Submitted:
23 June 2023
Posted:
23 June 2023
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
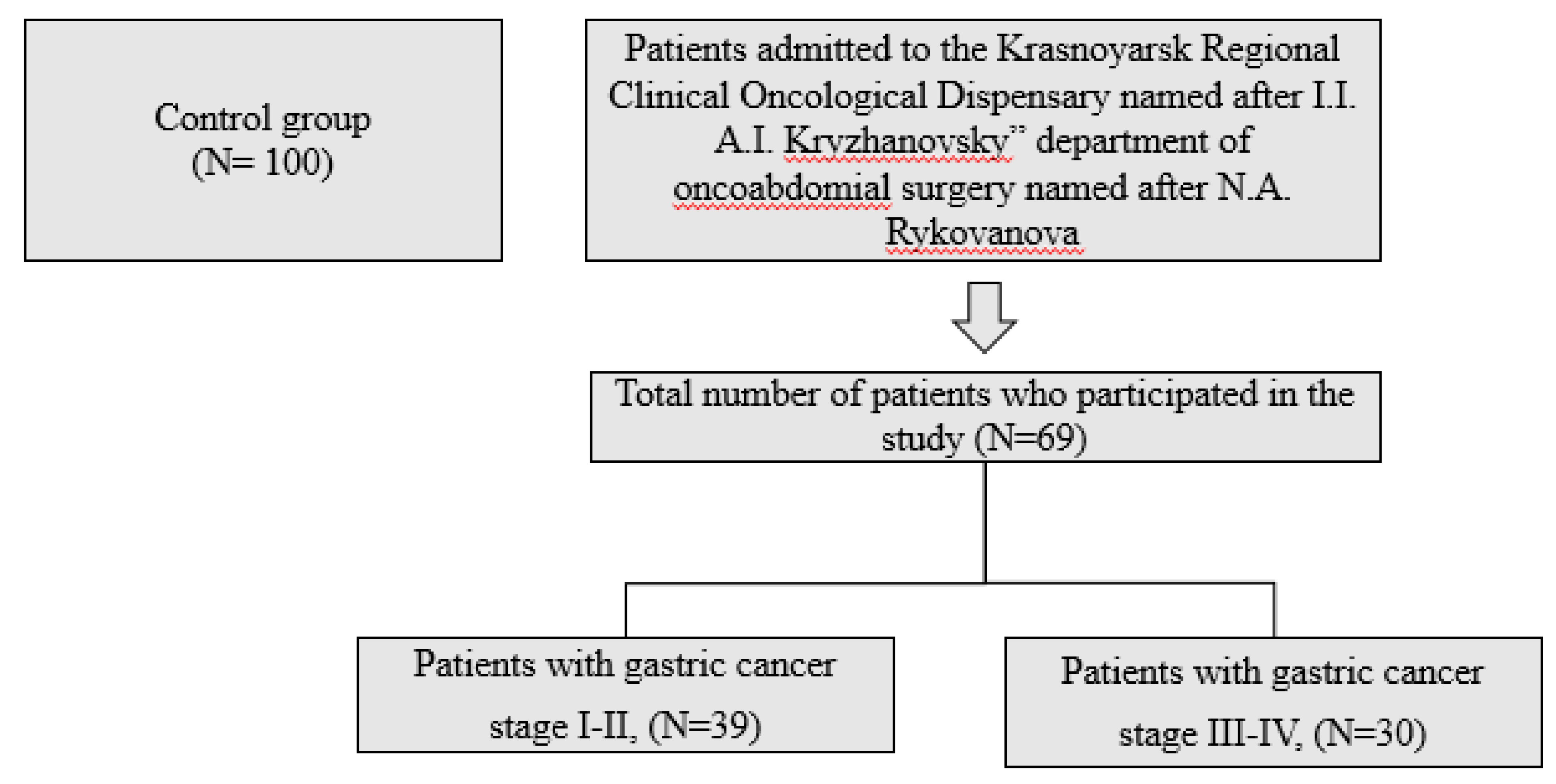
- 1.
