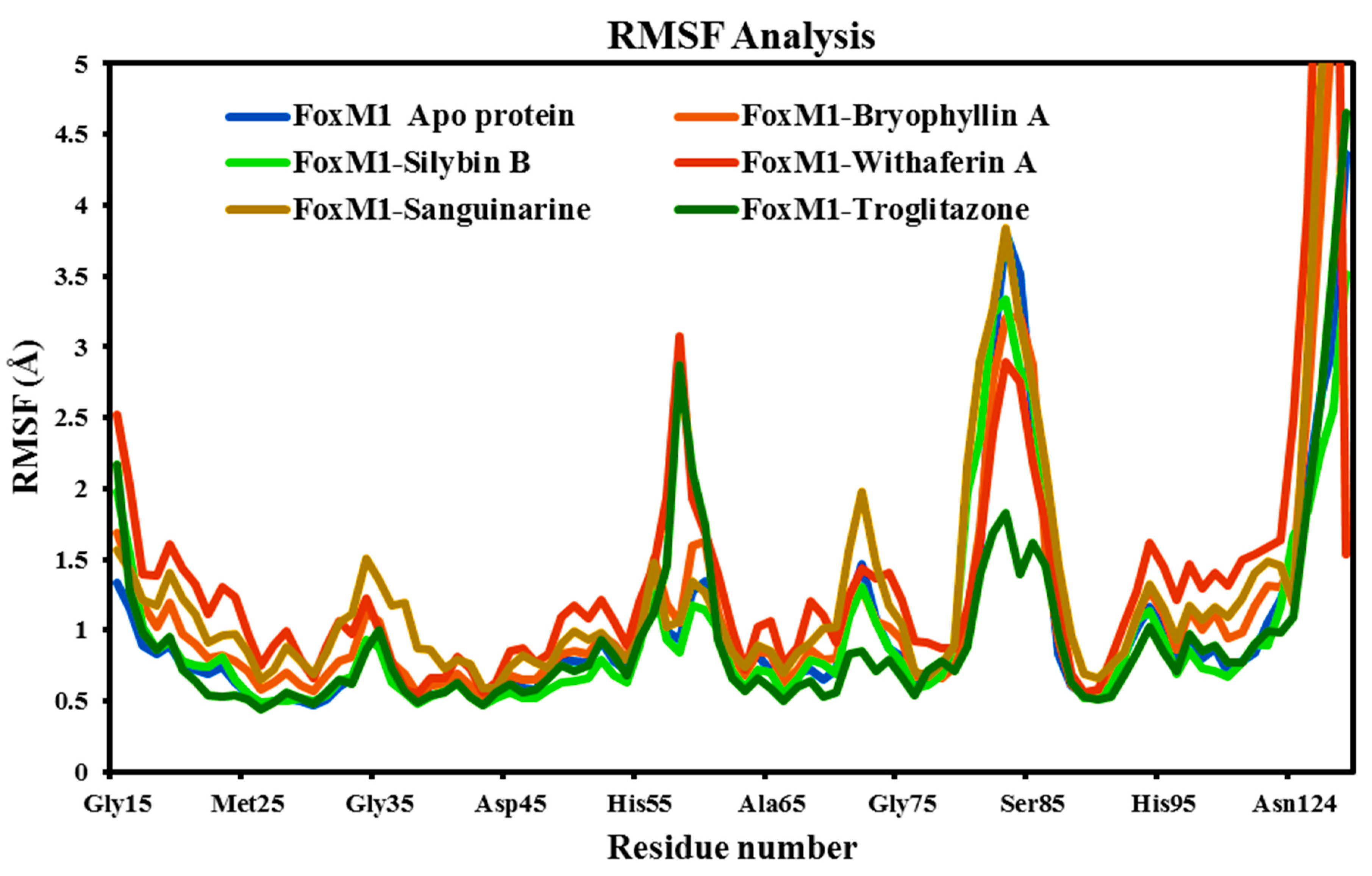
The RMSF determine the protein's residue-by-residue alterations during the course of the simulation. The RMSF value is critical for protein characterisation since it provides information about the protein's local changes as well as the protein chain. The RMSF focuses on residue fluctuations and reveals the importance of these fluctuations in the flexibility of functionally significant residues that affect the protein. In general, low RMSF values are associated with stability, while high RMSF values indicate higher flexibility. Significant alterations were seen throughout the simulation at the N-terminal regions, which are protein-flexible regions (Alsagaby et al., 2022; Ulyiye et al., 2022). During the simulation, all ligands shows interacted with around 42 amino acids of the FoxM1 protein, including Ala277, Arg236, Arg297, Asn283, Asn288, Asn302, Asp293, Asp328, Glu298, Gly280, Gly303, His287, His292, His311, Ile276, Leu259, Leu289, Leu291, Lys260, Lys278, Lys304, Met242, Met244, Phe273, Phe295, Phe307, Pro237, Pro238, Pro279, Ser240, Ser284, Ser290, Ser300, Ser306, Thr258, Thr299, Trp281, Trp308, Tyr239, Tyr241, Val296, and Val305.
The conformational changes of essential amino acids in the FoxM1 binding cavity (lowest RMSF value) demonstrated ligands capacity to generate stable interactions with the FoxM1 protein (
