Submitted:
25 January 2026
Posted:
26 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
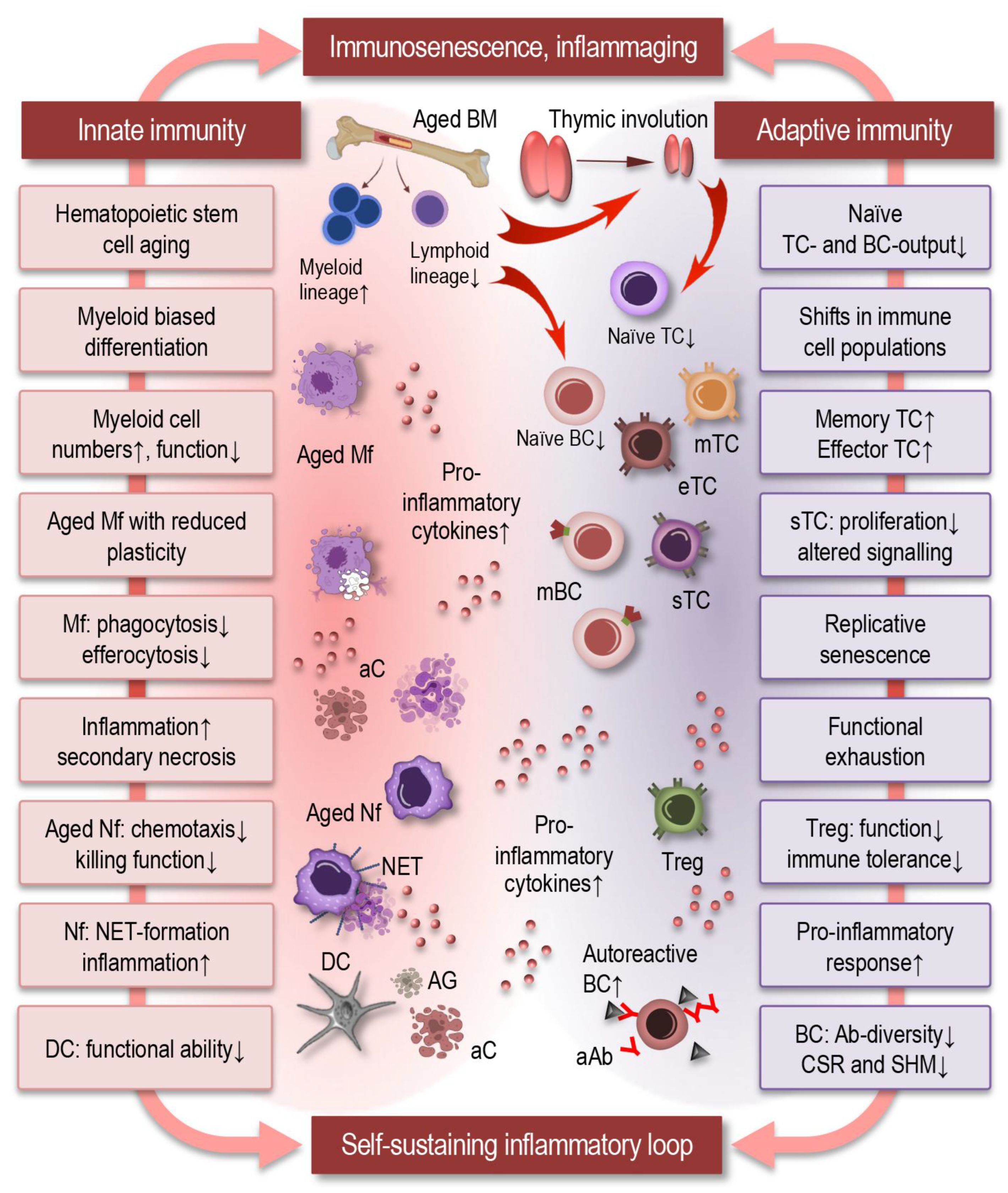
2. Hallmarks of Immune Dysfunction in Aging
2.1. Alterations in Innate Immunity
2.2. Remodeling of Adaptive Immunity
2.3. Systemic Consequences: Loss of Immune Homeostasis
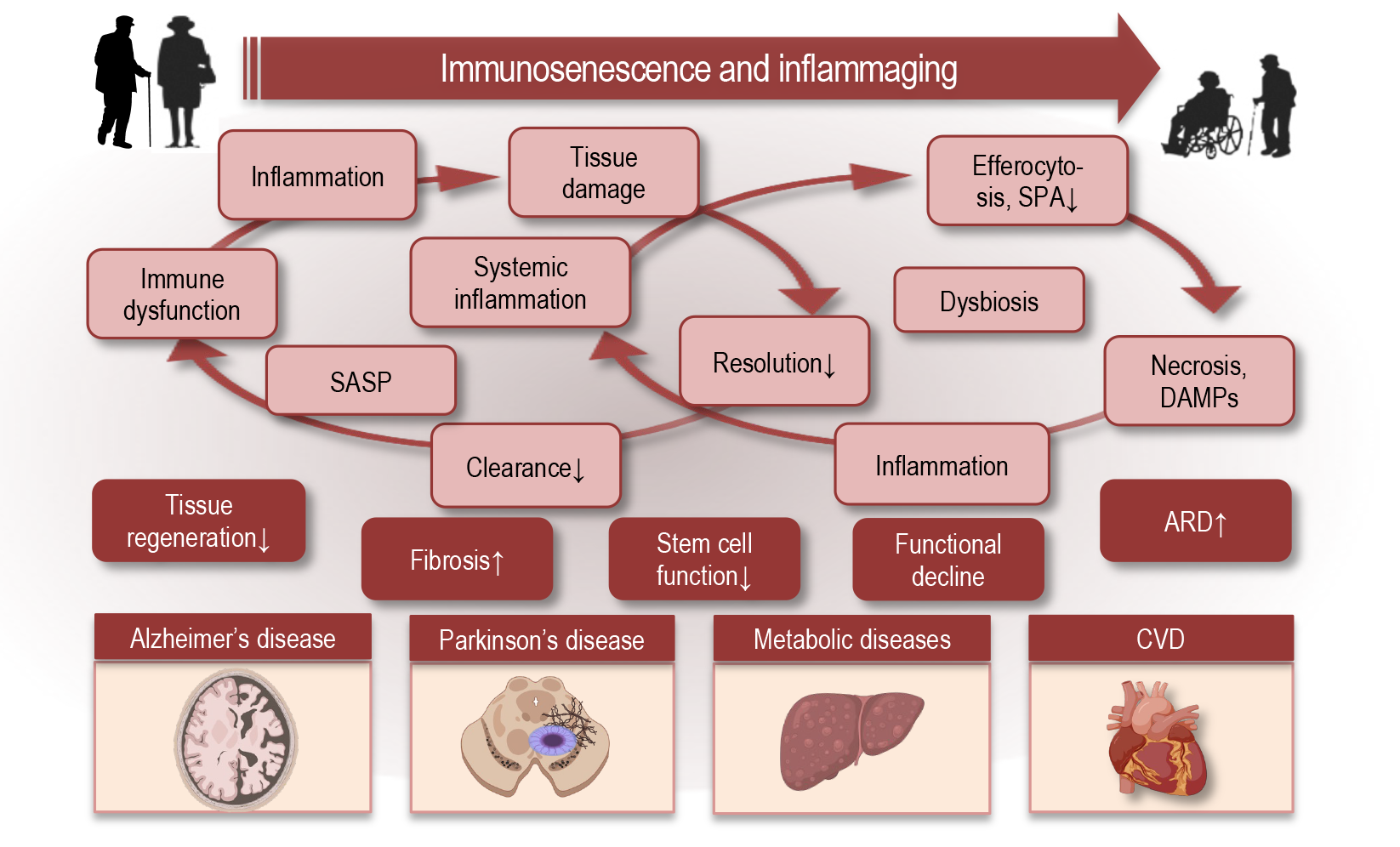
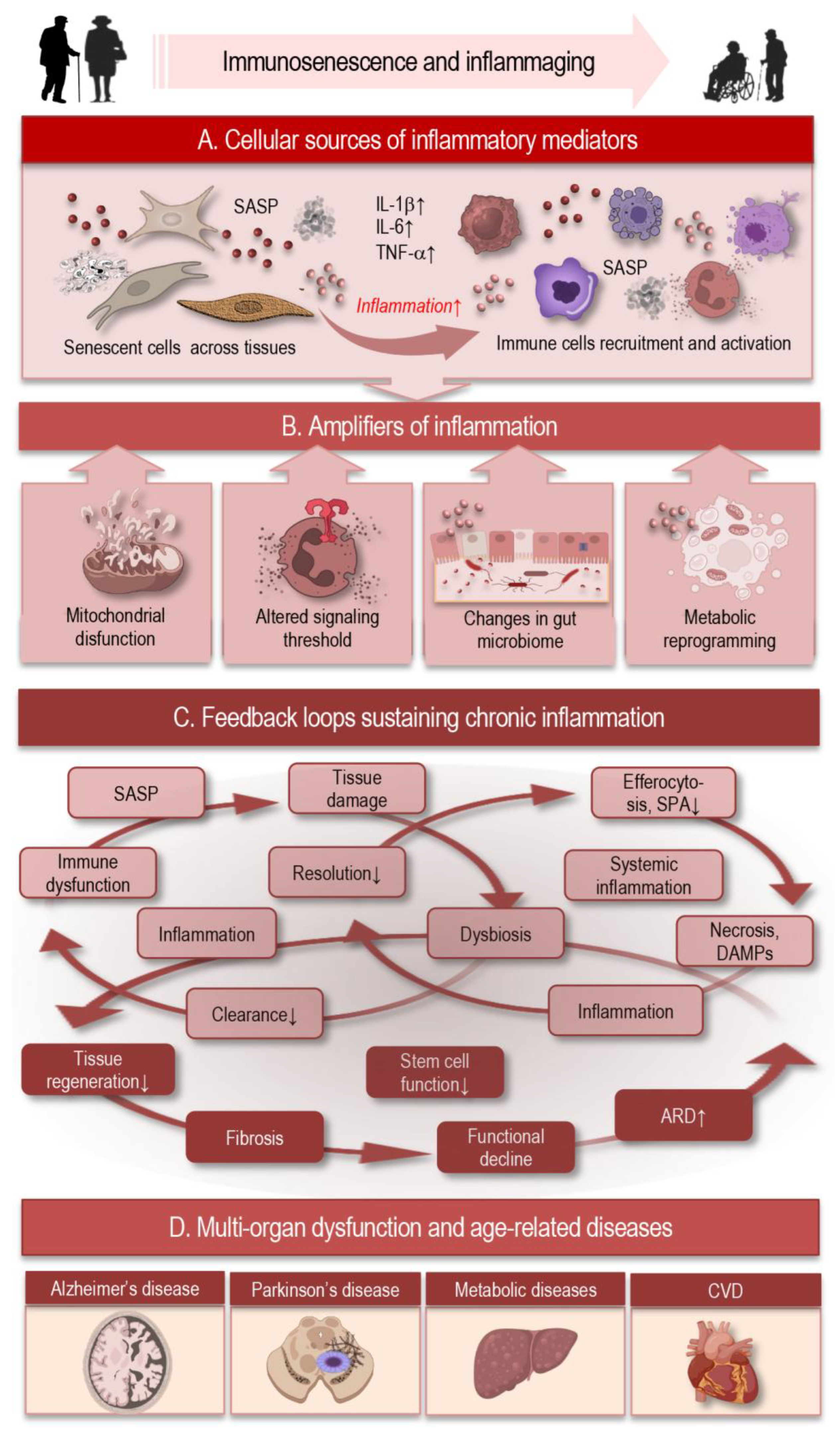
3. Inflammaging: Sources, Amplifiers, and Feedback Loops
3.1. Cellular Sources of Inflammatory Mediators
3.2. Amplifiers of Inflammation
3.3. Feedback Loops Sustaining Chronic Inflammation
3.4. Systemic and Organ-Level Consequences
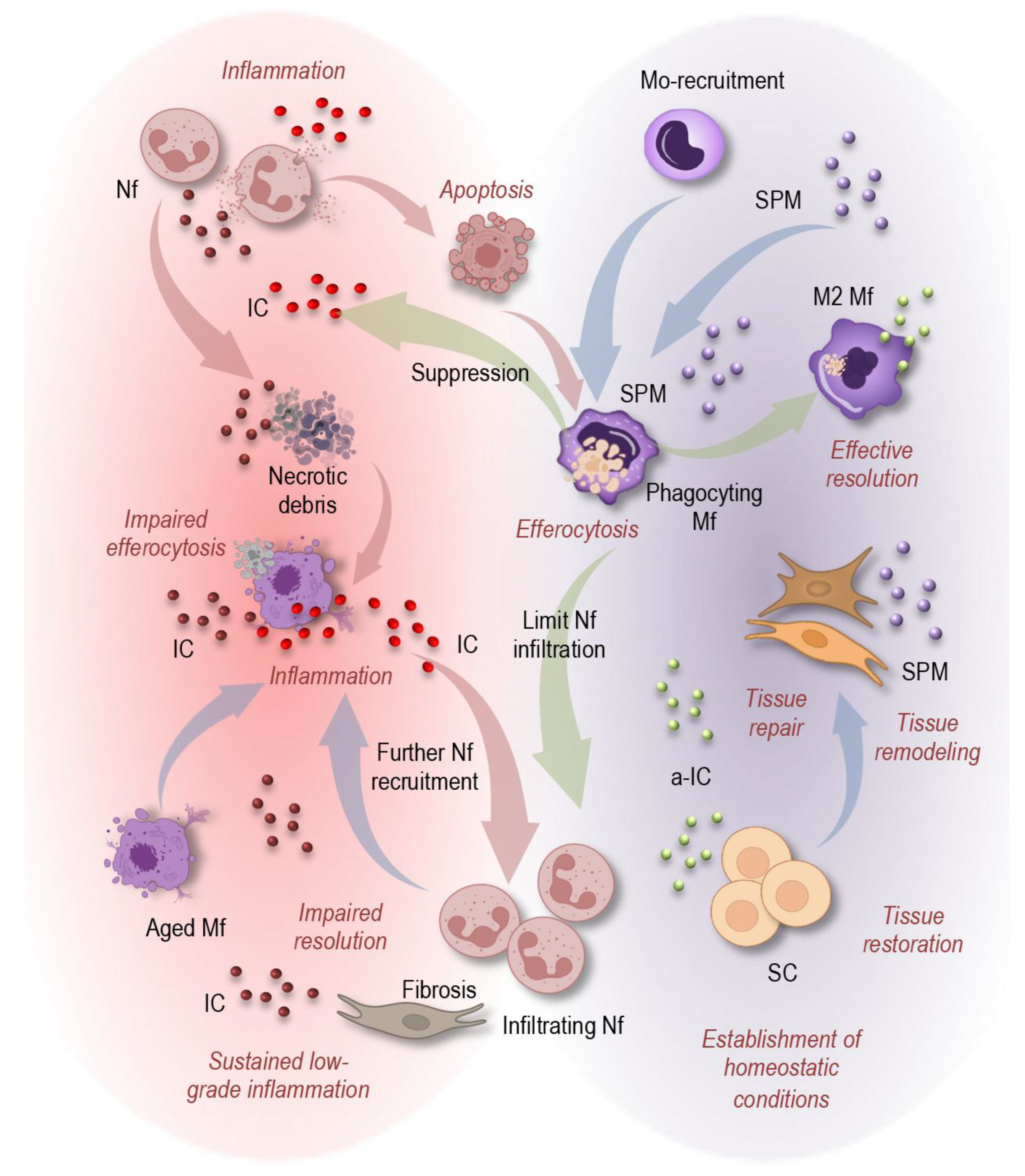
4. Impaired Resolution of Inflammation in Aging
4.1. Resolution as an Active, Regulated Process
4.2. Age-Related Defects in Resolution Pathways
4.3. Consequences of Failed Resolution
5. Defective Regeneration and Tissue Repair in the Aging Immune System
6. Immune Aging and the Pathophysiology of Age-Related Diseases
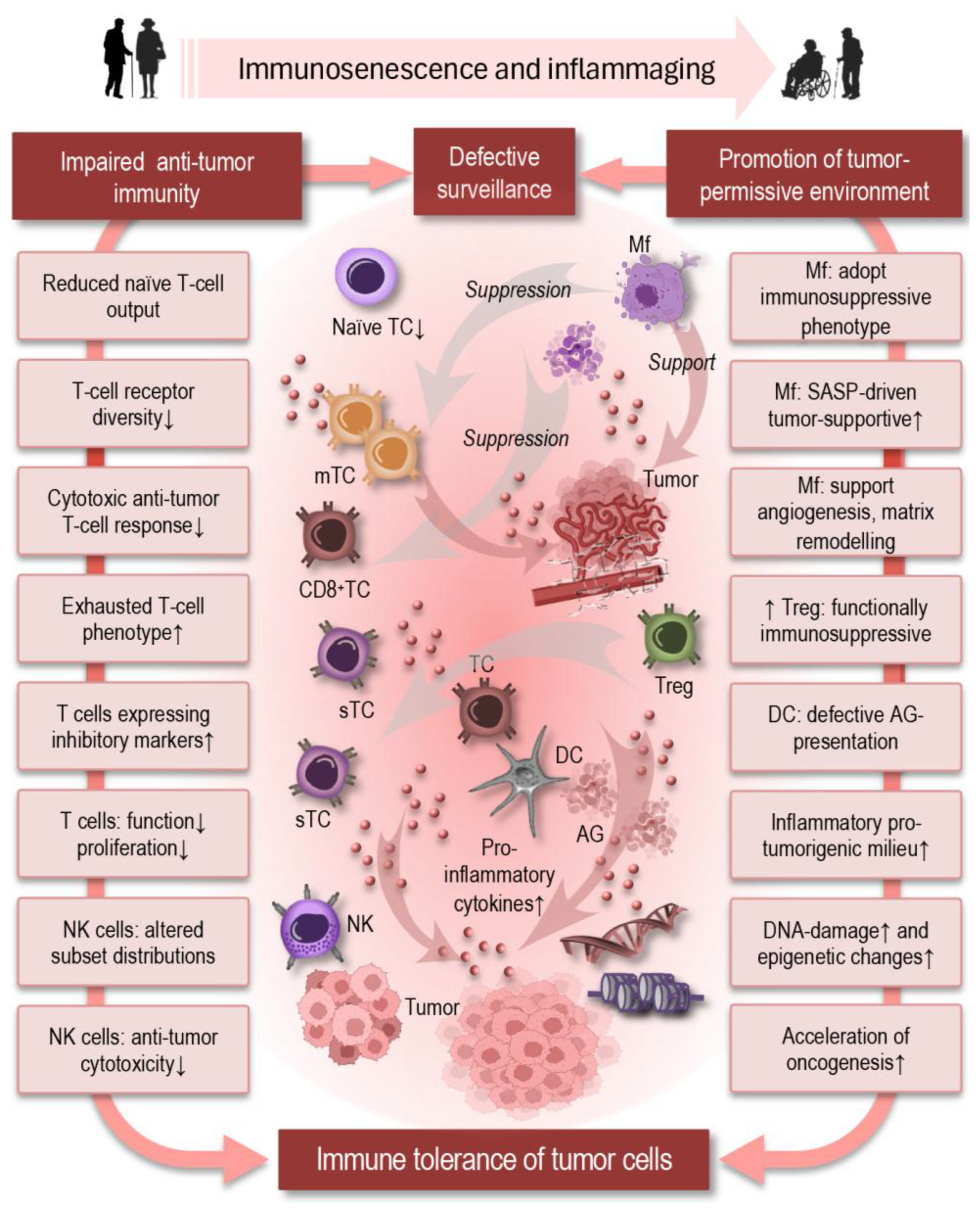
6.1. Cancer
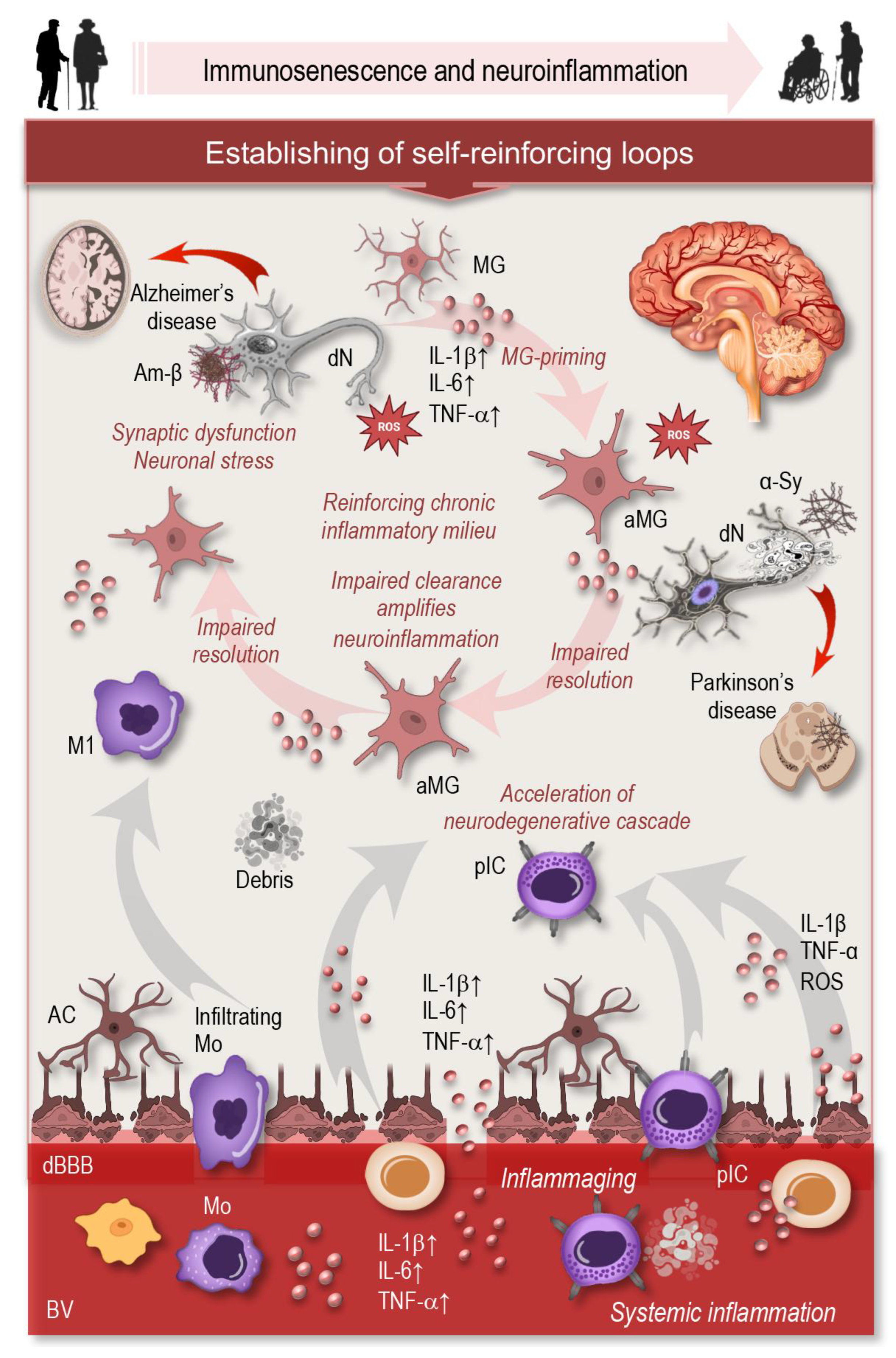
6.2. Neurodegenerative Disorders
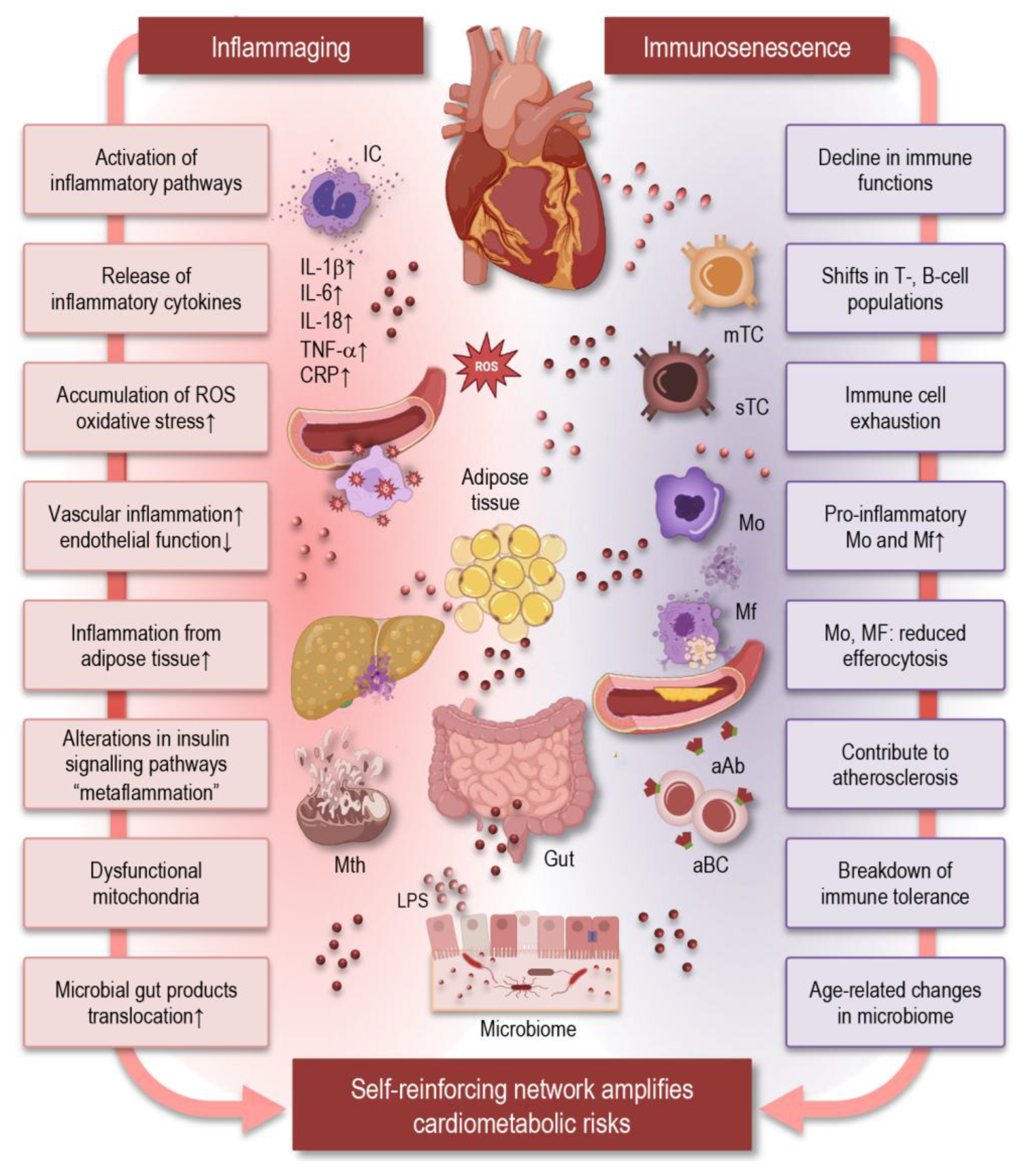
6.3. Cardiovascular and Metabolic Disorders
7. Therapeutic Strategies to Modulate Immune Aging
8. Systems-Level and Network Perspectives on Immune Aging
9. Knowledge Gaps and Future Directions
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goyani, P.; Christodoulou, R.; Vassiliou, E. Immunosenescence: Aging and Immune System Decline. Vaccines (Basel) 2024, 12. [Google Scholar] [CrossRef]
