Submitted:
06 February 2023
Posted:
27 February 2023
You are already at the latest version
Abstract
Keywords:
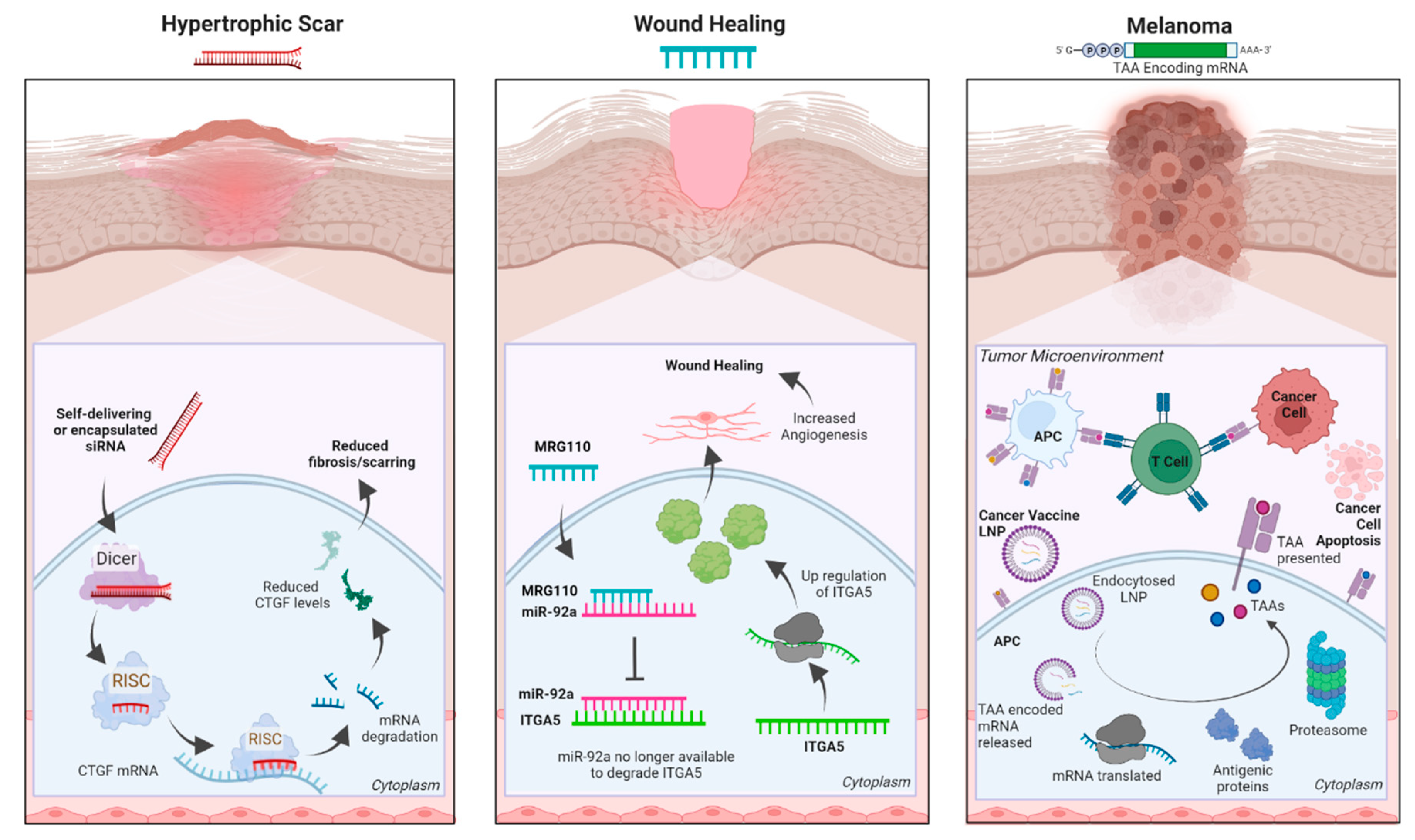
Introduction
RNA interference
Antisense Oligonucleotides (ASOs)
Messenger RNA (mRNA)
Summary
Acknowledgments and Funding
Conflicts of Interest
References
- Damase, T.R., Sukhovershin, R., Boada, C., Taraballi, F., Pettigrew, R.I., and Cooke, J.P. (2021). The Limitless Future of RNA Therapeutics. Front Bioeng Biotechnol 9, 628137. [CrossRef]
- Kalal, B.S., Upadhya, D., and Pai, V.R. (2017). Chemotherapy Resistance Mechanisms in Advanced Skin Cancer. Oncol Rev 11, 326. [CrossRef]
- Li, X., Ponandai-Srinivasan, S., Nandakumar, K.S., Fabre, S., Xu Landen, N., Mavon, A., and Khmaladze, I. (2021). Targeting microRNA for improved skin health. Health Sci Rep 4, e374. [CrossRef]
- Banerjee, J., Chan, Y.C., and Sen, C.K. (2011). MicroRNAs in skin and wound healing. Physiol Genomics 43, 543-556. [CrossRef]
- Bartel, D.P. (2018). Metazoan MicroRNAs. Cell 173, 20-51. [CrossRef]
- Hu, B., Zhong, L., Weng, Y., Peng, L., Huang, Y., Zhao, Y., and Liang, X.-J. (2020). Therapeutic siRNA: state of the art. Signal Transduction and Targeted Therapy 5, 101. [CrossRef]
- Lam, J.K., Chow, M.Y., Zhang, Y., and Leung, S.W. (2015). siRNA Versus miRNA as Therapeutics for Gene Silencing. Mol Ther Nucleic Acids 4, e252. [CrossRef]
- Zhu, Y., Zhu, L., Wang, X., and Jin, H. (2022). RNA-based therapeutics: an overview and prospectus. Cell Death Dis 13, 644. [CrossRef]
- Bejar, N., Tat, T.T., and Kiss, D.L. (2022). RNA Therapeutics: the Next Generation of Drugs for Cardiovascular Diseases. Curr Atheroscler Rep 24, 307-321. [CrossRef]
- Makino, K., Makino, T., Stawski, L., Lipson, K.E., Leask, A., and Trojanowska, M. (2017). Anti-connective tissue growth factor (CTGF/CCN2) monoclonal antibody attenuates skin fibrosis in mice models of systemic sclerosis. Arthritis Res Ther 19, 134. [CrossRef]
