Submitted:
06 June 2023
Posted:
07 June 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Collection of and Transportation of Samples
2.2. pH Measurement
2.3. Titratable Acidity of Fermented Milk Sample
- Vt= Volume of titrant (ml NaOH)
- N = Normality of titrant
- 90 = Equivalent weight for lactic acid
- Vs = Volume of sample used (ml yoghurt/milk)
2.4. Sample Preparation for Analysis
2.5. Microbial Analysis
Analytical Profile Index (API) Biochemical Test
| Medium for growth | Microorganisms | Time (Hours) | Growth condition and incubation Temperature | Growth condition and incubation Temperature |
|---|---|---|---|---|
| Plate Count Agar (Oxoid M325) | Total aerobic mesophilic aerobic bacteria | 48±2h | aerobic 30±1oC | aerobic 30±1oC |
| MRS agar, LAB098) | Mesophilic Lactobacilli | 48±2h | aerobic 35±1oC | aerobic 35±1oC |
| MRS agar (LAB 098 + Vancomycin) | Leuconostoc | 48±2h | anaerobic 30±1oC | anaerobic 30±1oC |
| MRS agar (pH 5.5) LAB098 | Thermophilic Lactobacilli | 48±2h | anaerobic 42±1oC | anaerobic 42±1oC |
| MRS agar (pH 6.) LAB098 | Thermophilic Lactococci | 48±2h | anaerobic 42±1oC | anaerobic 42±1oC |
| M17 agar (LAB 092) | Mesophilic Streptococci | 48±2h | anaerobic 30±1oC | aerobic 35±1oC |
| Violet Red Bile Lactose agar with MUG supplement BRO 71 E), | Non-Sorbitol E. coli | 24 ±2h | aerobic 37±1oC | aerobic 37±1oC |
| Violet Red Bile Agar (VRBA) | Total coliform | 24 ±2h | aerobic 30±1oC | aerobic 30±1oC |
| XLD | Salmonella and Shigella spp. | 24 ±2h | aerobic 37±1oC | aerobic 37±1oC |
| Baird–Parker’s medium (Oxoid CM 0275 + SR054C) | Staphylococcus aureus | 24 ±2h | aerobic 37±1oC | aerobic 37±1oC |
| Listeria Enrichment Broth A and B | Listeria. Monocytogenes | 24 ±2h | aerobic 30±1oC | aerobic 30±1oC |
| B. cereus agar | B. cereus | aerobic 30±1oC | aerobic 30±1oC | |
| 1.5% Malt Extract and Agar No. 2 | Yeast and mould | aerobic 25±1oC | aerobic 25±1oC | |
| PDA + chloramphenicol | Mould | aerobic 30±1oC | aerobic 30±1oC |
3. Results
3.1. The Physiochemical Properties
3.2. The Microbial Counts
| Medium | UG 1 | UG 2 | KE | Growth condition |
|---|---|---|---|---|
| Plate Count Agar (PCA) (for Total aerobic mesophilic bacteria) |
9.7 x 109 | 3.3 x 109 | 2.53 x 109 | aerobic 30±1oC |
| (MRS agar) (for Mesophilic Lactobacilli) | 1.74 x 108 | 2.12x 106 | 5.9 x 107 | aerobic 35±1oC |
| MRS agar + Vacomycine (for Leuconostoc) |
1.55 x 106 | 1.61x 105 | 6.2 x 107 | anaerobic 30±1oC |
| M17 agar (for Mesophilic lactococci) | 1.17 x 108 | 3.7 x 108 | 6.2 x 107 | anaerobic 30±1oC |
| MRS agar (Thermophilic Lactobacilli) | 2.87 x 107 | 1.54x 109 | 8.0 x 106 | anaerobic 42±1oC |
| MRS agar (for Thermophilic Lactococci | 2.63 x 106 | 1.25x 109 | 1.48 x 106 | anaerobic 42±1oC |
| M17 agar (for Streptococci) | 1.74 x 108 | 6.2 x 107 | 3.7 x 106 | aerobic 37±1oC |
| Violet Red Bile Lactose agar (for Non-Sorbitol E. coli) | 2.92 x 103 | 1.61x 103 | 4.1 x 103 | aerobic 37±1oC |
| Violet Red Bile Agar (VRBA), (coliforms counts) |
2.12 x 105 | 4.2 x 103 | 1.56 x 103 | aerobic 30±1oC |
| XLD (for Salmonella spp.) | 1.7 x 102 | 1.4 x 103 | 1.5 x 102 | aerobic 37±1oC |
| XLD (for Shigella spp.) | ND | ND | ND | aerobic 37±1oC |
| Baird–Parker’s medium (for S. aureus) | 1.18 x 103 | 1.4 x 105 | 1.11 x 103 | aerobic 37±1oC |