- Müller, L.; Fulop, T.; Pawelec, G. Immunosenescence in vertebrates and invertebrates. Immun Ageing 2013, 10, 12. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Campisi, J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J Gerontol A Biol Sci Med Sci 2014, 69 Suppl 1, S4–9. [Google Scholar] [CrossRef]
- Ajoolabady, A.; Pratico, D.; Tang, D.; Zhou, S.; Franceschi, C.; Ren, J. Immunosenescence and inflammaging: Mechanisms and role in diseases. Ageing Res Rev 2024, 101, 102540. [Google Scholar] [CrossRef]
- Fulop, T.; Larbi, A.; Dupuis, G.; Le Page, A.; Frost, E.H.; Cohen, A.A.; Witkowski, J.M.; Franceschi, C. Immunosenescence and Inflamm-Aging As Two Sides of the Same Coin: Friends or Foes? Front Immunol 2017, 8, 1960. [Google Scholar] [CrossRef]
- Müller, L.; Di Benedetto, S.; Pawelec, G. The Immune System and Its Dysregulation with Aging. Subcell Biochem 2019, 91, 21–43. [Google Scholar] [CrossRef] [PubMed]
- Di Benedetto, S.; Müller, L.; Wenger, E.; Duzel, S.; Pawelec, G. Contribution of neuroinflammation and immunity to brain aging and the mitigating effects of physical and cognitive interventions. Neurosci Biobehav Rev 2017, 75, 114–128. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Wang, B.; Alu, A.; Hong, W.; Lei, H.; He, X.; Shi, H.; Cheng, P.; Yang, X. Immunosenescence: signaling pathways, diseases and therapeutic targets. Signal Transduction and Targeted Therapy 2025, 10, 250. [Google Scholar] [CrossRef]
