2. Materials and Methods
2.1. Standards and Reagents
Promethazine Hydrochloride standard, purity of 99.5%, was purchased from the China National Institute for Food and Drug Control, China. Promethazine-d6 Hydrochloride standard with a chemical purity of 98% and an isotopic purity of 99.5%, Promethazine Sulfoxide (PMZSO) standard with a purity of 96%, Monodesmethyl-Promethazine Hydrochloride standard with a purity of 97% were all purchased from Toronto Research Chemicals, Canada.
HPLC grade acetonitrile (ACN) and methanol (MeOH) were purchased from Thermo Fisher Scientific, USA. HPLC grade formic acid was purchased from Shanghai Macklin Biochemical Co., Ltd., China. Analytical grade n-hexane was purchased from Tianjin Damao Chemical Reagent Factory, China. Ultrapure water was collected from a Milli-Q water purification system (Millipore, Billerica, MA, USA).
2.2. Instruments and Equipment
High performance liquid chromatography-tandem mass spectrometer (Shimadzu LC-30AD 220V liquid chromatograph, Japan), with ESI5500 tandem quadrupole mass spectrometer equipped with Turbo Ionspray electrospray interface, Analyst 1.6.3 software workstation (Applied Biosystems, ABI, Corporation, USA).
Chromatographic column was a Symmetry C18 (100 mm × 2.1 mm i.d., 3.5 µm), Waters Corporation, USA. Rotary evaporator (N-1300V-W, Tokyo Rikakikai Co., Ltd., Japan), high-speed desktop centrifuge (LEGEND MACH 1.6R, Thermo Corporation, USA), vortex mixer (Vortex 3000, VIGEN TECH Co., Ltd., Germany). Nylon syringe filters, 13 mm, 0.22µm disposable, purchased from Shanghai Ampu Company, China.
2.3. Preparation of Solution
Standard stock solution: The promethazine hydrochloride standard (calculated as PMZ, C17H20N2S), PMZSO standard (calculated as PMZSO, C17H20N2OS), and Monodesmethyl-Promethazine hydrochloride standard (calculated as Nor1PMZ, C16H18N2S) were weighed accurately and put into 50 mL volumetric flasks respectively, dissolved in HPLC grade ACN and made up to volume, obtaining the stock solution of each standard at 1000 μg/mL. All standard stock solutions were stored at -22°C.
PMZ-d6 standard stock solution: 10 mg of promethazine-d6 hydrochloride standard (C17H15D6ClN2S) was transferred to a 10 mL volumetric flask, dissolved in HPLC grade MeOH and made up to volume, obtaining the PMZ-d6 stock solution at 1000 μg/mL, sealed and stored at -22°C.
Mixed standard working solution: 1 mL each of PMZ, PMZSO, and Nor1PMZ standard stock solutions were took, mixed and diluted with HPLC grade ACN to obtain series working solutions at concentrations of 2.5 μg/mL, 1 μg/mL, 0.5 μg/mL, 0.25 μg/mL, 0.05 μg/mL, 0.025 μg/mL, 0.005 μg/mL, and 0.0025 μg/mL. The mixed standard working solutions were stored at 4°C.
PMZ-d6 working solution: An appropriate amount of PMZ-d6 standard stock solution was diluted with HPLC grade ACN to obtain an internal standard solution at a concentration of 1 μg/mL, sealed and stored at 4°C.
Acetonitrile saturated n-hexane: An appropriate amount of analytical grade n-hexane was added an appropriate amount of ACN, mixed well and allowed to stand until layered.
0.1% Formic Acid Solution in Water: 1.00 mL of HPLC grade formic acid was transferred, diluted to 1 L volume with ultrapure water and mix well.
0.1% Formic Acid in Acetonitrile: Transferred 1.00 mL of HPLC grade formic acid and added ACN to 1 L.
0.1% Formic Acid Solution in Water-Acetonitrile (80:20, v/v): Transferred 200 mL of HPLC grade ACN to a 1 L volumetric cylinder, added approximately 800 mL of 0.1% formic acid solution in water to 1 L, and mix well.