| Listeria Enrichment Broth A and B (for L. monocytogenes) |
1.7 x 102 | ND | 1.2 x 103 | aerobic 37±1oC |
| Bacillus cereus | 2.1 x 102 | 2.12x 103 | 1.35 x 103 | aerobic 30±1oC |
| Yeasts and moulds (on 1.5% Malt Extract and Agar No. 2)) | 2.07 x 107 | 5.4 x 109 | 3.9 x 1011 | aerobic 25±1oC |
| PDA + chloramphenicol (for Mould) | 4.0 x 108 | 6.7 x 107 | 2.16x 1010 | aerobic 30±1oC |
3.3. The Microbial Analysis
Identification of the Isolates with API Biochemical Analysis
| Isolate | Macro-colony morphology (margin, colour, elevation, cell appearance | UG1 | UG2 | KE |
|---|---|---|---|---|
| 1 | Cream, smooth, oval shape entire and ellipsoidal cell | √ | √ | √ |
| 2 | Undulating, white top with green base, slightly convex, spheroidal to short ellipsoidal. (Blue colony on Kluveymyces Differential Medium) | √ | √ | √ |
| 3 | Yellow-green, powdery and pale yellowish on reverse Aspergillus flavus | √ | ND | √ |
| 4 | Dirty white with yellow spores at the centre, base orange, slightly radially furrowed (Microsporum spp.) | √ | √ | √ |
| 5 | Cream-yellow, powdery, and pale yellowish on reverse, capsulate margin, slightly raised centre, filamentous cells, > 85 mm colony diameter. | √ | √ | √ |
| 6 | White to cream, yellowish, wrinkled, nearly flat elevation, oval cells & Ellipsoidal | √ | √ | ND |
| 7 | White to cream coloured, flat with aerial mycelium (Aspergillus spp) | √ | √ | √ |
| 8 | Green with a red base | √ | √ | √ |
| 9 | White at the base and black spores at the top | √ | √ | √ |
| 10 | White pin head, clear zones around the colony | √ | √ | √ |
| 11 | Black, yellow to pale cream in the centre (Aspergillus) | ND | ND | ND |
| 12 | White measuring 1-4 mm, opaque and flat. Ropy to the touch | √ | √ | √ |
| 13 | Straw cream at the centre, base orange, slightly radially furrowed | √ | √ | ND |
| 14 | Well-formed white colonies (grew well on M17 too) (Aspergillus spp)) | √ | √ | √ |
| 15 | Green and pale yellow on reverse (Penicillium) | ND | ND | ND |
| 16 | White base with black conidiophores | √ | √ | |
| 17 | Greenish black, white mycelia at the margin, white in the centre (Rhizopus sp.) | ND | ND | ND |
| 18 | Greenish with surrounded by creamy-white ring at the margin (Penicillium) | ND | √ | ND |
| 19 | White to cream, smooth, glaucous dark green on obverse and pale yellow on reverse, | √ | √ | √ |
| 20 | Cotton white to cream on the obverse and yellow to orange on the reverse with dark brown exudate | √ | √ | √ |
| 21 | White colony, opaque and flat | √ | √ | √ |
| 22 | Bright red colonies | ND | ND | ND |
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmad T., Butt M.Z., Aadil R.M., Abdallah Inam-ur-Raheem A., Bekhit M., El Din A. Guimaraes J.T., Balthaza, C.F., Rocha, R.S., Esmerino, E.A., Freitas, M.Q, Silva, M.C., Sameen, A., Cruz, A.G. Impact of nonthermal processing on different milk enzymes. Int J of Dairy Tech 2019, 481–495. [Google Scholar]
- Leone C, Thippareddi H, Ndiaye C., Niang I., Diallo Y, Singh M. Safety and Quality of Milk and Milk Products in Senegal-A Review. Foods. 2022, 2;11(21):3479. PMID: 36360092; PMCID: PMC9656659. [CrossRef]
- Moatsou G., Moschopoulou E.. Microbiology of raw milk. In: Özer BH, Akdemir-Evrendilek G, editors. Dairy microbiology and biochemistry: recent developments: Taylor & Francis Group, LLC; 2015:1–38.