- Müller, L.; Di Benedetto, S. Aging brain: exploring the interplay between bone marrow aging, immunosenescence, and neuroinflammation. Front Immunol 2024, 15, 1393324. [Google Scholar] [CrossRef]
- Müller, L.; Di Benedetto, S. Inflammaging, immunosenescence, and cardiovascular aging: insights into long COVID implications. Front Cardiovasc Med 2024, 11, 1384996. [Google Scholar] [CrossRef]
- Pawelec, G. Hallmarks of human “immunosenescence”: adaptation or dysregulation? Immun Ageing 2012, 9, 15. [Google Scholar] [CrossRef] [PubMed]
- Müller, L.; Di Benedetto, S.; Müller, V. From Homeostasis to Neuroinflammation: Insights into Cellular and Molecular Interactions and Network Dynamics. Cells 2025, 14. [Google Scholar] [CrossRef]
- Teissier, T.; Boulanger, E.; Cox, L.S. Interconnections between Inflammageing and Immunosenescence during Ageing. Cells 2022, 11. [Google Scholar] [CrossRef]
- Santoro, A.; Bientinesi, E.; Monti, D. Immunosenescence and inflammaging in the aging process: age-related diseases or longevity? Ageing Res Rev 2021, 71, 101422. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Savill, J. Resolution of inflammation: the beginning programs the end. Nature Immunology 2005, 6, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Julliard, W.A.; Myo, Y.P.A.; Perelas, A.; Jackson, P.D.; Thatcher, T.H.; Sime, P.J. Specialized pro-resolving mediators as modulators of immune responses. Semin Immunol 2022, 59, 101605. [Google Scholar] [CrossRef]