2.4. Chromatography and Mass Spectrometry Parameters
Symmetry C
18 (100 mm×2.1 mm i.d., 3.5 µm) was employed. Mobile phase composed of 0.1% phase A (0.1% formic acid solution in water) and phase B (acetonitrile, ACN). Flow rate 0.3 mL/min, gradient elution procedure as shown in
Table 1.
Mass spectrometer operated in positive ion mode (ESI+), mass spectrometric scan mode was multiple reaction monitoring (MRM). The main operating parameters were shown in
Table 2.
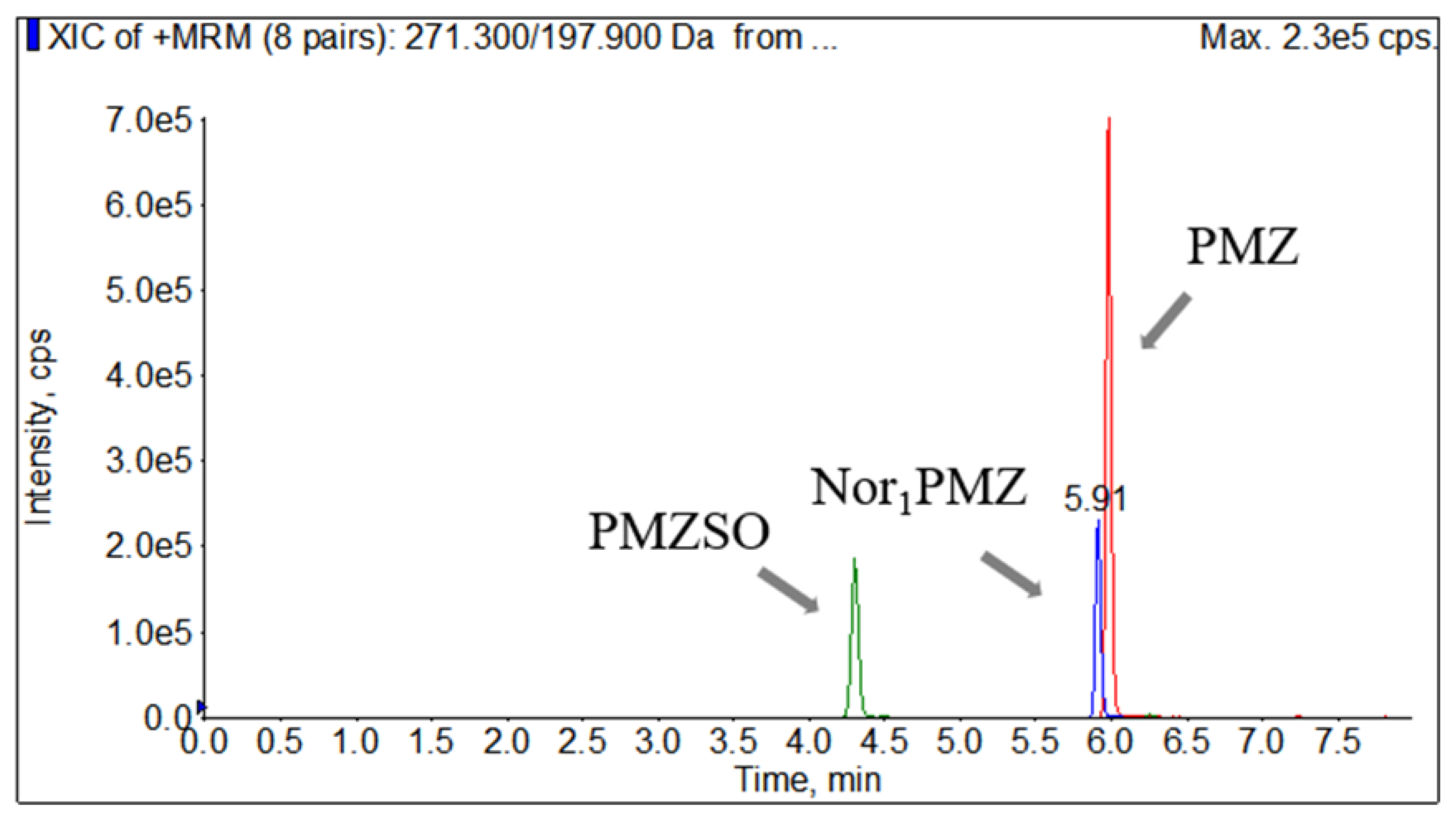
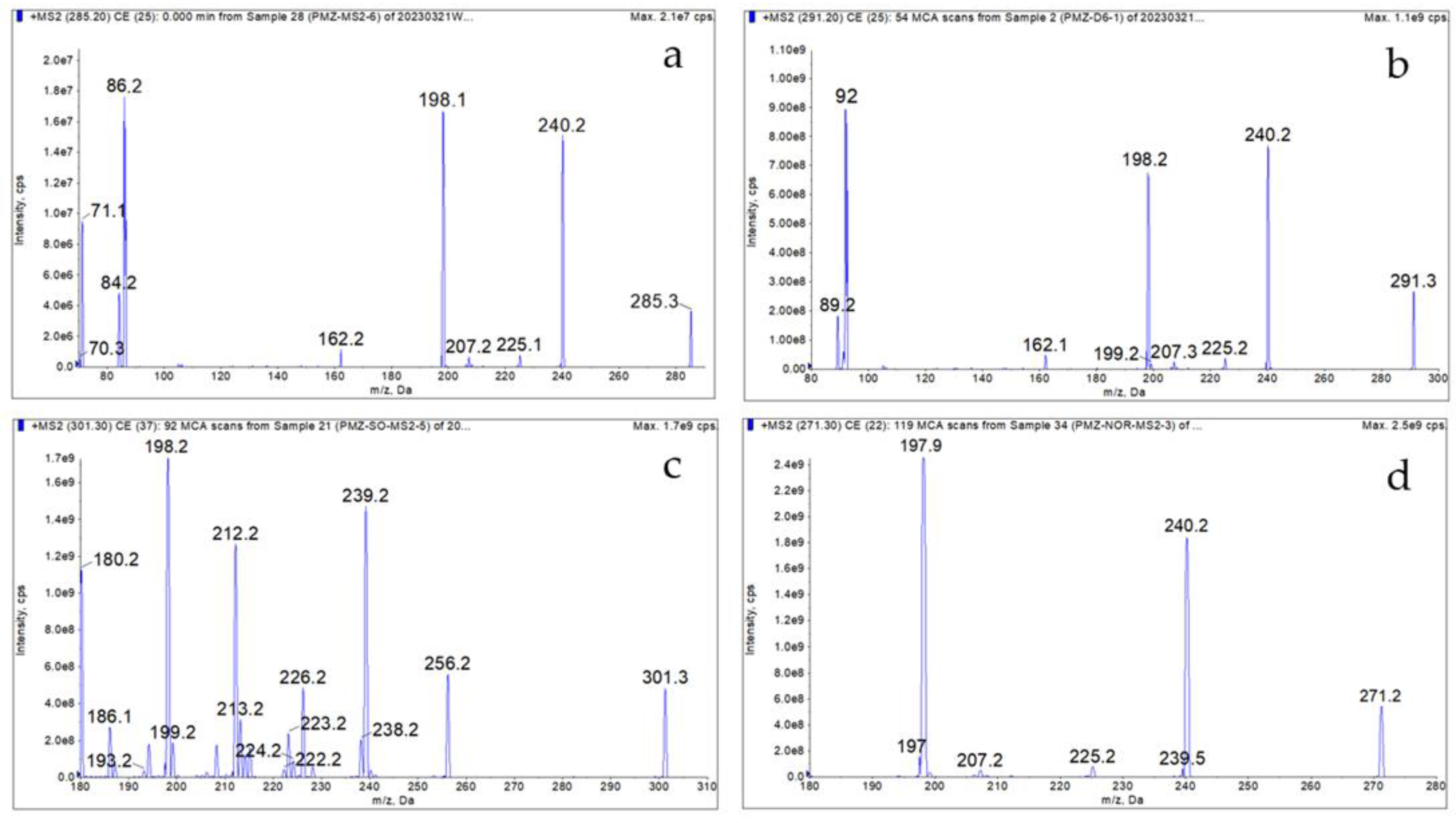
Standard stock solution from "2.3" was diluted respectively to 1 μg/mL with ACN, directly injected into the spectrometer for mass spectrometric optimization. The molecular ion peaks of target analytes and internal standard were determined by full-scan mass spectrometry in positive ion mode, where the m/z of PMZ was 285.2, PMZSO was 301.3, Nor
1PMZ was 271.3, and PMZ-d6 was 291.3. Each precursor ion was MS/MS scanned to determine and assess monitored ions for each analyte as quantitative ion and qualitative ion. Operation parameters of each ion was optimized using mass spectrometric scan mode of multiple reaction monitoring (MRM). Finally, the m/z of 86.2 and 198.1 were determined as the quantitative and qualitative ions for PMZ, 198.2 and 239.1 for PMZSO, 197.3 and 240.3 for Nor
1PMZ, and 92 and 240.3 for PMZ-d6. The qualitative and quantitative ion pairs, declustering potential, and collision energy for each target compound were shown in
Table 3. Quantification of PMZ and Nor
1PMZ were used PMZ-d6 as the internal standard, while quantification of PMZSO was used external standard method.