- Aliyo A, Teklemariam Z. Assessment of Milk Contamination, Associated Risk Factors, and Drug Sensitivity Patterns among Isolated Bacteria from Raw Milk of Borena Zone. Ethiopia. J Trop Med. 2022. PMID: 35769792; PMCID: PMC9236756. [CrossRef]
- Yambayamba K., E. & Zulu M. P. Influence of the milking environment on the microbial quality of raw milk produced by smallholder farmers in Magoye. Uni of Zambia J of Sci and Tech. 2011, 15, 37–43. [Google Scholar]
- Leone C, Thippareddi H, Ndiaye C., Niang I, Diallo Y, Singh M. Safety and Quality of Milk and Milk Products in Senegal-A Review. Foods. 2022. 2:11(21):3479. [CrossRef]
- Landis EA., Oliverio A.M., McKenney E.A., Nichols L.M., Kfoury N., Biango-Daniels M., Shell L.K., Madden A.A., Shapiro L., Sakunala S., Drake K., Robbat A., Booker M., Dunn, RR., Fiere N., Wolfe BE. The diversity and function of sourdough starter microbiomes. Ecology Micro and Inf Dis. 2021. [CrossRef]
- Deddefo A., mamo G., Asfaw M., and Amenu, K. Factors affecting the microbiological quality and contamination of farm bulk milk by Staphylococcus aureus in dairy farms in Asella, Ethiopia. BMC Micro. 2023. 2365. [CrossRef]
- Washaya, S. , Jakata C., Tagwira M., Mupofu T. Bacterial Milk Quality along the Value Chain in Smallholder Dairy Production. Scientific World Journal. 2022, 21, 7967569. [Google Scholar] [CrossRef]
- El-Ansary, M.A. Assessment of Microbiological Quality of Yoghurt Sold in El-Behera Governorate. Alexandria J of Vet Sci. 2014, 43, 52–57. [Google Scholar] [CrossRef]
- Mukisa, I.M., Kyoshabire. Microbiological, physicochemical, and sensorial quality of small-scale produced stirred yoghurt on the market in Kampala city, Uganda. Nutr. Food Sci 2010, 40, 409–418. [Google Scholar] [CrossRef]
- AOAC Official Methods of Analysis Chemist (18th ed.) 2006.
- Duncan S. E. , Yaun , and Sumner S. S., and Bruhn J., Tech. Comm. , “Chapter 09 Microbiological Methods for Dairy Products”, Standard Methods for the Examination of Dairy Products. American Pub Health Ass. Washington, D.C., USA. 2012. [CrossRef]
- Abebe B., Yilma Z., and Nurfeta. Hygienic and microbial quality of raw whole cow’s milk produced in Ezha district of Gurage Zone, Southern Ethiopia. Wudpecker J of Agric Res. 2012, 1, 178–187. [Google Scholar]
- IOS (International Organization for Standardization). Milk and Milk Products-General Guidance for the Preparation of Test Samples, Initial Suspensions and Decimal Dilutions for Microbiological Examination: Lait Et Produits Laitiers-Lignes Directrices Générales Pour la Préparaton Des Échantillons Pour Essai, de la Suspension Mère Et Des Dilutions Décimales en Vue de L'examen Microbiologique. 2001. ISO.
- De Man, J.C. , Rogosa, M., Sharpe E.M. A medium for the cultivation of Lactobacilli. J App Bact. 1960, 23, 130–135. [Google Scholar] [CrossRef]
- Terzaghi, B.E. , Sandine W.E. Improved medium for lactic Streptococci and their bacteriophages. Appl Microb. 1975, 29, 807–813. [Google Scholar] [CrossRef]
- Gebeyehu, A. , Taye M. & Abebe R. Isolation, molecular detection, and antimicrobial susceptibility profile of Salmonella from raw cow milk collected from dairy farms and households in southern Ethiopia. BMC Microbiol 2022, 22, 84. [Google Scholar] [CrossRef]
- Feng P., Weagant F.S., Grant M.A., Burhardt W. Enumeration of Escherichia coli and the Coliform Bacteria. Bacteriological Analytical Manual (BAM). 2020. Chapter 4.