Figure 8). All six systems (FoxM1 Apo protein, FoxM1-Bryophyllin A, FoxM1-Silybin B, FoxM1-Withaferin A, FoxM1-Sanguinarine, FoxM1-Troglitazone) demonstrated almost a similar pattern of fluctuation across the whole structure during simulation, except in FoxM1-Withaferin A and control FoxM1-Troglitazone where moderate fluctuation is observed at Glu58-Ile60 and Gly83. With a few exceptions, a slight fluctuation was observed, favoring the amino residues' stability in a dynamic state. The average RMSF values for the FoxM1 Apo protein, FoxM1-Bryophyllin A, FoxM1-Silybin B, FoxM1-Withaferin A, FoxM1-Sanguinarine, and FoxM1-Troglitazone complexes were 1.01 Å, 1.18 Å, 0.99 Å, 1.40 Å, 1.35 Å, and 0.94 Å, respectively (
Table 4). These results indicate that the studied docking complexes exhibit relatively minor conformational deviations. The lower fluctuations observed in the docking complexes suggest that the residues of FoxM1 surrounding the active region have significant interactions with Bryophyllin A, Silybin B, Withaferin A, and Sanguinarine. Overall, these findings indicate that Bryophyllin A, Silybin B, Withaferin A, and Sanguinarine have the ability to interact effectively with FoxM1, forming stable complexes with limited conformational deviations.