2.5. Sample Preparation
Approximately 500 g of muscle, liver, and kidney sample was removed connective tissue, blood vessels, and fat, then cut into pieces and chopped into uniform slurry using a homogenizer. Weighed 5.0 g±0.1 g of sample slurry in a 50 mL centrifuge tube, added 100 μL of PMZ-d6 internal standard working solution (1 μg/mL), vortex for 30 s and let stand for 30 min. Added 10 mL of 0.1% formic acid in acetonitrile, vortex for 1 min to mix and shook for 10 min using platform shaker at 100% speed, then centrifuged at 10000 rpm for 10 min. The supernatant was transferred to a pear-shaped bottle. Another 10 mL of 0.1% formic acid acetonitrile was added to the residue in the centrifuge tube, repeated the above operation to perform the second extraction. The two extraction liquids were collected in a pear-shaped bottle for purification and concentration.
Approximately 500 g of subcutaneous fat from pig was removed muscle and connective tissue, and homogenized with homogenizer to obtain a uniform slurry. Weighed 5.0 g±0.1 g of fat slurry in a 50 mL centrifuge tube and added 100 μL of PMZ-d6 internal standard working solution (1 μg/mL), vortex for 30 s and let stand for 30 min. Added 10 mL of acetonitrile saturated n-hexane, vortex until the fat is completely dissolved, let stand for 30 min. Added 10 mL of 0.1% formic acid in acetonitrile, vortex for 1 min, and shook for 10 min using platform shaker at 100% speed. After centrifuged at 10000 rpm for 10 min, the upper hexane layer was discarded. The lower extraction liquid was transferred to a new 50 mL centrifuge tube for purification.
The extraction liquid of muscle, liver, kidney and fat was added 10 mL of acetonitrile saturated n-hexane and vortex for about 30 s to mix, let stand for layering. The upper hexane layer was discarded. The lower extraction liquid was added 10 mL of anhydrous ethanol, and removed solvent using a rotary evaporator at 45℃. Then 5 mL of 0.1% formic acid water-acetonitrile was added and vortex for 30 s to dissolve the residue completely. Added 5 mL of n-hexane-saturated acetonitrile to the solution and vortex to mix, let stand for layering. Approximately 1 mL of the lower solution was transferred to a 1.5 mL centrifuge tube and centrifuged at 14000 r/min, 0℃ for 10 min. The clarified middle liquid was filtered with 0.22 μm nylon syringe filters, sealed in autosampler vial and stored at 4℃ for analysis.
2.6. Limit of detection and limit of quantification
Control blank tissue sample homogenate 5 g ± 0.1 g was added 100 μL of 1 μg/mL PMZ-d6 internal standard working solution and 100 μL of mixed standard working solution with concentrations of 0.05 μg/kg, 0.1 μg/kg, 0.5 μg/kg, and 1 μg/kg to prepare quality control samples. After processing by the method described in "2.5", detection was carried out according to the conditions in "2.4", and data analysis was performed using the Analyst 1.6.3 software workstation. The concentration of the sample with a signal-to-noise ratio (S/N) ≥ 3 was taken as the limit of detection (LOD), and the concentration of the sample with S/N ≥ 10 was taken as the limit of quantification (LOQ).