- Moushumi, B. and Prabir K.S. Microbiological quality of some retail spices in India. Food Res. Int. 2003, 469–474. [Google Scholar]
- IOS (International Organization for Standardization). Milk and Milk Products-General Guidance for the Preparation of Test Samples, Initial Suspensions and Decimal Dilutions for Microbiological Examination: Lait Et Produits Laitiers-Lignes Directrices Générales Pour la Préparaton Des Échantillons Pour Essai, de la Suspension Mère Et Des Dilutions Décimales en Vue de L'examen Microbiologique. 2001. ISO.
- Elbassiony, T.A. , Abd EL Mgeed A.S.M.; Ewida R.M. Prevalence of Some Spore Forming Food Poisoning Bacteria in Milk and Some Milk Products. J. Adv. Vet. Res. 2021, 11, 243–246. [Google Scholar]
- International Dairy Federation (IDF). International Dairy Federation. Milk and Milk-based Products — Enumeration of Staphylococcus aureus (IDF Standard 1990. 145: IDF, Brussels.
- Nero L.A, de Mattos M.R., Barros Mde A., Ortolani M.B., Beloti V., Franco B.D. Listeria monocytogenes and Salmonella spp. in raw milk produced in Brazil: Occurrence and interference of indigenous microbiota in their isolation and development. Zoonoses Public Health. 2008, 55, 299–305. [CrossRef]
- Gadaga, T.H. The occurrence and diversity of yeasts in Zimbabwean traditional fermented milk and their potential for use as starter cultures. PhD Thesis, Agricultural University of Norway. A°S Norway, ISBN 9-82-575-0444-0. 2000.
- Kunda, B. , Pandey G.S., and Muma J.B. Compositional and sanitary quality of raw milk produced by smallholder dairy farmers in Lusaka Province of Zambia. Livestock Research for Rural Development. 2015, 27. [Google Scholar]
- Makut, D. , Ogbonna A.I., Dalami H. An Assessment of the Bacteriological Quality of Different Brands of Yoghurt Sold in Keffi, Nasarawa State. Nigeria. J of Nat Sci and Res. 2014, 4, 19–22. [Google Scholar]
- Nduko, J.M. , Matofari J.W., Nandi Z.O., and Sichangi M.B. Spontaneously fermented Kenyan milk products. A review of the current state and future perspectives. African J of Food Sci. 2017, 11, 1–11. [Google Scholar]
- Ifeanyi, V.O. , Ihesiaba E.O., Muomaife O.M., & Ikenga C. Assessment of microbiological quality of yoghurt sold by street vendors in Onitsha metropolis, Anambra State, Nigeria. British Microbiology Research Journal. 2013, 3, 198. [Google Scholar]
- Digbabul B, Shember J, Amove J. Physicochemical, microbiological, and sensory evaluation of yoghurt sold in Makurdi metropolis. African Journal of Food Science and Technology 2014, 5, 129–135. [Google Scholar]
- Sulaiman I.M, Hsieh Y.H. Dairy in Human Health and Disease Across the Lifespan. Berlin, Germany: Springer. Foodborne pathogens in milk and dairy products: Genetic characterization and rapid diagnostic approach for food safety of public health importance. 2017: pp. 127–143.
- Health Protection Agency (HPA). Guidelines for Assessing the Microbiological Safety of Ready-to-Eat Foods London: Health Protection Agency. 2009.
