Submitted:
24 April 2023
Posted:
24 April 2023
Read the latest preprint version here
Abstract
Keywords:
1. Introduction & Background
2. Earlier Studies of Zanubrutinib in CLL
- Consistent with the favorable oral bioavailability observed in preclinical studies, oral administration of zanu achieves therapeutic plasma drug concentrations using the recommended phase II dose of 160 mg twice daily, with maintenance of drug levels above the IC50 required for full occupancy of the BTK binding site [25,27].
- Zanu is less prone to pharmacological interactions from food, drug-drug interactions with strong or moderate CYP3A inhibitors, and proton pump inhibitors [PPIs] leading to more consistent, sustained therapeutic exposures and improved dosing convenience. In addition, the clinical use of zanu is less sensitive than ibrutinib to impairments of liver function [25,27].
3. Phase III Clinical Trials of Zanubrutinib in CLL
3.1. Sequoia Trial
- Cohort 1 comprised 479 patients without del(17p) who were randomized 1:1 and either assigned to receive zanu (n=241)(until disease progression or unacceptable toxicity) or bendamustine and rituximab (BR) (n=238) for up to 6 cycles.
- Cohort 2 comprised 110 patients with del(17p) who were assigned to receive zanu as monotherapy as it was deemed unethical to randomize patients with del(17p) to BR.
- Cohort 3 comprised 80 patients with del(17p) or TP53 aberrations who were assigned to receive zanu in combination with venetoclax (ZV). This cohort was opened when Cohort 2 was fully enrolled to provide non-randomized treatment for patients with del(17p).
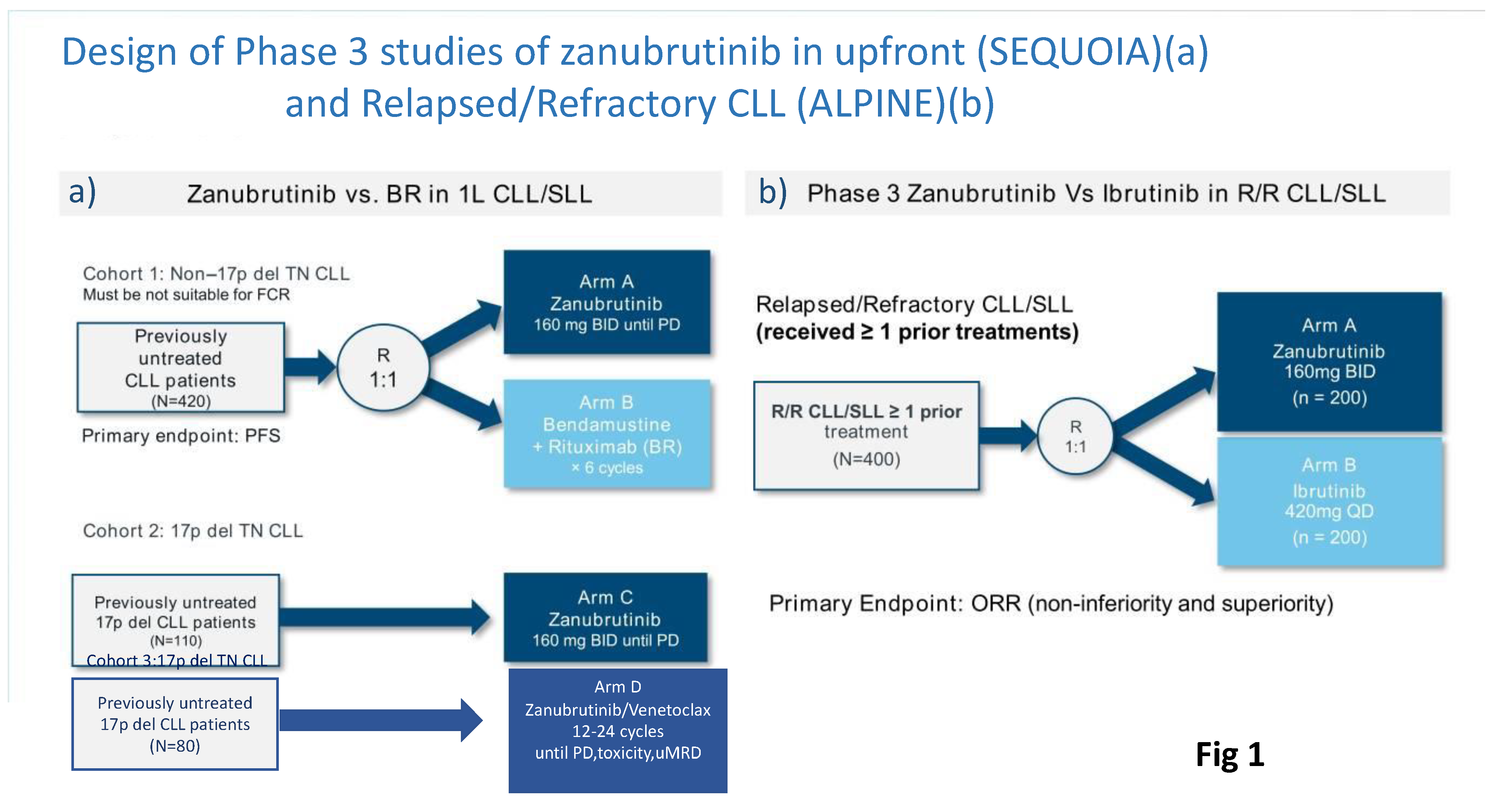
3.2. ALPINE Trial
4. Specific Aspects of Zanubrutib Therapy in CLL
4.1. Is It Possible to Simplify the Zanubrutinib Treatment Schedule?
4.2. Zanubrutinib after Discontinuation of a Covalent BTKi Because of Toxicity
4.3. Combining Zanubrutinib with Monoclonal Antibodies or Anti-BCL2 Agents
4.4. Three Drug Zanubrutinib Combinations
4.5. Mechanisms of Zanubrutinib Resistance in CLL
4.6. Zanubrutinib in Patients at Risk of Cardiovascular Complications

5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Byrd, J.C.; Furman, R.R.; Coutre, S.E.; Flinn, I.W.; Burger, J.A.; Blum, K.A.; Grant, B.; Sharman, J.P.; Coleman, M.; Wierda, W.G.; et al. Targeting BTK with Ibrutinib in Relapsed Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2013, 369, 32–42. [Google Scholar] [CrossRef]
