Submitted:
28 November 2023
Posted:
29 November 2023
You are already at the latest version
Abstract
Keywords:
Introduction

1. Beta-amyloid biomarkers
1.1. Two major beta-amyloid biomarkers: concentration of Aβ42 in the CSF and the density of amyloid aggregates in the brain
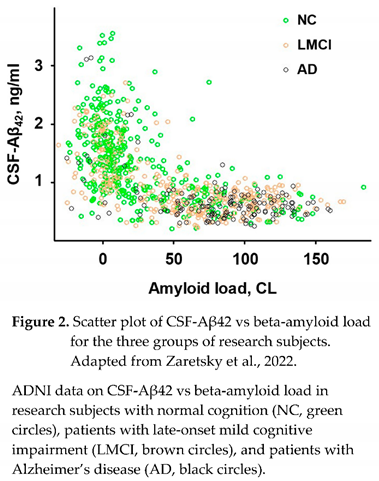

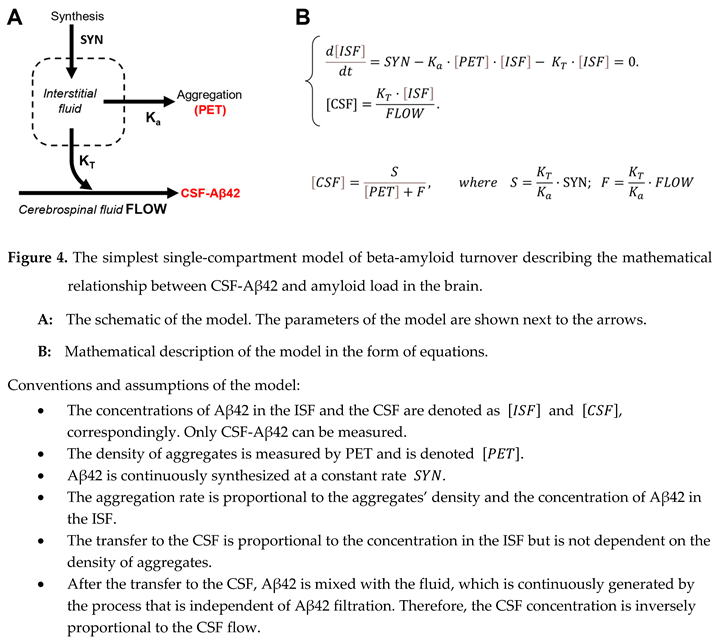
1.2. Characteristic distribution of beta-amyloid biomarkers can be described by a compartment kinetic model
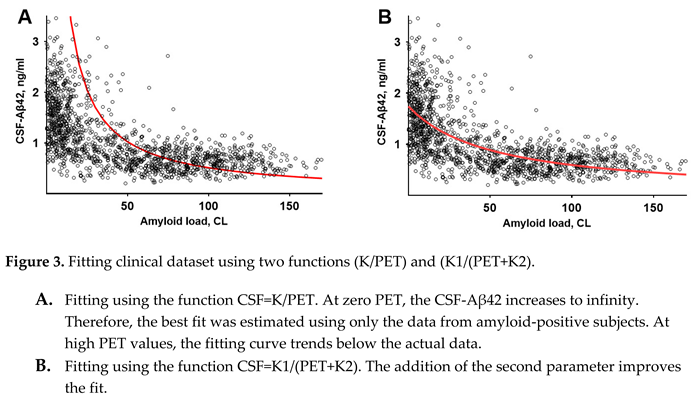

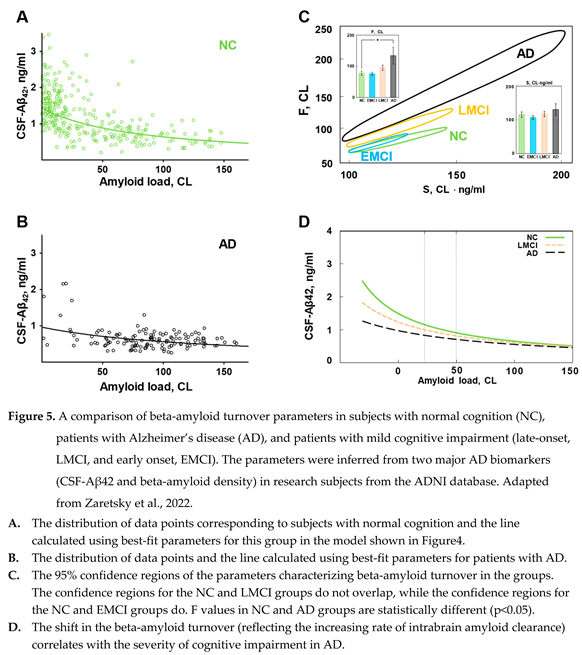
1.3. The distributions of beta-amyloid biomarkers in subjects with normal cognition and patients with AD: the evidence of increased intrabrain beta-amyloid removal rate
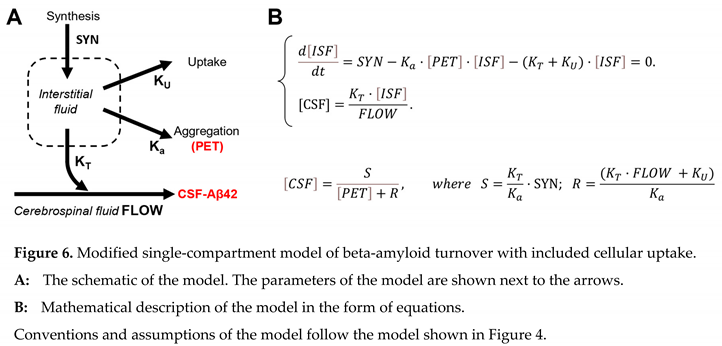

2. Amyloid biomarkers of AD: two biomarkers – two paradoxes
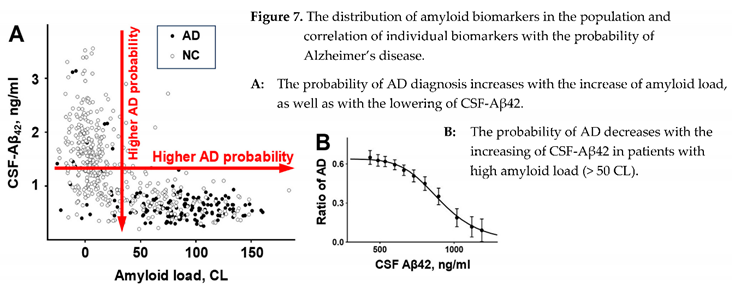

3. Aggregation and cytotoxicity of beta-amyloid require cellular uptake
3.1. How are amyloid deposits formed?


3.2. Is cellular uptake of beta-amyloid relevant to its cytotoxicity?

3.3. The dependence of both toxicity and aggregation of beta-amyloid on cellular uptake: relevance to clinical data on amyloid biomarkers

4. Molecular and cellular mechanisms of beta-amyloid-induced cytotoxicity
4.1. Lysosomal permeabilization by beta-amyloid – the mystery of a reality
- Intracellular concentrations of multiple ions change as synchronous waves.
- Waves of cytoplasmic acidification mirror calcium waves without any temporal offset.
- Waves are short, and the cytoplasmic con cen trations are typically reversed to pre-wave level.
- Ion responses to Aβ exposure are initiated with a noticeable delay of at least several minutes.

4.2. Beta-amyloid oligomers permeabilize membranes by forming channels
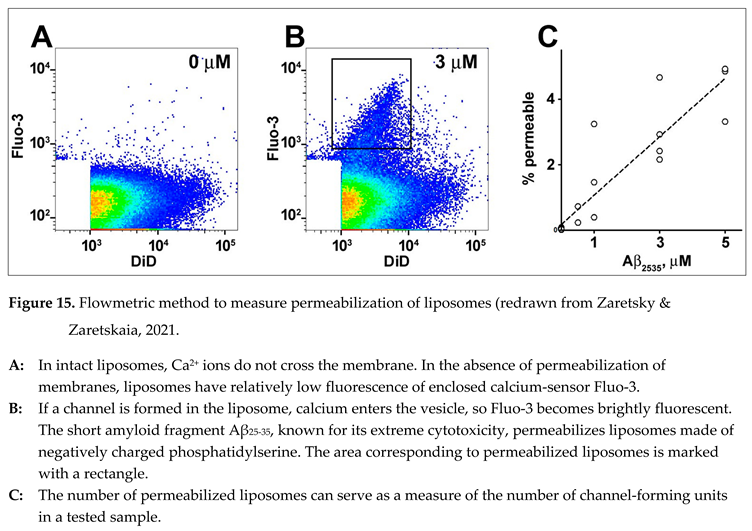
- Thousands of liposomes are permeabilized by short fragment Aβ25-35, known for its neurotoxicity, in a fashion that matches the formation of membrane channels.
- Aβ25-35 permeabilizes the membranes made of phosphatidylserine (acidic, negatively charged phospholipid) but not in the membranes made of phosphatidylcholine (neutral phospholipid, which does not carry a net electric charge).
- No permeabilization was observed by Aβ42 (full-length beta-amyloid, which is relevant to senile plaque formation and is the isoform that correlates with the progression of AD).
- Other short fragments, Aβ22-35 and Aβ31-35, do not permeabilize lipid membranes.
- In planar lipid bilayers, adding Aβ25-35, but not full-length beta-amyloid, resulted in effective channel formation [54]. In studies on lipid bilayers, full-length peptides usually produced only single channels [62,63]. In some studies using full-length peptides, the formation of channels was unreproducible [67].
- Permeabilization was observed only in nega tively charged membranes [70].
- Only some amyloid fragments, but not full-length beta-amyloid, can form membrane channels.
- The formation of single channels in experiments on planar lipid bilayers using full-length beta-amyloid could result from contamination with fragments. Synthetic peptides usually have a purity of less than 99%, so it is statistically possible that there was a sufficient concentration of channel-forming fragments. The dependence of results on the contaminants can explain why some laboratories could not reproduce the data.
- The fragment that can effectively form memb rane channels (Aβ25-35) is mostly lipophilic and carries a single positive charge, while the nega tively charged fragment cannot create channels.
- Positively charged fragments can form channels only in negatively charged membranes. Electrostatic inter action between the membrane and channel-forming unit is most likely needed to incorporate into the membrane.
