Submitted:
30 May 2023
Posted:
31 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
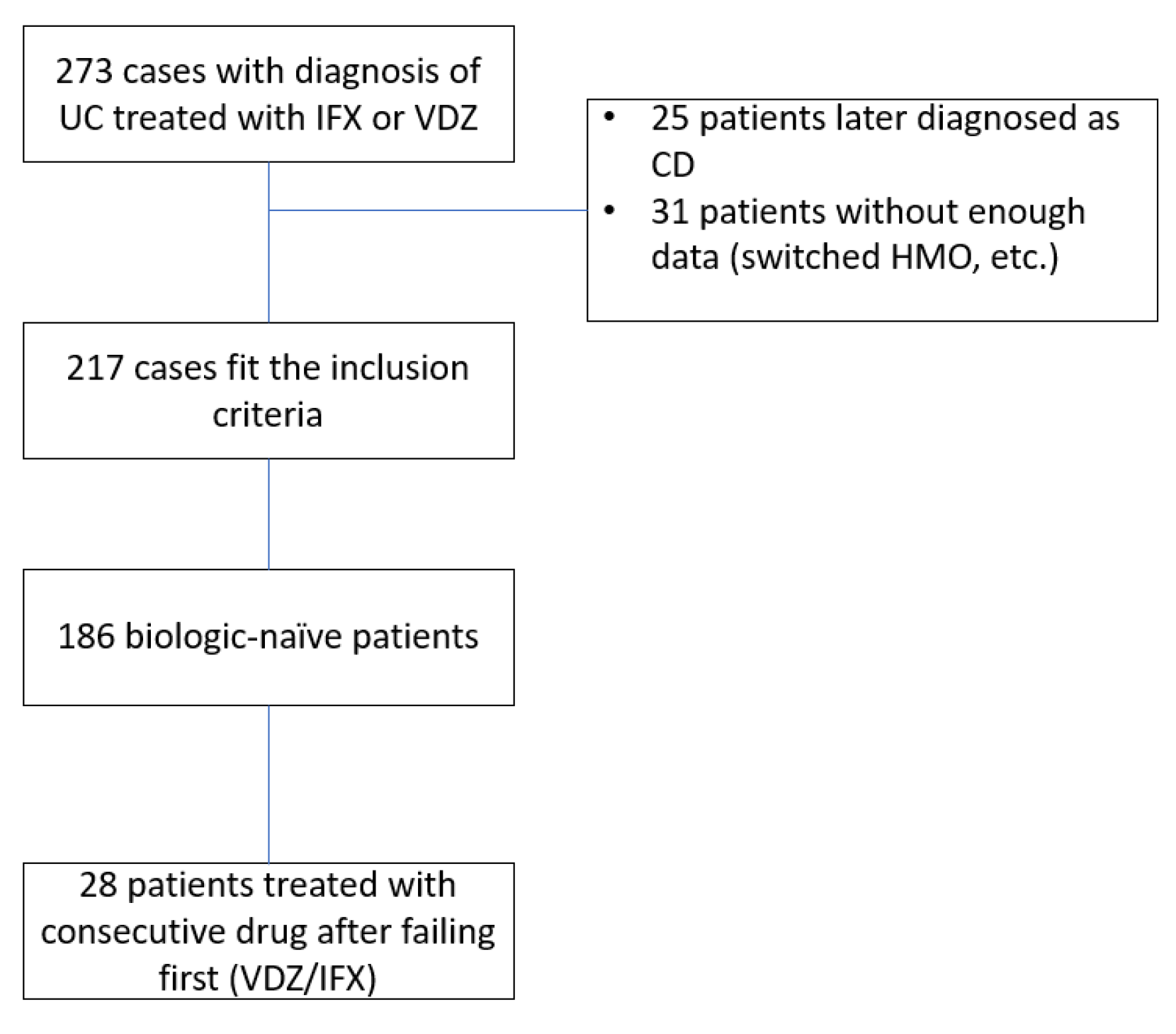
1.1. Patient Selection and Follow-Up Period
1.2. Outcomes
1.3. Data Collection
1.4. Statistical Analysis
3. Results
2.1. Baseline Population Characteristics
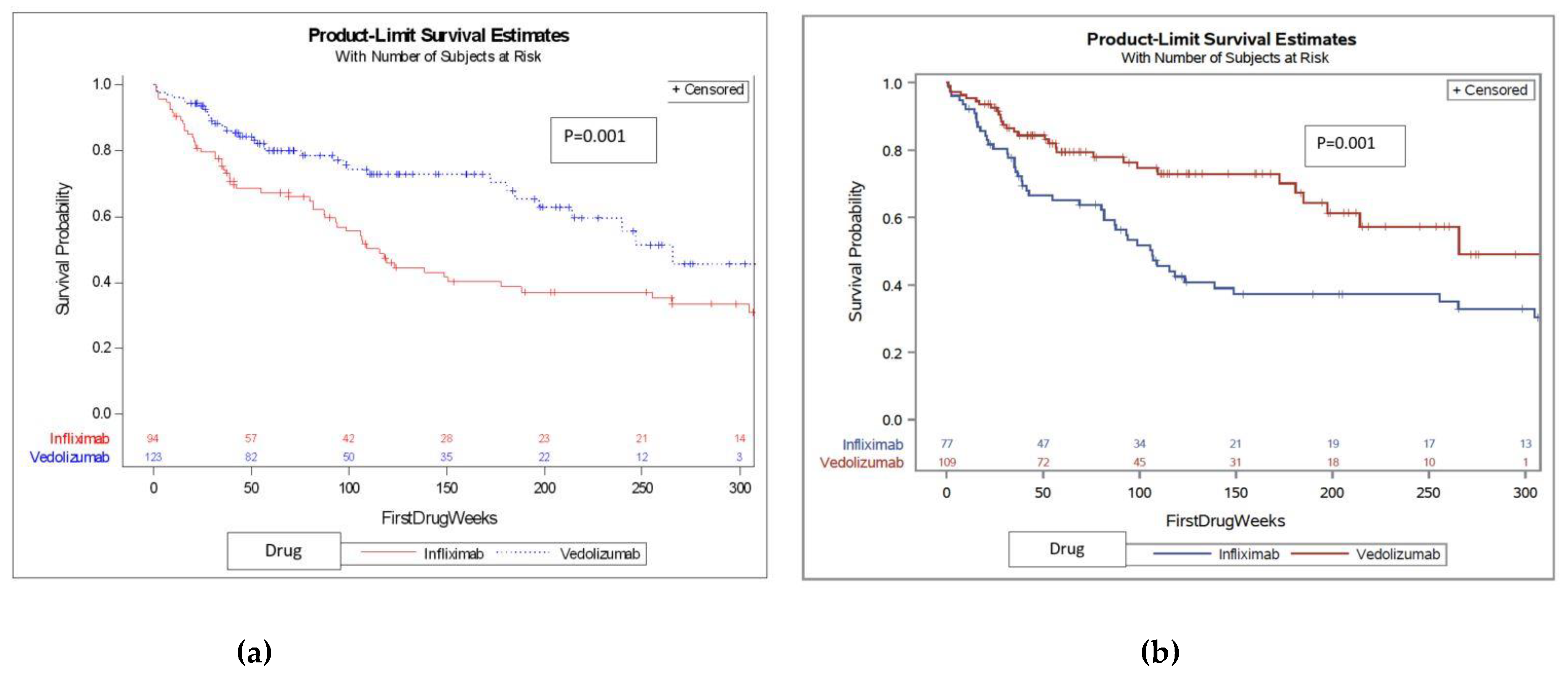
2.2. DS
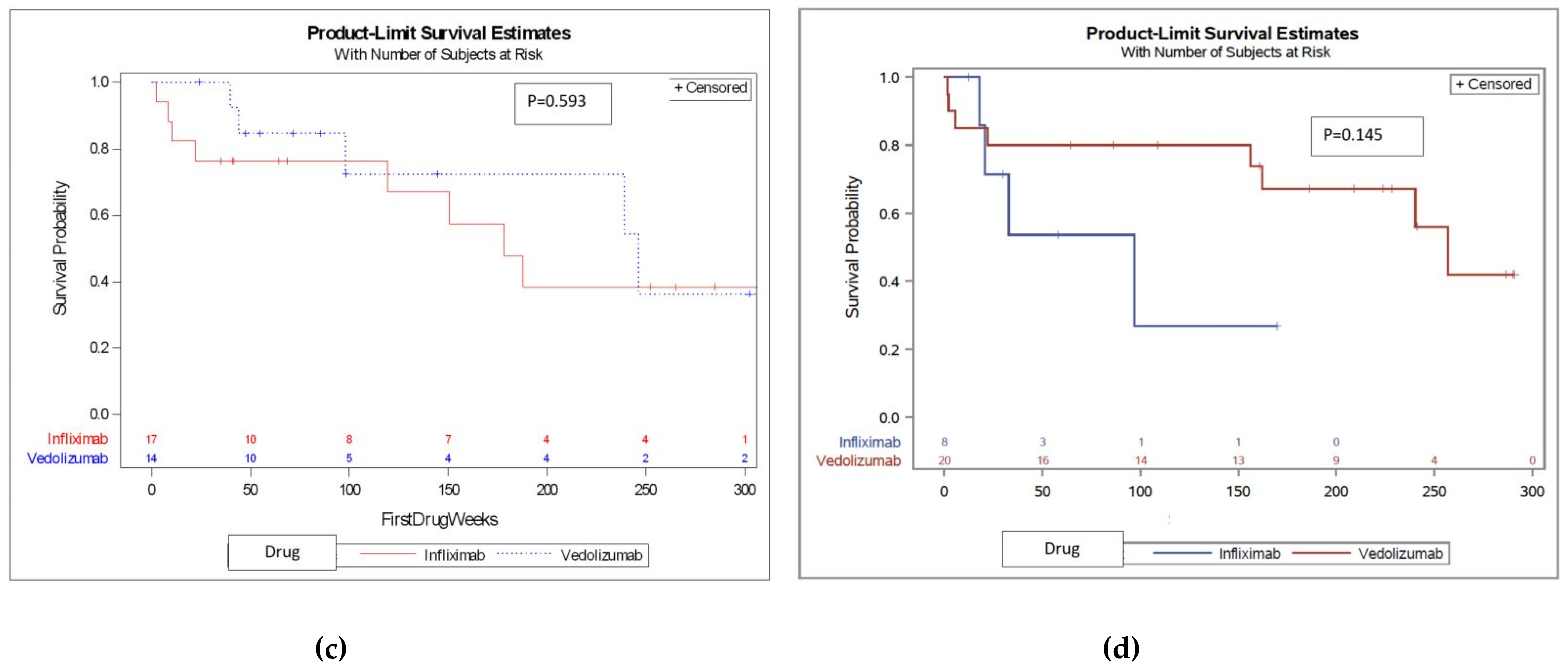
2.3. Subsequent Treatment with a Second Drug
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Molodecky, N.A.; Soon, I.S.; Rabi, D.M.; et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 2012, 142, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Peyrin-Biroulet, L.; Danese, S.; Argollo, M.; et al. Loss of Response to VDZ and Ability of Dose Intensification to Restore Response in Patients With Crohn's Disease or Ulcerative Colitis: A Systematic Review and Meta-analysis. Clin Gastroenterol Hepatol 2019, 17, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Roda, G.; Jharap, B.; Neeraj, N.; Colombel, J.F. Loss of Response to Anti-TNFs: Definition, Epidemiology, and Management. Clin Transl Gastroenterol 2016, 7, e135. [Google Scholar] [CrossRef] [PubMed]
- Allez, M.; Karmiris, K.; Louis, E.; et al. Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: Definitions, frequency and pharmacological aspects. J Crohns Colitis 2010, 4, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Macaluso, F.S.; Maida, M.; Grova, M.; et al. Head-to-head comparison of biological drugs for inflammatory bowel disease: From randomized controlled trials to real-world experience. Therap Adv Gastroenterol 2021, 14, 17562848211010668. [Google Scholar] [CrossRef] [PubMed]
- Baer, P.A.; Aumais, G.; Ewara, E.M.; et al. Patterns and predictors of long-term retention of inflammatory bowel or rheumatoid disease patients on innovator IFX: An analysis of a Canadian prescriptions claims database. Patient Prefer Adherence 2018, 12, 1805–1814. [Google Scholar] [CrossRef] [PubMed]