- Serhan, C.N. Pro-resolving lipid mediators are leads for resolution physiology. Nature 2014, 510, 92–101. [Google Scholar] [CrossRef]
- Chiang, N.; Serhan, C.N. Specialized pro-resolving mediator network: an update on production and actions. Essays Biochem 2020, 64, 443–462. [Google Scholar] [CrossRef]
- Doyle, R.; Sadlier, D.M.; Godson, C. Pro-resolving lipid mediators: Agents of anti-ageing? Seminars in Immunology 2018, 40, 36–48. [Google Scholar] [CrossRef]
- Rymut, N.; Heinz, J.; Sadhu, S.; Hosseini, Z.; Riley, C.O.; Marinello, M.; Maloney, J.; MacNamara, K.C.; Spite, M.; Fredman, G. Resolvin D1 promotes efferocytosis in aging by limiting senescent cell-induced MerTK cleavage. FASEB J 2020, 34, 597–609. [Google Scholar] [CrossRef]
- Wynn, T.A.; Vannella, K.M. Macrophages in Tissue Repair, Regeneration, and Fibrosis. Immunity 2016, 44, 450–462. [Google Scholar] [CrossRef]
- Eming, S.A.; Wynn, T.A.; Martin, P. Inflammation and metabolism in tissue repair and regeneration. Science 2017, 356, 1026–1030. [Google Scholar] [CrossRef]
- Hams, E.; Bermingham, R.; Fallon, P.G. Macrophage and Innate Lymphoid Cell Interplay in the Genesis of Fibrosis. Front Immunol 2015, 6, 597. [Google Scholar] [CrossRef] [PubMed]
- Bektas, A.; Schurman, S.H.; Sen, R.; Ferrucci, L. Aging, inflammation and the environment. Exp Gerontol 2018, 105, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.Q.T.; Cho, K.A. Targeting immunosenescence and inflammaging: advancing longevity research. Exp Mol Med 2025, 57, 1881–1892. [Google Scholar] [CrossRef]