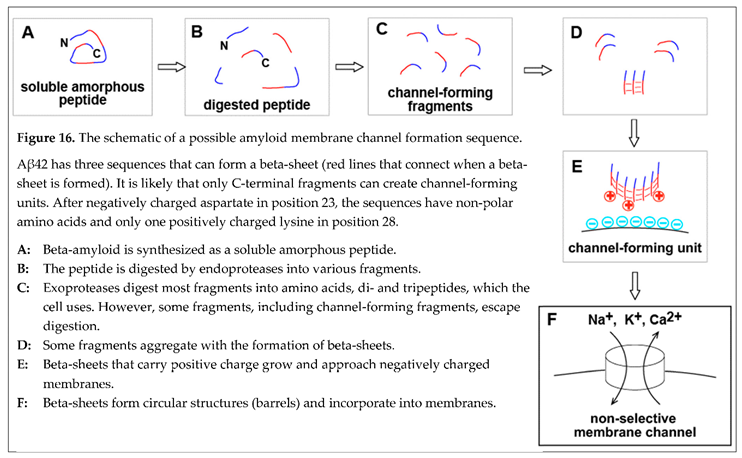
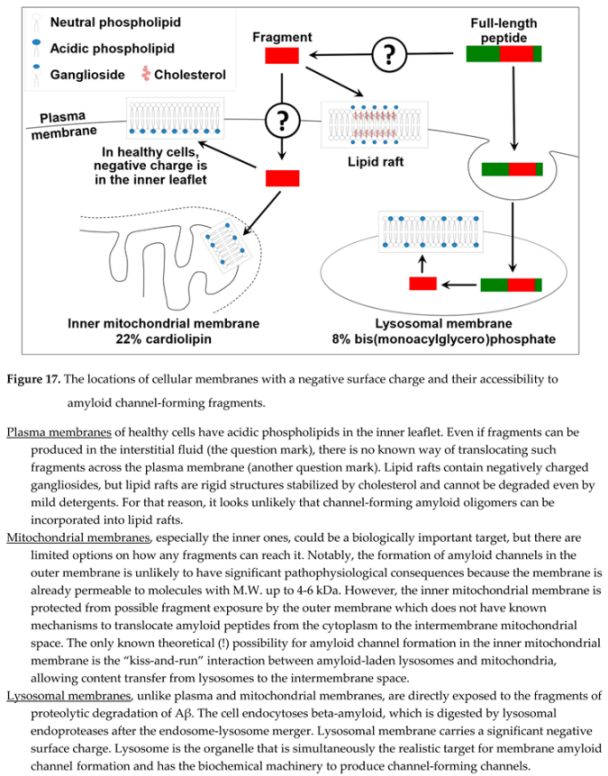
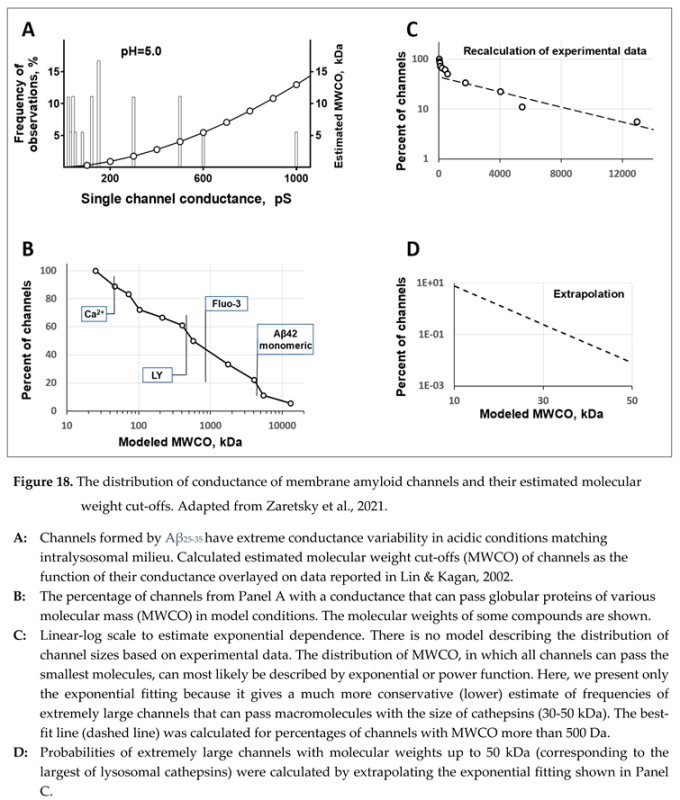

5. Independent clinical evidence of increased beta-amyloid uptake in AD

6. Cornerstones of the amyloid degradation hypothesis of Alzheimer’s disease
- 1)
- Amyloid membrane channels are formed by fragments of Aβ (such as Aβ25-35 but most likely other fragments as well), not full-length beta-amyloid (such as Aβ42 or Aβ40).
- 2)
- Incorporating amyloid channel-forming oligomers into lipid membranes requires that the membranes have a negative surface charge.
- 3)
- Lysosomal membranes are one of three cellular membranes with a negative surface charge (along with the inner leaflet of plasma membrane and mitochondrial membranes).
- 4)
- Lysosomes provide both the biochemical machinery, which creates channel-forming fragments from endocytosed beta-amyloid, and the membranes permeabilized by these channels.
- 5)
- Any (including the smallest) amyloid channels dissipate the lysosomal membrane’s pH gradient, resulting in lysosomal dysfunction due to the inhibition of acidic proteases.
- 6)
- Lysosomal dysfunction prevents normal quality control of organelles (mitochondria are the most sensitive to this disturbance due to high meta bolic damage associated with their function).
- 7)
- Largest amyloid channels can pass macro molecules, including cathepsins, which either directly mediate necrosis or activate apoptosis. Both necrosis and apoptosis result in cell death.
- 8)
- Membrane amyloid channel formation can be considered the primary molecular mechanism of beta-amyloid toxicity.
- 9)
- Membrane amyloid channels mediate lysosomal permeabilization.
- 10)
- Neuronal death is the cellular mechanism of cognitive impairment and dementia in AD.
- 11)
- The degree of cell death depends on the accu mulated Aβ toxicity, so the disease progresses over time.
- 12)
- The accumulated toxicity of Aβ, and as a result, the degree of neuronal death, has a positive correlation with the intensity of Aβ endocytosis.
- 13)
- The initial step of amyloid aggregation occurs intralysosomally. Therefore, the accumulation of amyloid aggregates positively correlates with the intensity of endocytosis of beta-amyloid.
- 14)
- Both cytotoxicity and intrabrain aggregation of beta-amyloid positively correlate with the intensity of endocytosis, which explains why the accumulation of extracellular amyloid aggregates is the biomarker of AD.
- 15)
- Both aggregation on existing amyloid aggregates and cellular uptake decrease interstitial concent rations of soluble Aβ, so a low concentration of soluble Aβ in the CSF serves as AD biomarker.
- 16)
- Higher cellular uptake results in faster neuro degeneration and lowers concentration of soluble Aβ independent of aggregation of existing aggregates, so lower soluble Aβ has a negative correlation with the progression of AD, which is independent of the density of deposits (measured by PET).
- 17)
- The severity of cognitive impairment in patients with late-onset AD positively correlates with the intensity of cellular Aβ uptake.
- 18)
- There is wide variability of sensitivity to amyloid endocytosis: patients with similar uptake can have significantly different levels of neuronal death.
- 19)
- The sensitivity to the endocytosed beta-amyloid depends on the balance between the rates of production and degradation of channel-forming fragments, the activity of biochemical pathways mediating neuronal death induced by lysosomal permeabilization, and cellular protection against consequences of lysosomal permeabilization.
- 20)
- Increased sensitivity to the endocytosed beta-amyloid could be one of the alternative mechanisms of AD: patients with early-onset AD do not have an increased Aβ uptake.