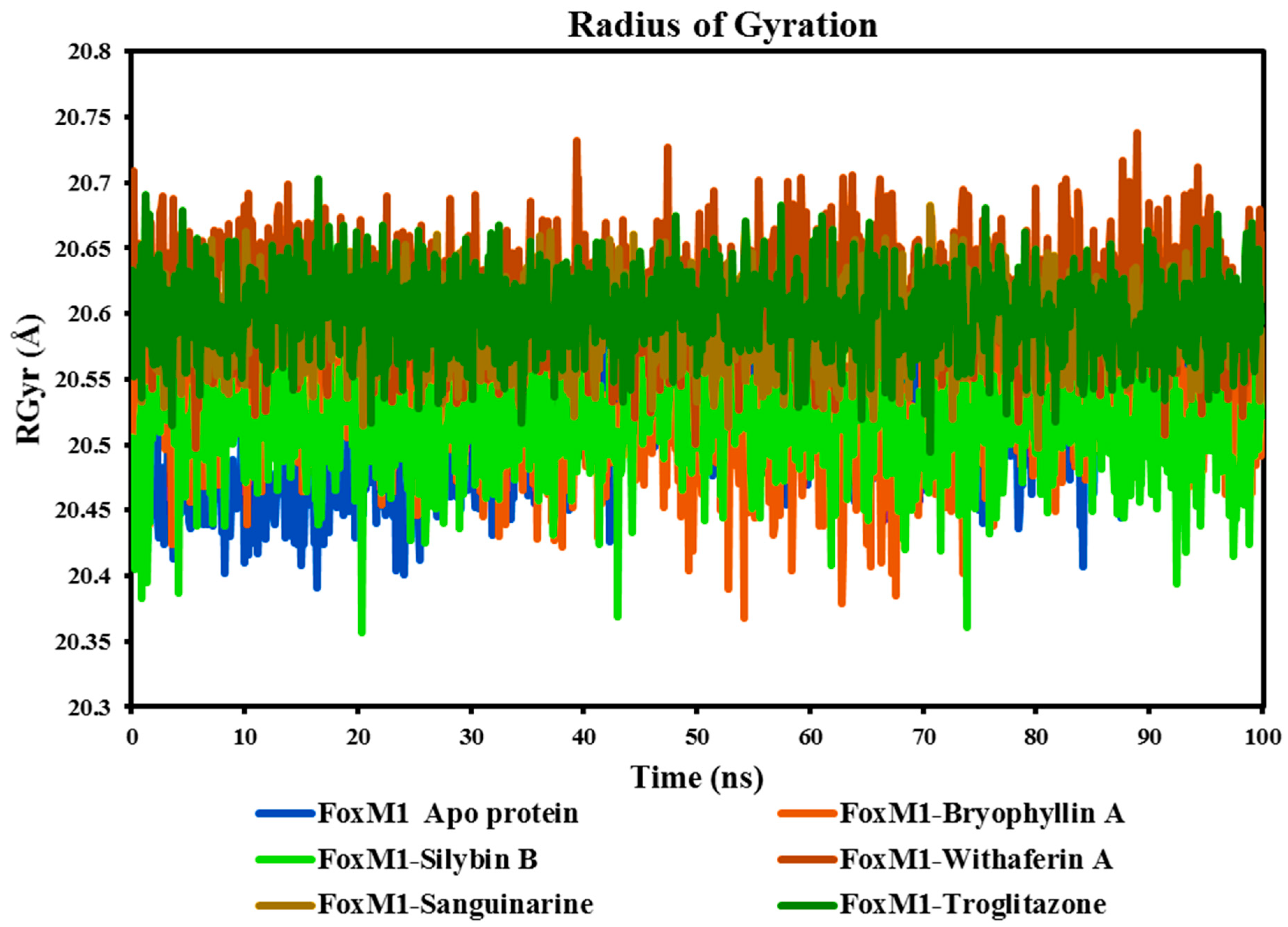
The RGyr parameter, determined from MD simulation trajectories, is an important measure for assessing the stability of protein-ligand complexes. It represents the protein's tertiary structure and overall size, which is important for understanding its compactness and folding (Ayipo et al., 2022). During MD simulation, a constant and consistent fluctuation of RGyr suggests a well-folded protein. The RGyr graph was generated for all complexes, and no aberrant or exceptional deviations were seen in protein complexes with ligands or free apo-protein. As shown in
Figure 9, the RGyr values obtained from the simulations for all three complexes were reported to be 20.3-20.74 Å. However, we observed a slight deviation in the protein-ligand complexes, which then achieved stability, indicating the system's compactness.