- Kang, S., Kim, J., Ahn, M., Kim, J., Heo, M.G., Min, D.H., and Won, C. (2020). RNAi nanotherapy for fibrosis: highly durable knockdown of CTGF/CCN-2 using siRNA-DegradaBALL (LEM-S401) to treat skin fibrotic diseases. Nanoscale 12, 6385-6393.
- Carswell, L., and Borger, J. (2022). Hypertrophic Scarring Keloids. In StatPearls. (Treasure Island (FL).
- Roberts, T.C., Langer, R., and Wood, M.J.A. (2020). Advances in oligonucleotide drug delivery. Nat Rev Drug Discov 19, 673-694. [CrossRef]
- Crooke, S.T., Baker, B.F., Crooke, R.M., and Liang, X.H. (2021). Antisense technology: an overview and prospectus. Nat Rev Drug Discov 20, 427-453. [CrossRef]
- Quemener, A.M., Bachelot, L., Forestier, A., Donnou-Fournet, E., Gilot, D., and Galibert, M.D. (2020). The powerful world of antisense oligonucleotides: From bench to bedside. Wiley Interdiscip Rev RNA 11, e1594. [CrossRef]
- Di Fusco, D., Dinallo, V., Marafini, I., Figliuzzi, M.M., Romano, B., and Monteleone, G. (2019). Antisense Oligonucleotide: Basic Concepts and Therapeutic Application in Inflammatory Bowel Disease. Front Pharmacol 10, 305. [CrossRef]
- Reese, C.B. (1978). The chemical synthesis of oligo-and poly-nucleotides by the phosphotriester approach. Tetrahedron 34, 3143-3179. [CrossRef]
- Schaller, H., Weimann, G., Lerch, B., and Khorana, H.G. (1963). Studies on Polynucleotides. XXIV.1 The Stepwise Synthesis of Specific Deoxyribopolynucleotides (4).2 Protected Derivatives of Deoxyribonucleosides and New Syntheses of Deoxyribonucleoside-3″ Phosphates3. Journal of the American Chemical Society 85, 3821-3827. [CrossRef]
- Orr, R.M. (2001). Technology evaluation: fomivirsen, Isis Pharmaceuticals Inc/CIBA vision. Curr Opin Mol Ther 3, 288-294.
