Submitted:
05 April 2023
Posted:
07 April 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Development of the Disease Definition
Consensus Method
3. Biological Definition of Parkinson’s Disease
3.1. PD Disease Concept
3.2. Genetics
- -
- The first level comprises the fully penetrant variants: SNCA triplications, SNCA missense variants, as well as bi-allelic PRKN, PINK1, and PARK7 missense, nonsense, small indels, and copy number variants.
- -
- The second level comprises variants that confer a strong predisposition to PD but not complete penetrance, including SNCA duplications and pathogenic variants in LRRK2, VPS35, and CHCHD2.
- -
- The third level, which results in an intermediate predisposition with highly reduced penetrance, consists of carriers of highly pathogenic GBA variants. GCH1 monoallelic pathogenic variants 29,30 and 22q11.2 deletion syndrome31 may also fall into this category, but are currently considered investigational due to limited knowledge.
- -
- Variants in SNCA32 and GBA25 unequivocally predispose to Parkinson’s type synucleinopathy.
- -
- LRRK2 monoallelic (or biallelic) pathogenic variants predispose to Parkinson’s type synucleinopathy in most cases, whereas neurodegeneration without synucleinopathy occurs in a minority30.
- -
- Biallelic variants in PRKN predispose to a Parkinson’s type synucleinopathy in approximately 20% of the cases33.
- -
- Only very few post-mortem reports are available for variants in PINK134,35, PARK736, and CHCHD237, and the 22q11.2 deletion syndrome31, some associated with Parkinson’s type synucleinopathy and others not, i.e. on a very low level of evidence.
- -
- For variants in VPS35 and GCH1, the predisposition for a Parkinson’s type synucleinopathy is currently unknown30.
3.3. Synucleinopathy
3.4. Neurodegeneration
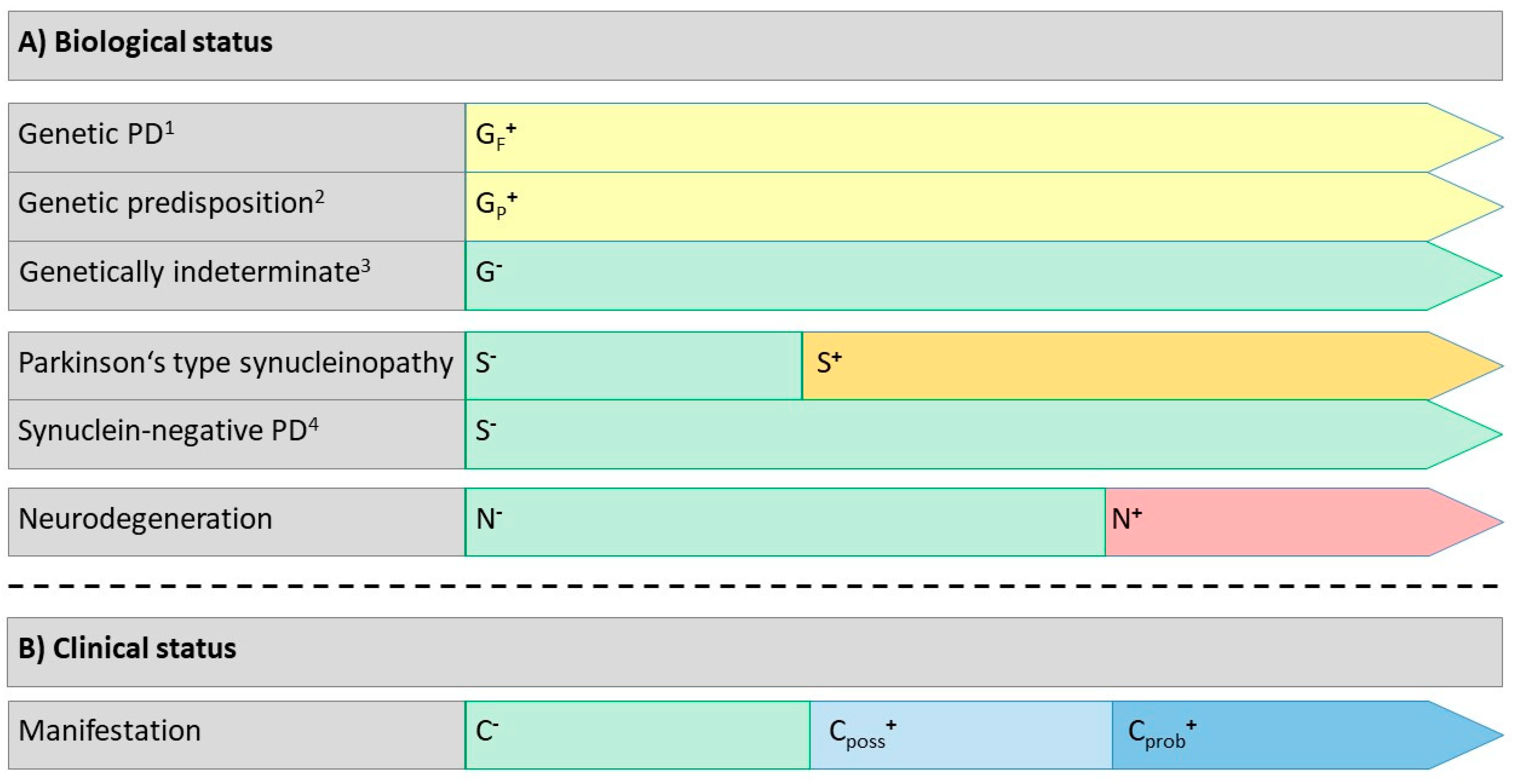
3.5. Biological Designations
3.6. Clinical Manifestations
4. Discussion
5. Limitations/Implementation/Future Directions
Contributors
Declaration of Interests
Acknowledgments
References
- Gibb, W.R.; Lees, A.J. The relevance of the Lewy body to the pathogenesis of idiopathic Parkinson's disease. J. Neurol. Neurosurg. Psychiatry 1988, 51, 745–752. [Google Scholar] [CrossRef] [PubMed]
- Nalls, M.A.; Blauwendraat, C.; Vallerga, C.L.; et al. Identification of novel risk loci, causal insights, and heritable risk for Parkinson's disease: A meta-analysis of genome-wide association studies. Lancet Neurol. 2019, 18, 1091–1102. [Google Scholar] [CrossRef] [PubMed]
- Polymeropoulos, M.H.; Higgins, J.J.; Golbe, L.I.; et al. Mapping of a gene for Parkinson's disease to chromosome 4q21-q23. Science 1996, 274, 1197–1199. [Google Scholar] [CrossRef] [PubMed]
- Ibanez, P.; Bonnet, A.M.; Debarges, B.; et al. Causal relation between alpha-synuclein gene duplication and familial Parkinson's disease. Lancet 2004, 364, 1169–1171. [Google Scholar] [CrossRef] [PubMed]
- Chartier-Harlin, M.C.; Kachergus, J.; Roumier, C.; et al. Alpha-synuclein locus duplication as a cause of familial Parkinson's disease. Lancet 2004, 364, 1167–1169. [Google Scholar] [CrossRef]