7. The description of the amyloid degradation toxicity hypothesis of Alzheimer’s disease at various levels
7.1. Molecular level
| Beta-amyloid is cleaved from a long precursor molecule (amyloid precursor peptide, APP) by proteases called secretases. |
| The predominant form is the 40-amino acid-long peptide (Aβ40). However, senile plaques in the brain, which are histological hallmarks of AD, contain mostly 42-amino acid-long Aβ42. Both amyloid forms are produced by consecutive action of beta-secretase and gamma-secretase. |
| Immediately after synthesis, beta-amyloid has no secondary structure and is soluble. |
| With time, the peptide can form a pleated beta-sheet (one of two major secondary protein structures) within the molecule. The formation of multimolecular beta-sheets produces beta-amyloid oligomers. |
| Elongation of oligomers and interaction between multiple oligomeric complexes form insoluble aggregates. Senile plaques are amyloid polymers that include other proteins. |
| Polymerization of beta-amyloid results in the formation of insoluble aggregates. Among beta-amyloid isoforms, Aβ42 is most prone to aggregation. |
| Soluble beta-amyloid is toxic to healthy cells in vitro, while aggregated beta-amyloid is non-toxic. |
| The oligomeric form of beta-amyloid mediates the peptide’s cytotoxicity. |
| Senile plaques are the hallmark of AD but are not the etiology of the disease. |
| No known receptor or ion channel can be activated by beta-amyloid peptide(s) in a way that such activation would result in cell death. |
| Absorption of beta-amyloid on lipid membranes can cause membrane damage only in concentrations exceeding 1 µM, while extracellular concentrations of beta-amyloid are in the nanomolar range. |
| Beta-amyloid peptide(s) can form membrane channels. |
| The formation of amyloid membrane channels is the only known primary molecular mechanism that can explain the cytotoxic action of endogenous beta-amyloid. |
| Some amyloid fragments form membrane channels much more effectively than full-length peptides. |
| Not all fragments have channel-forming ability. |
| Channels are formed in membranes carrying negative surface charge. |
| Multiple molecules of peptide form each channel (channels are oligomers). |
| Amyloid membrane channels are non-selective. |
| Channels are giant (with conductance up to several nanosiemens) and have various sizes. |
| The largest channels (conductance higher than 1 nS) can pass macromolecules. |
|
Beta-amyloid can induce cell damage if: 1. There is a production of channel-forming fragments 2. These fragments can target membranes carrying negative surface charge |
| Alzheimer’s disease is developing over multiple years. |
| The conductance of a single channel is sufficient to destroy the barrier function of the membrane of an organelle or whole cell. |
| In physiological conditions, the formation of amyloid channels, which can cause cell death, is rare. It is likely that not every instance of channel formation leads to cell death. |
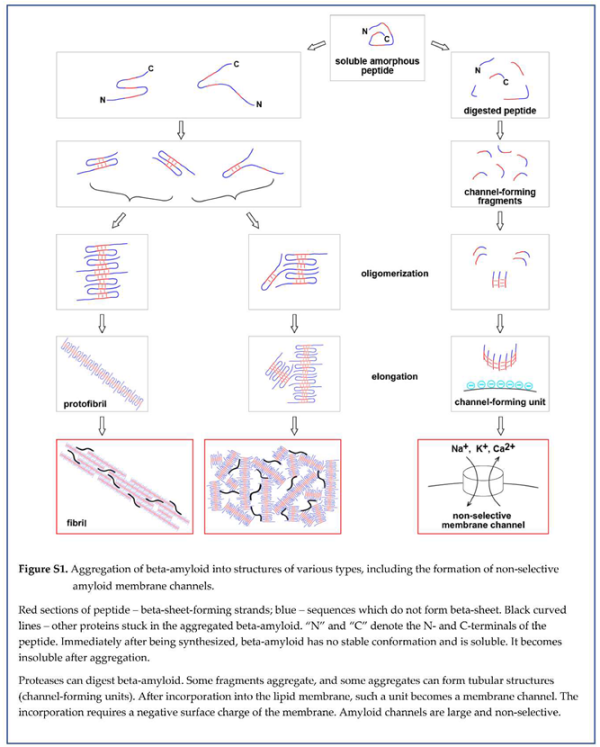
7.2. Organelle level
| The outer leaflet of the plasma membrane of healthy cells is not negatively charged. |
| No reliable experimental electrophysiological data supports the formation of giant amyloid channels in the plasma membrane of cultured cells in vitro. |
| Amyloid membrane channels are not formed in the cellular plasma membrane. |
| The inner mitochondrial membrane (IMM) is negatively charged, but known pathways require the involvement of other organelles (such as endosomes) to deliver channel-forming beta-amyloid fragments to the IMM. |
| The mitochondrial membrane is not a likely target for permeabilization by amyloid membrane channels. |
| Beta-amyloid is endocytosed by cells and is accumulated in lysosomes. |
| The presence of various lysosomal proteases suggests that at least some of them can generate channel-forming fragments. |
| Lysosomal membranes have a strong negative surface charge, which is the prerequisite for forming membrane amyloid channels. |
| Lysosomes are both the producers of channel-forming fragments and the targets of these toxic amyloid species. |
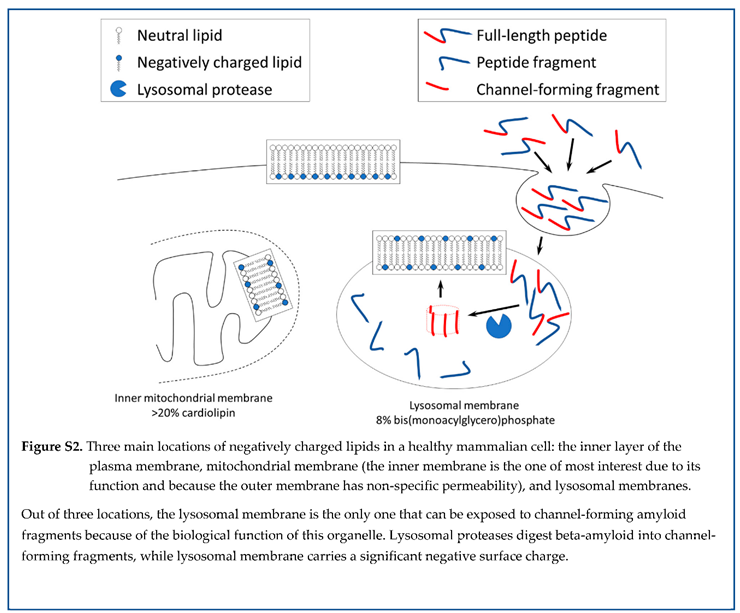
7.3. Cellular level
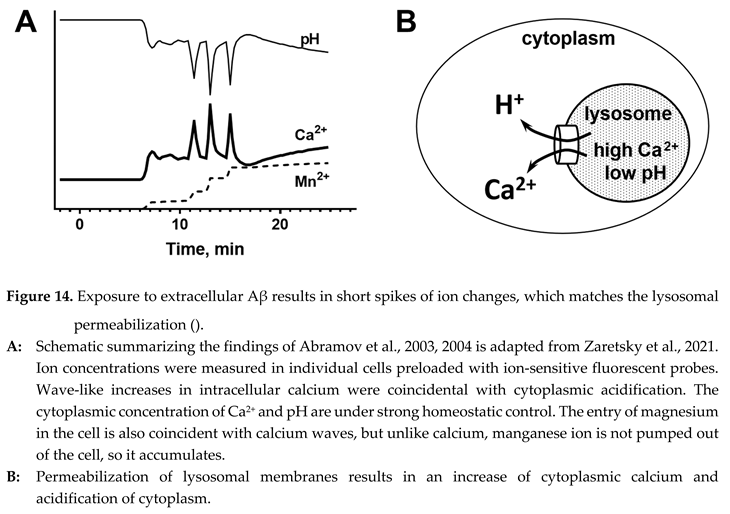
| Cells exposed to the Aβ42 demonstrate synchronous oscillations of concentrations of several intracellular ions (calcium, sodium, potassium, pH) that match the concept of a non-selective membrane channel opening. |
| The frequency of channel conductance oscillations observed electrophysiologically (millisecond range) does not match the time scale of oscillations in intracellular ion concentrations (seconds to minutes). |
| The conductance of amyloid membrane channels is very high. If a channel is formed in the plasma membrane, intracellular and extracellular ion concentrations will equilibrate fast. |
| The cellular ion concentrations return to their baseline after each wave induced by exposure to beta-amyloid. |
| The direction of ion disturbances and rapid recovery does not support the concept that channels are formed in the plasma membrane. |
| Amyloid membrane channels are not formed in the cellular plasma membrane. |
| Observed oscillations in intracellular ion concentra tions (an increase in calcium and potassium and a decrease in pH and sodium) match the ion distur bances that the permeabilization of lysosomes could cause. |
| The small volume of individual lysosomes allows for quick restoration of normal ion balance in the cell following the damage to a single lysosome. This results in the ion disturbance being observed as a wave. |
| The oscillations are caused by the permeabilization of multiple lysosomes. |
| Channels in lysosomal membranes explain intracellular ion changes induced by exposure to beta-amyloid. |
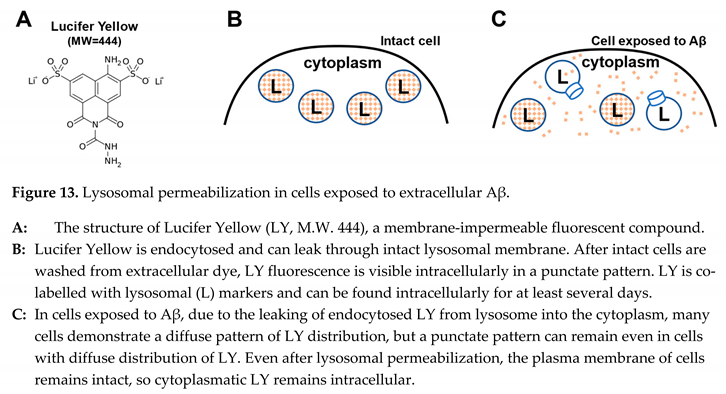
| Cells exposed to the Aβ42 demonstrate the content leakage from multiple lysosomes (but not all) without signs of plasma membrane damage. |
| Cells exposed to the Aβ42 demonstrate lysosomal content leakage before cell death. |
| In cells exposed to the beta-amyloid, lysosomes leak not only small molecules (such as Lucifer Yellow, M.W.444 or ethidium bromide, M.W.394) but also larger molecules such as cathepsin D and enzymes with M.W. 150 kDa. |
| The electrophysiological data suggests that the largest amyloid channels can pass macromolecules. |
| Channels in lysosomal membranes explain lysosomal permeabilization induced by exposure to beta-amyloid. |
| Channels in lysosomal membranes explain lysosomal permeabilization that occurs without damage to the plasma membrane. |
| Even a single channel is enough to equilibrate ion concentrations inside the entire organelle with the cytoplasm. |
| Amyloid membrane channel, regardless of its size, is permeable to protons and can cause lysosomal content to become non-acidic. |
| Neutralization of lysosomal content inactivates acidic proteases, prevents normal digestion of lysosomal cargo, and results in lysosomal failure. |
| Improper cellular recycling due to lysosomal failure prevents the removal of damaged lysosomes. |
| Channels formed in lysosomal membranes can explain lysosomal failure, which is the hallmark of AD. |
