Submitted:
28 May 2023
Posted:
29 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
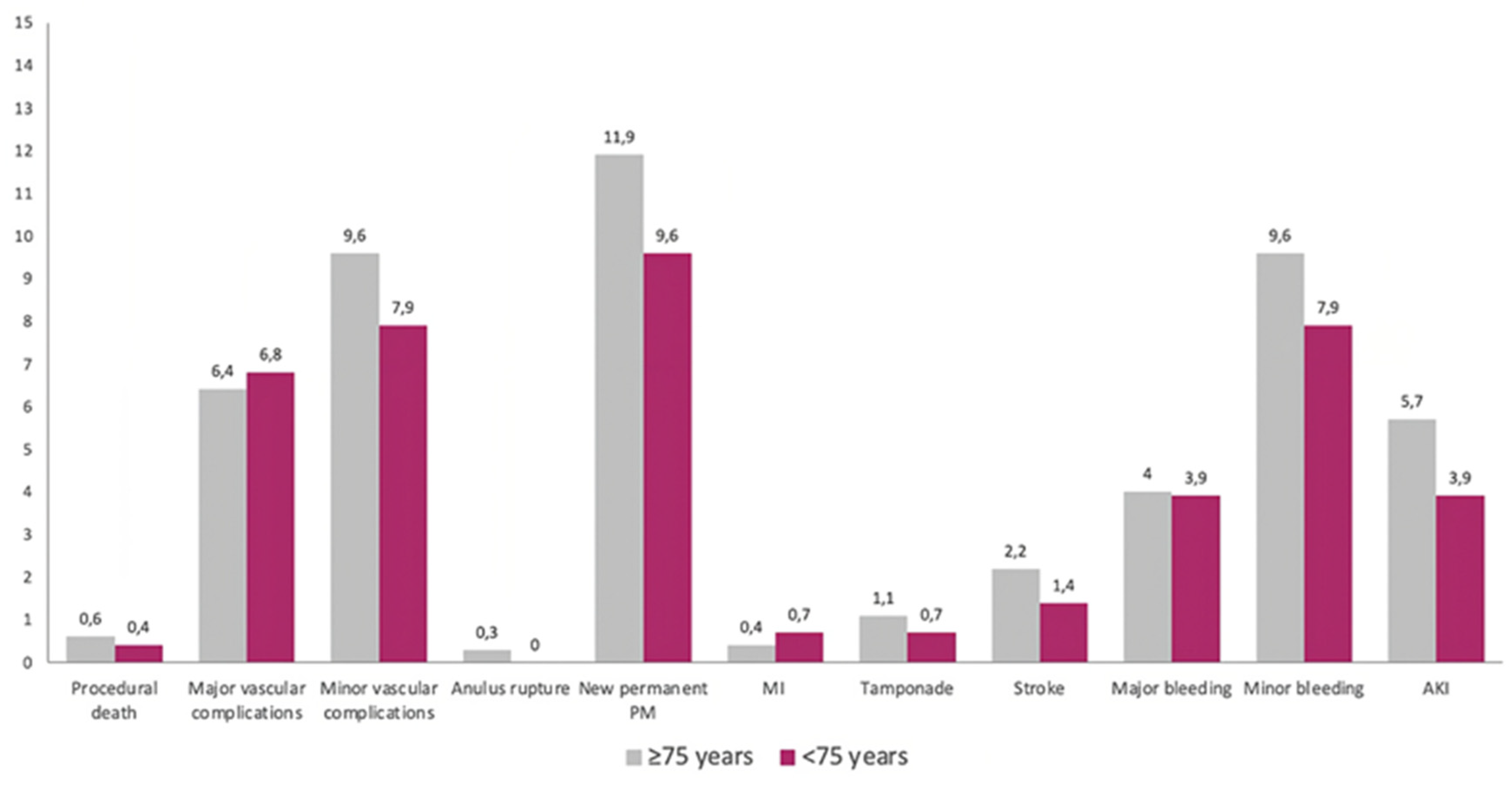
3.1. Baseline and Procedural Features
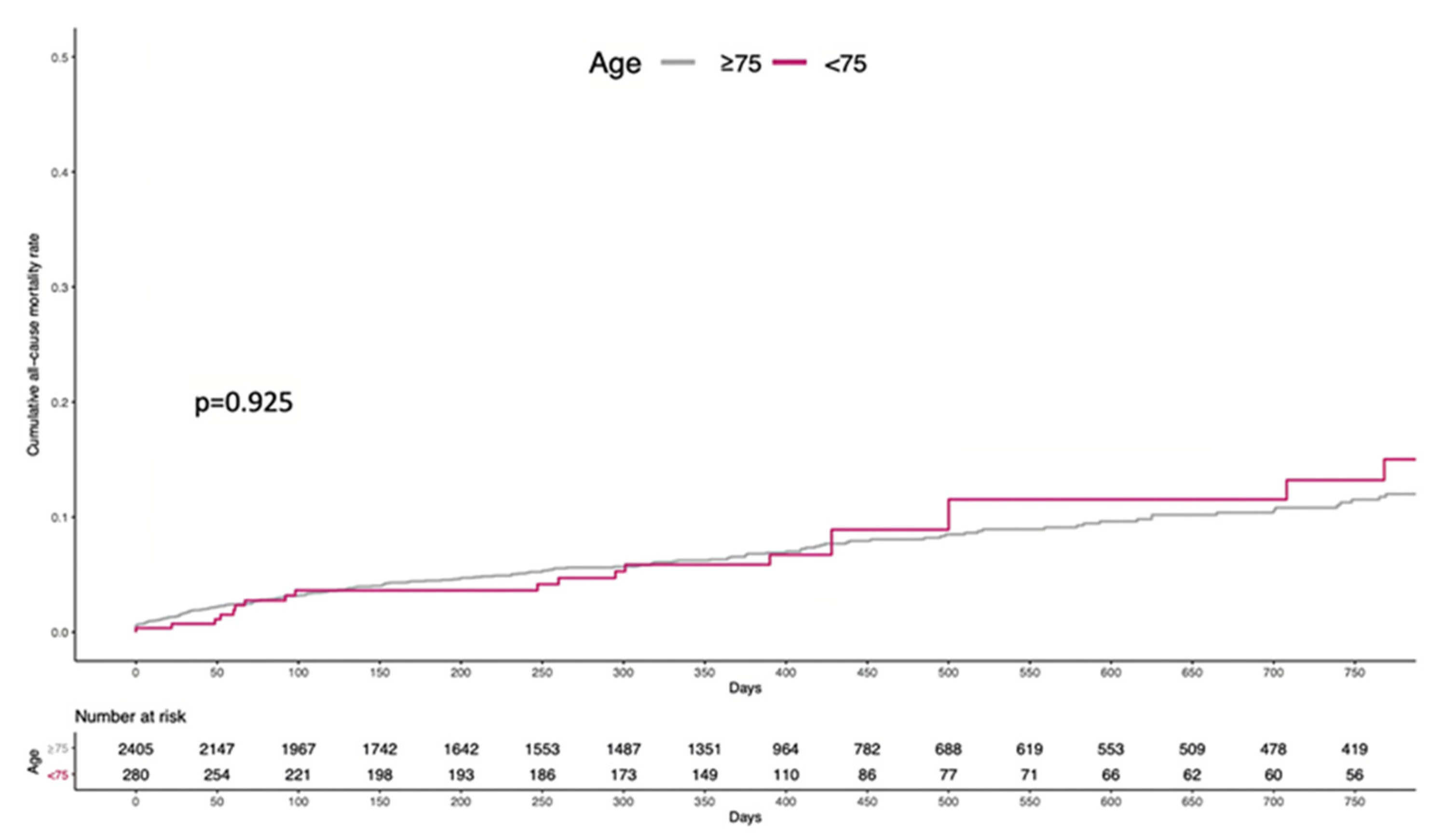
3.2. Clinical Outcomes
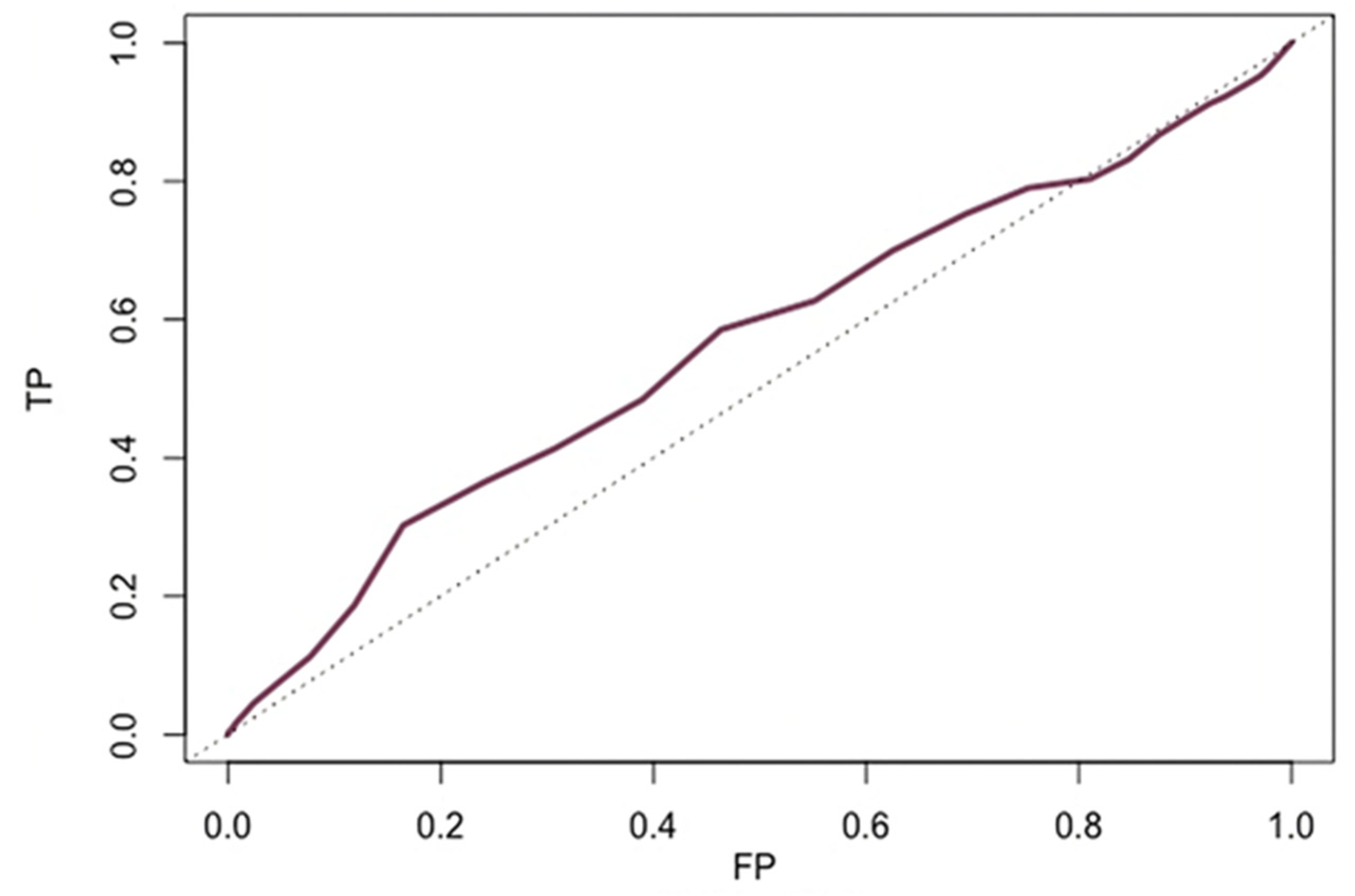
3.3. Accuracy of Age to Predict All-Cause Mortality
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Leon MB, Smith CR, Mack MJ, et al. Transcatheter or Surgical Aortic-Valve Replacement in Intermediate-Risk Patients. N Engl J Med. 2016;374(17):1609-1620. [CrossRef]
- Mack MJ, Leon MB, Thourani VH, et al. Transcatheter Aortic-Valve Replacement with a Balloon-Expandable Valve in Low-Risk Patients. N Engl J Med. 2019;380(18):1695-1705. [CrossRef]
- Reardon MJ, Van Mieghem NM, Popma JJ, et al. Surgical or Transcatheter Aortic-Valve Replacement in Intermediate-Risk Patients. N Engl J Med. 2017;376(14):1321-1331. [CrossRef]
- Popma JJ, Deeb GM, Yakubov SJ, et al. Transcatheter Aortic-Valve Replacement with a Self-Expanding Valve in Low-Risk Patients. N Engl J Med. 2019;380(18):1706-1715. [CrossRef]
- Landes U, Webb JG, De Backer O, et al. Repeat Transcatheter Aortic Valve Replacement for Transcatheter Prosthesis Dysfunction. J Am Coll Cardiol. 2020;75(16):1882-1893. [CrossRef]
- Søndergaard L, Steinbrüchel DA, Ihlemann N, et al. Two-Year Outcomes in Patients With Severe Aortic Valve Stenosis Randomized to Transcatheter Versus Surgical Aortic Valve Replacement: The All-Comers Nordic Aortic Valve Intervention Randomized Clinical Trial. Circ Cardiovasc Interv. 2016;9(6). [CrossRef]