- Subjects
- 2.
- Endoscopic examination, histologic examination, H. pylori test, and gastric juice sampling
- 3.
- Determination of the chemiluminescent activity of neutrophilic granulocytes
- 4.
- Statistical analysis
Results
- 1.
- Baseline patient characteristics
- 2.
- Correlation relationships between indicators of chemiluminescent activity of neutrophilic granulocytes and the system “lipid peroxidation - antioxidant defence”
- 3.
- Distribution of groups of patients with gastric cancer depending on the activity of neutrophilic granulocytes
- 4.
- Chemiluminescence of neutrophilic granulocytes in patients with gastric cancer at different stages of the disease
- 5.
- Indicators of the system “lipid peroxidation - antioxidant protection” (LPO-AOP) in gastric cancer, depending on the stage of the disease
Discussion
Conclusion
Author Contributions
Conflict of interest
Source of financing
Compliance with the principles of ethics
References
- Bray F, Ferlay J, Soerjomataran I, Siegel RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68: 394-424, 2018.
- Siegel R, Naishadham D and Jemal A: Cancer statistics, 2013. CA Cancer J Clin 63: 11-30, 2013.
- Katanoda K, Hori M, Matsuda T, Shibata A, Nishino Y, Hattori M, Soda M, Ioka A, Sobue T and Nishimoto H: An updated report on the trends in cancer incidence and mortality in Japan, 1958-2013. Jpn J Clin Oncol 45: 390-401, 2015.
- Plummer M, de Martel C, Vignat J, Ferlay J, Bray F and Franceschi S: Global burden of cancers attributable to infections in 2012: A synthetic analysis. Lancet Glob Health 4: e609-e616, 2016.
- Matsuda T, Ajiki W, Marugame T, Ioka A, Tsukuma H and Sobue T: Research group of population-based cancer registries of Japan: Population-based survival of cancer patients diagnosed between 1993 and 1999 in Japan: A chronological and international comparative study. Jpn J Clin Oncol 41: 40-51, 2011.
- Razzaghi R., Agarwal S., Kotlov N. et al. Compromised counterselection by FAS creates an aggressive subtype of germinal center lymphoma. J Exp Med. 2021. Vol. 218 (3). P. e20201173.
- Zhang Xin-Jia., Yong-Gang L., Xiao-Jun S., Xiao-Wu C., Dong Z., Da-Jian Z. The prognostic role of neutrophils to lymphocytes ratio and platelet count in gastric cancer: A meta-analysis. International Journal of Surgery. 2015. Vol. 21. P. 84–91.
- Keehoon Jung, Takahiro Heishi, Joao Incio, Dai Fukumura. Targeting CXCR4-dependent immunosuppressive Ly6Clow monocytes improves antiangiogenic therapy in colorectal cancer. PNAS. 2017. Vol. 114 (39). P. 10455–10460.
- Ustyanovska Avtenyuk, N., Choukrani G., Ammatuna E., Niki T., Cendrowicz E., Lourens H.J., Huls G., Wiersma V.R., Bremer E. Galectin-9 Triggers Neutrophil-Mediated Anticancer Immunity. Biomedicines. 2021. Vol. 10 (66). P. 10–16.
- Kraus R.F., Gruber M.A. Neutrophils-From Bone Marrow to First-Line Defense of the Innate Immune System. Front Immunol. 2021. Vol. 12 (767175). P. 1–35.
- Huang, H. Matrix Metalloproteinase-9 (MMP-9) as a Cancer Biomarker and MMP-9 Biosensors: Recent Advances. Sensors (Basel). 2018. Vol. 18 (10). P. 3249.
- Bui, T.M., Wiesolek H.L., Sumagin R. ICAM-1: A master regulator of cellular responses in inflammation, injury resolution, and tumorigenesis. J Leukoc Biol. 2020. Vol. 108 (3). P. 787–799.