| Though rare, the largest amyloid channels (with a con ductance of several nS) can transport macro mo lecules equal to or larger than lysosomal cathepsins. |
| The lysosomal cathepsins released into the cytoplasm can induce either necrosis or activate caspa ses, leading to apoptosis. In both cases, cell death occurs. |
| Lysosomal permeabilization due to the formation of channels in the lysosomal membrane can lead to cell death. |
| After exposure to beta-amyloid, the pathway leading to cell death is a multistep process that includes endocytosis, intralysosomal proteolysis, and the leakage of cathepsins into the cytoplasm. |
| The formation of a giant amyloid channel is a rare event. Only a tiny percentage of amyloid channels can kill the cell. |
| It could require prolonged exposure to beta-amyloid to produce a sufficient number of cytotoxic channels in a significant proportion of cells. |
| Cell death does not occur immediately after exposure to beta-amyloid. |
| The formation of small channels is a more frequent event, but it does not directly lead to cell death. |
| Cells with lysosomes carrying small amyloid channels survive but damaged lysosomes remain dysfunctional. |
| After exposure to beta-amyloid, cell death is primarily mediated by the formation of extremely large (giant) channels. |
| Lysosomal failure in AD is observed because many damaged neurons survive but cannot recycle damaged lysosomes. |
| The symptoms of AD depend on the number and functional status of remaining neurons. |
| Neurons that are damaged by the formation of small amyloid channels can recover. |
| The recovery requires the prevention of the formation of new channels. |
| It is feasible that some AD symptoms (specifically those that appear due to the dysfunction of surviving neurons) could be reversed if the formation of new amyloid channels is prevented. |
| The progression of AD depends on the rate of neuronal death. |
| Closing small amyloid channels is an ineffective way to prevent amyloid-induced neuronal death. |
| To slow the progression of AD, the cyto toxicity induced by the formation of giant amyloid channels should be prevented. |
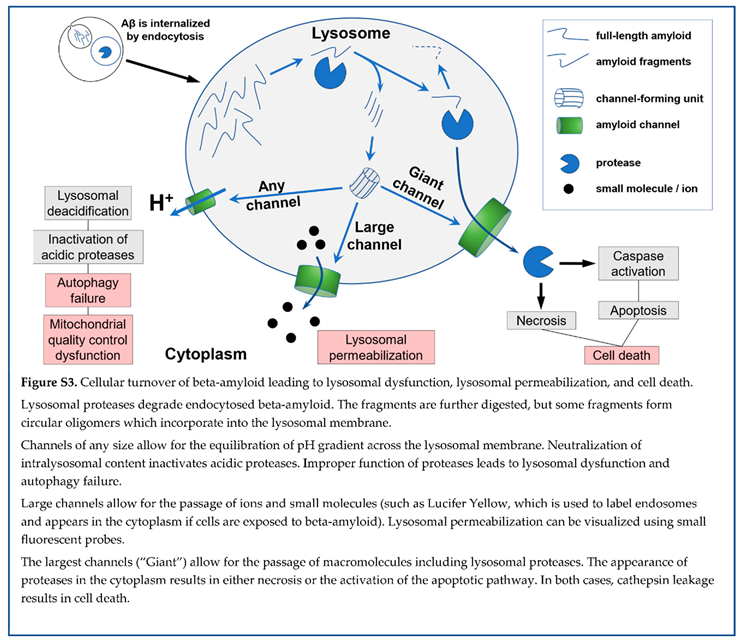
7.4. Tissue/Organ level
| Neurons are responsible for 70–80% of total brain energy consumption. | |
| Neuronal loss is a major contributor to lower brain metabolic activity in patients with AD. | |
| Lysosomal failure leads to improper recycling of various cell organelles. | |
| Mitochondria are the most sensitive to improper recycling due to the high rate of metabolic damage. | |
| Accumulation of damaged mitochondria results in an increased generation of reactive oxygen species, amplifying the damage to other organelles. | |
| Damaged but not recycled organelles occupy intracellular space and prevent normal mitogenesis. | |
| Mitophagy/mitogenesis disbalance in cells exposed to beta-amyloid explains higher production of toxic ROS and could be another contributor to lower brain metabolic activity in patients with AD. | |
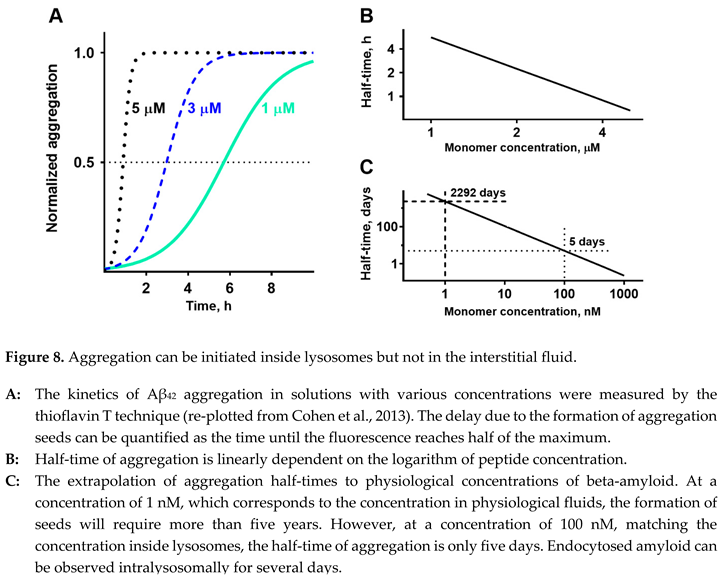
| The aggregation of Aβ42 cannot spontaneously initiate in the interstitial fluid due to a very low peptide concentration (around 1 nanoM). | |
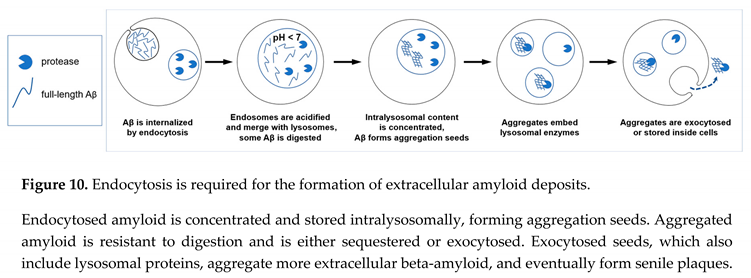
| Endocytosed Aβ42 is concentrated inside lysosomes up to 100-fold and can be observed intracellularly for more than 48 hours. | |
| Estimated intralysosomal Aβ42 concentration and the length of Aβ42 presence inside lysosomes allow for the initiation of amyloid aggregation. | |
| Aggregated Aβ42 is resistant to proteolytic digestion and can be sequestered inside cells. | |
| Intracellular amyloid aggregates are typical in AD brains. | |
| As an alternative to intracellular sequestration, the aggregated peptide is exocytosed along with remaining non-digested soluble Aβ42. | |
| When exposed to Aβ42, cultured cells in vitro promote beta-amyloid aggregation. | |
| Extracellular aggregation seeds are initially formed intralysosomally. | |
| Higher endocytosis intensity increases aggregation seeds’ production to the interstitial fluid. | |
| Faster accumulation of amyloid deposits will be observed in patients with a higher rate of Aβ42 endocytosis. |
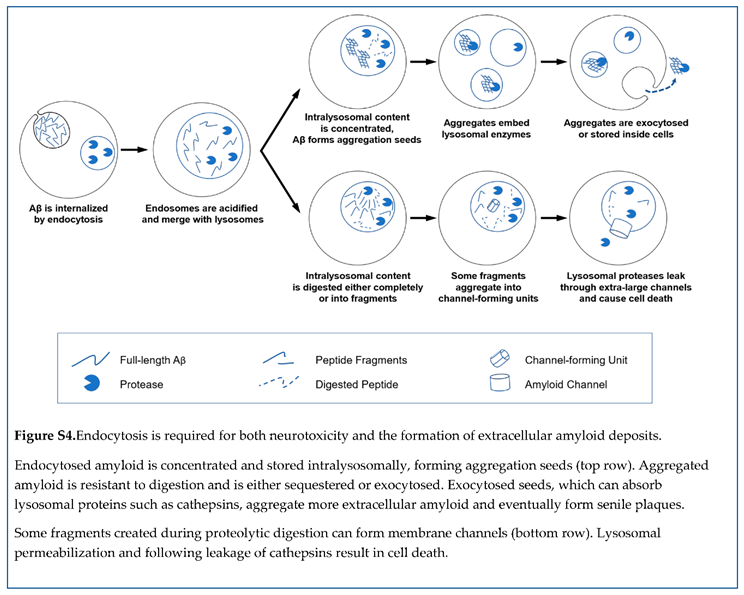
7.5. Organism level
| The cytotoxicity and the appearance of senile plaques are both the consequence of the same initial process - endocytosis of Aβ42. |
| The density of amyloid aggregates in the brain positively correlates with the level of neurodegeneration. |
| The density of amyloid deposits can be measured in living subjects by positron emission tomography (PET) using appropriate positron-emitting probes. |
| While the density of amyloid aggregates is relevant to AD pathophysiology, it is an indirect biomarker of AD. |
| In patients with AD, the concentration of Aβ42 in the CSF is lower than in the subjects with normal cognition. |
| Synthesis of Aβ42 in patients with AD is not differ rent from that of subjects with normal cognition. |
| In patients with AD, CSF flow is either the same or lower than in subjects with normal cognition. |
| Neither lower synthesis nor higher CSF flow explains the lower concentration of Aβ42 in the CSF in patients with AD. |
| Existing amyloid aggregates serve as aggregation centers for soluble Aβ42. |
| If the density of existing aggregates is high, more freshly synthesized Aβ42 aggregates. |
| If a higher ratio of freshly synthesized Aβ42 aggregates, the concentration of Aβ42 in the interstitial fluid decreases, so less Aβ42 is transported to the CSF. |
| Patients with AD have a higher density of amyloid deposits. |
| AD patients have lower levels of Aβ42 in the CSF due to the increased aggregation of Aβ42 on existing amyloid plaques. |
| The density of amyloid deposits, which is the biomarker of AD, is the primary determinant of the level of Aβ42 in the CSF. |
| The concentration of Aβ42 in the CSF is relevant to AD pathophysiology, but similarly to the density of amyloid deposits, it also is the indirect biomarker of AD. |
| The concentration of Aβ42 in the CSF as the standalone biomarker has a diagnostic value similar to PET measurements. |
| According to multivariate correlation analysis, CSF-Aβ42 levels and PET imaging data have independent predictive powers for AD diagnostics, even though these two biomarkers are highly correlated. |
| Combining two biomarkers (PET data and the concentration of Aβ42 in the CSF) provides more information than each biomarker alone. |
| If we compare the concentration of Aβ42 in the CSF of patients with AD and subjects with normal cognition with the same density of amyloid deposits, the Aβ42 concentration in patients with AD is significantly lower. |
| There is some aggregation-independent process that affects the concentration of Aβ42 in the interstitial fluid and has a rate that is different between patients with AD and subjects with normal cognition. |
| The rate of removal with CSF is not higher in patients with AD compared with subjects with normal cognition, so this additional process occurs inside the brain. |
| The synthesis rate of Aβ42 is not different between patients with AD and subjects with normal cognition. |
| The additional process is the aggregation-independent intrabrain removal of Aβ42. |