- Bloem, B.R.; Okun, M.S.; Klein, C. Parkinson's disease. Lancet 2021, 397, 2284–2303. [Google Scholar] [CrossRef] [PubMed]
- Spillantini, M.G.; Schmidt, M.L.; Lee, V.M.; Trojanowski, J.Q.; Jakes, R.; Goedert, M. Alpha-synuclein in Lewy bodies. Nature 1997, 388, 839–840. [Google Scholar] [CrossRef] [PubMed]
- Attems, J.; Toledo, J.B.; Walker, L.; et al. Neuropathological consensus criteria for the evaluation of Lewy pathology in post-mortem brains: A multi-centre study. Acta Neuropathol. 2021, 141, 159–172. [Google Scholar] [CrossRef]
- Irwin, D.J.; Grossman, M.; Weintraub, D.; et al. Neuropathological and genetic correlates of survival and dementia onset in synucleinopathies: A retrospective analysis. Lancet Neurol. 2017, 16, 55–65. [Google Scholar] [CrossRef]
- Postuma, R.B.; Berg, D.; Stern, M.; et al. MDS clinical diagnostic criteria for Parkinson's disease. Mov. Disord. 2015, 30, 1591–1601. [Google Scholar] [CrossRef]
- Berg, D.; Postuma, R.B.; Bloem, B.; et al. Time to redefine PD? Introductory statement of the MDS Task Force on the definition of Parkinson's disease. Mov. Disord. 2014, 29, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Berg, D.; Postuma, R.B.; Adler, C.H.; et al. MDS research criteria for prodromal Parkinson's disease. Mov. Disord. 2015, 30, 1600–1611. [Google Scholar] [CrossRef] [PubMed]
- Postuma, R.B.; Berg, D. Advances in markers of prodromal Parkinson disease. Nat. Rev. Neurol. 2016, 12, 622–634. [Google Scholar] [CrossRef] [PubMed]
- Heinzel, S.; Berg, D.; Gasser, T.; et al. Update of the MDS research criteria for prodromal Parkinson's disease. Mov. Disord. 2019, 34, 1464–1470. [Google Scholar] [CrossRef] [PubMed]
- REM sleep behaviour disorder. Nat. Rev. Dis. Primers 2018, 4, 20. [CrossRef] [PubMed]
- Coon, E.A.; Singer, W.; Low, P.A. Pure Autonomic Failure. Mayo Clin. Proc. 2019, 94, 2087–2098. [Google Scholar] [CrossRef]
- Gibb, W.R.; Lees, A.J. The significance of the Lewy body in the diagnosis of idiopathic Parkinson's disease. Neuropathol. Appl. Neurobiol. 1989, 15, 27–44. [Google Scholar] [CrossRef]
- Kalia, L.V.; Lang, A.E. Parkinson's disease. Lancet 2015, 386, 896–912. [Google Scholar] [CrossRef]
- Walker, Z.; Possin, K.L.; Boeve, B.F.; Aarsland, D. Lewy body dementias. Lancet 2015, 386, 1683–1697. [Google Scholar] [CrossRef]
- McKeith, I.G.; Ferman, T.J.; Thomas, A.J.; et al. Research criteria for the diagnosis of prodromal dementia with Lewy bodies. Neurology 2020, 94, 743–755. [Google Scholar] [CrossRef]
- McKeith, I.G.; Boeve, B.F.; Dickson, D.W.; et al. Diagnosis and management of dementia with Lewy bodies: Fourth consensus report of the DLB Consortium. Neurology 2017, 89, 88–100. [Google Scholar] [CrossRef] [PubMed]
- Skrahina, V.; Gaber, H.; Vollstedt, E.J.; et al. The Rostock International Parkinson's Disease (ROPAD) Study: Protocol and Initial Findings. Mov. Disord. 2021, 36, 1005–1010. [Google Scholar] [CrossRef]
- Lange, L.M.; Gonzalez-Latapi, P.; Rajalingam, R.; et al. Nomenclature of Genetic Movement Disorders: Recommendations of the International Parkinson and Movement Disorder Society Task Force - An Update. Mov. Disord. 2022, 37, 905–935. [Google Scholar] [CrossRef] [PubMed]