- Majoie G.Ã, Mousse W., Haziz S.I.N.., Farid B.A.D., Ahouissou O.R., Adjanohoun A., Lamine B.M. Microbial quality of artisanal yoghurt and Degue products collected in schools of Cotonou and Abomey-Calavi (Benin). Afr. J. Food Sci. 2020, 14, 112–118. [CrossRef]
- Abdalla M.O.M and Abdel Nabi, S.Z. Evaluation of microbiological quality of Sudanese fermented dairy product ‘mish’ during storage. Adv J of Food Sci and Tech 2010, 2, 155–158. [Google Scholar]
- Moonga, H. B. , Schoustra, S. E., Linnemann, A. R., Kuntashula, E., Shindano, J., & Smid, E. J. The art of mabisi production: A traditional fermented milk. PLOS ONE. 2019, 14, e0213541. [Google Scholar] [CrossRef]
- Benkirane G., Ananou S., Dumas E., Ghnimi S., and Gharsallaoui A. Moroccan Traditional Fermented Dairy Products: Current processing Practices and Physiochemical and Microbiological Properties. A Review. J of Micro, Biotech and Food Sci. 2022. [CrossRef]
- Maleke M. S., Adefisoye, M. A., Doorsamy, W., & Adebo, O. A. Processing, nutritional composition and microbiology of amasi: A Southern African fermented milk product. Sci Afri, 2021. 12. [CrossRef]
- Taye, Y. , Degu T., Fesseha H., and Mathewos M. Isolation and Identification of lactic acid bacteria from cow Milk and Milk products. The Sci World Jl. 2021. [CrossRef]
- Jans, C. , Meile L., Kaindi, D. W. M., Kogi-Makau, W., Lamuka, P., Renault, P.,... & Bonfoh, B. African fermented dairy products–overview of predominant technologically important microorganisms focusing on African Streptococcus infantarius variants and potential future applications for enhanced food safety and security. Int J of Food Micro. 2017, 250, 27–36. [Google Scholar]
- Gleeson, D. , O’Brien B., Flynn J., O’Callaghan E., Galli F. Effect of pre-milking teat preparation procedures on the microbial count on teats prior to cluster application. Ir Vet J. 2009, 62, 461. [Google Scholar] [CrossRef]
- Haile, W. , Yilma Z., and Teklegiorgis Y. Incidence of pathogenic and indicator bacteria in raw and pasteurized milk in Hawassa city, rift valley of Southern Ethiopia. Afr J of Food Sci, 2012, 7. [Google Scholar]
- Benkerroom, N. Traditional Fermented Foods of North African Countries: Technology and Food Safety Challenges with Regard to Microbiological Risks. Comp Rev in Food Sci and Food Saf. 2013, 12, 54–89. [Google Scholar] [CrossRef]
- Ismail Ahmed, A. Isolation and Identification of LAB from Sudanese Traditional Fermented Camel (Camelus dromedarius) Milk Gariss. Open J Nutr Food Sci. 2022, 4, 1022. [Google Scholar]
- Obodai M., Dodd, C.E.R. Characterization of dominant microbiota of a Ghanaian fermented milk product, nyarmie, by culture- and nonculture-based methods. J of App Micr. 2006, 100, 1355–1363. [Google Scholar] [CrossRef] [PubMed]
- Owusu-Kwarteng J. Tano-debrah K. Glover R.L.K., Akabanda F. Process characteristics and microbiology of fura produced in Ghana. Nat and Sci. 2010, 8, 41–51. [Google Scholar]
- Mathara, J.M. , Schillingera U., Kutima P.M., Mbugua S.K., Holzapfel W.H. Isolation, Identification, and characterisation of the dominant microorganisms of kule naoto: the Maasai traditional fermented milk in Kenya. Int J of Food Micro. 2004, 94, 269–27. [Google Scholar] [CrossRef] [PubMed]
- Teuber M., The genus Lactococcus. IN: Wood BJB., Holzapfel WH (ed). The Genera of Lactic acid Bacteria. The Lactic Acid Bacteria. 1995. Vol 2. Springer, Boston, MA.
- Togo, C.A. , Feresu, S.B., Mutukumira, A.N. Identification of Lactic Acid Bacteria isolated from Opaque beer (Chibuku) for potential use as a starter culture. The J of Food Tech in Afr. 2002, 7, 93–97. [Google Scholar]
- Webb, L.; Ma, L.; Lu, X. Impact of Lactic Acid Bacteria on the Control of Listeria monocytogenes in Ready-to-Eat Foods. Food Qual. Saf. 2022. [Google Scholar] [CrossRef]
- Nyambane, B. , Thari, W.M., Wangoh J., and. Njage P.M.K. Lactic acid bacteria and yeasts involved in the fermentation of amabere amaruranu, a Kenyan fermented milk. Food Sci & Nutri. 2014, 2, 692–699. [Google Scholar]
- Pyz-Lukasik, R. , Paszkiewicz W., Brodzki, P., Belkot, Z. Microbiological quality of milk sold directly from producers to consumers. J of Dairy Sci. 2015, 98, 4294–4301. [Google Scholar] [CrossRef]
- Hamama A. Moroccan traditional fermented dairy products. In: Ruskin, F.R. (Ed.). Applications of biotechnology to traditional fermented foods. National Academy Press, Washington DC. 1992. p: 75-79.
