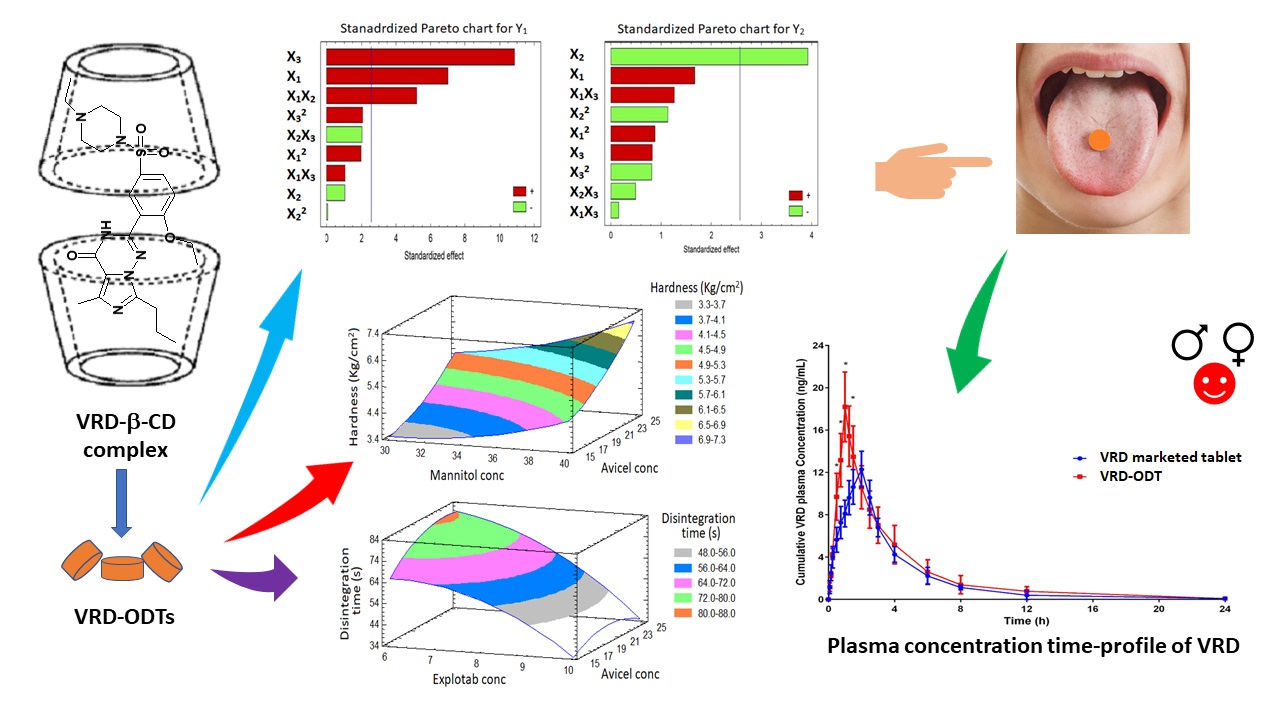
Because of lower solubility and considerable metabolism, vardenafil (VRD) bioavailability is 15 %. To get over this obstacle, this study aimed to increase the solubility, hasten the onset of action, and mask the unpleasant taste of VRD utilizing β-cyclodextrin (β-CD) and formulation of the inclusion complex as oral disintegrating tablets (ODTs). The solubility of the obtained complexes in various ratios has been studied. A Box-Behnken design (BBD) was utilized to investigate the influence of excipients on the quality of ODTs. The solubility of VRD was improved at 1:2 drug: β-CD ratio. The formulated VRD-ODTs exhibited satisfying results regarding the hardness and disintegration time. In addition, in vivo taste masking and disintegration time showed improved results, after placing the tablets in the oral cavity of the healthy volunteers. The pharmacokinetic parameters for the optimized VRD–ODTs exhibited a significant improvement with P < 0.05 in the maximum plasma concentration and reduction in the time needed to reach this concentration when compared with the marketed tablets. Finally, the optimized VRD-ODTs exhibited increased oral absorption of VRD and subsequent decreasing the time of onset of clinical effect and masking the unpleasant taste, which is favored for patients with erectile dysfunction.