Based on the average RGyr values, the FoxM1 Apo protein and its complexes with Bryophyllin A, Silybin B, Withaferin A, Sanguinarine, and Troglitazone exhibit similar sizes and shapes, with average RGyr values ranging from 20.51 Å to 20.61 Å. These values indicate that, on average, the protein-ligand complexes have a relatively compact and stable structure. The close proximity of the average RGyr values suggests that the binding of Bryophyllin A, Silybin B, Withaferin A, Sanguinarine, and Troglitazone to the FoxM1 protein does not significantly alter the overall size or shape of the protein. This indicates that the complexes maintain a similar overall structure compared to the Apo protein, even in the presence of different ligands. This stability is indicative of the protein's ability to accommodate these ligands without significant structural alterations, highlighting the potential for stable interactions between the ligands and the protein's binding site.
Figure 1.
Active site pocket obtained from BIOVIA Discovery Studio and AutoDock tool.
Figure 1.
Active site pocket obtained from BIOVIA Discovery Studio and AutoDock tool.
Figure 2.
Three-dimensional and Two-dimensional interaction analysis of Troglitazone with Protein. The bottom right corner of the figure displays all of the interactions, which are depicted by various colour patterns.
Figure 2.
Three-dimensional and Two-dimensional interaction analysis of Troglitazone with Protein. The bottom right corner of the figure displays all of the interactions, which are depicted by various colour patterns.
Figure 3.
Three-dimensional and Two-dimensional interaction analysis of Withaferin A with Protein. The bottom right corner of the figure displays all of the interactions, which are depicted by various colour patterns.
Figure 3.
Three-dimensional and Two-dimensional interaction analysis of Withaferin A with Protein. The bottom right corner of the figure displays all of the interactions, which are depicted by various colour patterns.
Figure 4.
Three-dimensional and Two-dimensional interaction analysis of Bryophyllin A with Protein. The bottom right corner of the figure displays all of the interactions, which are depicted by various colour patterns.
Figure 4.
Three-dimensional and Two-dimensional interaction analysis of Bryophyllin A with Protein. The bottom right corner of the figure displays all of the interactions, which are depicted by various colour patterns.
Figure 5.
Three-dimensional and Two-dimensional interaction analysis of Sanguinarine with Protein. The bottom right corner of the figure displays all of the interactions, which are depicted by various colour patterns.
Figure 5.
Three-dimensional and Two-dimensional interaction analysis of Sanguinarine with Protein. The bottom right corner of the figure displays all of the interactions, which are depicted by various colour patterns.
Figure 6.
Three-dimensional and Two-dimensional interaction analysis of Silybin B with Protein. The bottom right corner of the figure displays all of the interactions, which are depicted by various colour patterns.
Figure 6.
Three-dimensional and Two-dimensional interaction analysis of Silybin B with Protein. The bottom right corner of the figure displays all of the interactions, which are depicted by various colour patterns.
Figure 7.
Time-dependent RMSD of Cα atoms of Apo FoxM1 protein and its complexes with Bryophyllin A, Silybin B, Withaferin A, Sanguinarine, and the control drug Troglitazone.
Figure 7.
Time-dependent RMSD of Cα atoms of Apo FoxM1 protein and its complexes with Bryophyllin A, Silybin B, Withaferin A, Sanguinarine, and the control drug Troglitazone.
Figure 8.
RMSF of individual amino acids of Cα atoms of Apo FoxM1 protein and its complexes with Bryophyllin A, Silybin B, Withaferin A, Sanguinarine, and the control drug Troglitazone.
Figure 8.
RMSF of individual amino acids of Cα atoms of Apo FoxM1 protein and its complexes with Bryophyllin A, Silybin B, Withaferin A, Sanguinarine, and the control drug Troglitazone.
Figure 9.
Time-dependent Radius of Gyration (RGyr) of Apo FoxM1 protein and its complexes with Bryophyllin A, Silybin B, Withaferin A, Sanguinarine, and the control drug Troglitazone.
Figure 9.
Time-dependent Radius of Gyration (RGyr) of Apo FoxM1 protein and its complexes with Bryophyllin A, Silybin B, Withaferin A, Sanguinarine, and the control drug Troglitazone.
Table 2.
Docking results obtained from EasyDockVina.
Table 2.