- Gan-Or, Z.; Amshalom, I.; Kilarski, L.L.; et al. Differential effects of severe vs mild GBA mutations on Parkinson disease. Neurology 2015, 84, 880–887. [Google Scholar] [CrossRef] [PubMed]
- Hoglinger, G.; Schulte, C.; Jost, W.H.; et al. GBA-associated PD: Chances and obstacles for targeted treatment strategies. J. Neural. Transm (Vienna) 2022, 129, 1219–1233. [Google Scholar] [CrossRef] [PubMed]
- Cooper, D.N.; Krawczak, M.; Polychronakos, C.; Tyler-Smith, C.; Kehrer-Sawatzki, H. Where genotype is not predictive of phenotype: Towards an understanding of the molecular basis of reduced penetrance in human inherited disease. Hum. Genet. 2013, 132, 1077–1130. [Google Scholar] [CrossRef] [PubMed]
- Trinh, J.; Zeldenrust, F.M.J.; Huang, J.; et al. Genotype-phenotype relations for the Parkinson's disease genes SNCA, LRRK2, VPS35: MDSGene systematic review. Mov. Disord. 2018, 33, 1857–1870. [Google Scholar] [CrossRef] [PubMed]
- Wittke, C.; Petkovic, S.; Dobricic, V.; et al. Genotype-Phenotype Relations for the Atypical Parkinsonism Genes: MDSGene Systematic Review. Mov. Disord. 2021, 36, 1499–1510. [Google Scholar] [CrossRef]
- Mencacci, N.E.; Isaias, I.U.; Reich, M.M.; et al. Parkinson's disease in GTP cyclohydrolase 1 mutation carriers. Brain 2014, 137 Pt 9, 2480–2492. [Google Scholar] [CrossRef]
- Schneider, S.A.; Alcalay, R.N. Neuropathology of genetic synucleinopathies with parkinsonism: Review of the literature. Mov. Disord. 2017, 32, 1504–1523. [Google Scholar] [CrossRef]
- Butcher, N.J.; Kiehl, T.R.; Hazrati, L.N.; et al. Association between early-onset Parkinson disease and 22q11.2 deletion syndrome: Identification of a novel genetic form of Parkinson disease and its clinical implications. JAMA Neurol. 2013, 70, 1359–1366. [Google Scholar] [CrossRef] [PubMed]
- Goedert, M. Alpha-synuclein and neurodegenerative diseases. Nat. Rev. Neurosci. 2001, 2, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Madsen, D.A.; Schmidt, S.I.; Blaabjerg, M.; Meyer, M. Interaction between Parkin and alpha-Synuclein in PARK2-Mediated Parkinson's Disease. Cells 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Samaranch, L.; Lorenzo-Betancor, O.; Arbelo, J.M.; et al. PINK1-linked parkinsonism is associated with Lewy body pathology. Brain 2010, 133 Pt 4, 1128–1142. [Google Scholar] [CrossRef]
- Takanashi, M.; Li, Y.; Hattori, N. Absence of Lewy pathology associated with PINK1 homozygous mutation. Neurology 2016, 86, 2212–2213. [Google Scholar] [CrossRef]
- Taipa, R.; Pereira, C.; Reis, I.; et al. DJ-1 linked parkinsonism (PARK7) is associated with Lewy body pathology. Brain 2016, 139 Pt 6, 1680–1687. [Google Scholar] [CrossRef]
- Ikeda, A.; Nishioka, K.; Meng, H.; et al. Mutations in CHCHD2 cause alpha-synuclein aggregation. Hum. Mol. Genet. 2019, 28, 3895–3911. [Google Scholar] [CrossRef]
- Poewe, W.; Seppi, K.; Tanner, C.M.; et al. Parkinson disease. Nat. Rev. Dis. Primers 2017, 3, 17013. [Google Scholar] [CrossRef]
- Kalia, L.V.; Lang, A.E.; Hazrati, L.N.; et al. Clinical correlations with Lewy body pathology in LRRK2-related Parkinson disease. JAMA Neurol. 2015, 72, 100–105. [Google Scholar] [CrossRef]