| A high ratio of patients with a high density of amyloid aggregates also have advanced neurodegeneration, but some of them have no cognitive deficiencies at all. |
| A maximal concentration of Aβ42 in the CSF can be observed for each given density of amyloid deposits. |
| Subjects with the highest possible concentrations of Aβ42 in the CSF for a given amyloid deposit density have a very low AD probability. |
| Higher aggregation-independent intrabrain removal of Aβ42 corresponds to the decrease of Aβ42 concentration in the CSF. |
| The aggregation-independent intrabrain removal of Aβ42 is more intense in the brains of AD patients. |
| Most of intrabrain Aβ42 removal is mediated by proteolytic degradation. |
| Most proteases are located intracellularly. |
| To be degraded by proteases, Aβ42 needs to be internalized by cells. |
| The primary way of Aβ42 cellular uptake is endocytosis. |
| Aβ42 endocytosis is increased in the brains of AD patients. |
| Endocytosis is the initial step of beta-amyloid neurotoxicity. |
| Increased Aβ42 endocytosis is involved in higher neurodegeneration in AD patients. |
| The stable isotope labeling kinetics (SILK) technique enables the investigation of beta-amyloid turnover in the brains of living patients. |
| SILK studies showed increased irreversible intrabrain removal of Aβ42 in AD patients, which can be observed even in the absence of existing plaques. |
| The data generated by the studies of beta-amyloid turnover supports the hypothesis about higher Aβ42 endocytosis in patients with AD. |
| SILK studies also demonstrated that patients with mutation-induced AD are characterized by the pre sence of an “exchange pool” of freshly synthesized Aβ42. In subjects with normal cognition, the size of such a pool is not statistically different from zero. |
| Increased endocytosis of Aβ42 is associated with the exocytosis of non-aggregated intralysosomal Aβ42, which is exocytosed along with amyloid aggregation seeds. |
| Dramatically increased endocytosis of Aβ42 fits the concept of “exchange pool.” |
| Increased endocytosis explains both increased irreversible intrabrain removal of Aβ42 and the larger size of its “exchange pool.” |
| Increased cellular uptake of Aβ42 is one of the reasons for the increased production of channel-forming fragments, but there could be other reasons. |
| Increased activity of proteases producing channel-forming fragments and/or decreased activity of proteases degrading fragments would result in higher concentrations of toxic fragments. Such disbalance would increase beta-amyloid toxicity without increased uptake. |
| Cellular Aβ42 uptake in patients with early-onset AD is not different from uptake in subjects with normal cognition. The early-onset AD is most likely characterized by higher toxicity of endocytosed Aβ42. |
| The amyloid degradation toxicity hypothesis provides the framework for the pathophysiology-based classification of various AD forms. |
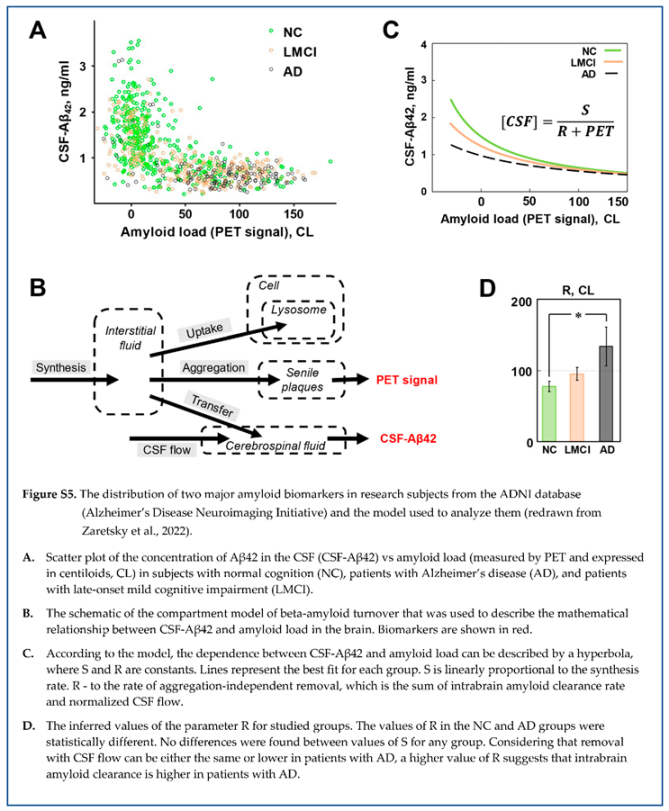
7.6. Population level
| The probability of the disease in a particular patient increases with increased accumulated cellular uptake of Aβ42. |
| The probability of AD increases with age. |
| Cellular uptake defines the toxic insult made by beta-amyloid. Still, the level of neuronal death, the severity of clinical symptoms, and eventually the diagnosis of AD depends on the individual relative toxicity of endocytosed beta-amyloid. |
| The ability of lysosomal proteases to produce and degrade channel-forming fragments defines the relative toxicity of endocytosed amyloid. Also, the ability of cells to resist the consequences of lysosomal permeabilization has individual variability (for example, due to different levels of cytoplasmatic protease inhibitors). |
| There is a wide variability of sensitivity (resistance) to the toxic action of beta-amyloid. |
| Patients with a high rate of endocytosis have a higher probability of advanced neurodegeneration and AD diagnosis, but some of them can have no cognitive deficiencies. |
| High endocytosis of beta-amyloid leads to increased accumulation of amyloid aggregates (higher amyloid load, higher amyloid PET signal) |
| On average, patients with high amyloid load have a higher rate of neurodegeneration and a higher probability of AD diagnosis. Still, some patients with high values of amyloid PET signal can have normal cognition. |
| The same process - endocytosis of Aβ42 – initiates both neurotoxicity and amyloid aggregation. |
| Both accumulated cytotoxicity and the density of senile plaques are increasing over time (and, correspondingly, with age). |
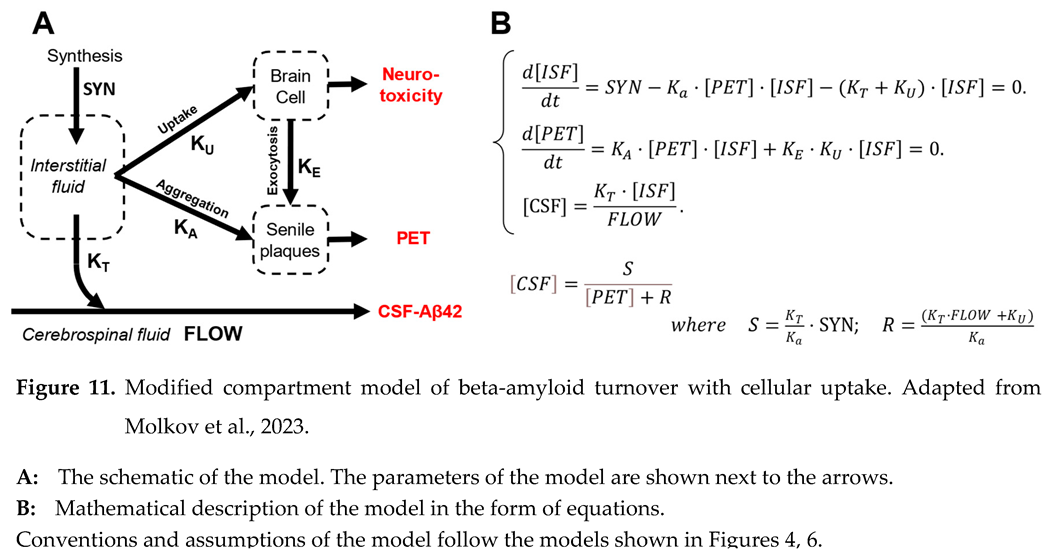
| The model, which describes the turnover of Aβ42 as the interaction between synthesis, transfer to the CSF, aggregation on existing plaques, and cellular uptake of the peptide, can be expressed as a system of differential equations. |
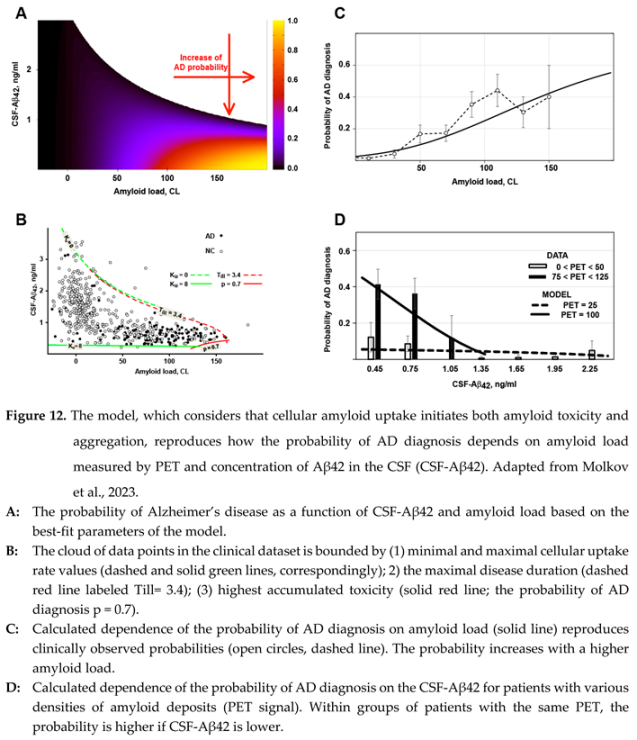
| In modeling, the probability of AD diagnosis was approximated as a sigmoid function of accumulated endocytosed beta-amyloid. At low accumulated uptakes, until some threshold is reached, the neurotoxicity can be compensated, so the probability of AD diagnosis is close to zero. At the highest accumulated uptakes, neuronal death reaches levels when the probability of AD diagnosis approaches 100% and cannot be increased anymore. |
| The clinical dataset, which contains values of amyloid deposits density and levels of Aβ42 in the CSF in populations of patients with AD and subjects with normal cognition (ADNI database), was used to infer the parameters of the model. |
| The model reproduces the distribution of late-onset AD in the population if the best-fit set of parameters is used. |
| A goodness-of-fit test confirms that the model reproduces clinical data accurately. |
| The distribution of two major amyloid biomarkers in the human population supports the amyloid degradation toxicity hypothesis of Alzheimer’s disease. |
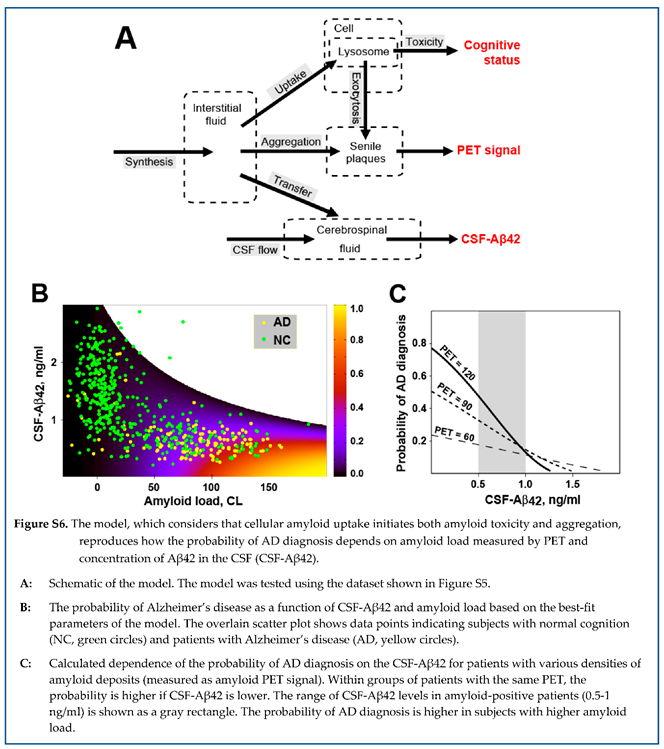
7.7. Multiscale summary of amyloid degradation toxicity hypothesis
| Molecular Level |
| Beta-amyloid is the neurotoxin that causes neurodegeneration in AD. The primary molecular interaction underlying beta-amyloid cytotoxicity is the formation of non-selective giant membrane channels. Amyloid membrane channels are formed by amyloid fragments with a positive charge in membranes carrying a negative surface charge. Spontaneous aggregation of beta-amyloid cannot be initiated in the intercellular space because of extremely low concentration, but existing amyloid aggregates grow extracellularly by absorbing soluble beta-amyloid. |
| Organelle Level |
| Lysosomes are the primary target in the cytotoxic action of beta-amyloid. Intralysosomal digestion of beta-amyloid produces channel-forming amyloid fragments. Lysosomal membrane carries a significant negative surface charge required for the formation of membrane amyloid channels. |
| Cellular Level |
| Endocytosis of beta-amyloid is the first step required for cytotoxicity. Membrane channels of any size cause lysosomal dysfunction, while extremely large channels allow for the leakage of lysosomal proteins to the cytoplasm. Leaked cathepsins induce either necrosis or apoptosis. Lysosomal permeabilization and lysosomal failure are the origins of other cellular hallmarks of AD – mitochondrial insufficiency, increased production of reactive oxygen species, the appearance of dysfunctional lysosomes, tau-protein accumulation, intracellular ion disturbances, etc. Beta-amyloid, which is stored intralysosomally, is the origin of aggregation seeds. Seeds of aggregated beta-amyloid are exocytosed and then grow extracellularly. Endocytosis of beta-amyloid is required for the appearance of senile plaques. |