- Chan, J.H., Lim, S., and Wong, W.S. (2006). Antisense oligonucleotides: from design to therapeutic application. Clin Exp Pharmacol Physiol 33, 533-540. [CrossRef]
- Juliano, R.L., and Carver, K. (2015). Cellular uptake and intracellular trafficking of oligonucleotides. Adv Drug Deliv Rev 87, 35-45. [CrossRef]
- Liang, X.H., Sun, H., Nichols, J.G., and Crooke, S.T. (2017). RNase H1-Dependent Antisense Oligonucleotides Are Robustly Active in Directing RNA Cleavage in Both the Cytoplasm and the Nucleus. Mol Ther 25, 2075-2092. [CrossRef]
- Mulhbacher, J., St-Pierre, P., and Lafontaine, D.A. (2010). Therapeutic applications of ribozymes and riboswitches. Curr Opin Pharmacol 10, 551-556. [CrossRef]
- Desterro, J., Bak-Gordon, P., and Carmo-Fonseca, M. (2020). Targeting mRNA processing as an anticancer strategy. Nat Rev Drug Discov 19, 112-129. [CrossRef]
- Juliano, R.L. (2016). The delivery of therapeutic oligonucleotides. Nucleic Acids Res 44, 6518-6548. [CrossRef]
- Younis, H.S., Templin, M.V., Whiteley, L.O., Kornbrust, D.J., Kim, T.-W., and Henry, S.P. (2013). Overview of the Nonclinical Development Strategies and Class-Effects of Oligonucleotide-Based Therapeutics. In. (. [CrossRef]
- Wan, J., Bauman, J.A., Graziewicz, M.A., Sazani, P., and Kole, R. (2013). Oligonucleotide therapeutics in cancer. Cancer Treat Res 158, 213-233. [CrossRef]
- Crooke, S.T., Vickers, T.A., and Liang, X.-h. (2020). Phosphorothioate modified oligonucleotide–protein interactions. Nucleic Acids Research 48, 5235-5253. [CrossRef]
- Sikes, R.A. (2007). Chemistry and pharmacology of anticancer drugs. British Journal of Cancer 97, 1713-1713. [CrossRef]
- Agrawal, S., Jiang, Z., Zhao, Q., Shaw, D., Cai, Q., Roskey, A., Channavajjala, L., Saxinger, C., and Zhang, R. (1997). Mixed-backbone oligonucleotides as second generation antisense oligonucleotides: in vitro and in vivo studies. Proc Natl Acad Sci U S A 94, 2620-2625. [CrossRef]
- Sardone, V., Zhou, H., Muntoni, F., Ferlini, A., and Falzarano, M.S. (2017). Antisense Oligonucleotide-Based Therapy for Neuromuscular Disease. Molecules 22. [CrossRef]
- Wahlestedt, C., Salmi, P., Good, L., Kela, J., Johnsson, T., Hokfelt, T., Broberger, C., Porreca, F., Lai, J., Ren, K., et al. (2000). Potent and nontoxic antisense oligonucleotides containing locked nucleic acids. Proc Natl Acad Sci U S A 97, 5633-5638. [CrossRef]
- Havens, M.A., and Hastings, M.L. (2016). Splice-switching antisense oligonucleotides as therapeutic drugs. Nucleic Acids Res 44, 6549-6563. [CrossRef]
- Prakash, T.P. (2011). An overview of sugar-modified oligonucleotides for antisense therapeutics. Chem Biodivers 8, 1616-1641. [CrossRef]
- Hagedorn, P.H., Persson, R., Funder, E.D., Albæk, N., Diemer, S.L., Hansen, D.J., Møller, M.R., Papargyri, N., Christiansen, H., Hansen, B.R., et al. (2018). Locked nucleic acid: modality, diversity, and drug discovery. Drug Discov Today 23, 101-114. [CrossRef]
- Gallant-Behm, C.L., Piper, J., Dickinson, B.A., Dalby, C.M., Pestano, L.A., and Jackson, A.L. (2018). A synthetic microRNA-92a inhibitor (MRG-110) accelerates angiogenesis and wound healing in diabetic and nondiabetic wounds. Wound Repair Regen 26, 311-323. [CrossRef]
- Huang, C.K., Kafert-Kasting, S., and Thum, T. (2020). Preclinical and Clinical Development of Noncoding RNA Therapeutics for Cardiovascular Disease. Circ Res 126, 663-678. [CrossRef]
- Wolff, J.A., Malone, R.W., Williams, P., Chong, W., Acsadi, G., Jani, A., and Felgner, P.L. (1990). Direct gene transfer into mouse muscle in vivo. Science 247, 1465-1468. [CrossRef]
- Weide, B., Carralot, J.P., Reese, A., Scheel, B., Eigentler, T.K., Hoerr, I., Rammensee, H.G., Garbe, C., and Pascolo, S. (2008). Results of the first phase I/II clinical vaccination trial with direct injection of mRNA. J Immunother 31, 180-188. [CrossRef]
- Gu, Y., Duan, J., Yang, N., Yang, Y., and Zhao, X. (2022). mRNA vaccines in the prevention and treatment of diseases. MedComm (2020) 3, e167. [CrossRef]
- van Dulmen, M., and Rentmeister, A. (2020). mRNA Therapies: New Hope in the Fight against Melanoma. Biochemistry 59, 1650-1655. [CrossRef]
- National Library of Medicine. In. (National Library of Medicine.
- (2023). AdisInsight. In. (Springer.
- Sahin, U., Oehm, P., Derhovanessian, E., Jabulowsky, R.A., Vormehr, M., Gold, M., Maurus, D., Schwarck-Kokarakis, D., Kuhn, A.N., Omokoko, T., et al. (2020). An RNA vaccine drives immunity in checkpoint-inhibitor-treated melanoma. Nature 585, 107-112. [CrossRef]
- BioNTech. (2023). In.
- BioNTech. (2021). In. (BioNTech.
- Kang, K., Xie, F., Mao, J., Bai, Y., and Wang, X. (2020). Significance of Tumor Mutation Burden in Immune Infiltration and Prognosis in Cutaneous Melanoma. Front Oncol 10, 573141. [CrossRef]
- Vitale, G. (2023). Chemical & Engineering News. In. (Chemical & Engineering News, American Chemical Society.
- Moderna. (2022). In. (Moderna Inc.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