- Lukin, D.; Faleck, D.; Xu, R.; et al. Comparative Safety and Effectiveness of VDZ to Tumor Necrosis Factor Antagonist Therapy for Ulcerative Colitis. Clin Gastroenterol Hepatol 2020. [Google Scholar] [CrossRef]
- Visuri, I.; Eriksson, C.; Olen, O.; et al. Predictors of drug survival: A cohort study comparing anti-tumour necrosis factor agents using the Swedish inflammatory bowel disease quality register. Aliment Pharmacol Ther 2021, 54, 931–943. [Google Scholar] [CrossRef] [PubMed]
- Cholapranee, A.; Hazlewood, G.S.; Kaplan, G.G.; Peyrin-Biroulet, L.; Ananthakrishnan, A.N. Systematic review with meta-analysis: Comparative efficacy of biologics for induction and maintenance of mucosal healing in Crohn's disease and ulcerative colitis controlled trials. Aliment Pharmacol Ther 2017, 45, 1291–1302. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Fumery, M.; Sandborn, W.J.; Murad, M.H. Systematic review with network meta-analysis: First- and second-line pharmacotherapy for moderate-severe ulcerative colitis. Aliment Pharmacol Ther 2018, 47, 162–175. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.; McParland, P.; Dodd, S.; et al. Comparative effectiveness of antitumour necrosis factor agents and VDZ in ulcerative colitis. Eur J Gastroenterol Hepatol 2019, 31, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Rundquist, S.; Sachs, M.C.; Eriksson, C.; et al. Drug survival of anti-TNF agents compared with VDZ as a second-line biological treatment in inflammatory bowel disease: Results from nationwide Swedish registers. Aliment Pharmacol Ther 2021, 53, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Hartzema, A.G.; Xiao, H.; et al. Real-world Pattern of Biologic Use in Patients With Inflammatory Bowel Disease: Treatment Persistence, Switching, and Importance of Concurrent Immunosuppressive Therapy. Inflamm Bowel Dis 2019, 25, 1417–1427. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.S.; Han, M.; Park, S.; Cheon, J.H. Biologic Use Patterns and Predictors for Non-persistence and Switching of Biologics in Patients with Inflammatory Bowel Disease: A Nationwide Population-Based Study. Dig Dis Sci 2020, 65, 1436–1444. [Google Scholar] [CrossRef] [PubMed]
- Bressler, B.; Yarur, A.; Silverberg, M.S.; et al. VDZ and Anti-Tumour Necrosis Factor alpha Real-World Outcomes in Biologic-Naive Inflammatory Bowel Disease Patients: Results from the EVOLVE Study. J Crohns Colitis 2021, 15, 1694–1706. [Google Scholar] [CrossRef] [PubMed]
- Pudipeddi, A.; Ko, Y.; Paramsothy, S.; Leong, R.W. VDZ has longer persistence than IFX as a first-line biological agent but not as a second-line biological agent in moderate-to-severe ulcerative colitis: Real-world registry data from the Persistence Australian National IBD Cohort (PANIC) study. Therap Adv Gastroenterol 2022, 15, 17562848221080793. [Google Scholar] [CrossRef] [PubMed]
- Narula, N.; Wong, E.C.L.; Marshall, J.K.; Colombel, J.F.; Dulai, P.S.; Reinisch, W. Comparative Efficacy for IFX Vs VDZ in Biologic Naive Ulcerative Colitis. Clin Gastroenterol Hepatol 2021. [Google Scholar] [CrossRef]
| Parameter | IFX | VDZ | All | p-value | |
|---|---|---|---|---|---|
| Male | 47 (61%) | 60 (55%) | 107 (57.5%) | 0.454 | |
| Disease extent | 0.149 | ||||
| Disease extent | Extensive Colitis | 28 (36.3%) | 31 (28.4%) | 59 (31.7%) | 0.267 |
| Left-sided UC | 41 (53.2%) | 53 (48.6%) | 94 (50.4%) | 0.555 | |
| Proctitis | 8 (10.3%) | 24 (22%) | 32 (17.2%) | 0.048 | |