- Smith CR, Leon MB, Mack MJ, et al. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med. 2011;364(23):2187-2198. [CrossRef]
- Ruparelia N, Latib A, Buzzatti N, et al. Long-Term Outcomes After Transcatheter Aortic Valve Implantation from a Single High-Volume Center (The Milan Experience). Am J Cardiol. 2016;117(5):813-819. [CrossRef]
- Vahanian A, Beyersdorf F, Praz F, et al. 2021 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2022;43(7):561-632. [CrossRef]
- Gallo F, Gallone G, Kim WK, et al. Horizontal Aorta in Transcatheter Self-Expanding Valves: Insights From the HORSE International Multicentre Registry. Circ Cardiovasc Interv. 2021;14(9). [CrossRef]
- World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310(20):2191-2194. [CrossRef]
- Kappetein AP, Head SJ, Généreux P, et al. Updated standardized endpoint definitions for transcatheter aortic valve implantation: the Valve Academic Research Consortium-2 consensus document. Eur Heart J. 2012;33(19). [CrossRef]
- Arsalan M, Szerlip M, Vemulapalli S, et al. Should Transcatheter Aortic Valve Replacement Be Performed in Nonagenarians?: Insights From the STS/ACC TVT Registry. J Am Coll Cardiol. 2016;67(12):1387-1395. [CrossRef]