- Byrd JC, Brown JR, O’Brien S, et al. RESONATE Investigators. Ibrutinib versus ofatumumab in previously treated chronic lymphoid leukemia. N Engl J Med. 2: 2014;371(3), 2014. [CrossRef]
- Burger, J.A.; Tedeschi, A.; Barr, P.M.; Robak, T.; Owen, C.; Ghia, P.; Bairey, O.; Hillmen, P.; Bartlett, N.L.; Li, J.; et al. Ibrutinib as Initial Therapy for Patients with Chronic Lymphocytic Leukemia. New Engl. J. Med. 2015, 373, 2425–2437. [Google Scholar] [CrossRef] [PubMed]
- Shanafelt TD, Wang XV, Kay NE, et al. Ibrutinib- Rituximab or Chemoimmunotherapy for Chronic Lymphocytic Leukemia. N Engl J Med. 2019 Aug 1;381(5):432-443. [CrossRef]
- Woyach JA, Ruppert AS, Heerema NA, et al. Ibrutinib Regimens versus Chemoimmunotherapy in Older Patients with Untreated CLL. N Engl J Med. 2018 Dec 27;379(26):2517-2528. [CrossRef]
- Moreno C, Greil R, Demirkan F, et al. Ibrutinib plus obinutuzumab in first-line treatment of chronic lymphocytic leukaemia (iLLUMINATE): a multicenter, randomized, open-label, phase 3 trial. Lancet Oncol. 2019 Jan;20(1):43-56.
- Munir T, Brown JR, O’Brien S,et al Final analysis from RESONATE: Up to six years of follow-up on ibrutinib in patients with previously treated chronic lymphocytic leukemia or small lymphocytic lymphoma. Am J Hematol 2019 Dec;94(12):1353-1363. [CrossRef]
- Barr PM, Owen C, Robak T,et al. Up to 8-year follow-up from RESONATE-2: first-line ibrutinib treatment for patients with chronic lymphocytic leukemia. Blood Adv. 2022 Oct 25;6(20):5641-5654. [CrossRef]
- Molica, S.; Matutes, E.; Tam, C.; Polliack, A. Ibrutinib in the treatment of chronic lymphocytic leukemia: 5 years on. Hematol. Oncol. 2019, 38, 129–136. [Google Scholar] [CrossRef]
- Lipsky, A.; Lamanna, N. Managing toxicities of Bruton tyrosine kinase inhibitors. Hematology 2020, 2020, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Byrd JC, Hillmen P, Ghia P, Kater AP, et al. Acalabrutinib Versus Ibrutinib in Previously Treated Chronic Lymphocytic Leukemia: Results of the First Randomized Phase III Trial. J Clin Oncol. 2021 Nov 1;39(31):3441-3452. [CrossRef]
- Project Orbis: FDA approves acalabrutinib for CLL and SLL. FDA website. Posted , 2019. https://bit.ly/35idpJM. Accessed , 2019. 21 November.