Docking results obtained from EasyDockVina.
| S.No. |
PubChem ID |
Compound name |
Binding affinity |
| 1 |
5271805 |
Ginkgetin |
-7.6 |
| 2 |
10621 |
Hesperidin |
-7.2 |
| 3 |
5318767 |
Kaempferol-3-O-beta-rutinoside |
-7.1 |
| 4 |
442428 |
Naringin |
-7.0 |
| 5 |
65727 |
Solanidine |
-6.9 |
| 6 |
265237 |
Withaferin A |
-6.9 |
| 7 |
5488801 |
Bryophyllin A |
-6.9 |
| 8 |
1548994 |
Silybin B |
-6.8 |
| 9 |
5154 |
Sanguinarine |
-6.7 |
| 10 |
259846 |
Lupeol |
-6.7 |
| 11 |
5591 |
Troglitazone (Control compound) |
-6.6 |
| 12 |
24360 |
Camptothecin |
-6.6 |
| 13 |
64945 |
Ursolic acid |
-6.6 |
| 14 |
5280805 |
Rutin |
-6.6 |
| 15 |
5742590 |
Daucosterol |
-6.5 |
| 16 |
11541511 |
Oleandrin |
-6.5 |
| 17 |
72307 |
Sesamin |
-6.4 |
| 18 |
107876 |
Procyanidin |
-6.4 |
| 19 |
5281647 |
Mangiferin |
-6.4 |
| 20 |
5318645 |
Isorhamnetin-3-O-glucoside |
-6.4 |
| 21 |
6918260 |
Larotaxel |
-6.4 |
| 22 |
11250133 |
Procyanidin B1 |
-6.4 |
| 23 |
74978268 |
Quercetin 3'-methyl ether 4'-rhamnosyl-(1->2)-glucoside |
-6.4 |
| 24 |
3084131 |
Paulownin |
-6.3 |
| 25 |
168928 |
Beta-boswellic acid |
-6.2 |
| 26 |
10607 |
Podofilox |
-6.2 |
| 27 |
16216869 |
17-hydroxy-campesta-4,6-dien-3-one |
-6.2 |
| 28 |
175267221 |
Β-carotene |
-6.2 |
| 29 |
3503 |
Gossypol |
-6.1 |
| 30 |
443654 |
Peonidin-3-O-glucoside |
-6.1 |
| 31 |
5281813 |
Wedelolactone |
-6.1 |
| 32 |
5281855 |
Ellagic acid |
-6.1 |
| 33 |
15895316 |
Triterpene |
-6.1 |
| 34 |
163184362 |
13,14-seco-stigmasta-9(11),14-dien-3alpha-ol |
-6.1 |
| 35 |
65064 |
Epigallocatechin-3-gallate |
-6.0 |
| 36 |
92097 |
Taraxerol |
-6.0 |
| 37 |
5175738 |
FDI 6 (Control compound) |
-6.0 |
| 38 |
5280441 |
Vitexin |
-6.0 |
| 39 |
5280445 |
Luteolin |
-6.0 |
| 40 |
969516 |
Curcumin |
-5.9 |
| 41 |
1794427 |
Chlorogenic acid |
-5.9 |
| 42 |
5281316 |
Cucurbitacin B |
-5.9 |
| 43 |
5490064 |
Avicularin |
-5.9 |
| 44 |
42607963 |
8-c-Glucopyranosyleriodictylol |
-5.9 |
| 45 |
932 |
Naringenin |
-5.8 |
| 46 |
9064 |
Cianidanol |
-5.8 |
| 47 |
439533 |
Taxifolin |
-5.8 |
| 48 |
5280343 |
Quercetin |
-5.8 |
| 49 |
5280443 |
Apigenin |
-5.8 |
| 50 |
5281605 |
Baicalein |
-5.8 |
| 51 |
5281672 |
Myricetin |
-5.8 |
| 52 |
2353 |
Berberine |
-5.7 |
| 53 |
23307 |
