Submitted:
15 May 2023
Posted:
15 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
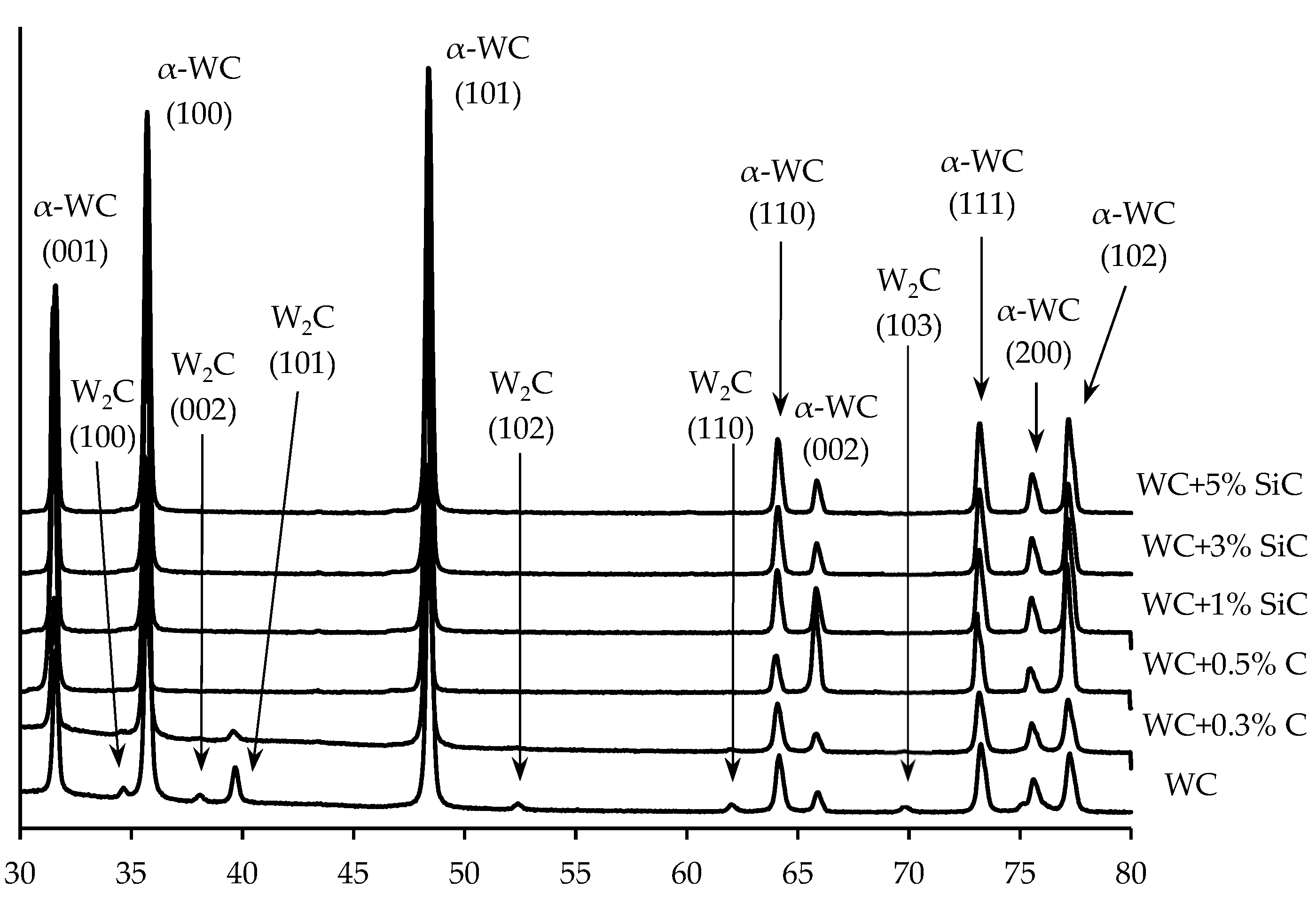
3.1. Characteristics of initial powders
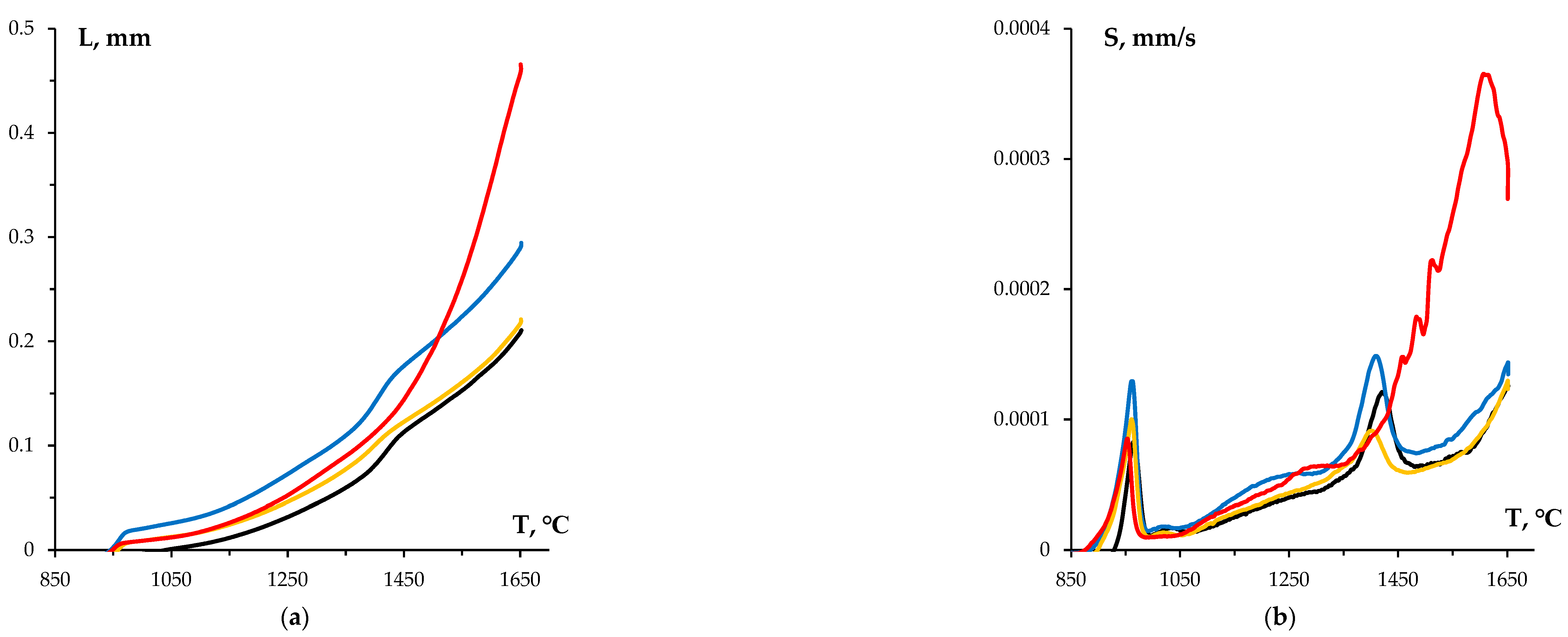
3.2. Sintering of α-WC nanopowders
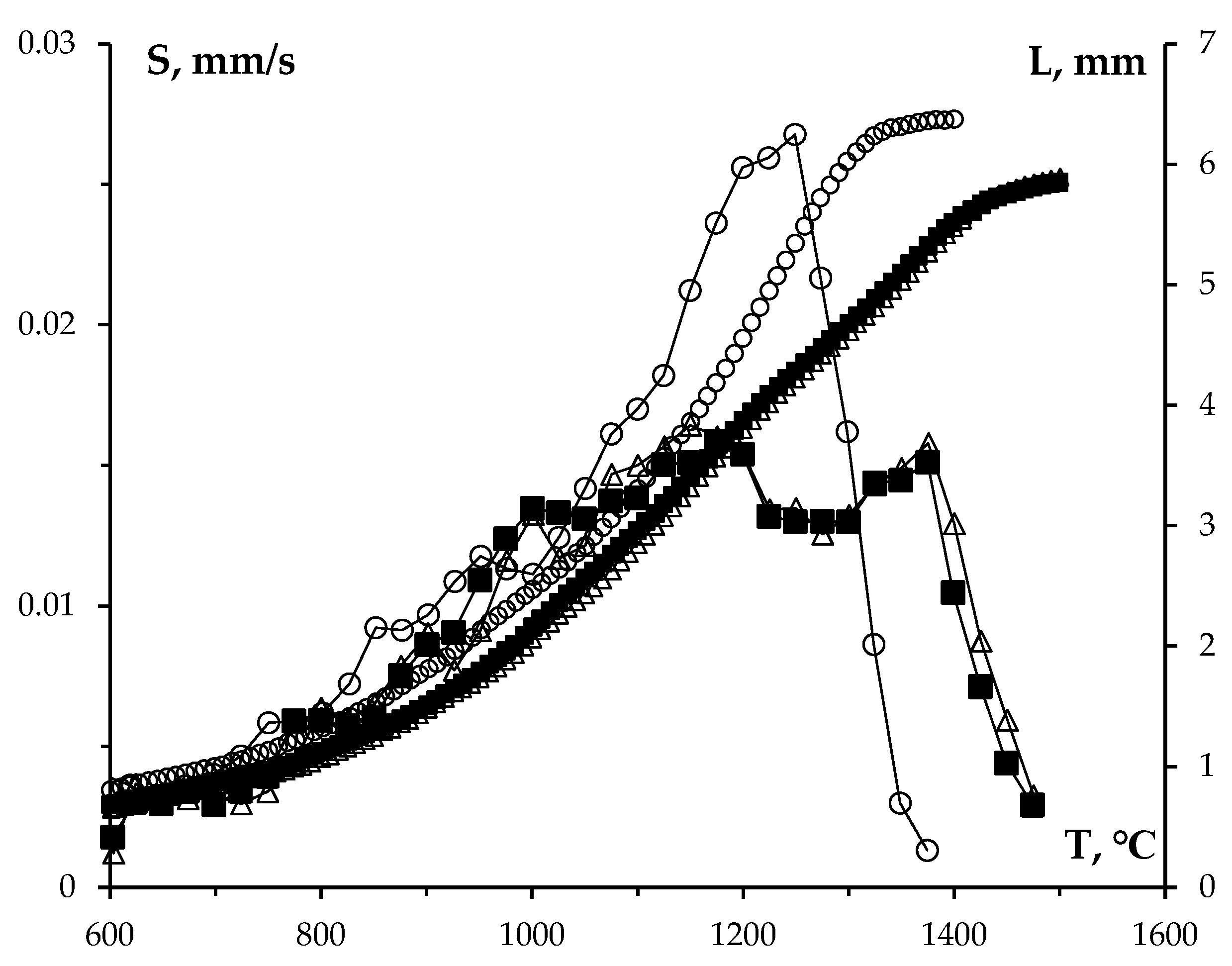
3.3. Effect of graphite on sintering kinetics of tungsten carbide nanopowders
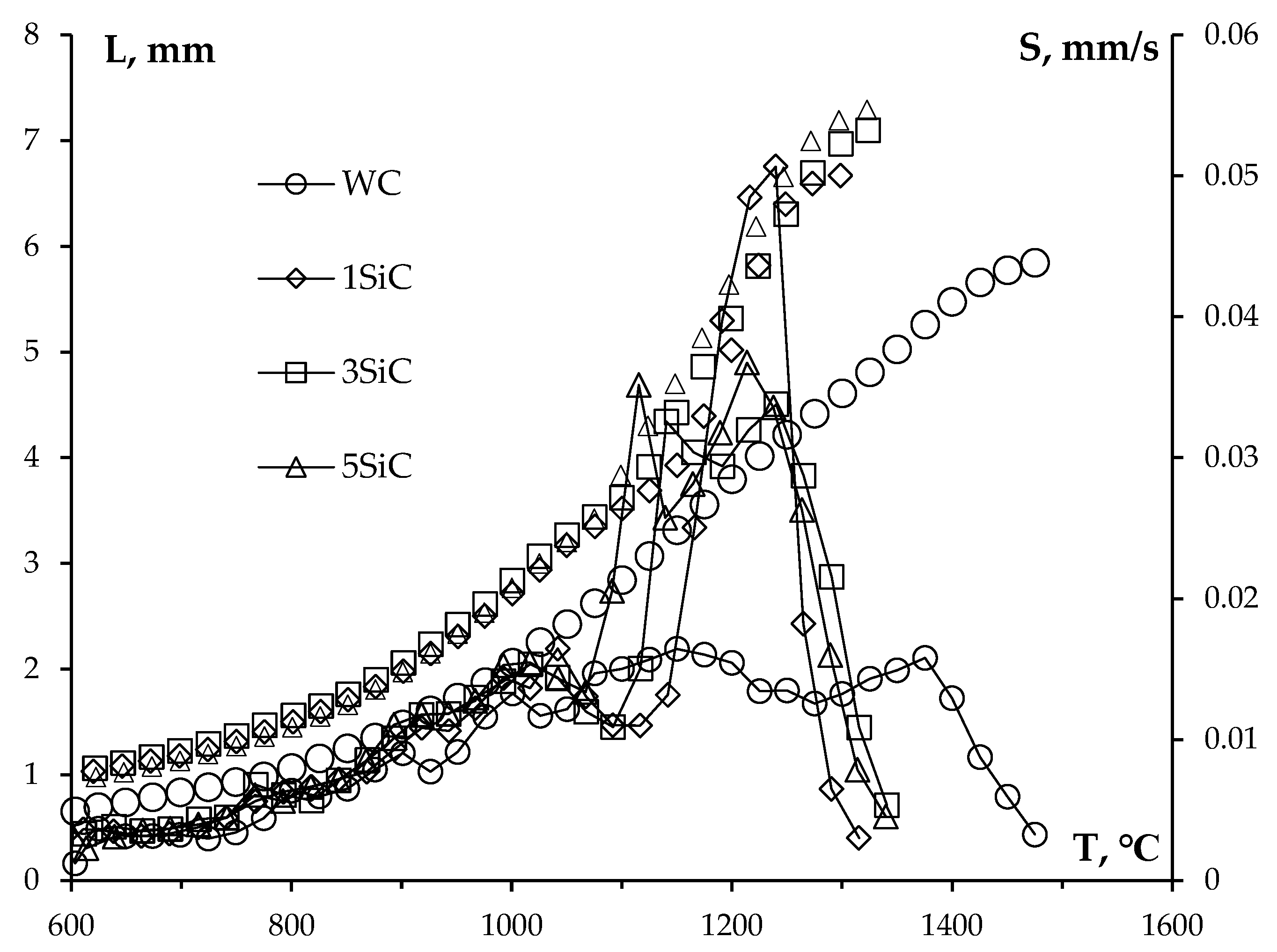
3.4. Sintering of α-WC nanopowders
4. Discussion
5. Conclusions
- 1.
- Prepressing of tungsten carbide nanopowders (350 MPa), reduction of heating rates from 100 to 10℃/min as well as degassing at 950 ℃ are shown to lead neither to an increase in density nor to changes in the microstructure of tungsten carbide samples. Fine-grained binderless tungsten carbide ceramics with relative density of ~99% were obtained by heating nanopowders at 10 ℃/min to 1520 ℃, with exposure at this temperature lasting for 3 min. The ceramic is characterized by high mechanical characteristics: hardness HV = 28.7 GPa and fracture toughness coefficient KIC = 5.0 MPa·m1/2.
