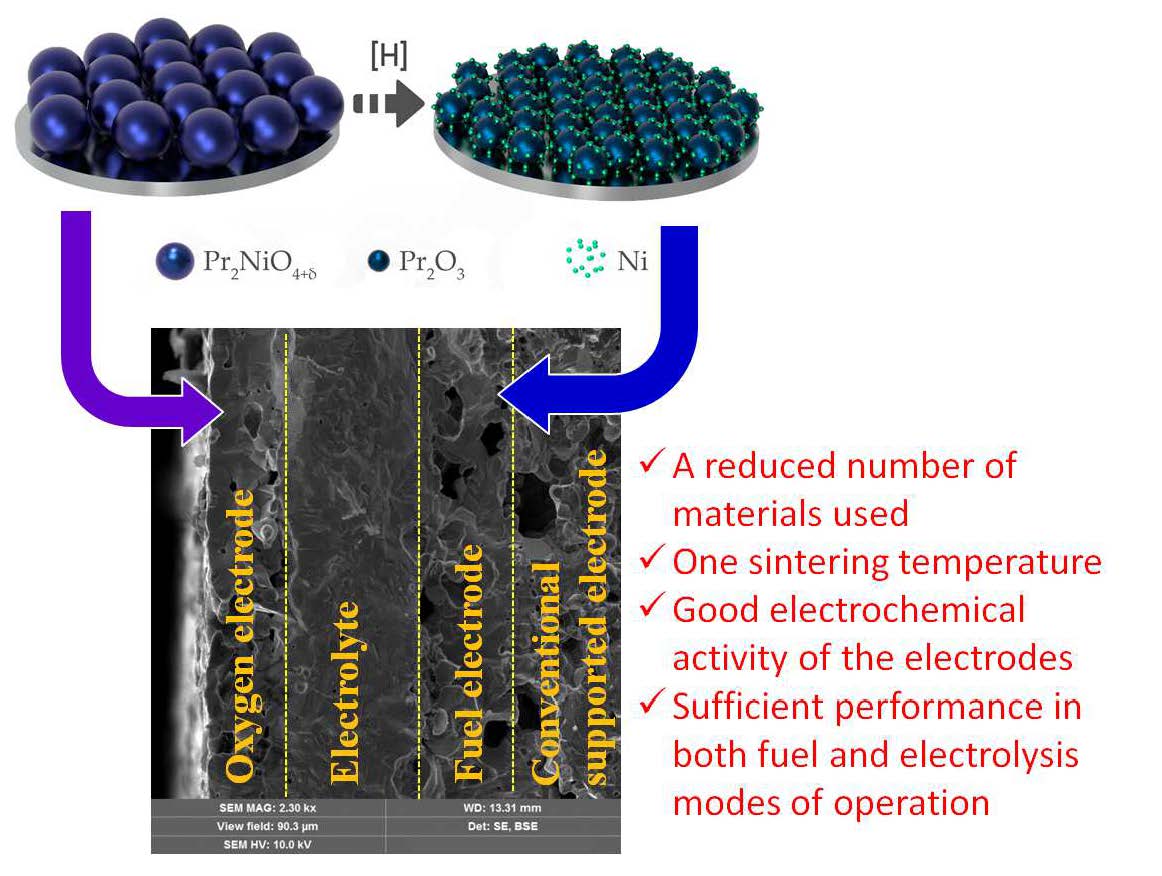
Reversible protonic ceramic cells (rPCCs) combine two different operation regimes, fuel and electrolysis modes, which allow reversible chemical-to-electrical energy conversion at reduced temperatures with high efficiency and performance. Here we present novel technological and materials science approaches, enabling a rPCC with symmetrical functional electrodes to be prepared using a single sintering step. The response of the cell fabricated on the basis of P–N–BCZD|BCZD|PBN–BCZD (where BCZD = BaCe0.5Zr0.3Dy0.2O3–δ, PBN = Pr1.9Ba0.1NiO4+δ, P = Pr2O3, N = Ni) is studied at different temperatures and water vapor partial pressures by means of volt-ampere measurements, electrochemical impedance spectroscopy and distribution of relaxation times analyses. The obtained results demonstrate that symmetrical electrodes exhibit classical mixed-ionic/electronic conducting behavior with no hydration capability at 750 °C; therefore, increasing the pH2O values in both reducing and oxidizing atmospheres leads to some deterioration of their electrochemical activity. At the same time, the electrolytic properties of the BCZD membrane are improved, positively affecting the rPCC’s efficiency. The electrolysis mode of the rPCC is found to be more efficient than the fuel cell mode under highly humidified atmospheres, since its performance is determined by the ohmic resistance, which decreases under respectively less humid conditions.