Coralyne |
-5.7 |
| 54 |
64981 |
Arctigenin |
-5.7 |
| 55 |
222284 |
Beta-sitosterol |
-5.7 |
| 56 |
638024 |
Piperine |
-5.7 |
| 57 |
5280961 |
Genistein |
-5.7 |
| 58 |
5319688 |
4,4′-dihydroxy-2’-methoxy-chalcone |
-5.7 |
| 59 |
5281708 |
Daidzein |
-5.6 |
| 60 |
5281792 |
Rosmarinic acid |
-5.6 |
| 61 |
10807249 |
Caesaldekarin J |
-5.6 |
| 62 |
5316262 |
Cycloolivil |
-5.5 |
| 63 |
68079 |
Isopimpinellin |
-5.4 |
| 64 |
11414799 |
Verubulin |
-5.3 |
| 65 |
10205 |
Plumbagin |
-5.2 |
| 66 |
145858 |
Anthocyanins |
-5.2 |
| 67 |
442793 |
Gingerol |
-5 |
| 68 |
445154 |
Resveratrol |
-5.0 |
| 69 |
72 |
Protocatechuic acid |
-4.8 |
| 70 |
2214 |
Apocynin |
-4.8 |
| 71 |
3469 |
2,5-Dihydroxybenzoic acid |
-4.8 |
| 72 |
8468 |
Vanillic acid |
-4.8 |
| 73 |
10742 |
Syringic acid |
-4.8 |
| 74 |
370 |
Gallic acid |
-4.7 |
| 75 |
2153 |
Theophylline |
-4.7 |
| 76 |
10207 |
Aloe-emodin |
-4.7 |
| 77 |
445858 |
Ferulic acid |
-4.7 |
| 78 |
637540 |
O-Coumaric acid |
-4.7 |
| 79 |
637775 |
Sinapic acid |
-4.7 |
| 80 |
689043 |
Caffeic acid |
-4.7 |
| 81 |
9922008 |
Pipataline |
-4.7 |
| 82 |
31211 |
Zingerone |
-4.6 |
| 83 |
637542 |
P-Coumaric acid |
-4.6 |
| 84 |
8655 |
Syringaldehyde |
-4.5 |
| 85 |
5281515 |
Caryophyllene |
-4.5 |
| 86 |
1183 |
Vanillin |
-4.4 |
| 87 |
10364 |
Carvacrol |
-4.4 |
| 88 |
167551 |
6-Pentadecyl salicylic acid |
-4.4 |
| 89 |
444539 |
Cinnamic acid |
-4.4 |
| 90 |
135 |
4-Hydroxybenzoic acid |
-4.3 |
| 91 |
10281 |
Thymoquinone |
-4.3 |
| 92 |
332 |
4-Vinylguaiacol |
-4.2 |
| 93 |
938 |
3-pyridinecarboxylic acid |
-4.2 |
| 94 |
126 |
P-Hydroxybenzaldehyde |
-3.9 |
| 95 |
289 |
Catechol |
-3.9 |
| 96 |
460 |
Guaiacol |
-3.9 |
| 97 |
68148 |
Chavicol |
-3.9 |
| 98 |
9793905 |
S-Allyl cysteine |
-3.8 |
| 99 |
5386591 |
Ajoene |
-3.4 |
| 100 |
65036 |
Allicin |
-3.1 |
Table 3.
Docking results obtained from AutoDock Vina.
Table 3.
Docking results obtained from AutoDock Vina.
| S.No. |
PubChem ID |
Compound name |
Binding affinity |
| 1 |
265237 |
Withaferin A |
-7.0 |
| 2 |
5488801 |
Bryophyllin A |
-6.9 |
| 3 |
1548994 |
Silybin B |
-6.8 |
| 4 |
5154 |
Sanguinarine |
-6.7 |
| 5 |
5591 |
Troglitazone (Control compound) |
-6.6 |
| 6 |
24360 |
Camptothecin |
-6.5 |
Table 3.