- Franceschi, C.; Garagnani, P.; Parini, P.; Giuliani, C.; Santoro, A. Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat Rev Endocrinol 2018, 14, 576–590. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Huo, T.; Lu, M.; Zhao, Y.; Zhang, J.; He, W.; Chen, H. Recent Advances in Aging and Immunosenescence: Mechanisms and Therapeutic Strategies. Cells 2025, 14. [Google Scholar] [CrossRef]
- Kovtonyuk, L.V.; Fritsch, K.; Feng, X.; Manz, M.G.; Takizawa, H. Inflamm-Aging of Hematopoiesis, Hematopoietic Stem Cells, and the Bone Marrow Microenvironment. Front Immunol 2016, 7, 502. [Google Scholar] [CrossRef]
- Bleve, A.; Motta, F.; Durante, B.; Pandolfo, C.; Selmi, C.; Sica, A. Immunosenescence, Inflammaging, and Frailty: Role of Myeloid Cells in Age-Related Diseases. Clin Rev Allergy Immunol 2023, 64, 123–144. [Google Scholar] [CrossRef]
- Sendama, W. The effect of ageing on the resolution of inflammation. Ageing Research Reviews 2020, 57, 101000. [Google Scholar] [CrossRef]
- Arnardottir, H.H.; Dalli, J.; Colas, R.A.; Shinohara, M.; Serhan, C.N. Aging delays resolution of acute inflammation in mice: reprogramming the host response with novel nano-proresolving medicines. J Immunol 2014, 193, 4235–4244. [Google Scholar] [CrossRef]
- Papayannopoulos, V. Neutrophil extracellular traps in immunity and disease. Nature Reviews Immunology 2018, 18, 134–147. [Google Scholar] [CrossRef]
- Mahbub, S.; Brubaker, A.L.; Kovacs, E.J. Aging of the Innate Immune System: An Update. Curr Immunol Rev 2011, 7, 104–115. [Google Scholar] [CrossRef]