2.7. Calibration curve and linearity
Homogenized blank tissue samples of 5 g ± 0.1 g were taken, and 100 μL of PMZ-d6 internal standard working solution at 1 μg/mL and 100 μL of mixed standard working solution from "2.3" were added separately to obtain spiked samples with PMZ and PMZSO concentrations ranging from 0.1 μg/kg to 50 μg/kg and Nor1PMZ concentration ranging from 0.5 μg/kg to 50 μg/kg. The samples were processed using the method described in "2.5" and analyzed under the conditions specified in "2.4" using the Analyst 1.6.2 software workstation. The calibration curve and correlation coefficient (r) were obtained using a weighted least-squares method with the ratio of the concentration of PMZ, Nor1PMZ, and PMZ-d6 as the abscissa and the peak area ratio of the quantitative ion pairs of PMZ, Nor1PMZ, and PMZ-d6 as the ordinate, with the weight chosen as 1/X2. The calibration curve and correlation coefficient of PMZSO were obtained using a weighted least-squares method with the concentration of PMZSO as the abscissa and the peak area of the PMZSO quantitative ion pair as the ordinate, with the weight chosen as 1/X2. The experiment was repeated in triplicate.
2.8. Recovery and Precision
Homogenized blank tissue samples of 5 g ± 0.1 g were taken, then spiked 100μL of low, medium, and high concentration mixed standard working solutions to obtain spiked tissue samples with compound contents of 0.5 μg/kg, 5 μg/kg, and 50 μg/kg, respectively. Set up 6 parallels for each concentration. Follow the method described in "2.5" and test under the conditions in "2.4". Analyze the data using Analyst 1.6.2 software workstation, and calculate the recovery and relative standard deviation (RSD) of sample determination values. RSD is used to indicate precision. Repeat the experiment for three batches to test intra-day precision.
2.9. Investigation of matrix effects
Homogenized blank tissue samples of 5 g ± 0.1 g were taken, processed using the method described in "2.5" to obtain a sample matrix solution. Then 100 μL of mixed standard working solution from "2.3" was added separately into the sample matrix solution to prepare matrix-matched samples with concentrations of 0.1 μg/kg, 0.5 μg/kg, 1 μg/kg, 5 μg/kg, 10 μg/kg, 20 μg/kg, and 50 μg/kg. The samples were processed and analyzed, and the curve of the matrix-matched samples was obtained. The experiment was repeated in triplicate.
The mixed standard working solution from "2.3" was diluted with methanol to concentrations of 0.1 μg/L, 0.5 μg/L, 1 μg/L, 5 μg/L, 10 μg/L, 20 μg/L, and 50 μg/L, and analyzed under the conditions specified in "2.4", and the curve of the standard working solution was obtained.
Matrix effect refers to the influence of the sample matrix on the determination of the target compound. It is evaluated by comparing the slope of the curve of matrix-matched samples with that of the standard working solution of the same concentration. If ME>0, it indicates matrix enhancement, while if ME<0, it indicates matrix suppression. When 0≤|ME|≤20%, the matrix has low signal interference and can be ignored. When 20%<|ME|<50%, it indicates moderate matrix interference. When |ME|≥50%, it indicates strong matrix interference.
The formula for calculating matrix effect is as follows:
ME: Matrix Effect
Sm: Slope of the curve of matrix-matched samples
Ss: Slope of the curve of standard working solution
2.10. Stability Test
Homogenized 5g±0.1g of blank tissue sample was mixed with a low or high concentration of mixed standard working solution to obtain QC sample. The QC samples with target drug concentrations of 0.5 µg/kg and 50 µg/kg were processed according to the method described in section "2.5". The stability of the samples was evaluated after 30 days of storage at -22℃, 1 week of storage at 4℃, freeze-thaw cycle for 3 times, and 24 hours of exposure to room temperature and light. Three replicates were made for each concentration. The actual measured concentration was compared with the theoretical added concentration, and the deviation between the mean value of each concentration and the theoretical concentration was calculated to observe whether the RSD was within 15%.