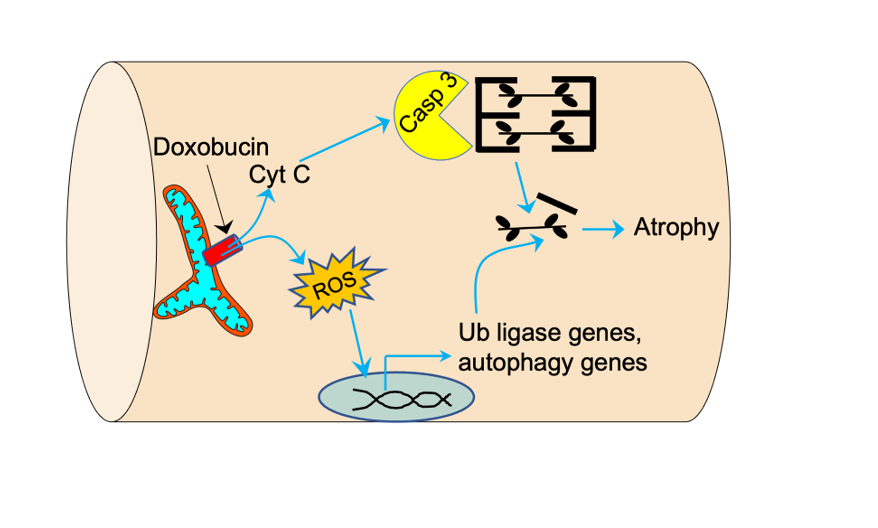
Doxorubicin (Dox) is a commonly used chemotherapeutic that can adversely affect skeletal muscle, including causing muscle atrophy. Dox is known to induce an event known as mitochondrial permeability transition (MPT) in cardiac muscle and this plays an important role in Dox-mediated cardiac toxicity. Further to this, recent evidence identifies MPT as a mechanism of atrophy in skeletal muscle, suggesting that MPT may underlie some of the Dox-related toxicity in skeletal muscle. To test this hypothesis, we used cultured human primary myotubes, C2C12 myotubes, and single adult mouse flexor digitorum brevis (FDB) muscle fibers in experiments involving Dox treatment with or without inhibitors of MPT. Dox treatment of myotubes caused myonuclear translocation of the mitochondrial protein apoptosis inducing factor (AIF) and increased mitochondrial reactive oxygen species (mROS), consistent with the known consequences of MPT. Furthermore, Dox caused atrophy in C2C12 myotubes grown on patterned plates, human primary myotubes, and single muscle fibers from adult mice. Notably, Dox-induced atrophy could be prevented by a wide variety of agents that inhibit MPT, as well as by inhibiting mROS or Caspase 3. In conclusion, our results indicate that MPT plays an important role in driving Dox-mediated skeletal muscle atrophy.