- Müller, L.; Pawelec, G. As we age: Does slippage of quality control in the immune system lead to collateral damage? Ageing Res Rev 2015, 23, 116–123. [Google Scholar] [CrossRef]
- Solana, R.; Tarazona, R.; Gayoso, I.; Lesur, O.; Dupuis, G.; Fulop, T. Innate immunosenescence: effect of aging on cells and receptors of the innate immune system in humans. Semin Immunol 2012, 24, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Kim, S.J.; Lei, Y.; Wang, S.; Wang, H.; Huang, H.; Zhang, H.; Tsung, A. Neutrophil extracellular traps in homeostasis and disease. Signal Transduction and Targeted Therapy 2024, 9, 235. [Google Scholar] [CrossRef]
- Di Benedetto, S.; Derhovanessian, E.; Steinhagen-Thiessen, E.; Goldeck, D.; Müller, L.; Pawelec, G. Impact of age, sex and CMV-infection on peripheral T cell phenotypes: results from the Berlin BASE-II Study. Biogerontology 2015, 16, 631–643. [Google Scholar] [CrossRef]
- Thomas, R.; Wang, W.; Su, D.M. Contributions of Age-Related Thymic Involution to Immunosenescence and Inflammaging. Immun Ageing 2020, 17, 2. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y. Immune Aging and Its Implication for Age-Related Disease Progression. Physiology 2025, 40, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Czesnikiewicz-Guzik, M.; Lee, W.W.; Cui, D.; Hiruma, Y.; Lamar, D.L.; Yang, Z.Z.; Ouslander, J.G.; Weyand, C.M.; Goronzy, J.J. T cell subset-specific susceptibility to aging. Clin Immunol 2008, 127, 107–118. [Google Scholar] [CrossRef]