Interaction study of selected ligand using Biovia Discovery Studio.
Table 3.
Interaction study of selected ligand using Biovia Discovery Studio.
| Ligand |
Interacting residues |
Residues showing Vander waals’ force of attraction |
Residues showing Alkyl interaction |
Residues showing Carbon-Hydrogen bonding |
| Troglitazone (Control Compound) |
Glu267, Lys282, Lys260, Val305, Thr 258, Tyr 263, Leu259, Ser306, and Thr264 |
Val305, and Thr 258 |
Thr 258, Leu259, and Lys260 |
Glu267, Lys282, and Ser306 |
| Withaferin A |
Ser284, Lys278, Trp281, Gly289, Ala277, Tyr272, Phe273, Tyr241, Ser240, Met242, His292, Leu291, His287, Arg236, and Asn288 |
Ser284, Lys278, Trp281, Gly289, Ala277, Tyr272, Phe273, Tyr241, Ser240, Met242, His292, Leu291, and His287 |
- |
Arg236, and Asn288 |
| Silybin B |
His287, Leu291, Thr258, Arg297, Trp308, Lys260, Arg286, Ser306, Ser290, and Leu259 |
His287, Leu291, Thr258, Arg297, and Trp308 |
Lys260 |
Arg297, Arg286, Ser306, Ser290, and Leu259 |
| Bryophyllin A |
Val305, Thr258, Lys260, Arg297, Leu289, Ser290, Ser306, Leu259, Arg286, and Trp308 |
Val305, Thr258, Lys260, Arg297, Leu289, and Ser290 |
Leu259, and Arg286 |
Ser306 |
| Camptothecin |
Leu289, Ser290, Ser306, Thr258, Asp261, Leu259, Trp308, Lys260, and Arg286 |
Leu289, Ser290, Ser306, Thr258, andAsp261 |
Lys260, and Arg286 |
Leu259, and Trp308 |
| Sanguinarine |
Lys278, Tyr272, Ala277, Tyr241, Arg236, Ser284, Gly280, and Trp281 |
Tyr272, Tyr241, Arg236, Ser284, Trp281, and Gly280 |
Ala277 |
Lys278 |
Table 4.
The Minimum, maximum and average values of different parameters, RMSD, RMSF, RGyr, and Hydrogen Bonding of studied complexes.
Table 4.
The Minimum, maximum and average values of different parameters, RMSD, RMSF, RGyr, and Hydrogen Bonding of studied complexes.
| |
FoxM1 Apo protein |
FoxM1-Bryophyllin A |
FoxM1-Silybin B |
FoxM1-Withaferin A |
FoxM1-Sanguinarine |
FoxM1-Troglitazone |
| Root-mean-square deviation Å (RMSD) |
| Minimum |
0.95 |
1.03 |
0.80 |
0.82 |
0.84 |
1.11 |
| Maximum |
3.10 |
3.27 |
3.66 |
3.05 |
3.73 |
2.83 |
| Average |
2.18 |
2.28 |
2.79 |
1.88 |
2.93 |
1.93 |
| Root-mean-square fluctuation Å (RMSF) |
| Minimum |
0.46 |
0.52 |
0.47 |
0.55 |
0.59 |
0.44 |
| Maximum |
4.36 |
7.43 |
3.51 |
8.73 |
8.71 |
4.65 |
| Average |
1.01 |
1.18 |
0.99 |
1.40 |
1.35 |
0.94 |
| The radius of gyration Å (RGyr) |
| Minimum |
20.39 |
20.37 |
20.36 |
20.50 |
20.50 |
20.50 |
| Maximum |
20.63 |
20.62 |
20.62 |
20.74 |
20.68 |
20.70 |
| Average |
20.51 |
20.52 |
20.52 |
20.61 |
20.59 |
20.60 |