- Magalhaes, P.; Lashuel, H.A. Opportunities and challenges of alpha-synuclein as a potential biomarker for Parkinson's disease and other synucleinopathies. NPJ Parkinsons Dis. 2022, 8, 93. [Google Scholar] [CrossRef]
- Zubelzu, M.; Morera-Herreras, T.; Irastorza, G.; Gomez-Esteban, J.C.; Murueta-Goyena, A. Plasma and serum alpha-synuclein as a biomarker in Parkinson's disease: A meta-analysis. Parkinsonism Relat. Disord. 2022, 99, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Bellomo, G.; De Luca, C.M.G.; Paoletti, F.P.; Gaetani, L.; Moda, F.; Parnetti, L. alpha-Synuclein Seed Amplification Assays for Diagnosing Synucleinopathies: The Way Forward. Neurology 2022, 99, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Concha-Marambio, L.; Pritzkow, S.; Shahnawaz, M.; Farris, C.M.; Soto, C. Seed amplification assay for the detection of pathologic alpha-synuclein aggregates in cerebrospinal fluid. Nat. Protoc. 2023. [Google Scholar] [CrossRef] [PubMed]
- Yoo, D.; Bang, J.I.; Ahn, C.; et al. Diagnostic value of alpha-synuclein seeding amplification assays in alpha-synucleinopathies: A systematic review and meta-analysis. Parkinsonism Relat. Disord. 2022, 104, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Iranzo, A.; Fairfoul, G.; Ayudhaya, A.C.N.; et al. Detection of alpha-synuclein in CSF by RT-QuIC in patients with isolated rapid-eye-movement sleep behaviour disorder: A longitudinal observational study. Lancet Neurol. 2021, 20, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Singer, W.; Schmeichel, A.M.; Shahnawaz, M.; et al. Alpha-Synuclein Oligomers and Neurofilament Light Chain Predict Phenoconversion of Pure Autonomic Failure. Ann. Neurol. 2021, 89, 1212–1220. [Google Scholar] [CrossRef] [PubMed]
- Bartl, M.; Dakna, M.; Galasko, D.; et al. Biomarkers of neurodegeneration and glial activation validated in Alzheimer's disease assessed in longitudinal cerebrospinal fluid samples of Parkinson's disease. PLoS ONE 2021, 16, e0257372. [Google Scholar] [CrossRef] [PubMed]
- Schulz, I.; Kruse, N.; Gera, R.G.; et al. Systematic Assessment of 10 Biomarker Candidates Focusing on alpha-Synuclein-Related Disorders. Mov. Disord. 2021, 36, 2874–2887. [Google Scholar] [CrossRef]
- Mitchell, T.; Lehericy, S.; Chiu, S.Y.; Strafella, A.P.; Stoessl, A.J.; Vaillancourt, D.E. Emerging Neuroimaging Biomarkers Across Disease Stage in Parkinson Disease: A Review. JAMA Neurol. 2021, 78, 1262–1272. [Google Scholar] [CrossRef]
- Antonini, A.; Leenders, K.L.; Vontobel, P.; et al. Complementary PET studies of striatal neuronal function in the differential diagnosis between multiple system atrophy and Parkinson's disease. Brain 1997, 120 Pt 12, 2187–2195. [Google Scholar] [CrossRef]
- Holtbernd, F.; Gagnon, J.F.; Postuma, R.B.; et al. Abnormal metabolic network activity in REM sleep behavior disorder. Neurology 2014, 82, 620–627. [Google Scholar] [CrossRef] [PubMed]