- Moller, S.H.; Hsueh, P.C.; Yu, Y.R.; Zhang, L.; Ho, P.C. Metabolic programs tailor T cell immunity in viral infection, cancer, and aging. Cell Metab 2022, 34, 378–395. [Google Scholar] [CrossRef]
- Raynor, J.; Lages, C.S.; Shehata, H.; Hildeman, D.A.; Chougnet, C.A. Homeostasis and function of regulatory T cells in aging. Curr Opin Immunol 2012, 24, 482–487. [Google Scholar] [CrossRef]
- Ratliff, M.; Alter, S.; Frasca, D.; Blomberg, B.B.; Riley, R.L. In senescence, age-associated B cells secrete TNFalpha and inhibit survival of B-cell precursors. Aging Cell 2013, 12, 303–311. [Google Scholar] [CrossRef]
- Weksler, M.E. Changes in the B-cell repertoire with age. Vaccine 2000, 18, 1624–1628. [Google Scholar] [CrossRef]
- Frasca, D.; Diaz, A.; Romero, M.; Garcia, D.; Blomberg, B.B. B Cell Immunosenescence. Annu Rev Cell Dev Biol 2020, 36, 551–574. [Google Scholar] [CrossRef]
- Huntington, N.D.; Gray, D.H. Immune homeostasis in health and disease. Immunol Cell Biol 2018, 96, 451–452. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Mu, W.C.; Markov, N.T.; Fuentealba, M.; Halaweh, H.; Senchyna, F.; Manwaring-Mueller, M.N.; Winer, D.A.; Furman, D. Immunological biomarkers of aging. J Immunol 2025, 214, 889–902. [Google Scholar] [CrossRef] [PubMed]
- Müller, L.; Di Benedetto, S. Immunosenescence and inflammaging: Mechanisms and modulation through diet and lifestyle. Front Immunol 2025, 16, 1708280. [Google Scholar] [CrossRef] [PubMed]