- Attinger-Toller A, Ferrari E, Tueller D, et al. Age-Related Outcomes After Transcatheter Aortic Valve Replacement: Insights From the SwissTAVI Registry. JACC Cardiovasc Interv. 2021;14(9):952-960. [CrossRef]
- Vlastra W, Chandrasekhar J, Vendrik J, et al. Transfemoral TAVR in Nonagenarians: From the CENTER Collaboration. JACC Cardiovasc Interv. 2019;12(10):911-920. [CrossRef]
- Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021;143(5):E72-E227. [CrossRef]
- Eggebrecht H, Bestehorn K, Rassaf T, et al. In-hospital outcomes after transcatheter or surgical aortic valve replacement in younger patients less than 75 years old: a propensity-matched comparison. EuroIntervention. 2018;14(1):50-57. [CrossRef]
- Witberg G, Landes U, Codner P, et al. Clinical outcomes of transcatheter aortic valve implantation in patients younger than 70 years rejected for surgery: the AMTRAC registry. EuroIntervention. 2022;17(16):1289-1297. [CrossRef]
- Johnston DR, Soltesz EG, Vakil N, et al. Long-term durability of bioprosthetic aortic valves: implications from 12,569 implants. Ann Thorac Surg. 2015;99(4):1239-1247. [CrossRef]
- Yerasi C, Rogers T, Forrestal BJ, et al. Transcatheter Versus Surgical Aortic Valve Replacement in Young, Low-Risk Patients With Severe Aortic Stenosis. JACC Cardiovasc Interv. 2021;14(11):1169-1180. [CrossRef]
- 21Otto, CM. Alignment and divergence in European and North American aortic stenosis guidelines. EuroIntervention. 2022;17(14):E1123-E1125. [CrossRef]