- Kotomin, I.; Korotkov, A.; Solnyshkina, I.; Didur, M.; Cherednichenko, D.; Kireev, M. Parkinson's Disease-Related Brain Metabolic Pattern Is Expressed in Schizophrenia Patients during Neuroleptic Drug-Induced Parkinsonism. Diagnostics (Basel) 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.C.; Poston, K.L.; Eckert, T.; et al. Differential diagnosis of parkinsonism: A metabolic imaging study using pattern analysis. Lancet Neurol. 2010, 9, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Kashihara, K.; Imamura, T.; Shinya, T. Cardiac 123I-MIBG uptake is reduced more markedly in patients with REM sleep behavior disorder than in those with early stage Parkinson's disease. Parkinsonism Relat. Disord. 2010, 16, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Bohnen, N.I.; Yarnall, A.J.; Weil, R.S.; et al. Cholinergic system changes in Parkinson's disease: Emerging therapeutic approaches. Lancet Neurol. 2022, 21, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Nahimi, A.; Kinnerup, M.B.; Sommerauer, M.; Gjedde, A.; Borghammer, P. Molecular Imaging of the Noradrenergic System in Idiopathic Parkinson's Disease. Int. Rev. Neurobiol. 2018, 141, 251–274. [Google Scholar] [PubMed]
- de Natale, E.R.; Wilson, H.; Politis, M. Serotonergic imaging in Parkinson's disease. Prog. Brain Res. 2021, 261, 303–338. [Google Scholar] [PubMed]
- Tabrizi, S.J.; Schobel, S.; Gantman, E.C.; et al. A biological classification of Huntington's disease: The Integrated Staging System. Lancet Neurol. 2022, 21, 632–644. [Google Scholar] [CrossRef]
- Jack, C.R., Jr.; Bennett, D.A.; Blennow, K.; et al. A/T/N: An unbiased descriptive classification scheme for Alzheimer disease biomarkers. Neurology 2016, 87, 539–547. [Google Scholar] [CrossRef]
- Norcliffe-Kaufmann, L.; Kaufmann, H.; Palma, J.A.; et al. Orthostatic heart rate changes in patients with autonomic failure caused by neurodegenerative synucleinopathies. Ann. Neurol. 2018, 83, 522–531. [Google Scholar] [CrossRef]
| Designation | Biomarker Status | Abnormality | Pathogenic effect | Presdisposing for |
|---|---|---|---|---|
| GF+ | Recommended | SNCA monoallelic triplication30,32 | fully penetrant | Parkinson’s type synucleinopathy |
| GF+ | Recommended | SNCA monoallelic pathogenic single nucleotide variants30,32 | fully penetrant | Parkinson’s type synucleinopathy |
| GF+ | Recommended | PRKN biallelic pathogenic variants30,33 | fully penetrant | Neurodegeneration without synucleinopathy in ~80% and Parkinson’s type synucleinopathy in ~20% of the cases |
| GF+ | Recommended | PINK1 biallelic pathogenic variants30,34,35 | fully penetrant | Parkinson’s type synucleinopathy in a single case and neurodegeneration without synucleinopathy in another |
| GF+ | Recommended | PARK7 biallelic pathogenic variants30,36 | fully penetrant | Parkinson’s type synucleinopathy in the single case published |
| GP+ | Recommended | SNCA monoallelic duplication30,32 | strong predisposition | Parkinson’s type synucleinopathy |
| GP+ | Recommended | LRRK2 monoallelic (or biallelic) pathogenic variants30 | strong predisposition | Parkinson’s type synucleinopathy in most cases; neurodegeneration without synucleinopathy in a minority |
| GP+ | Recommended | VPS35 monoallelic pathogenic variants30 | strong predisposition | unknown |