- 2.
- When carbon concentration in tungsten carbide nanopowders increases, shrinkage curves during SPS shift towards lower temperatures. Tungsten carbide with an increased carbon content demonstrates an abnormal grain growth, which leads to a noticeable decrease in mechanical characteristics of the ceramics. During SPS of tungsten carbide nanopowders, the addition of 0.3% graphite is optimal since it helps to deintensify the formation of undesirable W2C particles. The introduction of 0.3% graphite into the nanopowders increases the hardness of tungsten carbide to 28.8 GPa with a simultaneous decrease in fracture toughness to KIC = 3.6 MPa·m1/2.
- 3.
- WC + SiC + 0.3% С samples with high relative density (95.4-98.1%) were obtained by SPS. These ceramics have a homogeneous UFG microstructure with a grain size of 0.1-0.2 μm and improved mechanical properties. The introduction of SiC particles into the plasma chemical tungsten carbide nanopowder reduces the shrinkage completion temperature during SPS by ~150℃ and allows to reduce the volume fraction of abnormally large grains. The compaction kinetics of WC + SiC + 0.3% C powders has a three-stage character; the powder sintering kinetics at Stage II (medium temperatures) and Stage III (high temperatures) is controlled by grain boundary and volume carbon diffusion in tungsten carbide, respectively. UFG WC + 1% SiC + 0.3% C ceramic samples have the following characteristics: hardness HV = 19.1 GPa, fracture toughness coefficient KIC = 5.2 MPa·m1/2, relative density 97.4%.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Panov, V.S.; Chuvilin, A.M. Technology and Properties of Sintered Hard Alloys and Their Products; MISIS: Moscow, Russia, 2001; p. 428. (In Russian) [Google Scholar]
- Kurlov, A.S.; Gusev, A.I. Tungsten Carbides. Structure, Properties and Application in Hardmetals; Springer: Cham, Switzerland, 2013; p. 242. [Google Scholar] [CrossRef]
- Kramer, G.S. Strength of hard alloys; Metallurgy: Moscow, Russia, 1971; p. 247. (In Russian) [Google Scholar]
- Tretyakov, V.I. Fundamentals of metal science and technology of production of sintered hard alloys; Metallurgy: Moscow, Russia, 1976; p. 527. (In Russian) [Google Scholar]
- Wachowicz, J.; Dembiczak, T.; Stradomski, G.; et al. Properties of WC-Co composites produced by the SPS method intended for cutting tools for machining of wood-based materials. Materials 2021, 14, 2618. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Zhao, J.; Huang, Z.; et al. A Review on Binderless Tungsten Carbide: Development and Application. Nanomicro Lett. 2020, 12, 13. [Google Scholar] [CrossRef] [PubMed]
- Cha, S.I.; Hong, S.H. Microstructures of binderless tungsten carbides sintered by spark plasma sintering process. Mater. Sci. Eng. A 2003, 356, 381–389. [Google Scholar] [CrossRef]
- Sivaprahasam, D.; Chandrasekar, S.B.; Sundaresan, R. Microstructure and mechanical properties of nanocrystalline WC-12Co consolidated by spark plasma sintering. Int. J. Refract. Met. Hard Mater. 2007, 25, 144–152. [Google Scholar] [CrossRef]
- Eriksson, M.; Radwan, M.; Shen, Zh. Spark plasma sintering of WC, cemented carbide and functional graded materials. Int. J. Refract. Met. Hard Mater. 2013, 36, 31–37. [Google Scholar] [CrossRef]
- Nino, A.; Nakaibayashi, Y.; Sugiyama, S.; et al. Microstructure and Mechanical Properties of WC-SiC Composites. Mater. Trans. 2011, 52, 1641–1645. [Google Scholar] [CrossRef]
- Nino, A.; Sekine, T.; Sugawara, K.; et al. Effect of Added Cr3C2 on the Microstructure and Mechanical Properties of WC–SiC Ceramics. Key Eng. Mater. 2015, 656–657, 33–38. [Google Scholar] [CrossRef]
- Nino, A.; Nakaibayashi, Y.; Sugiyama, S.; et al. Effect of Mo2C addition on the microstructures and mechanical properties of WC–SiC ceramics. Int. J. Refract. Met. Hard Mater. 2017, 64, 35–39. [Google Scholar] [CrossRef]
- Isaeva, N.V.; Blagoveshchenskii, Yu.V.; Blagoveshchenskaya, N.V.; et al. Preparation of nanopowders of carbides and hard-alloy mixtures applying low-temperature plasma. Russ. J. Non-Ferr. Met. 2014, 55, 585–591. [Google Scholar] [CrossRef]
- Chuvil’deev, V.N.; Blagoveshchenskii, Yu.V.; Nokhrin, A.V.; et al. Spark plasma sintering of tungsten carbide nanopowders obtained through DC arc plasma synthesis. J. Alloys Compd. 2017, 708, 547–561. [Google Scholar] [CrossRef]
- Tokita, M. Progress of Spark Plasma Sintering (SPS) Method, Systems, Ceramics Applications an Industrialization. Ceramics 2021, 4, 160–198. [Google Scholar] [CrossRef]
- Olevsky, E.A.; Dudina, D.V. Field-Assisted Sintering: Science and Applications; Springer: Cham, Switzerland, 2018; p. 425. [Google Scholar] [CrossRef]
- Zheng, D.; Li, X.; Li, Y.; et al. ZrO2 (3Y) toughened WC composites prepared by spark plasma sintering. J. Alloys Compd. 2013, 572, 62–67. [Google Scholar] [CrossRef]
- Wang, J.; Zuo, D.; Zhu, L.; et al. Effects and influence of Y2O3 addition on the microstructure and mechanical properties of binderless tungsten carbide fabricated by spark plasma sintering. Int. J. Refract. Met. Hard Mater. 2018, 71, 167–174. [Google Scholar] [CrossRef]
- Kumar, A.N.; Watabe, M.; Kurokawa, K. The sintering kinetics of ultrafine tungsten carbide powders. Ceram. Int. 2011, 37, 2643–2654. [Google Scholar] [CrossRef]
- Ren, X.; Peng, Z.; Wang, Ch.; et al. Effect of ZrC nano-powder addition on the microstructure and mechanical properties of binderless tungsten carbide fabricated by spark plasma sintering. Int. J. Refract. Met. Hard Mater. 2015, 48, 398–407. [Google Scholar] [CrossRef]
- Demirskyi, D.; Borodianska, H.; Agrawal, D.; et al. Peculiarities of the neck growth process during initial stage of spark-plasma, microwave and conventional sintering of WC spheres. J. Alloys Compd. 2012, 523, 1–10. [Google Scholar] [CrossRef]
- Poetschke, J.; Richter, V.; Holke, R. Influence and effectivity of VC and Cr3C2 grain growth inhibitors on sintering of binderless tungsten carbide. Int. J. Refract. Met. Hard Mater. 2012, 31, 218–223. [Google Scholar] [CrossRef]
- Munir, Z.A.; Anselmi-Tamburini, U.; Ohyanagi, M. The effect of electric field and pressure on the synthesis and consolidation materials: A review of the spark plasma sintering method. J. Mater. Sci. 2006, 41, 763–777. [Google Scholar] [CrossRef]
- Lantsev, E.A.; Malekhonova, N.V.; Chuvil’deev, V.N.; et al. Study of high-speed sintering of fine-grained hard alloys based on tungsten carbide with ultralow cobalt content: Part I. Pure tungsten carbide. Inorg. Mater. Appl. Res. 2022, 13, 761–774. [Google Scholar] [CrossRef]
- Chuvildeev, V.N.; Boldin, M.S.; Dyatlova, Ya.G.; et al. Comparative Study of Hot Pressing and High-Speed Electropulse Plasma Sintering of Al2O3 / ZrO2 / Ti(C,N) Powders. Russ. J. Inorg. Chem. 2015, 60, 987–993. [Google Scholar] [CrossRef]
- Krasovskii, P.V.; Malinovskaya, O.S.; Samokhin, A.V.; et al. XPS study of surface chemistry of tungsten carbides nanopowders produced through DC thermal plasma/hydrogen annealing process. Appl. Surf. Sci. 2015, 339, 46–54. [Google Scholar] [CrossRef]
- Krasovskii, P.V.; Blagoveshchenskii, Yu.V.; Grigorovich, K.V. Determination of oxygen in W-C-Co nanopowders. Inorg. Mater. 2008, 44, 954–959. [Google Scholar] [CrossRef]
- Rahaman, M.N. Ceramic processing and sintering, 2nd ed.; Marcel Dekker: New York, USA, 2003; p. 876. [Google Scholar] [CrossRef]
- Coble, R.L. Sintering Crystalline Solids. I. Intermediate and Final State Diffusion Models. J. Appl. Phys. 1961, 32, 787–792. [Google Scholar] [CrossRef]
- Johnson, D.L. New Method of Obtaining Volume, Grain-Boundary, and Surface Diffusion Coefficients from Sintering Data. J. Appl. Phys. 1969, 40, 192–200. [Google Scholar] [CrossRef]
- Kingery, W.D.; Berg, M. Study of the Initial Stages of Sintering Solids by Viscous Flow, Evaporation-Condensation, and Self-Diffusion. J. Appl. Phys. 1955, 26, 1205–1212. [Google Scholar] [CrossRef]
- Young, W.S.; Culter, I.B. Initial sintering with constant rates of heating. J. Am. Ceram. Soc. 1970, 53, 659–663. [Google Scholar] [CrossRef]
- Nanda Kumar, A.K.; Watabe, M.; Kurokawa, K. The sintering kinetics of ultrafine tungsten carbide powders. Ceram. Int. 2011, 37, 2643–2654. [Google Scholar] [CrossRef]
- Lantsev, E.A.; Chuvildeev, V.N.; Nokhrin, A.V.; et al. Kinetics of Spark Plasma Sintering of WC–10% Co Ultrafine-Grained Hard Alloy. Inorg. Mater. Appl. Res. 2020, 11, 586–597. [Google Scholar] [CrossRef]
- Buhsmer, C.; Crayton, P. Carbon self-diffusion in tungsten carbide. J. Mater. Sci. 1971, 6, 981–988. [Google Scholar] [CrossRef]
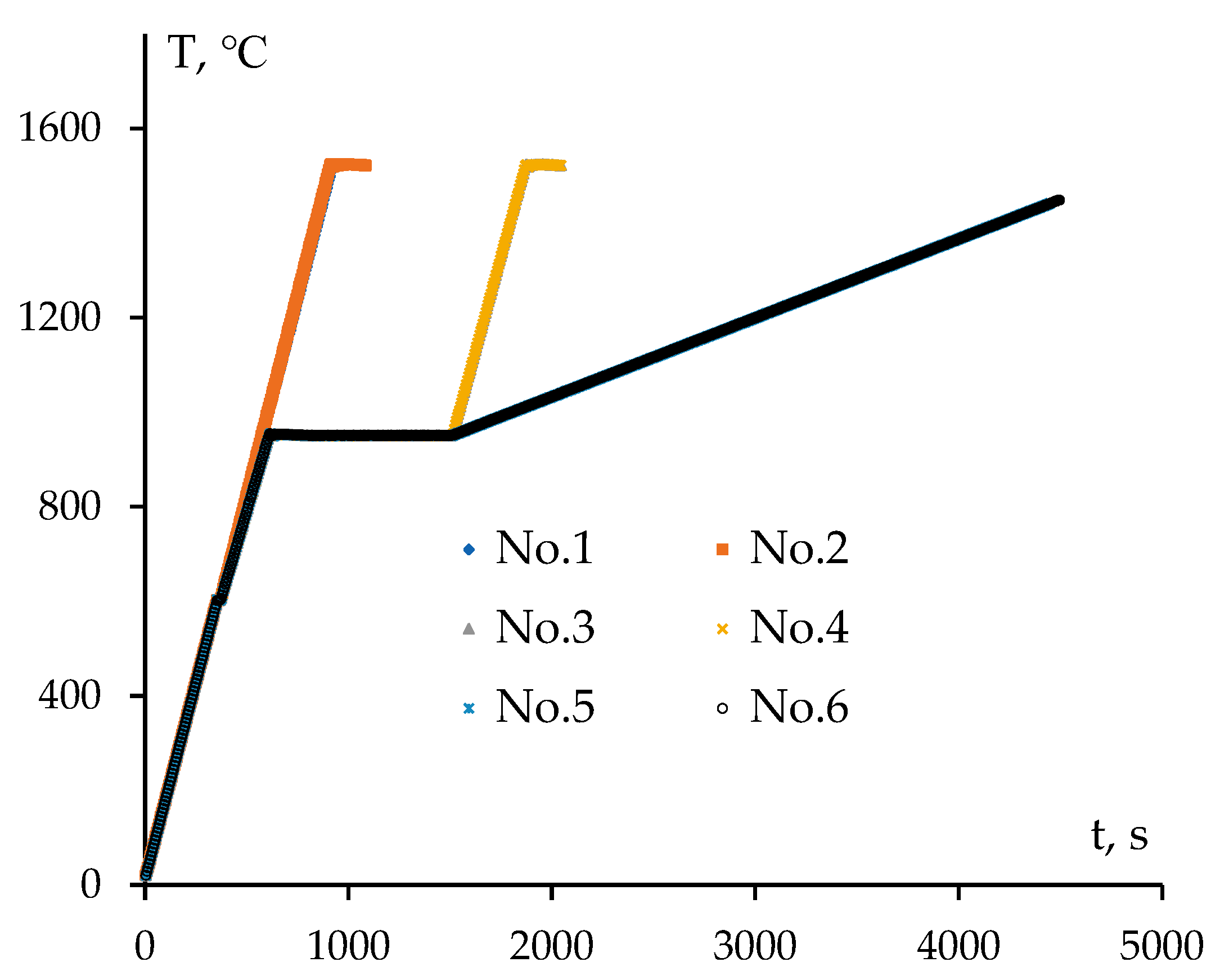

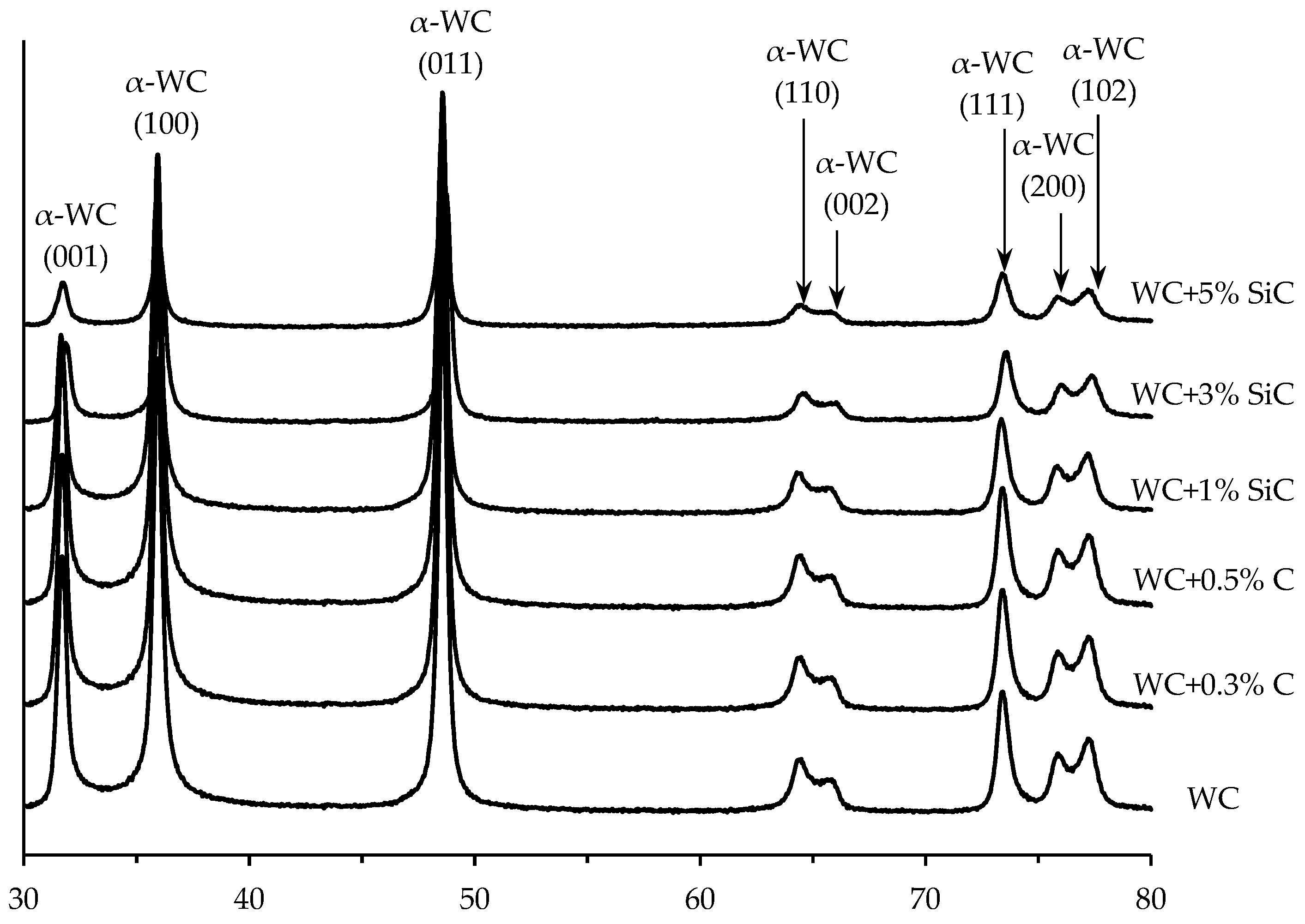
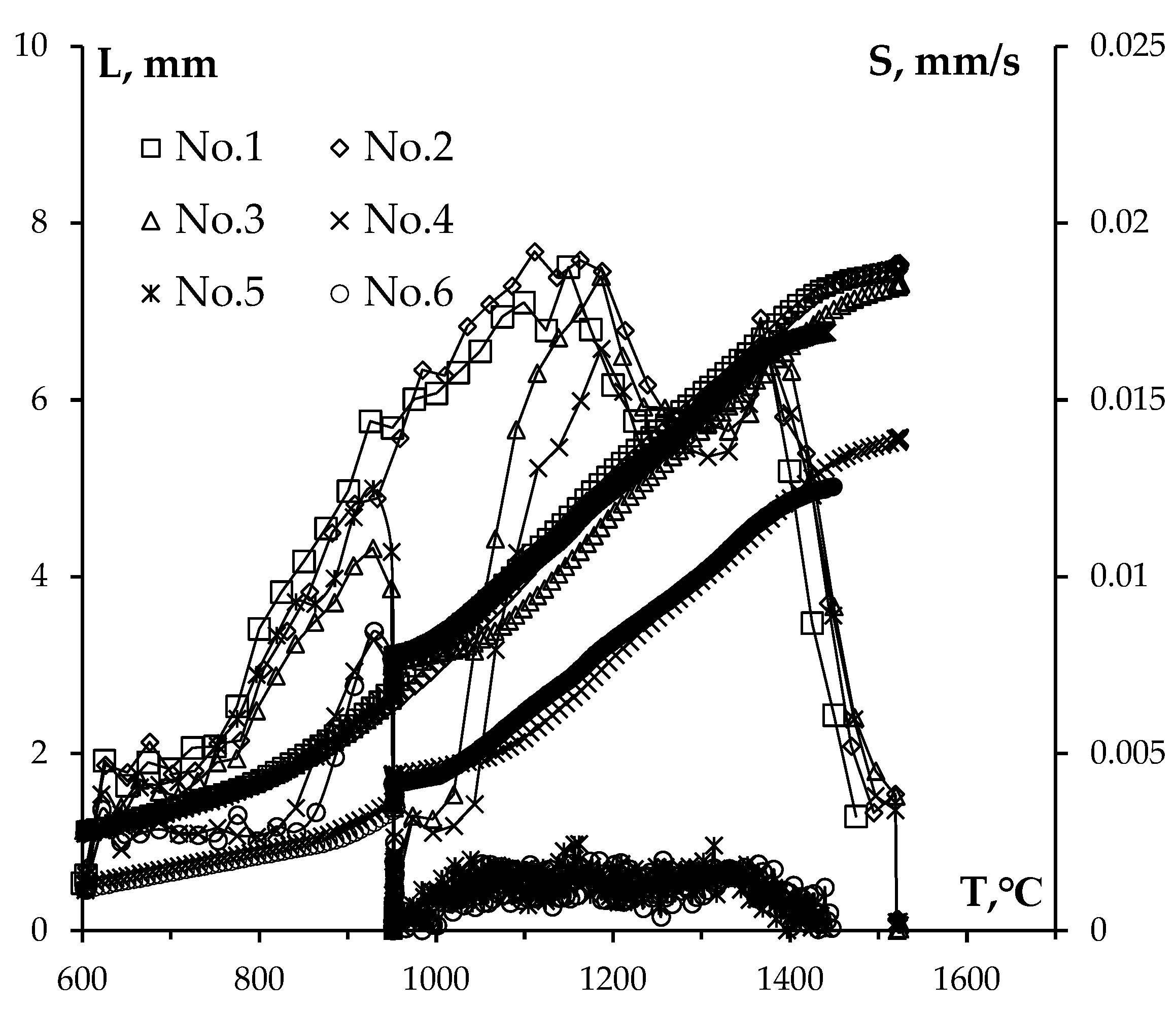

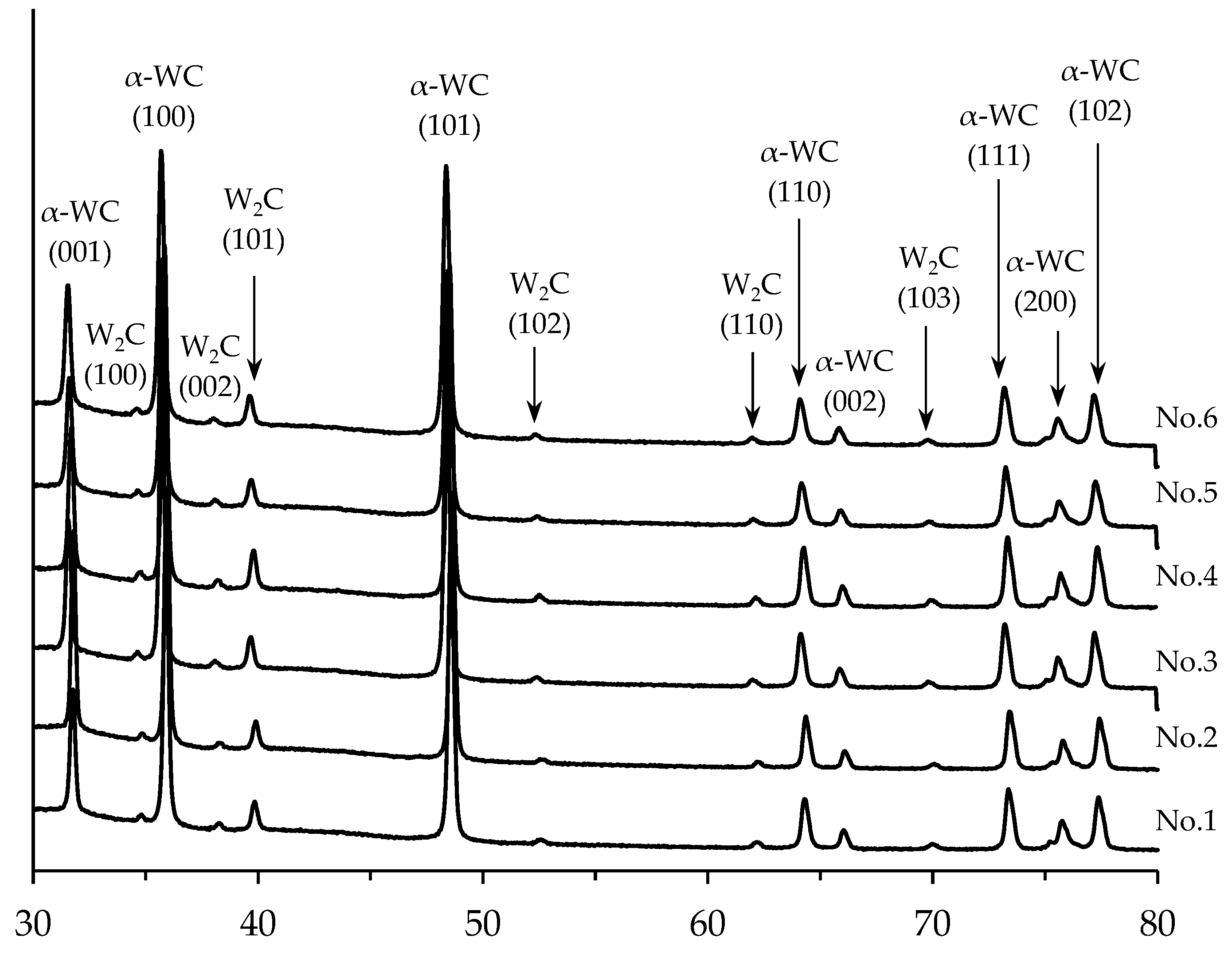


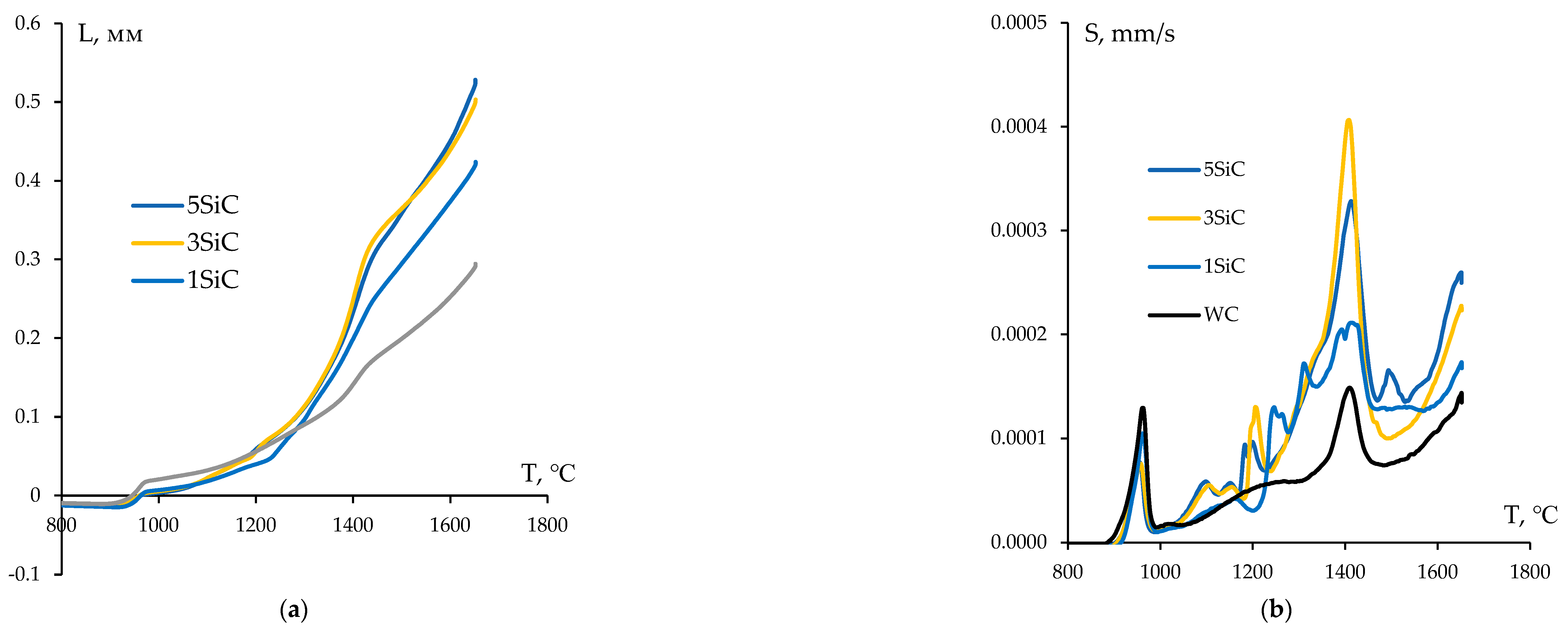


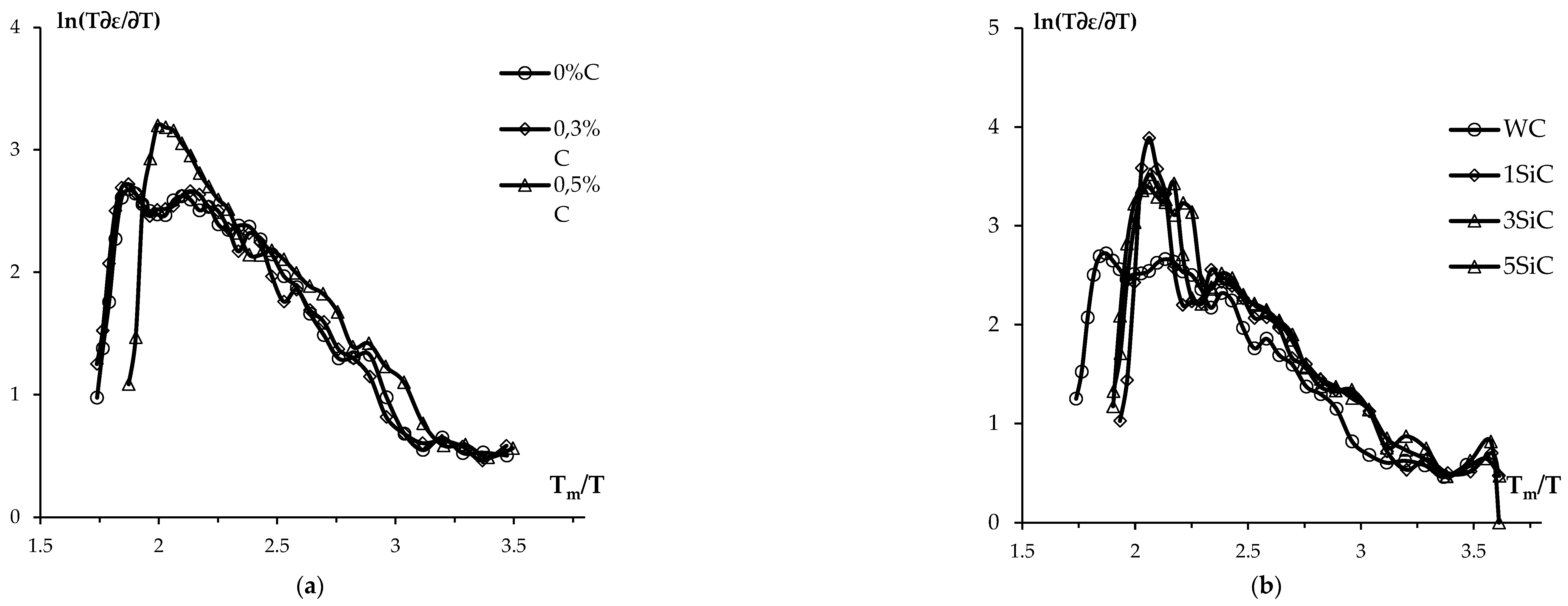
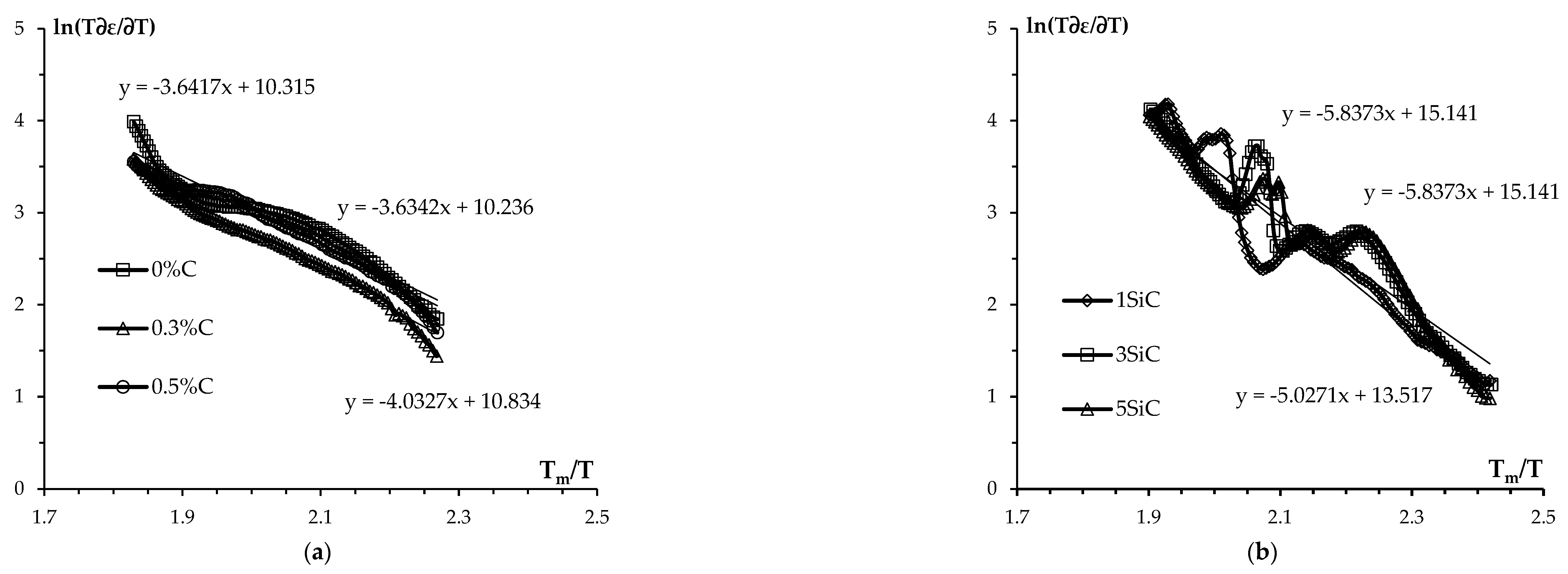
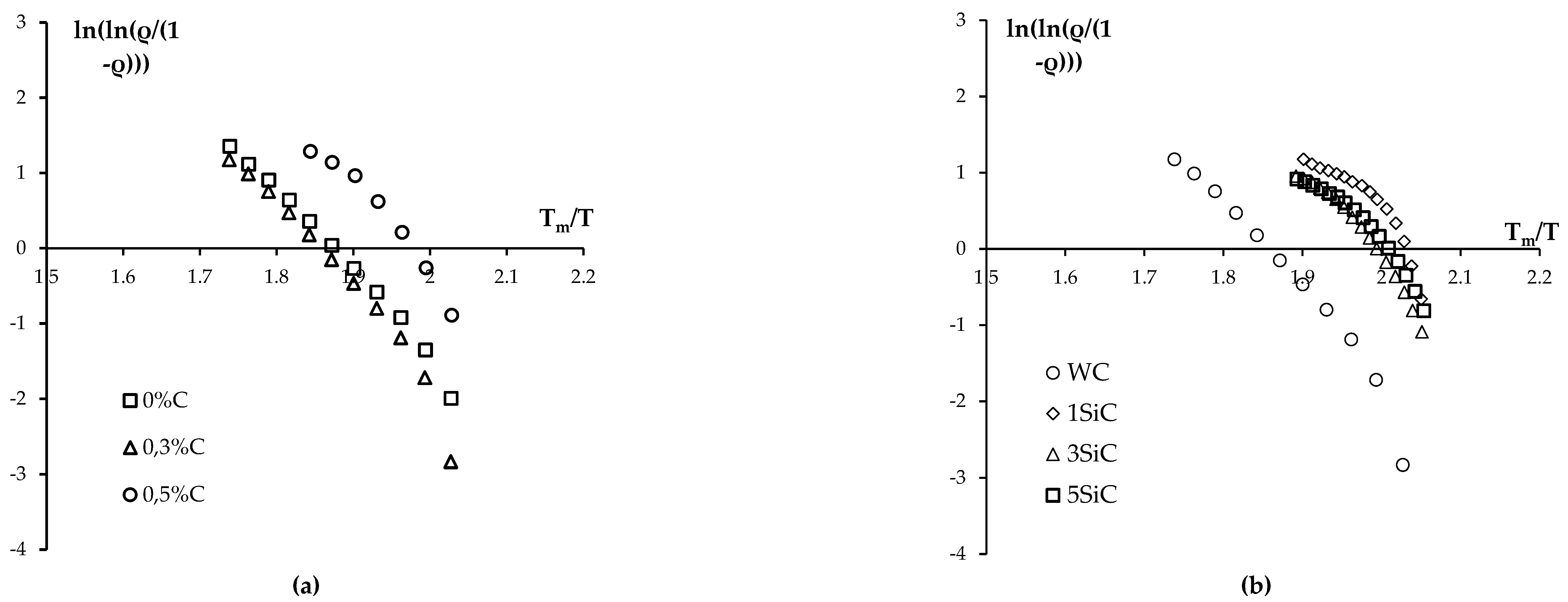
| Ceramics No. | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| Powder characteristics | ||||||
| Carbon concentration, % wt. | 6.26 | |||||
| Oxygen concentration, % wt. | 0.64 | |||||
| α-WC content, % wt. | 100 | |||||
| SPS modes | ||||||
| Stress during prepressing, MPa | 0 | 0 | 0 | 350 | 0 | 350 |
| Applied stress, MPa | 70 | 70 | 70 | 70 | 70 | 70 |
| Heating rate, ℃/min | 100 | 100 | 100 | 100 | 10 | 10 |
| Holding time at 950℃, min | 0 | 0 | 15 | 15 | 15 | 15 |
| Sintering completion temperature Ts, ℃ | 1520 | 1520 | 1520 | 1520 | 1440 | 1440 |
| Holding time at Ts, min | 0 | 3 | 3 | 3 | 0 | 0 |
| Ceramics characteristics | ||||||
| W2C content, % wt. | 7.4 | 7.0 | 7.5 | 7.8 | 7.7 | 8.3 |
| Absolute density, g/cm3 | 15.65 | 15.71 | 15.71 | 15.71 | 15.50 | 15.55 |
| Relative density, % | 98.59 | 99.06 | 98.97 | 98.98 | 97.69 | 97.93 |
| Grain size d, µm | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
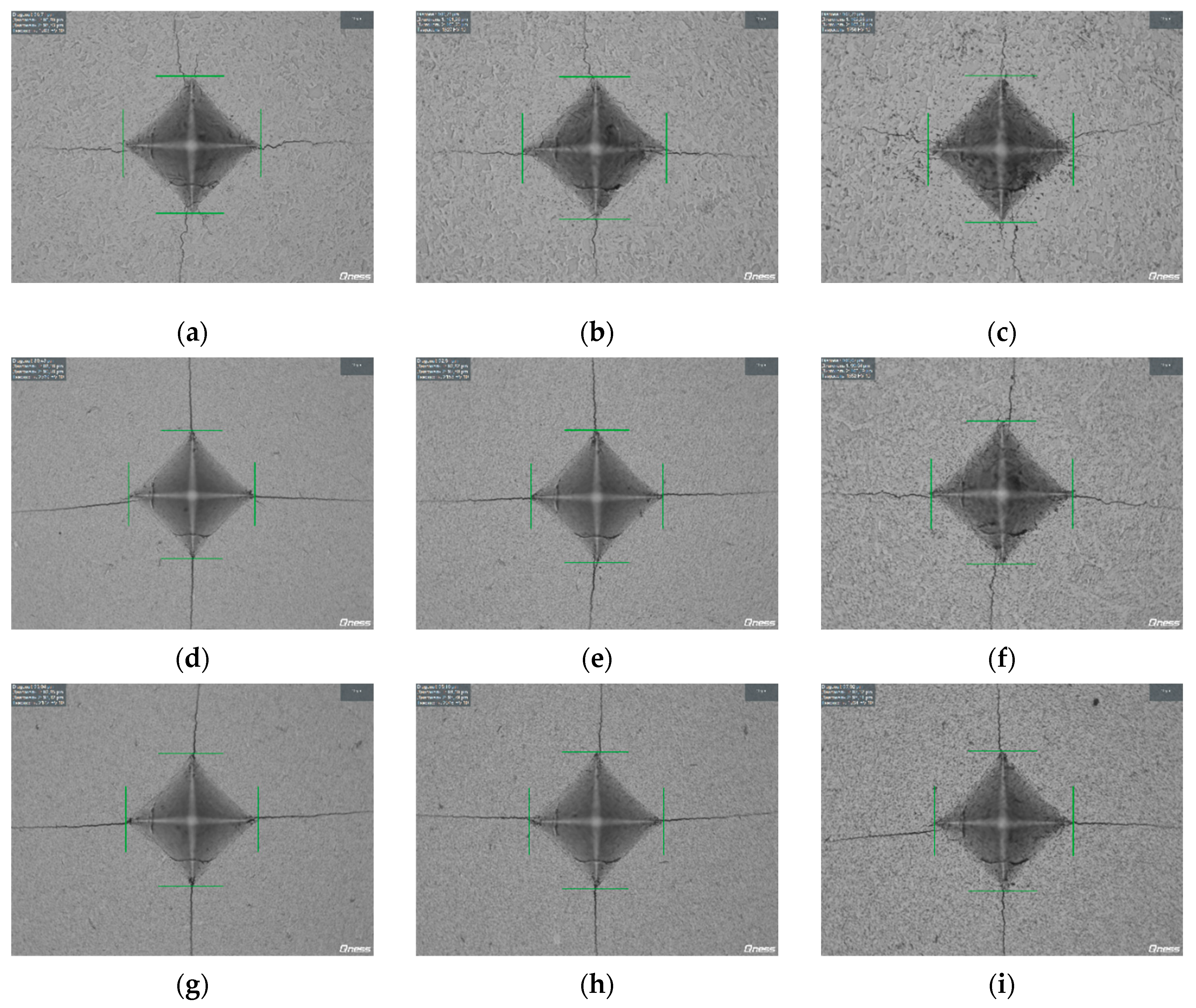
| HV, GPa (±0.2) | 28.7 | 28.0 | 27.9 | 27.4 | 28.0 | 27.4 |
| KIC, MPa·m1/2 (±0.2) | 5.0 | 4.9 | 4.4 | 4.9 | 4.2 | 4.3 |
| C, % wt. | t, min | Ts, ℃ | ρ/ρth, % (±0.05%) |
d, μm | W2C, % wt. | HV, GPa (±0.2) |
KIC, MPa·m1/2 (±0.2) | SPS activation energy | |
|---|---|---|---|---|---|---|---|---|---|
| mQsII, kTm (kJ/mol) | QsIII, kTm (kJ/mol) | ||||||||
| 0 | 0 | 1500 | 99.4 | 0.1 | 7.5 | 27.8 | 4.0 | 1.3 (33) | 11.0 (279) |
| 3 | 99.7 | 0.15 | 28.2 | 3.9 | |||||
| 30 | 99.6 | 3.0 | 25.3 | 5.0 | |||||
| 0.3 | 0 | 1500 | 98.5 | 0.1 | 2.5 | 28.8 | 3.6 | 2.0 (51) | 13.0 (330) |
| 3 | 99.1 | 0.2 | 27.6 | 4.1 | |||||
| 30 | 98.6 | 5.5 | 24.1 | 5.2 | |||||
| 0.5 | 0 | 1400 | 97.7 | 22 | 0 | 12.9 | 9.0 | 2.3 (58) | 12.0 (304) |
| 3 | 97.7 | 85 | 12.3 | 8. | |||||
| 30 | 97.9 | 200 | 11.7 | 8.5 | |||||
| SiC, % wt. | t, min | Ts, ℃ | ρ/ρth, % (±0.05%) |
d, μm | W2C, % wt. | HV, GPa (±0.2) | KIC, MPa·m1/2 (±0.2) | SPS activation energy | |
|---|---|---|---|---|---|---|---|---|---|
| mQsII, kTm (kJ/mol) | QsIII, kTm (kJ/mol) | ||||||||
| 1 | 0 | 1350 | 97.4 | 7 | 0 | 19.2 | 5.9 | 2.2 (56) | 10.0 (250) |
| 3 | 97.3 | 10 | 18.1 | 6.1 | |||||
| 30 | 97.7 | 16 | 17.3 | 6.2 | |||||
| 3 | 0 | 1350 | 95.5 | 0.5 | 0 | 22.6 | 3.8 | 2.2 (56) | 12.0 (304) |
| 3 | 95.4 | 1.2 | 21.1 | 4.1 | |||||
| 30 | 96.7 | 12 | 18.3 | 4.4 | |||||
| 5 | 0 | 1350 | 95.1 | 0.3 | 0 | 21.5 | 3.9 | 2.15 (55) | 10.0 (250) |
| 3 | 96.1 | 1 | 20.1 | 4 | |||||
| 30 | 95.8 | 3.2 | 18.7 | 4.4 | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