- Khokhar AA, Ponticelli F, Zlahoda-Huzior A, et al. Coronary access following ACURATE neo implantation for transcatheter aortic valve-in-valve implantation: Ex vivo analysis in patient-specific anatomies. Front Cardiovasc Med. 2022;9. [CrossRef]
| All N=2685 |
≥75 years N=2405 |
<75 years N=280 |
P value | |
|---|---|---|---|---|
| Clinical characteristics | ||||
| Age, years | 82 (79-86) | 83 (80-86) | 72 (69-73) | <0.001 |
| Male sex | 960 (36) | 835 (35) | 125 (45) | 0.001 |
| Body mass index, kg/m2 | 26.9 (24-31) | 26.7 (24-30) | 28.5 (24-33) | <0.001 |
| Hypertension | 2334 (87) | 2092 (87) | 242 (86) | 0.813 |
| Dyslipidemia | 1035 (52) | 925 (52) | 110 (51) | 0.933 |
| Diabetes | 705 (26) | 613 (25) | 92 (33) | 0.010 |
| Smoke | 228 (15) | 176 (7) | 52 (19) | <0.001 |
| Prior myocardial infarction | 482 (18) | 441 (18) | 41 (15) | 0.144 |
| Prior percutaneous coronary intervention | 731 (27) | 656 (27) | 75 (27) | 0.905 |
| Prior stroke | 287 (11) | 261 (11) | 26 (9) | 0.478 |
| Chronic obstructive pulmonary disease | 450 (17) | 381 (16) | 69 (25) | <0.001 |
| PM or ICD | 276 (10) | 259 (11) | 17 (6) | 0.019 |
| Atrial fibrillation | 2334 (87) | 795 (33) | 65 (23) | 0.010 |
| Chronic kidney disease | 1645 (61) | 1562 (65) | 83 (30) | <0.001 |
| Baseline creatinine, mg/dl | 1.10 (0.61) | 1.09 (0.55) | 1.19 (0.98) | 0.010 |
| Peripheral arterial disease | 333 (12) | 297 (12) | 36 (13) | 0.890 |
| NYHA III-IV | 1787 (67) | 1607 (67) | 180 (64) | 0.390 |
| STS score, % | 3.60 (2.50-5.03) | 3.73 (2.60-5.10) | 2.55 (1.75 4.10) | <0.001 |
| Low risk | 1520 (57) | 1318 (55) | 202 (72) | <0.001 |
| Intermediate risk | 1165 (43) | 1087 (45) | 78 (28) | <0.001 |
| Echocargiographic data | ||||
| Mean aortic valve gradient, mmHg | 45.1 (16.2) | 45.2 (16.3) | 44.9 (15.6) | 0.821 |
| Aortic valve area, mm2 | 0.76 (2.73) | 0.77 (2.87) | 0.73 (0.19) | 0.850 |
| Left ventricular ejection fraction, % | 60 (55.0-65) | 60 (55-65) | 0.006 | |
| Moderate-severe aortic regurgitation | 56 (3) | 51 (3) | 5 (2) | 0.966 |
| MDCT data | ||||
| Perimeter, mm | 70 (26-76) | 70 (26-76) | 71 (26-77) | 0.367 |
| Moderate-severe aortic valve calcification | 1173 (44) | 1055 (44) | 118 (42) | 0.971 |
| Moderate/severe LVOT calcification | 446 (16) | 408 (17) | 38 (14) | 0.224 |
| Porcelain aorta | 271 (10) | 225 (9) | 46 (16) | <0.001 |
| All N=2685 |
≥75 years N=2405 |
<75 years N=280 |
P value | |
|---|---|---|---|---|
| Predilatation | 1452 (54) | 1309 (54) | 143 (51) | 0.309 |
| Valve type | ||||
| Evolute R | 1068 (40) | 963 (40) | 105 (38) | 0.449 |
| Evolute PRO | 301 (11) | 253 (11) | 48 (17) | 0.001 |
| ACURATE neo | 1316 (49) | 1189 (49) | 127 (45) | 0.219 |
| Valve size, mm | 0.025 | |||
| <23 | 423 (16) | 381 (16) | 42 (15) | |
| 23-26 | 1021 (38) | 933 (39) | 88 (31) | |