| GP+ | Recommended | CHCHD2 monoallelic pathogenic variants30,37 | strong predisposition | unknown |
| GP+ | Recommended | GBA monoallelic severely pathogenic variants25,30 | medium predisposition | Parkinson’s type synucleinopathy |
| G- | Investigational | GCH1 monoallelic pathogenic variants30 | medium predisposition | Parkinson’s type synucleinopathy in the single case published |
| G- | Investigational | 22q11.2 deletion syndrome30,31 | medium predisposition | Neurodegeneration without synucleinopathy in 1/3 and Parkinson’s type synucleinopathy in 2/3 published cases |
| Designation | Biomarker Status | Abnormality | Sensitivity* | Specificity* |
|---|---|---|---|---|
| S+ | Recommended | α-syn SAA in CSF | High | High |
| S+ | Recommended | α-syn SAA in skin | High | High |
| S+ | Recommended | α-syn ICH/ICF in skin | Moderate | High |
| S+ | Investigational | α-syn SAA in neuronal exosomes from plasma | Insufficient evidence | Insufficient evidence |
| S+ | Investigational | α-syn SAA in plasma | Insufficient evidence | Insufficient evidence |
| S+ | Investigational | α-syn SAA in submandibular gland | Insufficient evidence | Insufficient evidence |
| Exclusion criterion ruling out S+ | For S+ testing unable to differentiate PD from MSA (e.g., selected SAA methodologies in CSF and SAA in skin to date) | Elevated Neurofilament Light chain (NfL) | High for atypical parkinsonism (e.g., MSA) | High for MSA but low for specific diagnosis (e.g., also elevated in PSP but these cases would be S- in the absence of copathology) |
| Exclusion criterion ruling out S+ | For S+ testing unable to differentiate PD from MSA* | Neuroimaging features of MSA (e.g., characteristic changes in the putamen, cerebellum and pons) | Moderate | High |
| Designation | Biomarker Status | Examination | Interpretation | Sensitivity* | Specificity* |
|---|---|---|---|---|---|
| N+ | Recommended | DAergic PET/SPECT | Striatal dopaminergic deficit | High | Low |
| N+ | Recommended | Metabolic FDG PET | PD related brain metabolic pattern | High | High |
| N+ | Recommended | Cardiac MIBG SPECT | Sympathetic cardiac denervation | Moderate to high | Moderate |
| N+ | Investigational | Neuromelanin MRI | Limited test-retest stability | Moderate to high | Low |
| N+ | Investigational | Iron-sensitive MRI | Sophisticated method restricted to specialized centers, may not directly prove neurodegeneration | Moderate to high | Low |
| N+ | Investigational | Substantia nigra free water MRI | Sophisticated method restricted to specialized centers, may not directly prove neurodegeneration | High | Moderate to high if applied to extra-nigral sites |
| N+ | Investigational | Structural MRI (T1) | Sophisticated method restricted to specialized centers | Low | Moderate to high |
| N+ | Investigational | Diffusion tensor imaging | Sophisticated method restricted to specialized centers | Low to moderate | High |
| N+ | Investigational | Multimodal MRI | Sophisticated method restricted to specialized centers | Moderate to high | High |
| Exclusion criterion ruling out N+ | Recommended | Structural MRI | Findings characteristic of atypical parkinsonism: e.g. PSP (midbrain/superior cerebellar atrophy); e.g. MSA (pontine atrophy, hot-cross bun sign, cerebellar atrophy, increased basal ganglia iron with putaminal rim), e.g. CBS (parietal atrophy) | Moderate, stage dependent | High |
| Exclusion criterion ruling out N+ | Recommended | FDG PET | PSP and MSA each have characteristic patterns that are distinct from PD | High | High |
| Exclusion criterion ruling out N+ | Investigational | 11C-raclopride PET or 123I-iodobenzamide SPECT | Major reductions in dopamine receptor binding in early disease are more suggestive of PSP or MSA (modest reductions can be seen in treated PD) | Moderate | High |
| Gene pathogenic variant | Synucleinopathy | Neurodegeneration | Biological designation |
|---|---|---|---|
| G- | S+ | N+ | Sporadic PD |
| N- | Sporadic Parkinson’s type synucleinopathy | ||
| S- | N+ | Non-PD neurodegeneration (or false negative S test) |
|
| N- | No evidence for PD | ||
| GF+ | S+ or S- | N+ or N- | Genetic PD (e.g. SNCA-PD) |
| GP+ | S+ | N+ | Genetic PD (e.g. GBA-PD) |
| N- | Genetic Parkinson’s type synucleinopathy (e.g. GBA-Parkinson’s type synucleinopathy) |
||
| S- | N+ (gene predisposing for either Parkinson’s type synucleinopathy or non-synucleinopathy) | Genetic synuclein-negative PD (e.g. LRRK2-PD, PRKN-PD) |
|
| N+ (gene consistently predisposing for Parkinson’s type synucleinopathy) | Non-PD neurodegeneration (or false neg. S-test) |
||
| N- | Genetic predisposition for PD (e.g. GBA-predisposition for PD) |
| A. Clinical abnormalities possibly related to PD (Cposs+) |
|---|
| - Option 1: If S+ or N+ *: at least 1 feature from 1 of the following categories - Option 2: If isolated G+ (S- and N-): at least 1 feature from 2 of the following categories |
| 1. Motor |
| a. A single cardinal manifestation of parkinsonism (i.e. expert-examined bradykinesia, or rest tremor) b. Abnormal quantitative motor testing (>1 standard deviation below age-adjusted normal motor speed) |
| 2. Sensory |
| a. Olfactory loss |
| 3. Autonomic |
| a. Chronic constipation |
| b. Urinary dysfunction |
| c. Severe erectile dysfunction (onset at an age <60) |
| d. Likely neurogenic orthostatic hypotension (i.e. heart rate increase <0.5 bpm/mmHg systolic BP drop)60 |
| 4. Sleep |
| History of REM sleep behavior disorder (without polysomnographic confirmation) |
| b. Excessive daytime somnolence |
| 5. Affective / Cognitive |
| a. Depression |
| b. Mild cognitive impairment |
| B. Clinical abnormalities probably related to PD (Cprob+) |
| - Option 1: If S+ or N+ *: at least 1 feature from at least 2 of the categories of A, or - Option 2: If isolated G+ (S- and N-): at least 1 feature from at least 3 of the categories of A, or - Option 3: If G+, S+, or N+: at least 1 of the following features: |
| a. Parkinsonism10 (bradykinesia plus one of rigidity or rest tremor) |
| b. Dementia |
| c. REM sleep behavior disorder (polysomnography-confirmed) |
| d. Neurogenic orthostatic hypotension (≥20/10 mmHg blood pressure drop within 3 minutes of standing or head-up tilt test)60 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