- Calquence approved in the EU for the treatment of chronic lymphocytic leukaemia. News release. AstraZeneca. , 2020. Accessed November 10, 2020. https://bit.ly/3kiCkUT. 9 November.
- Shirley, D. How does zanubrutinib fare in treatment of B-cell malignancies? Lancet Haematol. 2023 Jan;10(1):e5-e6. [CrossRef]
- https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-zanubrutinib-mantle-cell-lymphoma.
- https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-zanubrutinib-marginal-zone-lymphoma.
- https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-zanubrutinib-waldenstroms-macroglobulinemia.
- Tam CS, Opat S, D’Sa S, Jurczak W, et al. A randomized phase 3 trial of zanubrutinib vs ibrutinib in symptomatic Waldenström macroglobulinemia: the ASPEN study. Blood. 2020 Oct 29;136(18):2038-2050. [CrossRef]
- Tam CS, Brown JR, Kahl BS, et al. Zanubrutinib versus bendamustine and rituximab in untreated chronic lymphocytic leukaemia and small lymphocytic lymphoma (SEQUOIA): a randomised, controlled, phase 3 trial. Lancet Oncol. 2022 Aug;23(8):1031-1043. [CrossRef]
- Brown JR, Eichhorst B, Hillmen P, et al. Zanubrutinib or Ibrutinib in Relapsed or Refractory Chronic Lymphocytic Leukemia. N Engl J Med 2023 Jan 26;388(4):319-332. [CrossRef]
- https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-zanubrutinib-chronic-lymphocytic-leukemia-or-small-lymphocytic-lymphoma.
- https://www.ema.europa.eu/en/medicines/human/EPAR/brukinsa.
- Gradishar, W.J.; Moran, M.S.; Abraham, J.; Abramson, V.; Aft, R.; Agnese, D.; Allison, K.H.; Anderson, B.; Burstein, H.J.; Chew, H.; et al. NCCN Guidelines® Insights: Breast Cancer, Version 4.2023. J. Natl. Compr. Cancer Netw. 2023, 21, 594–608. [Google Scholar] [CrossRef] [PubMed]
- https://www.onkopedia.com/de/onkopedia/guidelines/chronische-lymphatische-leukaemie-cll/@@guideline/html/index.html.
- Tam, C.S.; Trotman, J.; Opat, S.; Burger, J.A.; Cull, G.; Gottlieb, D.; Harrup, R.; Johnston, P.B.; Marlton, P.; Munoz, J.; et al. Phase 1 study of the selective BTK inhibitor zanubrutinib in B-cell malignancies and safety and efficacy evaluation in CLL. Blood 2019, 134, 851–859. [Google Scholar] [CrossRef]
- Brander, DM. The use of zanubrutinib in chronic lymphocytic leukemia. Clin Adv Hematol Oncol. 2022 Dec;20(12):705-708.
- Muñoz, J.; Wang, Y.; Jain, P.; Wang, M. Zanubrutinib in lymphoproliferative disorders: a comprehensive review. Ther. Adv. Hematol. 2022, 13. [Google Scholar] [CrossRef]
- Cull G, Burger JA, Opat S, et al. Zanubrutinib for treatment-naïve and relapsed/refractory chronic lymphocytic leukaemia: long-term follow-up of the phase I/II AU-003 study. Br J Haematol. 2022 Mar;196(5):1209-1218. [CrossRef]
- Tam CS, Giannopoulos K, Jurczak W, et al. SEQUOIA: results of a phase 3 randomized study of zanubrutinib versus bendamustine + rituximab in patients with treatment-naïve chronic lymphocytic leukemia/small lymphocytic lymphoma [ASH abstract 396]. Blood. 2021;138(suppl 1). [CrossRef]
- Sharman, J.P.; Egyed, M.; Jurczak, W.; Skarbnik, A.; Pagel, J.M.; Flinn, I.W.; Kamdar, M.; Munir, T.; Walewska, R.; Corbett, G.; et al. Efficacy and safety in a 4-year follow-up of the ELEVATE-TN study comparing acalabrutinib with or without obinutuzumab versus obinutuzumab plus chlorambucil in treatment-naïve chronic lymphocytic leukemia. Leukemia 2022, 36, 1171–1175. [Google Scholar] [CrossRef]
- Ghia, P, Barnes G, Yang K, et al. Patient-reported outcomes from phase 3 randomized study of zanubrutinib versus bendamustine and rituximab (BR) in patients with treatment-naïve (TN) CLL/SLL. HemaSphere 6():p 560-561, 22. 20 June.
- Tam CS, Robak T, Ghia P, et al. Zanubrutinib monotherapy for patients with treatment naïve chronic lymphocytic leukemia and 17p deletion. Haematologica 2020 Oct 13;106(9):2354-2363. [CrossRef]
- Tedeschi, A.; Ferrant, E.; Flinn, I.W.; Tam, C.S.; Ghia, P.; Robak, T.; Brown, J.R.; Ramakrishnan, V.; Tian, T.; Kuwahara, S.B.; et al. Zanubrutinib in Combination with Venetoclax for Patients with Treatment-Naïve (TN) Chronic Lymphocytic Leukemia (CLL) or Small Lymphocytic Lymphoma (SLL) with del(17p): Early Results from Arm D of the SEQUOIA (BGB-3111-304) Trial. 63rd Annual Meeting and Exposition of the American-Society-of-Hematology (ASH). LOCATION OF CONFERENCE, COUNTRYDATE OF CONFERENCE; pp. 67–67.
- Hillmen P, Eichhorst B, Brown JR, et al. Zanubrutinib Versus Ibrutinib in Relapsed/Refractory Chronic Lymphocytic Leukemia and Small Lymphocytic Lymphoma: Interim Analysis of a Randomized Phase III Trial. J Clin Oncol. 2023 Feb 10;41(5):1035-1045. [CrossRef]
- Brown, J.R.; Eichhorst, B.; Hillmen, P.; Jurczak, W.; Kaźmierczak, M.; Lamanna, N.; O’brien, S.M.; Tam, C.S.; Qiu, L.; Zhou, K.; et al. Zanubrutinib or Ibrutinib in Relapsed or Refractory Chronic Lymphocytic Leukemia. New Engl. J. Med. 2023, 388, 319–332. [Google Scholar] [CrossRef]
- Brown JR, Eichhorst B, Hillmen P, et al. Zanubrutinib demonstrates superior progression-free survival (PFS) compared with ibrutinib for treatment of relapsed/refractory chronic lymphocytic leukemia and small lymphocytic lymphoma (R/R CLL/SLL): results from final analysis of ALPINE randomized phase 3 study [ASH abstract LBA-6]. Blood. 2022;140(Suppl 1). [CrossRef]
- Tam, C.S.; Ou, Y.C.; Trotman, J.; Opat, S. Clinical pharmacology and PK/PD translation of the second-generation Bruton’s tyrosine kinase inhibitor, zanubrutinib. Expert Rev. Clin. Pharmacol. 2021, 14, 1329–1344. [Google Scholar] [CrossRef]
- Hillmen, P.; Brown, J.; Lamanna, N.; O’brien, S.; Tam, C.; Qiu, L.; Yang, K.; Barnes, G.; Wu, K.; Salmi, T.; et al. P663: HEALTH-RELATED QUALITY OF LIFE OUTCOMES ASSOCIATED WITH ZANUBRUTINIB VS IBRUTINIB MONOTHERAPY IN PATIENTS WITH RELAPSED/REFRACTORY (RR) CLL/SLL: RESULTS FROM THE RANDOMIZED PHASE 3 ALPINE TRIAL. HemaSphere 2022, 6, 561–562. [Google Scholar] [CrossRef]
- Ou, Y.C.; Tang, Z.; Novotny, W.; Cohen, A.; Wang, K.; Liu, L.; Gao, Y.; Sahasranaman, S. Rationale for once-daily or twice-daily dosing of zanubrutinib in patients with mantle cell lymphoma. Leuk. Lymphoma 2021, 62, 2612–2624. [Google Scholar] [CrossRef]
- Shadman M, Flinn IW, Levy MY, et al. Zanubrutinib in patients with previously treated B-cell malignancies intolerant of previous Bruton tyrosine kinase inhibitors in the USA: a phase 2, open-label, single-arm study. Lancet Haematol. 2023 Jan;10(1):e35-e45. [CrossRef]
- Mato AR, Nabhan C, Thompson MC, et al. Toxicities and outcomes of 616 ibrutinib-treated patients in the United States: a real-world analysis. Haematologica 2018 May;103(5):874-879. [CrossRef]
- Byrd, J.C.; Woyach, J.A.; Furman, R.R.; Martin, P.; O’brien, S.; Brown, J.R.; Stephens, D.M.; Barrientos, J.C.; Devereux, S.; Hillmen, P.; et al. Acalabrutinib in treatment-naive chronic lymphocytic leukemia. Blood 2021, 137, 3327–3338. [Google Scholar] [CrossRef] [PubMed]
- Rogers KA, Thompson PA, Allan JN, et al. Phase II study of acalabrutinib in ibrutinib-intolerant patients with relapsed/refractory chronic lymphocytic leukemia. Haematologica. 2021 Sep 1;106(9):2364-2373. [CrossRef]
- Burger, J.A.; Sivina, M.; Jain, N.; Kim, E.; Kadia, T.; Estrov, Z.; Nogueras-Gonzalez, G.M.; Huang, X.; Jorgensen, J.; Li, J.; et al. Randomized trial of ibrutinib vs ibrutinib plus rituximab in patients with chronic lymphocytic leukemia. Blood 2019, 133, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Tam CS, Quach H, Nicol A, et al. Zanubrutinib (BGB-3111) plus obinutuzumab in patients with chronic lymphocytic leukemia and follicular lymphoma. Blood Adv 2020 Oct 13;4(19):4802-4811. [CrossRef]
- Soumerai, J.D.; Mato, A.R.; Dogan, A.; E Seshan, V.; Joffe, E.; Flaherty, K.; Carter, J.; Hochberg, E.; A Barnes, J.; Hamilton, A.M.; et al. Zanubrutinib, obinutuzumab, and venetoclax with minimal residual disease-driven discontinuation in previously untreated patients with chronic lymphocytic leukaemia or small lymphocytic lymphoma: a multicentre, single-arm, phase 2 trial. Lancet Haematol. 2021, 8, e879–e890. [Google Scholar] [CrossRef] [PubMed]
- Davids, M.S.; Lampson, B.L.; Tyekucheva, S.; Wang, Z.; Lowney, J.C.; Pazienza, S.; Montegaard, J.; Patterson, V.; Weinstock, M.; Crombie, J.L.; et al. Acalabrutinib, venetoclax, and obinutuzumab as frontline treatment for chronic lymphocytic leukaemia: a single-arm, open-label, phase 2 study. Lancet Oncol. 2021, 22, 1391–1402. [Google Scholar] [CrossRef] [PubMed]
- Woyach JA, Ruppert AS, Guinn D, et al. BTKC481S-Mediated Resistance to Ibrutinib in Chronic Lymphocytic Leukemia. J Clin Oncol 2017 ;35(13):1437-1443. 1 May. [CrossRef]
- Zhu H, Sha Y, Miao Y, et al. Integrating Multi-Omics to Reveal the Clonal Evolutionary Characteristics in CLL Patients with Zanubrutinib Resistance. [ASH abstract 3104]. Blood. 2022;140(Suppl 1). [CrossRef]
- Blombery, P.; Thompson, E.R.; Lew, T.E.; Tiong, I.S.; Bennett, R.; Cheah, C.Y.; Lewis, K.L.; Handunnetti, S.M.; Tang, C.P.S.; Roberts, A.; et al. Enrichment of BTK Leu528Trp mutations in patients with CLL on zanubrutinib: potential for pirtobrutinib cross-resistance. Blood Adv. 2022, 6, 5589–5592. [Google Scholar] [CrossRef] [PubMed]
- Ahn IE, Brown JR. Selecting initial therapy in CLL. Hematology Am Soc Hematol Educ Program. 2022 Dec 9;2022(1):323-328. [CrossRef]
- Shadman, M. Diagnosis and Treatment of Chronic Lymphocytic Leukemia: A Review. JAMA. 2023 Mar 21;329(11):918-932. [CrossRef]
- Gordon MJ, Kaempf A, Sitlinger A, et al. The Chronic Lymphocytic Leukemia Comorbidity Index (CLL-CI): A Three-Factor Comorbidity Model. Clin Cancer Res. 2021 Sep 1;27(17):4814-4824. [CrossRef]
- Dickerson, T.; Wiczer, T.; Waller, A.; Philippon, J.; Porter, K.; Haddad, D.; Guha, A.; Rogers, K.A.; Bhat, S.; Byrd, J.C.; et al. Hypertension and incident cardiovascular events following ibrutinib initiation. Blood 2019, 134, 1919–1928. [Google Scholar] [CrossRef] [PubMed]
- Shanafelt TD, Parikh SA, Noseworthy PA, et al. Atrial fibrillation in patients with chronic lymphocytic leukemia (CLL). Leuk Lymphoma 2017 Jul;58(7):1630-1639. [CrossRef]
- Awan FT, Addison D, Alfraih F, et al. International consensus statement on the management of cardiovascular risk of Bruton’s tyrosine kinase inhibitors in CLL. Blood Adv (2022) 6 (18): 5516–5525. [CrossRef]
- Bhat SA, Gambril J, Azali L, et al. Ventricular arrhythmias and sudden death events following acalabrutinib initiation. Blood 2022 Nov 17;140(20):2142-2145. [CrossRef]
- Lampson, B.L.; Yu, L.; Glynn, R.J.; Barrientos, J.C.; Jacobsen, E.D.; Banerji, V.; Jones, J.A.; Walewska, R.; Savage, K.J.; Michaud, G.F.; et al. Ventricular arrhythmias and sudden death in patients taking ibrutinib. Blood 2017, 129, 2581–2584. [Google Scholar] [CrossRef]
- Hampel, P.J.; Parikh, S.A. Chronic lymphocytic leukemia treatment algorithm 2022. Blood Cancer J. 2022, 12, 1–10. [Google Scholar] [CrossRef]
- Kuss B, Nagarajan C, Hsieh WS, et al. Practical management of chronic lymphocytic leukemia with acalabrutinib. Leuk Lymphoma 2022 Dec;63(12):2785-2794. [CrossRef]
- Sharma, S.; Pepin, X.; Burri, H.; Zheng, L.; Kuptsova-Clarkson, N.; de Jong, A.; Yu, T.; MacArthur, H.L.; Majewski, M.; Byrd, J.C.; et al. Bioequivalence and Relative Bioavailability Studies to Assess a New Acalabrutinib Formulation That Enables Coadministration With Proton-Pump Inhibitors. Clin. Pharmacol. Drug Dev. 2022, 11, 1294–1307. [Google Scholar] [CrossRef] [PubMed]
- Davids, M.S.; Sharman, J.P.; Eyre, T.A.; Woyach, J.A.; de Miranda, P.A.P.; Shahkarami, M.; Butturini, A.; Emeribe, U.; Byrd, J.C. Contribution of Obinutuzumab to Acalabrutinib Therapy in Patients with Treatment-Naive Chronic Lymphocytic Leukemia: Analysis of Survival Outcomes By Genomic Features. 64th Annual Meeting and Exposition of the American-Society-of-Hematology (ASH). LOCATION OF CONFERENCE, COUNTRYDATE OF CONFERENCE; pp. 4173–4175.
- Tam, C.S.; Allan, J.N.; Siddiqi, T.; Kipps, T.J.; Jacobs, R.W.; Opat, S.; Barr, P.M.; Tedeschi, A.; Trentin, L.; Bannerji, R.; et al. Fixed-duration ibrutinib plus venetoclax for first-line treatment of CLL: primary analysis of the CAPTIVATE FD cohort. Blood 2022, 139, 3278–3289. [Google Scholar] [CrossRef] [PubMed]
- Kater, A.P.; Owen, C.; Moreno, C.; Follows, G.; Munir, T.; Levin, M.-D.; Benjamini, O.; Janssens, A.; Osterborg, A.; Robak, T.; et al. Fixed-Duration Ibrutinib-Venetoclax in Patients with Chronic Lymphocytic Leukemia and Comorbidities. NEJM Évid. 2022, 1. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