- Fernandez Maestre, I.; Harris, A.S.; Amor, C. Aging and immunity: the age-old tango. Genes Dev 2025, 39, 948–974. [Google Scholar] [CrossRef]
- Franceschi, C.; Olivieri, F.; Moskalev, A.; Ivanchenko, M.; Santoro, A. Toward precision interventions and metrics of inflammaging. Nature Aging 2025, 5, 1441–1454. [Google Scholar] [CrossRef]
- Kamroo, A.; Kakroudi, M.H.; Sarmadian, A.J.; Firouzabadi, A.; Mousavi, S.; Yazdanpanah, N.; Saleki, K.; Rezaei, N. Immunosenescence and organoids: pathophysiology and therapeutic opportunities. Immunity & Ageing 2025, 22, 46. [Google Scholar] [CrossRef] [PubMed]
- Karpuzoglu, E.; Holladay, S.D.; Gogal, R.M., Jr. Inflammaging: triggers, molecular mechanisms, immunological consequences, sex differences, and cutaneous manifestations. Front Immunol 2025, 16, 1704203. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Loygorri, J.I.; Villarejo-Zori, B.; Viedma-Poyatos, A.; Zapata-Munoz, J.; Benitez-Fernandez, R.; Frutos-Lison, M.D.; Tomas-Barberan, F.A.; Espin, J.C.; Area-Gomez, E.; Gomez-Duran, A.; et al. Mitophagy curtails cytosolic mtDNA-dependent activation of cGAS/STING inflammation during aging. Nat Commun 2024, 15, 830. [Google Scholar] [CrossRef]
- Gulen, M.F.; Samson, N.; Keller, A.; Schwabenland, M.; Liu, C.; Glück, S.; Thacker, V.V.; Favre, L.; Mangeat, B.; Kroese, L.J.; et al. cGAS–STING drives ageing-related inflammation and neurodegeneration. Nature 2023, 620, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Hu, Z.; Xu, D.; Bian, H.; Lv, J.; