Submitted:
20 April 2023
Posted:
21 April 2023
You are already at the latest version
Abstract
Keywords:
Introduction
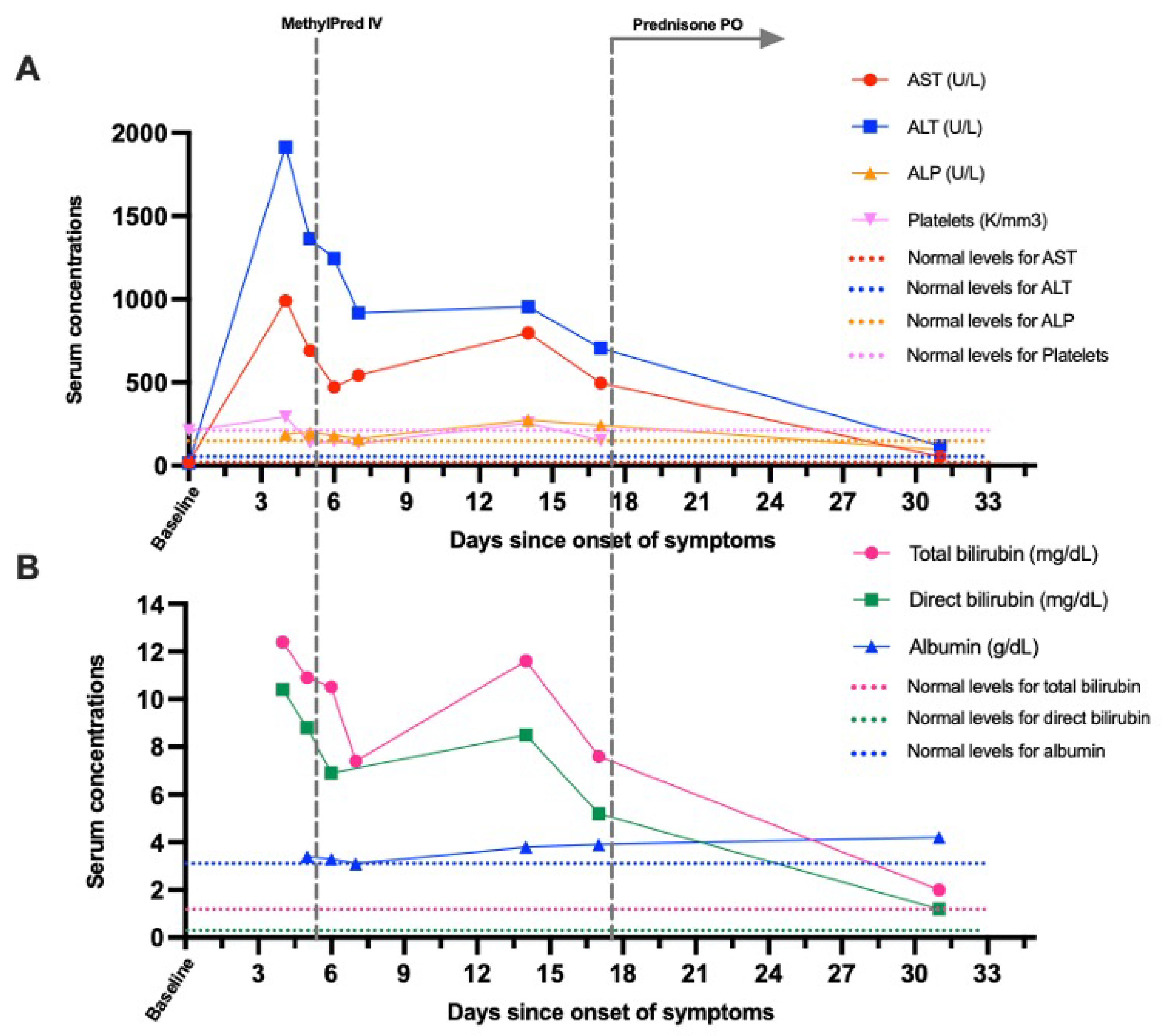
Case Report
Discussion
Conclusion
Financial Support or Competing Interests
Data Availability
Consent
Abbreviations
References
- Reuben A, Koch DG, Lee WM. Drug-induced acute liver failure: results of a U.S. multicenter, prospective study. Hepatology. 2010;52:2065-2076.
- Hoofnagle JH, Björnsson ES. Drug-induced liver injury—types and phenotypes. New England Journal of Medicine. 2019;381(3):264-73.
- Björnsson, ES. Drug-induced liver injury: Hy's rule revisited. Clinical Pharmacology & Therapeutics. 2006;79:521-528.
- Calabresi PA. B-Cell Depletion - A Frontier In Monoclonal Antibodies For Multiple Sclerosis. New England Journal of Medicine. 2017 Jan 19;376(3):280-282.
- Montalban, X, Hauser, SL, Kappos, L. Ocrelizumab versus placebo in primary progressive multiple sclerosis. New England Journal of Medicine. 2017;376: 209–220.
- Ocrevus Medication Guides. US Food & Drug Administration. 2022, August 8. Retrieved December 31, 2022, directly downloaded from: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=medguide.page.
- Ng HS, Rosenbult CL, & Tremlett H. Safety profile of ocrelizumab for the treatment of multiple sclerosis: a systematic review. Expert Opinion on Drug Safety. 2020;19(9):1069-1094.
- Nicolini LA, Canepa P, Caligiuri P. Fulminant Hepatitis Associated With Echovirus 25 During Treatment With Ocrelizumab For Multiple Sclerosis. JAMA Neurology. 2019;76(7):866-867.
- Ciardi MR, Iannetta M, Zingaropoli MA, Salpini R, Aragri M, Annecca R, Pontecorvo S, Altieri M, Russo G, Svicher V, Mastroianni CM, Vullo V. Reactivation of Hepatitis B Virus With Immune-Escape Mutations After Ocrelizumab Treatment for Multiple Sclerosis. Open Forum Infect Diseases. 2019;6(1):ofy356.
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Ocrelizumab. 2012. Directly downloaded from: https://www.ncbi.nlm.nih.gov/books/NBK548145/.
- Kaplowitz, N. Idiosyncratic drug hepatotoxicity. Nature Reviews Drug Discovery. 2005;4,489-499.
- Reuben, A. et al. Outcomes in adults with acute liver failure between 1998 and 2013: An observational cohort study. Annals of Internal Medicine. 2016;164,724-732.
- Chen M, Borlak J, and Tong W. High lipophilicity and high daily dose of oral medications are associated with significant risk for drug-induced liver injury. Hepatology. 2013;58,388-96.
- Robles-Diaz, M. et al. Use of Hy’s law and a new composite algorithm to predict acute liver failure in patients with drug-induced liver injury. Gastroenterology. 2014;147,109–118.
- Hayashi, PH. et al. Death and liver transplantation within 2 years of onset of drug-induced liver injury. Hepatology. 2017;66,1275-1285.
- Castiella A, Zapata E, Lucena MI, Andrade RJ. Drug-induced autoimmune liver disease: A diagnostic dilemma of an increasingly reported disease. World Journal of Hepatology. 2014;6(4):160-168.
- Liu ZX, Kaplowitz N. Immune-mediated drug-induced liver disease. Clinal Liver Disease. 2002;6:755-774.
- Alla V, Abraham J, Siddiqui J, Raina D, Wu GY, Chalasani NP, Bonkovsky HL. Autoimmune hepatitis triggered by statins. Journal of Clinical Gastroenterology. 2006;40:757-761.
- Grasset L, Guy C, Ollagnier M. Cyclines and acne: pay attention to adverse drug reactions! A recent literature review. Revue de Medicine Interne. 2003;24:305-31.
- Ibrahim AM, Jafri SM. A Rare Case of Refractory Drug Induced Liver Injury Following Ocrelizumab Use for Multiple Sclerosis. The American Journal of Gastroenterology. 2022;117:2914-2917. DOI 10.14309/01.ajg.0000868304.44094.3b.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



