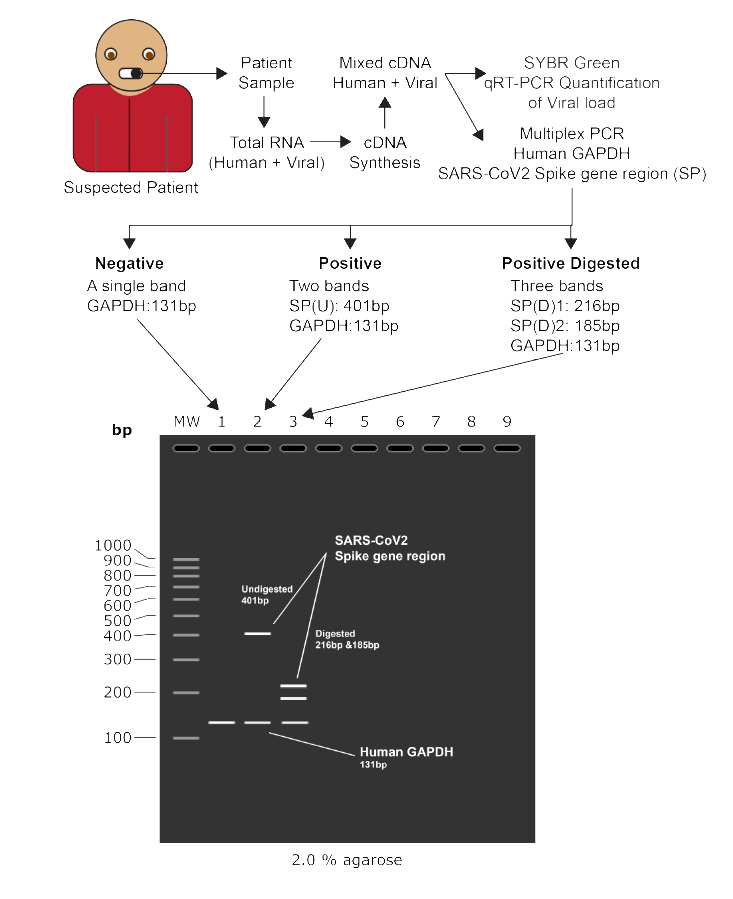
SARS-CoV 2 also known as COVID-19 is a fast spreading coronavirus related disease that emerged from China in December 2019 and is currently attained the status of a pandemic. There are currently no drugs/ vaccines against the same and moreover limited diagnostic tests to identify the infection. Additionally, these tests are expensive and hence are exclusive for very highly suspected cases of the disease especially in developing countries. This is causing an under-diagnosis which is an alarming state of affairs, as even a single missed SARS-CoV 2 case would spread the disease exponentially and keep it in the community. Through this entirely in silico study, we have developed a cheaper and faster diagnostic method based on simple PCR and restriction enzyme digestion, commonly used in restriction fragment length polymorphism (RFLP) tests. Through comparative genomics, we found the closest neighbours of SARS-CoV 2 then found the highly conserved regions of the genome which were absent in SARS-CoV 1, its closest neighbour. Then we found restriction sites for various enzymes followed by designing of PCR primers flanking those sites. We have found the primer pair to produce a 401 bp amplicon and when digested by SwaI enzyme, it produces two fragments of lengths 216 bp and 185 bp. As an internal control, GAPDH primers are pooled with the SARS-CoV 2 primers as the patient sample will also include human RNA mixed with the viral RNA. This primer pair gives an amplicon of 131 bp and hence a negative sample should show a single band of 131 bp while a positive digested sample will give three bands of 401 bp, 216 bp and 131 bp. The primers are specific to SARS-CoV 2 only and can additionally be used for SYBR green based real time quantification of viral load. The developed tests have not yet been tested in vitro due to stressed out working hours in the only pathogenic virus handling laboratory in our institute. Nonetheless, this study works as a head start for other laboratories to rapidly test the suggested protocols in vitro and make available a cheaper alternative test for SARS-CoV 2 which would especially be beneficial for the lower to middle income countries.