| Combination therapy | 11 (14.2%) | 3 (2.7%) | 14 (7.5%) | 0.004 | |
| Age at Diagnosis ( years median, IQR) | 32 (20-48) | 32 (21-46) | 32 (21-47) | 0.869 | |
| Age at treatment start (years, median, IQR) | 37 (25-55) | 42 (28-57) | 40.5 (26-56) | 0.293 | |
| Diagnosis length at treatment start (years, median, IQR) | 2 (0-6) | 4 (1-12) | 2 (1-11) | 0.104 | |
| Calprotectin (µg/g, median, IQR) | 4425 (1350-5870) | 529 (161-1339) | 890 (196-3230) | 0.001 | |
| Hemoglobin (g/dL, years, median, IQR) | 11 (9.5-12.9) | 13.1 (12-14.1) | 12.7 (10.8-13.8) | <.001 | |
| Platelets (K/micl, median, IQR) | 309 (242-396) | 284 (225-366) | 298 (231-376) | 0.187 | |
| Hematocrit (%, median, IQR) | 33.8 (30.5-38.9) | 40.9 (38-43) | 38.8 (33.9-42.5) | <.001 | |
| Hb/Hct ratio (median, IQR) | 0.32 (0.31-0.33) | 0.32 (0.31-0.33) | 0.32 (0.31-0.33) | 0.640 | |
| Eosinophils (K/micl, median, IQR) | 0.1 (0.0-3) | 0.2 (0.1-0.8) | 0.1 (0.0-3) | 0.589 | |
| Neutrophils (K/micl, median, IQR) | 6 (4.6-9.1) | 5.2 (4-7.3) | 5.5 (4.1-7.9) | 0.203 | |
| Lymphocytes (K/micl, median, IQR) | 1.4 (1-2) | 1.8 (1.3-2.4) | 1.6 1.2-2.3) | 0.009 | |
| Neutrophil-to-lymphocyte ratio (median, IQR) | 4.2 (2.7-7) | 2.7 (2-4.6) | 3.2 (2.2-5.4) | 0.000 | |
| Leukocytes (K/micl, median, IQR) | 9 (6.7-11.3) | 7.9 (6.3-10.1) | 8.2 (6.3-10.6) | 0.497 | |
| Albumin (g/dL, median, IQR) | 3.4 (2.8-4.1) | 4.3 (4.1-4.6) | 4.2 (3.5-4.5) | <.001 | |
| Creatinine (mg/dL, median, IQR) | 0.6 (0.5-0.9) | 0.8 (0.7-0.9) | 0.8 (0.6-0.9) | 0.002 | |
| CRP (mg/dL, median, IQR) | 1.3 (0.4-3.5) | 0.5 (0.2-0.9) | 0.6 (0.2-1.8) | 0.002 | |
| CPK (U/L, median, IQR) | 43 (30-76) | 59 (43-115) | 53 (31-85) | 0.843 | |
| Phosphorus (mg/dL, median, IQR) | 3.3 (2.9-3.8) | 3.4 (2.9-3.8) | 3.3 (2.9-3.8) | 0.425 | |
| Total protein (g/dL, median, IQR) | 6.2 (5.4-7) | 7.2 (6.9-7.5) | 7 (6.3-7.4) | <.001 | |