- Taya, M., Garcia-Hernandez M.L., Rangel-Moreno J., Minor B., Gibbons E., Hammes S.R. Neutrophil elastase from myeloid cells promotes TSC2-null tumor growth. Endocr Relat Cancer. 2020. Vol. 27 (4). P. 261–274.
- Dimitriou, N., Felekouras E., Karavokyros I., Alexandrou A., Pikoulis E, Griniatsos J. Neutrophils to lymphocytes ratio as a useful prognosticator for stage II colorectal cancer patients. BMC Cancer. 2018. Vol. 18 (1). P. 1202.
- Drake IM, Davies MJ, Mapstone NP, Dixon MF, Schorah CJ, White KL, Chalmers DM, Axon AT. Ascorbic acid may protect against human gastric cancer by scavenging mucosal oxygen radicals. Carcinogenesis. 1996 Mar;17(3):559-562. [CrossRef]
- Na HK, Woo JH. Helicobacter pylori Induces Hypermethylation of CpG Islands Through Upregulation of DNA Methyltransferase: Possible Involvement of Reactive Oxygen/Nitrogen Species. J Cancer Prev. 2014;19(4):259-264. [CrossRef]
- Zhuang XQ, Lin SR. Research of Helicobacter pylori infection in precancerous gastric lesions. World J Gastroentero 2000; 6: 428-429.
- Prinz C, Schoniger M, Rad R, Becker I, Keiditsch E, Wagenpfeil S, ClassenM, Rosch T, ScheppW, GerhardM. Key importance of the Helicobacter pylori adherence factor blood group antigen binding adhesin during chronic gastric inflammation. Cancer Res 2001; 61: 1903-1909.
- Zhu Y, Lin J, Li D, Du Q, Qian K, Wu Q, Zheng S. Helicobacter pylori antigen and its IgG, IgA-type specific immunocomplexes in sera from patients with Helicobacter pylori infection. ChinMed J (Engl) 2002; 115: 381-383.
- Futagami S, TakahashiH, Norose Y, KobayashiM. Systemic and local immune responses against Helicobacter pylori urease in patients with chronic gastritis: distinct IgA and IgG productive sites. Gut 1998; 43: 168-175.
- Xu H, Chaturvedi R, Cheng Y, Bussière FI, Asim M, Yao MD, et al. Spermine oxidation induced by Helicobacter pylori results in apoptosis and DNA damage: implications for gastric carcinogenesis. Cancer Res 2004; 64:8521-5; PMID:15574757. [CrossRef]
- Domellof, L. Reversal of gastric atrophy after Helicobacter pylori eradication: is it possible or not? Am J Gastroenterol 1998, 93: 1407-1408.
- Meining A, Stolte M. Close correlation of intestinal metaplasia and corpus gastritis in patients infected with Helicobacter pylori. Z Gastroenterol 2002; 40: 557-560.
- Zhu Y, Lin J, Li D, Du Q, Qian K, Wu Q, Zheng S. Helicobacter pylori antigen and its IgG, IgA-type specific immunocomplexes in sera from patients with Helicobacter pylori infection. ChinMed J (Engl) 2002; 115: 381-383.
- Figueroa G, Faundez G, Troncoso M, Navarrete P, Toledo MS. Immunoglobulin G antibody response to infection with coccoid forms of Helicobacter pylori. Clin Diagn Lab Immunol 2002; 9: 1067-1071.
- Futagami S, TakahashiH, Norose Y, KobayashiM. Systemic and local immune responses against Helicobacter pylori urease in patients with chronic gastritis: distinct IgA and IgG productive sites. Gut 1998; 43: 168-175.
- Drake IM, Davies MJ, Mapstone NP, Dixon MF, Schorah CJ, White KL, Chalmers DM, Axon AT. Ascorbic acid may protect against human gastric cancer by scavenging mucosal oxygen radicals. Carcinogenesis. 1996 Mar;17(3):559-562. [CrossRef]
- Na HK, Woo JH. Helicobacter pylori Induces Hypermethylation of CpG Islands Through Upregulation of DNA Methyltransferase: Possible Involvement of Reactive Oxygen/Nitrogen Species. J Cancer Prev. 2014;19(4):259-264. [CrossRef]
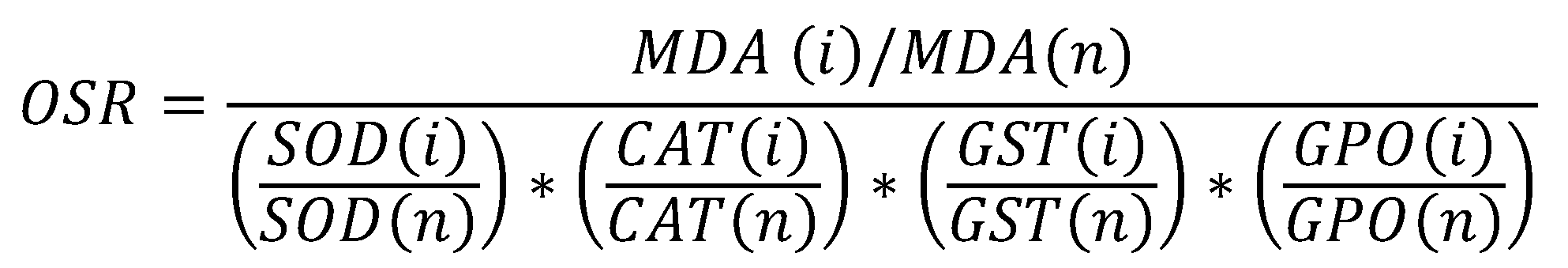
| Parameter | Control group, N=100 | GC I-II,N=39 | GC III-IV,N=30 | p-value |
|---|---|---|---|---|
| Gender (n,%)MaleFemale |
58 (58%) |
23 (59%) |
19 (63%) |
0.325 |
| 42 (42%) | 16 (41%) | 11 (37%) | ||
| Age, (y) | 47.54±12.65 | 57.3±9.65 | 49.87±8.34 | 0.414 |
| Weight, (kg) | 58,4(±5,3) | 56,1(±3,2) | 53,2(±4,1) | 0.256 |
| Height, (m) | 1,75 (±0,05) | 1,7(±0,08) | 1,69(±0,05) | 0.465 |
| BMI, (kg/m2) | 22.69±3.71 | 19.52±3.33 | 17.22±3.89 | 0.398 |
| Alcohol drink1.Never2.Past3.Current |
16 (16%) |
9 (23%) |
7 (23%) |
0.472 |
| 41 (41%) | 16 (41%) | 13 (44%) | ||
| 43 (43%) | 14 (36%) | 10 (33%) | ||
| Smoking1.Never2.Past3.Current |
50 (50%) |
12(31%) |
11(37%) |
0.371 |
| 18 (18%) | 7 (18%) | 11 (37%) | ||
| 32 (32%) | 20 (51%) | 8 (26%) |
| MDA | SOD | CAT | GST | GPO | CP | |
|---|---|---|---|---|---|---|
| Imax spon. |
r=0,72 (p=0,03) |
r=0,78 (p=0,002) |
r=0,71 (p=0,05) |
r=0,78 (p=0,01) |
r=0,78 (p=0,02) |
r=0,84 (p<0,001) |
| I max induced |
r=0,79 (p=0,013) |
r=0,73 (p=0,021) |
r=0,74 (p=0,012) |
r=0,73 (p=0,03) |
r=0,7 (p=0,041) |
r=0,72 (p=0,004) |
| MDA | SOD | CAT | GST | GPO | CP | |
|---|---|---|---|---|---|---|
| Imax spon. |
r=0,74 (p=0,002) |
r=0,7 (p=0,002) |
r=0,75 (p=0,05) |
r=0,71 (p=0,01) |
r=0,75 (p<0,001) |
r=0,8 (p=0,03) |
| I max induced |
r=0,7 (p=0,03) |
r=0,8 (p=0,03) |
r=0,74 (p=0,004) |
r=0,73 (p=0,02) |
r=0,74 (p=0,023) |
r=0,7 (p=0,02) |
| Activity of neutrophilic granulocytes | Gastric cancer I-II N=39 |
Gastric cancer III-IV N=30 |
|---|---|---|
| Normal activity of neutrophilic granulocytes (abs., %) | 10 – patients (26%) | 10 – patients (33%) |