- Foster, J.W. Low pH Adaptation and the Acid ~Tolerance Response of Salmonella typhimurium. Cri Reviews in Micro. 2008, 21, 215–237. [Google Scholar] [CrossRef] [PubMed]
- Liyuwork, T. , Biruhalem T., Sefinew A., Hailleleul N. Prevalence and antimicrobial resistance profile of Salmonella isolates from dairy products in Addis Ababa, Ethiopia. Afr. J of Micro Res 2013, 7943, 5045–5050. [Google Scholar]
- Bedassa, A. , Nahusenay H., Asefa Z., Sisay T; Girmay G., Kovac J., Vipham J.L., and Zewdu A. Prevalence and associated risk factors for Salmonella enterica contamination of cow milk and cottage cheese in Ethiopia. Int J Food Cont. 2023, 10, 2. [Google Scholar]
- Chatti A., Daghfous D., Landoulsi A. Acid resistance of Salmonella isolated from animals, food and wastewater in Tunisia. Ann Saudi Med. 2007, 27, 195–198. [CrossRef]
- Halkman HBD., Halkman, A.K. Indicator Organisms. Encyclopaedia of Food Microbiology (Second Edition). 2014.
- D’Amico and Donelly, C.W. Microbiological quality of raw milk used for small-scale artisan cheese production in Vermont: Effect of farm characteristics and practices. J of Dairy Sci. 2010, 9, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Somaratne, N.; Hallas, G. Review of Risk Status of Groundwater Supply Wells by Tracing the Source of Coliform Contamination. Water 2015, 7, 3878–3905. [Google Scholar] [CrossRef]
- Fleet, G.H. Yeasts in dairy products. A Review. J. of App Bact. 1990, 68, 199–211. [Google Scholar] [CrossRef]
- Akabanda, F. , Owusu-Kwarteng, J., Glover, R.L.K., Tano-Debrah, K. Microbiological characteristics of Ghanaian traditional fermented milk product. Nunu. Nature Sci. 2010, 8, 178–187. [Google Scholar]
- Savova, L. and Nikolova, M. Isolation and Taxonomic study of yeast strain from Bulgarian Dairy products. J of Culture Coll. 2000, 3, 59–65. [Google Scholar]
- Bùchi NR., and Seller H. Yeast and moulds: yeasts in milk and dairy products. 2011. On ResearchGate. [CrossRef]
- Getachew, A., Tadie, A., Chercos, D.H., and Guadu, T. Level of Faecal Coliform Contamination of Drinking Water Sources and Its Associated Risk Factors in Rural Settings of North Gondar Zone, Ethiopia: A Cross-Sectional Community Based Study. Ethiop J Health Sci. 2018, 28, 227–223. [CrossRef]
- Carpentier, B.; Cerf, O. Review-Persistence of Listeria monocytogenes in Food Industry Equipment and Premises. Int. J. Food Micro. 2011, 145, 1–8. [Google Scholar] [CrossRef] [PubMed]
- ZelalemY. Quality factors that affect Ethiopian milk business: Experiences from selected dairy potential areas. Netherlands Development Organization, Addis Ababa, Ethiopia. 2010.
- Jans, C. , Meile L., Wambua D., Kaindi M., Kogi-Makau W., Lamuka P., Renault, P., Kreikemeyer B., Lacroix C., Hattendorf J., Zinsstag J., Schelling E.,m Fokou g., Bonfoh B. African fermented dairy products – Overview of predominant technologically important microorganisms focusing on African Streptococcus infantarius variants and potential future applications for enhanced food safety and security. Int J of Food Micro. 2017, 250, 27–36. [Google Scholar]
- Bereda A, Yesuf Kurtu M, Yilma Z. Handling, processing and utilization of Milk and Milk products in Ethiopia: a review. World J Dairy Food Sci. 2014, 9, 105–112. [Google Scholar]
- Mekonnen, Z. , Kidemu M. Abebe H., Semere M., Gebreyesus M., Workuy A., Tesfaye M., Chernet A. Traditional knowledge and institutions for sustainable climate change adaptation in Ethiopia. Current Res in Env Sust. 2021, 3, 100080. [Google Scholar]
- Okello AL, Bardosh K, Smith J, Welburn SC. One Health: Past successes and future challenges in three African contexts. PLoS Negl Trop Dis. 2014, 8, e2884. [Google Scholar]
- Food and Agriculture Organisation of the United Nations (FAO). Guide to good dairy farming practice: Milking Hygiene. 2023.
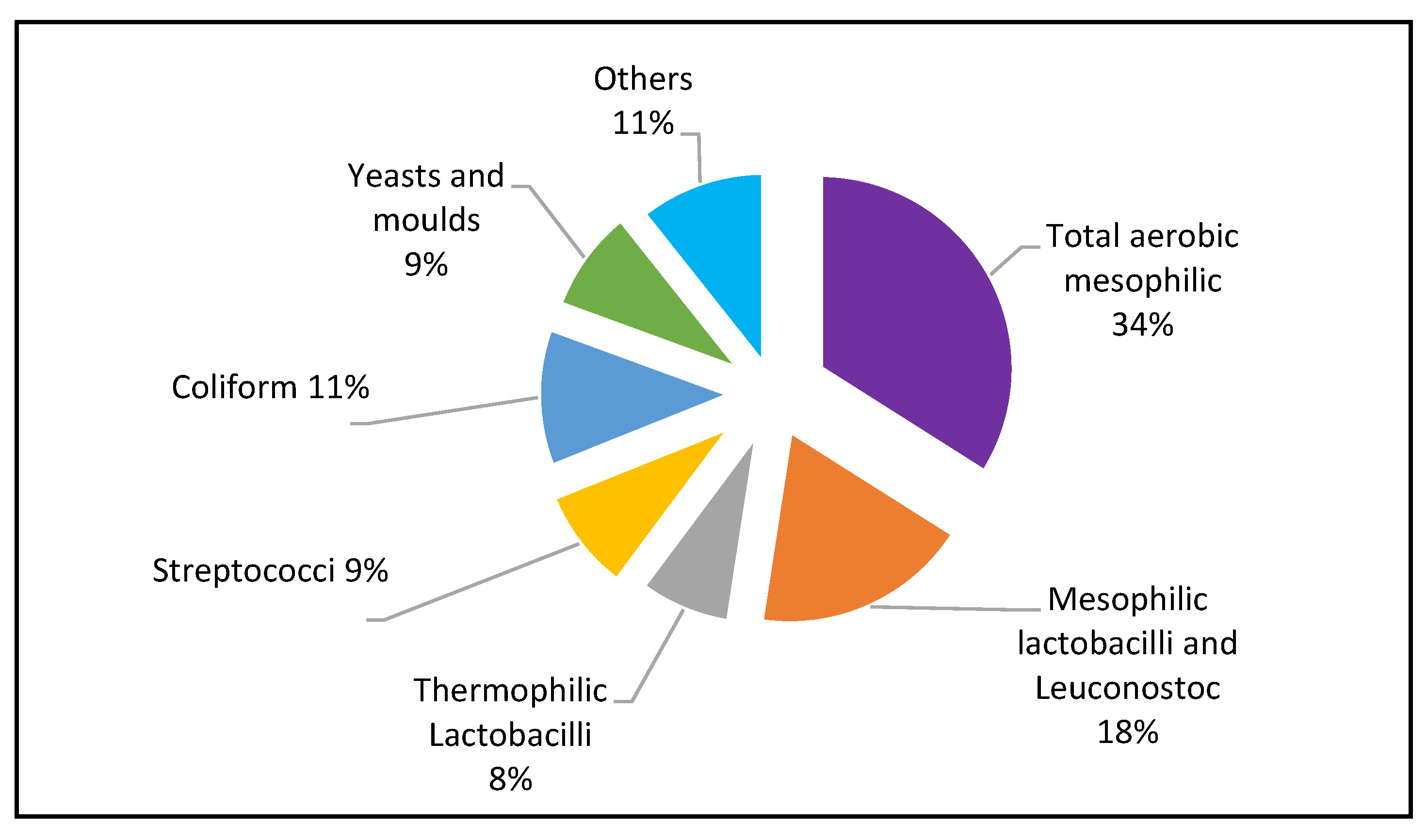
| Samples | pH | Titratable acidity (%) |
|---|---|---|
| UG 1 | 2.9 ± 0.01 | 1.26 ± 0.1 |
| UG 2 | 3.4 ± 0.01 | 0.71 ± 0.1 |
| KE | 3.6± 0.01 | 0.92 ± 0.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