| Tissue/Organ Level |
| Cellular death explains gross brain pathology in AD (especially in advanced stages of the dise ase): parenchymal atrophy, low metabolism, etc. The common origin of both aggregation and cytotoxicity – cellular uptake of beta-amyloid - explains why the presence of senile plaques is the hallmark of AD. Therefore, the density of amyloid deposits is a pathophysiology-relevant but indirect biomarker of AD. |
| Organism Level |
| Patients with late-onset AD have higher cellular uptake of Aβ42 than subjects with normal cogni tion. As both neurodegeneration and beta-amyloid aggregation are initiated by the same process - cellular amyloid uptake - higher amyloid uptake is the reason for faster neurodegeneration and high er density of amyloid deposits in patients with AD. The concentration of Aβ42 in the CSF is predominantly defined by the aggregation of this toxic soluble peptide on existing amyloid depo sits, while the density of deposits correlates with neurodegeneration. For that reason, CSF-Aβ42 can serve as a standalone AD biomarker, which is similar in value to the density of amyloid deposits. Therefore, CSF-Aβ42 is also the pathophysiology-relevant but indirect biomarker of AD. However, in patients with the same density of beta-amyloid deposits, a higher cellular uptake rate results in lower CSF-Aβ42. At a predefined density of deposits, lower CSF-Aβ42 indicates higher cellular amyloid uptake and faster neurodegeneration. Therefore, CSF-Aβ42 has a diagnostic value independent of the diagnostic value of the density of amyloid deposits. Using two major amyloid biomarkers together is advantageous until direct pathophysiology-relevant biomarkers are developed. |
| Population Level |
| The probability of AD diagnosis in a particular patient depends on the accumulated neurotoxicity of Aβ42, which in turn depends on the accumu lated amount of endocytosed Aβ42. However, the neurotoxicity of endocytosed Aβ42 also depends on the relative toxicity of this pep tide. Relative toxicity can be different between patients. The mathematical model that considers all these conditions accurately reproduces clinical data on the distribution of biomarkers and the probability of AD in the population. Currently used biomarkers (the density of amyloid deposits and the concentration of Aβ42 in the CSF) are related to AD pathobiology and correlate well with the disease. However, their non-direct nature limits the ability to use them to diagnose and, more importantly, predict disease progression.Methods that allow for the estimation of cellular amyloid uptake (such as the stable isotope labeling kinetics technique, SILK) would be more optimal for developing biomarkers and predictors of AD. Methods to estimate and modulate the relative neurotoxicity of beta-amyloid in patients are also the way to address the unmet need in the treatment of Alzheimer’s disease. |

7.5. The amyloid degradation toxicity hypothesis is the integrative theory of Alzheimer’s disease
| The hypothesis was described at all levels of bio logical organization – from molecular to population. |
| The hypothesis suggests the etiology and pathophysiology of the disease. |
| The amyloid degradation toxicity hypothesis is an integrative theory. |
There are two major paradoxes in any amyloid-centric theory of AD:
|
| AD is characterized by lysosomal failure, lysosomal permeabilization, activation of apoptosis, accumulation of intracellular amyloid aggregates, mitochondrial dysfunction, increased reactive oxygen species production, lower brain metabolism, accumulation of tau protein, etc. |
| The general human population is characterized by a particular distribution of amyloid biomarkers. |
| The amyloid degradation toxicity hypo thesis interprets multiple phenomena and paradoxes associated with AD. |
| The hypothesis explains why currently used amyloid-based biomarkers of AD (brain amyloid load and level of Aβ42 in the CSF) are associated with the pathophysiology of AD. |
| Both biomarkers provide indirect measures of parameters relevant to the etiology of AD. |
| The hypothesis interprets why there is a significant overlap between two amyloid biomarkers and what independent information can be found if these two biomarkers are considered together. |
7.8. Amyloid degradation toxicity hypothesis provides a framework for future research
| The hypothesis identifies processes that are relevant to AD pathophysiology and can be quantitatively characterized in a patient. |
| The rate of cellular amyloid uptake is likely to be the most informative parameter. There are tested approaches to measure this rate in clinical practice. |
| The hypothesis suggests novel biomarkers that are directly related to AD pathophysiology. |
| The hypothesis identifies the etiology of AD and involved biochemical and cellular pathways. |
| The hypothesis explains why currently used treatments do not prevent disease progression. |
| The hypothesis explains why dissolving amyloid deposits did not result in a reliable clinical effect. |
| The etiology and pathways involved in AD contain druggable targets (for example, proteases can be inhibited). |
| The hypothesis provides the framework for the development of novel medica tions to treat, slow the progression, or prevent the development of Alzheimer’s disease. |
8. Summary: etiology and pathogenesis of Alzheimer’s disease
- The toxicity of soluble beta-amyloid is the driving force in the development of Alzheimer’s disease.
- Amyloid membrane channel formation is the primary molecular mechanism of beta-amyloid toxicity and can be considered the etiology of AD.
- Lysosomal permeabilization due to membrane amyloid channel formation is the initial cellular damage.
- Lysosomes digest endocytosed Aβ, producing fragments, some of which can form membrane channels, while lysosomal membranes have characteristics required to incorporate channel-forming amyloid oligomers.
- Neuronal death is the result of activation of necrosis and/or apoptosis due to the leakage of lysosomal proteases through extremely large amyloid membrane channels, which are formed with low, but non-zero, probability.
- Dissipation of the pH gradient on the lysosomal membrane through amyloid membrane channels of any size results in lysosomal dysfunction and corresponding autophagy failure.
- The progression of AD includes functional (potentially reversible) cellular changes and cell death (irreversible, so the disease progresses over time).
- The degree of neuronal death depends on the accumulated Aβ toxicity and, therefore, on the intensity of Aβ endocytosis and time.
- Humans have very wide variability of sensitivity to the endocytosed Aβ because the pathway from Aβ endocytosis to cell death includes multiple steps.
- The sensitivity to endocytosed Aβ can be high, which can explain why some forms of AD are not associated with increased cellular amyloid uptake.