| Parameter | Hazard ratio (CI 95%) | P-value (Chi-Sq) | Adjusted* HR (CI 95%) | Adjusted* p-value | |
|---|---|---|---|---|---|
| VDZ (compared with IFX) | 0.48 (0.3-0.76) | 0.002 | 0.55 (0.3-0.98) | 0.042 | |
| Male | 0.87 (0.55-1.36) | 0.546 | 1.05 (0.6-1.84) | 0.851 | |
| Disease extent | Proctitis | 0.77 (0.39-1.53) | 0.469 | -- | -- |
| Left-sided | 0.81 (0.5-1.32) | 0.412 | -- | -- | |
| Extensive | 1.25 (0.79-1.99) | 0.827 | -- | -- | |
| Combination therapy | 1.59 (0.09-26.78) | 0.352 | 0.97 (0.44-2.13) | 0.939 | |
| Age at diagnosis | 0.98 (0.97-1.00) | 0.119 | -- | -- | |
| Age at treatment start | 0.98 (0.97-0.99) | 0.032 | 0.98 (0.97-1) | 0.091 | |
| Disease length | 0.97 (0.95-1) | 0.166 | -- | -- | |
| Calprotectin | 1 (1-1) | 0.070 | -- | -- | |
| Hemoglobin (Hb) | 0.89 (0.8-0.99) | 0.037 | 0.73 (0.46-1.16) | 0.187 | |
| Platelets | 1 (1-1) | 0.062 | -- | -- | |
| Hematocrit (Hct) | 0.96 (0.93-1) | 0.051 | 1.11 (0.94-1.3) | 0.195 | |
| Hb/Hct Ratio | 0 (0-135.09) | 0.188 | -- | -- | |
| Eosinophils absolute number | 0.38 (0.11-1.31) | 0.126 | 0.39 (0.11-1.36) | 0.142 | |
| Neutrophils absolute number | 1.04 (0.98-1.12) | 0.164 | -- | -- | |
| Lymphocytes absolute number | 1.29 (0.99-1.68) | 0.059 | -- | -- | |
| Neutrophil-to-lymphocyte ratio | 0.99 (0.96-1.02) | 0.835 | 0.98 (0.94-1.02) | 0.503 | |
| White blood count | 1.04 (0.98-1.12) | 0.164 | -- | -- | |
| Albumin | 0.68 (0.52-0.9) | 0.007 | 0.74 (0.44-1.24) | 0.603 | |
| Creatinine | 0.31 (0.11-0.88) | 0.028 | -- | -- | |
| CRP | 1 (0.94-1.07) | 0.821 | 0.97 (0.9-1.06) | 0.603 | |
| CPK | 0.99 (0.99-1) | 0.609 | -- | -- | |
| Phosphorus | 1.37 (1.00-1.89) | 0.049 | -- | -- | |
| Total Protein | 0.77 (0.62-0.97) | 0.026 | 1.07 (0.7-1.64) | 0.101 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