| Increased activity of neutrophilic granulocytes (abs., %) | 8 – patients (21%) | 13 – patients (44%) |
| Reduced activity of neutrophilic granulocytes (abs., %) | 21 – patients (54%) | 7 – patients (23%) |
| Indicators | Control group (N=100), (1) | GC I-II, reduced activity of neutrophilic granulocytes (N=21), (2) |
GC III-IV, increased activity of neutrophilic granulocytes (N=13), (3) |
|---|---|---|---|
| Imax spontaneous (у.е.) |
10.730 [3.262 – 21.997] |
11.583 [4590-17473] |
25.285 [24472 – 26787] |
| p1-3=0,03; p2-3=0,02 | |||
| Squr spontaneous (*106) |
0,22 [0,15-0,4] |
3,4 [0,9-3,7] |
9,6 [7,2 – 9,9] |
| p1-2<0,001 | p1-3<0,001; p2-3<0,001 | ||
| T max spont. (s.) |
969 [615-1753] |
1.336[1.267 -1436] |
1.278 [1.187 – 2.116] |
| Imax induced (у.е.) |
19.904 [7281,5 - 32.121] |
17.625 [16.525-30530] |
33.135,5 [32.635,5 – 63.446,50] |
| p1-3=0,04; p2-3=0,01 | |||
| Squr induced (*106) |
0,4 [0,15-0,6] |
5,4 [4,9-6,1] |
12,8 [11,7-13,5] |
| p1-2<0,001 | p1-3<0,001; p2-3<0,001 | ||
| T max induced (s.) |
1.380,8 [796-1.586] |
1.404 [1.304 -1.525] |
1.456 [1.105 – 1.505] |
| Activation index |
1,3 [0,9-2,0] |
2,61 [1,01-3,02] |
2,1 [1,8-2,7] |
| p1-2<0,001 | p1-3=0,001 |
| Indicators | Control group (N=100), (1) | GC I-II, reduced activity of neutrophilic granulocytes (N=21), (2) |
GC III-IV, increased activity of neutrophilic granulocytes (N=13), (3) |
|---|---|---|---|
| MDA, µmol/1 g of protein |
1,6 [0,96-2,24] |
47,83 [36,7-81,1] |
55,3 [50,3 - 66,75 ] |
| p1-2=0,001 | p1-3=0,001 | ||
| SOD, U/min/1 g protein |
204,41 [151,05-250,3] |
267,1 [185,8-421,6] |
367,2 [272,2 - 431,3] |
| p1-2=0,03 | p1-3=0,002 | ||
| CAT, µmol/s/1 g of protein |
0,27 [0,16-0,39] |
0,77 [0,61-0,88] |
0,62 [0,5 - 0,8] |
| p1-2=0,03 | p1-3=0,03 | ||
| GST, mmol/min/1 g protein |
41,3 [37,7-42,64] |
76,3 [52,1-93,6] |
64,2 [55,6 - 83,2] |
| p1-2=0,04 | p1-3=0,046 | ||
| GPO, µmol/1 g protein |
107,9 [81,19-162,38] |
174,7 [147,3-200,1] |
150,8 [134,7 - 156,2] |
| p1-2=0,01 | p1-3=0,023 | ||
| CP, mg/l |
192,5 [157,5-227,5] |
481,5 [321,5-729,1] |
767,1 [649,5 - 806,8] |
| p1-2<0,001 | p1-3<0,001; p2-3<0,001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