Acknowledgments
Abbreviations
References
- Alzheimer, A. über eigenartige Krankheitsfälle des späteren Alters. Zeitschrift für die gesamte Neurologie und Psychiatrie 1911, 4, 356. [Google Scholar] [CrossRef]
- Graeber, M.B.; Mehraein, P. Reanalysis of the first case of Alzheimer’s disease. Eur. Arch. Psychiatry Clin. Neurosci. 1999, 249 (Suppl. 3), 10–13. [Google Scholar] [CrossRef] [PubMed]
- Glenner, G.G. Amyloid beta protein and the basis for Alzheimer’s disease. Prog. Clin. Biol. Res. 1989, 317, 857–868. [Google Scholar] [PubMed]
- Glenner, G.G.; Wong, C.W. Alzheimer’s disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem. Biophys. Res. Commun. 1984, 120, 885–890. [Google Scholar] [CrossRef] [PubMed]
- Demuro, A.; Mina, E.; Kayed, R.; Milton, S.C.; Parker, I.; Glabe, C.G. Calcium Dysregulation and Membrane Disruption as a Ubiquitous Neurotoxic Mechanism of Soluble Amyloid Oligomers. J. Biol. Chem. 2005, 280, 17294–17300. [Google Scholar] [CrossRef] [PubMed]
- Cline, E.N.; Bicca, M.A.; Viola, K.L.; Klein, W.L. The Amyloid-β Oligomer Hypothesis: Beginning of the Third Decade. J. Alzheimer’s Dis. JAD 2018, 64, S567–S610. [Google Scholar] [CrossRef] [PubMed]
- Lambert, M.P.; Barlow, A.K.; Chromy, B.A.; Edwards, C.; Freed, R.; Liosatos, M.; Morgan, T.E.; Rozovsky, I.; Trommer, B.; Viola, K.L.; et al. Diffusible, nonfibrillar ligands derived from Abeta1-42 are potent central nervous system neurotoxins. Proc. Natl. Acad. Sci. USA 1998, 95, 6448–6453. [Google Scholar] [CrossRef] [PubMed]
- Knafo, S.; Alonso-Nanclares, L.; Gonzalez-Soriano, J.; Merino-Serrais, P.; Fernaud-Espinosa, I.; Ferrer, I.; DeFelipe, J. Widespread changes in dendritic spines in a model of Alzheimer’s disease. Cereb. Cortex 2009, 19, 586–592. [Google Scholar] [CrossRef] [PubMed]
- Tsai, J.; Grutzendler, J.; Duff, K.; Gan, W.B. Fibrillar amyloid deposition leads to local synaptic abnormalities and breakage of neuronal branches. Nat. Neurosci. 2004, 7, 1181–1183. [Google Scholar] [CrossRef]
- Bittner, T.; Fuhrmann, M.; Burgold, S.; Ochs, S.M.; Hoffmann, N.; Mitteregger, G.; Kretzschmar, H.; LaFerla, F.M.; Herms, J. Multiple Events Lead to Dendritic Spine Loss in Triple Transgenic Alzheimer’s Disease Mice. PLoS ONE 2010, 5, e15477. [Google Scholar] [CrossRef]
- Mattsson, N.; Insel, P.S.; Landau, S.; Jagust, W.; Donohue, M.; Shaw, L.M.; Trojanowski, J.Q.; Zetterberg, H.; Blennow, K.; Weiner, M. Diagnostic accuracy of CSF Ab42 and florbetapir PET for Alzheimer’s disease. Ann. Clin. Transl. Neurol. 2014, 1, 534–543. [Google Scholar] [CrossRef]
- Ong, K.T.; Villemagne, V.L.; Bahar-Fuchs, A.; Lamb, F.; Langdon, N.; Catafau, A.M.; Stephens, A.W.; Seibyl, J.; Dinkelborg, L.M.; Reininger, C.B.; et al. Aβ imaging with 18F-florbetaben in prodromal Alzheimer’s disease: a prospective outcome study. J. Neurol. Neurosurg. Psychiatry 2015, 86, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Selkoe, D.J.; Hardy, J. The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Mol. Med. 2016, 8, 595–608. [Google Scholar] [CrossRef] [PubMed]
- Ricciarelli, R.; Fedele, E. The Amyloid Cascade Hypothesis in Alzheimer’s Disease: It’s Time to Change Our Mind. Curr. Neuropharmacol. 2017, 15, 926–935. [Google Scholar] [CrossRef] [PubMed]
- Elbert, D.L.; Patterson, B.W.; Bateman, R.J. Analysis of a compartmental model of amyloid beta production, irreversible loss and exchange in humans. Math. Biosci. 2015, 261, 48–61. [Google Scholar] [CrossRef] [PubMed]
- Patterson, B.W.; Elbert, D.L.; Mawuenyega, K.G.; Kasten, T.; Ovod, V.; Ma, S.; Xiong, C.; Chott, R.; Yarasheski, K.; Sigurdson, W.; et al. Age and amyloid effects on human central nervous system amyloid-beta kinetics. Ann. Neurol. 2015, 78, 439–453. [Google Scholar] [CrossRef] [PubMed]
- Potter, R.; Patterson, B.W.; Elbert, D.L.; Ovod, V.; Kasten, T.; Sigurdson, W.; Mawuenyega, K.; Blazey, T.; Goate, A.; Chott, R.; et al. Increased in vivo amyloid-β42 production, exchange, and loss in presenilin mutation carriers. Sci. Transl. Med. 2013, 5, 189ra177. [Google Scholar] [CrossRef] [PubMed]
- Zaretsky, D.V.; Zaretskaia, M.V.; Molkov, Y.I. Patients with Alzheimer’s disease have an increased removal rate of soluble beta-amyloid-42. PLoS ONE 2022, 17, e0276933. [Google Scholar] [CrossRef] [PubMed]
- Mawuenyega, K.G.; Sigurdson, W.; Ovod, V.; Munsell, L.; Kasten, T.; Morris, J.C.; Yarasheski, K.E.; Bateman, R.J. Decreased clearance of CNS beta-amyloid in Alzheimer’s disease. Science 2010, 330, 1774. [Google Scholar] [CrossRef]
- Silverberg, G.D.; Heit, G.; Huhn, S.; Jaffe, R.A.; Chang, S.D.; Bronte-Stewart, H.; Rubenstein, E.; Possin, K.; Saul, T.A. The cerebrospinal fluid production rate is reduced in dementia of the Alzheimer’s type. Neurology 2001, 57, 1763–1766. [Google Scholar] [CrossRef]
- Fishman, R.A. The cerebrospinal fluid production rate is reduced in dementia of the Alzheimer’s type. Neurology 2002, 58, 1866. [Google Scholar] [CrossRef] [PubMed]
- Abraham, C.R. Potential roles of protease inhibitors in Alzheimer’s disease. Neurobiol. Aging 1989, 10, 463–465. [Google Scholar] [CrossRef] [PubMed]
- Sturchio, A.; Dwivedi, A.K.; Young, C.B.; Malm, T.; Marsili, L.; Sharma, J.S.; Mahajan, A.; Hill, E.J.; Andaloussi, S.E.L.; Poston, K.L. High cerebrospinal amyloid-β 42 is associated with normal cognition in individuals with brain amyloidosis. EClinicalMedicine 2021, 100988. [Google Scholar] [CrossRef] [PubMed]
- Mattsson, N.; Insel, P.S.; Donohue, M.; Landau, S.; Jagust, W.J.; Shaw, L.M.; Trojanowski, J.Q.; Zetterberg, H.; Blennow, K.; Weiner, M.W. Independent information from cerebrospinal fluid amyloid-β and florbetapir imaging in Alzheimer’s disease. Brain 2015, 138, 772–783. [Google Scholar] [CrossRef] [PubMed]
- Motter, R.; Vigo-Pelfrey, C.; Kholodenko, D.; Barbour, R.; Johnson-Wood, K.; Galasko, D.; Chang, L.; Miller, B.; Clark, C.; Green, R.; et al. Reduction of beta-amyloid peptide42 in the cerebrospinal fluid of patients with Alzheimer’s disease. Ann. Neurol. 1995, 38, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Andreasen, N.; Hesse, C.; Davidsson, P.; Minthon, L.; Wallin, A.; Winblad, B.; Vanderstichele, H.; Vanmechelen, E.; Blennow, K. Cerebrospinal Fluid β-Amyloid(1-42) in Alzheimer Disease: Differences Between Early- and Late-Onset Alzheimer Disease and Stability During the Course of Disease. Arch. Neurol. 1999, 56, 673–680. [Google Scholar] [CrossRef]
- Espay, A.J.; Herrup, K.; Kepp, K.P.; Daly, T. The Proteinopenia Hypothesis: Loss of Aβ42 and the Onset of Alzheimer’s Disease. Ageing Res. Rev. 2023, 102112. [Google Scholar] [CrossRef] [PubMed]
- Espay, A.J.; Sturchio, A.; Schneider, L.S.; Ezzat, K. Soluble Amyloid-β Consumption in Alzheimer’s Disease. J. Alzheimer’s Dis. JAD 2021. [Google Scholar] [CrossRef] [PubMed]
- Espay, A.J.; Lafontant, D.-E.; Poston, K.L.; Caspell-Garcia, C.; Marsili, L.; Cho, H.R.; McDaniel, C.; Kim, N.; Coffey, C.S.; Mahajan, A.; et al. Low soluble amyloid-β 42 is associated with smaller brain volume in Parkinson’s disease. Park. Relat. Disord. 2021, 92, 15–21. [Google Scholar] [CrossRef]
- Terakawa, M.S.; Yagi, H.; Adachi, M.; Lee, Y.H.; Goto, Y. Small liposomes accelerate the fibrillation of amyloid β (1-40). J. Biol. Chem. 2015, 290, 815–826. [Google Scholar] [CrossRef]
- Cohen, S.I.A.; Linse, S.; Luheshi, L.M.; Hellstrand, E.; White, D.A.; Rajah, L.; Otzen, D.E.; Vendruscolo, M.; Dobson, C.M.; Knowles, T.P.J. Proliferation of amyloid-β42 aggregates occurs through a secondary nucleation mechanism. Proc. Natl. Acad. Sci. USA 2013, 110, 9758–9763. [Google Scholar] [CrossRef] [PubMed]
- Wesén, E.; Jeffries, G.D.M.; Matson Dzebo, M.; Esbjörner, E.K. Endocytic uptake of monomeric amyloid-β peptides is clathrin- and dynamin-independent and results in selective accumulation of Aβ(1-42) compared to Aβ(1-40). Sci. Rep. 2017, 7, 2021. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Crick, S.L.; Bu, G.; Frieden, C.; Pappu, R.V.; Lee, J.-M. Amyloid seeds formed by cellular uptake, concentration, and aggregation of the amyloid-beta peptide. Proc. Natl. Acad. Sci. USA 2009, 106, 20324–20329. [Google Scholar] [CrossRef] [PubMed]
- Knauer, M.F.; Soreghan, B.; Burdick, D.; Kosmoski, J.; Glabe, C.G. Intracellular accumulation and resistance to degradation of the Alzheimer amyloid A4/beta protein. Proc. Natl. Acad. Sci. USA 1992, 89, 7437–7441. [Google Scholar] [CrossRef] [PubMed]
- Burdick, D.; Kosmoski, J.; Knauer, M.F.; Glabe, C.G. Preferential adsorption, internalization and resistance to degradation of the major isoform of the Alzheimer’s amyloid peptide, A beta 1-42, in differentiated PC12 cells. Brain Res. 1997, 746, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Cataldo, A.M.; Thayer, C.Y.; Bird, E.D.; Wheelock, T.R.; Nixon, R.A. Lysosomal proteinase antigens are prominently localized within senile plaques of Alzheimer’s disease: evidence for a neuronal origin. Brain Res. 1990, 513, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Fu, H.; Li, J.; Du, P.; Jin, W.; Gao, G.; Cui, D. Senile plaques in Alzheimer’s disease arise from Aβ- and Cathepsin D-enriched mixtures leaking out during intravascular haemolysis and microaneurysm rupture. FEBS Lett. 2023, 597, 1007–1040. [Google Scholar] [CrossRef] [PubMed]