| ≥27 | 1241 (46) | 1091 (45) | 150 (54) | |
| Postdilatation | 892 (33) | 788 (33) | 104 (37) | 0.153 |
| Contrast dose, ml | 110 (80-160) | 110 (80-160) | 110 (80-150) | 0.612 |
| Fluoroscopy time, minutes | 16 (10-24) | 16 (10-24) | 15 (10-23) | 0.244 |
| Univariable | Multivariable | |||
|---|---|---|---|---|
| Variable | HR (95% CI) | P value | HR (95% CI) | P value |
| Age (1-year increase) | 1.01 (0.99-1.04) | 0.294 | ||
| Male sex | 1.14 (0.87-1.50) | 0.336 | ||
| Prior MI | 1.19 (0.84-1.68) | 0.323 | ||
| Diabetes | 1.05 (0.78-1.42) | 0.746 | ||
| NYHA III-IV | 1.92 (1.40-2.63) | <0.001 | 1.68 (1.23-2.35) | 0.001 |
| COPD | 1.12 (0.80-1.56) | 0.502 | ||
| Prior stroke | 1.02 (0.67-1.57) | 0.928 | ||
| PAD | 1.37 (0.96-.95) | 0.082 | ||
| CKD | 1.75 (1.30-2.37) | <0.001 | 1.47 (1.05-2.08) | 0.026 |
| AF | 1.48 (1.13-1.93) | 0.004 | 1.40 (1.06-1.84) | 0.016 |
| Baseline creatinine, mg/dl (1-unit increase) | 1.28 (1.09-1.50) | 0.002 | 1.12 (0.91-1.38) | 0.272 |
| Permanent PM or ICD | 1.20 (0.81-1.79) | 0.359 | ||
| STS score (1% increase) | 1.16 (1.08-1.26) | <0.001 | 1.12 (1.03-1.21) | 0.007 |
| EF (1% increase) | 0.99 (0.98-1.00) | 0.229 | ||
| Predilatation | 1.06 (0.82-1.39) | 0.648 | ||
| Postdilatation | 1.02 (0.77-1.35) | 0.874 | ||
| Valve size, mm (vs. <23) | ||||
| 23-26 | 0.99 (0.65-1.53) | 0.993 | ||
| ≥27 | 1.28 (0.86-1.94) | 0.227 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