- Zaretsky, D.V.; Zaretskaia, M.V.; Molkov, Y.I. Membrane channel hypothesis of lysosomal permeabilization by beta-amyloid. Neurosci. Lett. 2021, 136338. [Google Scholar] [CrossRef] [PubMed]
- Yang, A.J.; Chandswangbhuvana, D.; Margol, L.; Glabe, C.G. Loss of endosomal/lysosomal membrane impermeability is an early event in amyloid Abeta1-42 pathogenesis. J. Neurosci. Res. 1998, 52, 691–698. [Google Scholar] [CrossRef]
- Ji, Z.S.; Miranda, R.D.; Newhouse, Y.M.; Weisgraber, K.H.; Huang, Y.; Mahley, R.W. Apolipoprotein E4 potentiates amyloid beta peptide-induced lysosomal leakage and apoptosis in neuronal cells. J. Biol. Chem. 2002, 277, 21821–21828. [Google Scholar] [CrossRef]
- Molkov, Y.I.; Zaretskaia, M.V.; Zaretsky, D.V. Towards The Integrative Theory of Alzheimer’s Disease: Linking Molecular Mechanisms of Neurotoxicity, Beta-Amyloid Biomarkers, And The Diagnosis. Curr. Alzheimer Res. 2023. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.S.; Mamun, A.A.; Labu, Z.K.; Hidalgo-Lanussa, O.; Barreto, G.E.; Ashraf, G.M. Autophagic dysfunction in Alzheimer’s disease: Cellular and molecular mechanistic approaches to halt Alzheimer’s pathogenesis. J. Cell. Physiol. 2019, 234, 8094–8112. [Google Scholar] [CrossRef] [PubMed]
- Peric, A.; Annaert, W. Early etiology of Alzheimer’s disease: tipping the balance toward autophagy or endosomal dysfunction? Acta Neuropathol. 2015, 129, 363–381. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, D.M.; Lee, J.H.; Kumar, A.; Lee, S.; Orenstein, S.J.; Nixon, R.A. Autophagy failure in Alzheimer’s disease and the role of defective lysosomal acidification. Eur. J. Neurosci. 2013, 37, 1949–1961. [Google Scholar] [CrossRef] [PubMed]
- Nixon, R.A.; Yang, D.S. Autophagy failure in Alzheimer’s disease--locating the primary defect. Neurobiol. Dis. 2011, 43, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Nixon, R.A.; Cataldo, A.M. A.; Cataldo, A.M. Lysosomal system pathways: genes to neurodegeneration in Alzheimer’s disease. J. Alzheimer’s Dis. JAD 2006, 9, 277–289. [Google Scholar] [CrossRef]
- Nixon, R.A.; Wegiel, J.; Kumar, A.; Yu, W.H.; Peterhoff, C.; Cataldo, A.; Cuervo, A.M. Extensive involvement of autophagy in Alzheimer disease: an immuno-electron microscopy study. J. Neuropathol. Exp. Neurol. 2005, 64, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Kavčič, N.; Pegan, K.; Turk, B. Lysosomes in programmed cell death pathways: from initiators to amplifiers. Biol. Chem. 2017, 398, 289–301. [Google Scholar] [CrossRef] [PubMed]
- McMahon, S.M.; Jackson, M.B. An Inconvenient Truth: Calcium Sensors Are Calcium Buffers. Trends Neurosci. 2018, 41, 880–884. [Google Scholar] [CrossRef]
- Abramov, A.Y.; Canevari, L.; Duchen, M.R. Calcium signals induced by amyloid β peptide and their consequences in neurons and astrocytes in culture. Biochim. Biophys. Acta (BBA) Mol. Cell Res. 2004, 1742, 81–87. [Google Scholar] [CrossRef]
- Abramov, A.Y.; Canevari, L.; Duchen, M.R. Changes in intracellular calcium and glutathione in astrocytes as the primary mechanism of amyloid neurotoxicity. J. Neurosci. Off. J. Soc. Neurosci. 2003, 23, 5088–5095. [Google Scholar] [CrossRef] [PubMed]
- Zaretsky, D.V.; Zaretskaia, M.V. Intracellular ion changes induced by the exposure to beta-amyloid can be explained by the formation of channels in the lysosomal membranes. Biochim. Biophys. Acta. Mol. Cell Res. 2021, 119145. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.-c.A.; Kagan, B.L. Electrophysiologic properties of channels induced by Abeta25-35 in planar lipid bilayers. Peptides 2002, 23, 1215–1228. [Google Scholar] [CrossRef] [PubMed]
- Mirzabekov, T.; Lin, M.C.; Yuan, W.L.; Marshall, P.J.; Carman, M.; Tomaselli, K.; Lieberburg, I.; Kagan, B.L. Channel formation in planar lipid bilayers by a neurotoxic fragment of the beta-amyloid peptide. Biochem. Biophys. Res. Commun. 1994, 202, 1142–1148. [Google Scholar] [CrossRef]
- Quist, A.; Doudevski, I.; Lin, H.; Azimova, R.; Ng, D.; Frangione, B.; Kagan, B.; Ghiso, J.; Lal, R. Amyloid ion channels: a common structural link for protein-misfolding disease. Proc. Natl. Acad. Sci. USA 2005, 102, 10427–10432. [Google Scholar] [CrossRef]
- Kourie, J.I.; Culverson, A.L.; Farrelly, P.V.; Henry, C.L.; Laohachai, K.N. Heterogeneous amyloid-formed ion channels as a common cytotoxic mechanism: implications for therapeutic strategies against amyloidosis. Cell Biochem. Biophys. 2002, 36, 191–207. [Google Scholar] [CrossRef]
- Kagan, B.L.; Hirakura, Y.; Azimov, R.; Azimova, R.; Lin, M.C. The channel hypothesis of Alzheimer’s disease: current status. Peptides 2002, 23, 1311–1315. [Google Scholar] [CrossRef]
- Bhatia, R.; Lin, H.; Lal, R. Fresh and globular amyloid beta protein (1-42) induces rapid cellular degeneration: evidence for AbetaP channel-mediated cellular toxicity. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2000, 14, 1233–1243. [Google Scholar]
- Lin, H.; Zhu, Y.J.; Lal, R. Amyloid beta protein (1-40) forms calcium-permeable, Zn2+-sensitive channel in reconstituted lipid vesicles. Biochemistry 1999, 38, 11189–11196. [Google Scholar] [CrossRef]
- Pollard, H.B.; Arispe, N.; Rojas, E. Ion channel hypothesis for Alzheimer amyloid peptide neurotoxicity. Cell. Mol. Neurobiol. 1995, 15, 513–526. [Google Scholar] [CrossRef]
- Pollard, H.B.; Rojas, E.; Arispe, N. A new hypothesis for the mechanism of amyloid toxicity, based on the calcium channel activity of amyloid beta protein (A beta P) in phospholipid bilayer membranes. Ann. N. Y. Acad. Sci. 1993, 695, 165–168. [Google Scholar] [CrossRef] [PubMed]
- Arispe, N.; Rojas, E.; Pollard, H.B. Alzheimer disease amyloid beta protein forms calcium channels in bilayer membranes: blockade by tromethamine and aluminum. Proc. Natl. Acad. Sci. USA 1993, 90, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Arispe, N.; Pollard, H.B.; Rojas, E. Giant multilevel cation channels formed by Alzheimer disease amyloid beta-protein [A beta P-(1-40)] in bilayer membranes. Proc. Natl. Acad. Sci. USA 1993, 90, 10573–10577. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.; Good, T. A rapid method to measure beta-amyloid induced neurotoxicity in vitro. J. Neurosci. Methods 2007, 161, 1–10. [Google Scholar] [CrossRef]
- Arispe, N.; Pollard, H.B.; Rojas, E. beta-Amyloid Ca(2+)-channel hypothesis for neuronal death in Alzheimer disease. Mol. Cell Biochem. 1994, 140, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Valincius, G.; Heinrich, F.; Budvytyte, R.; Vanderah, D.J.; McGillivray, D.J.; Sokolov, Y.; Hall, J.E.; Lösche, M. Soluble amyloid beta-oligomers affect dielectric membrane properties by bilayer insertion and domain formation: implications for cell toxicity. Biophys. J. 2008, 95, 4845–4861. [Google Scholar] [CrossRef]
- Sokolov, Y.; Kozak, J.A.; Kayed, R.; Chanturiya, A.; Glabe, C.; Hall, J.E. Soluble amyloid oligomers increase bilayer conductance by altering dielectric structure. J. Gen. Physiol. 2006, 128, 637–647. [Google Scholar] [CrossRef] [PubMed]
- Zaretsky, D.V.; Zaretskaia, M.V. Flow cytometry method to quantify the formation of beta-amyloid membrane ion channels. Biochim. Biophys. Acta. Biomembr. 2020, 183506. [Google Scholar] [CrossRef]
- Zaretsky, D.V.; Zaretskaia, M. Degradation Products of Amyloid Protein: Are They The Culprits? Curr. Alzheimer Res. 2020, 17, 869–880. [Google Scholar] [CrossRef]
- Alarcon, J.M.; Brito, J.A.; Hermosilla, T.; Atwater, I.; Mears, D.; Rojas, E. Ion channel formation by Alzheimer’s disease amyloid beta-peptide (Abeta40) in unilamellar liposomes is determined by anionic phospholipids. Peptides 2006, 27, 95–104. [Google Scholar] [CrossRef]
- Das, A.; Nag, S.; Mason, A.B.; Barroso, M.M. Endosome-mitochondria interactions are modulated by iron release from transferrin. J. Cell Biol. 2016, 214, 831–845. [Google Scholar] [CrossRef] [PubMed]
- Hamdi, A.; Roshan, T.M.; Kahawita, T.M.; Mason, A.B.; Sheftel, A.D.; Ponka, P. Erythroid cell mitochondria receive endosomal iron by a “kiss-and-run” mechanism. Biochim. Biophys. Acta (BBA) Mol. Cell Res. 2016, 1863, 2859–2867. [Google Scholar] [CrossRef] [PubMed]
- Repnik, U.; Stoka, V.; Turk, V.; Turk, B. Lysosomes and lysosomal cathepsins in cell death. Biochim. Biophys. Acta (BBA) Proteins Proteom. 2012, 1824, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Turk, B.; Stoka, V.; Rozman-Pungercar, J.; Cirman, T.; Droga-Mazovec, G.; Oresić, K.; Turk, V. Apoptotic pathways: involvement of lysosomal proteases. Biol. Chem. 2002, 383, 1035–1044. [Google Scholar] [CrossRef]
- Goel, P.; Chakrabarti, S.; Goel, K.; Bhutani, K.; Chopra, T.; Bali, S. Neuronal cell death mechanisms in Alzheimer’s disease: An insight. Front. Mol. Neurosci. 2022, 15, 937133. [Google Scholar] [CrossRef]

| Biomarker | Toxicity of amyloid form | Concentration in patients with AD |
| Aggregated Aβ | inert | increased |
| Soluble Aβ42 | toxic | decreased |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




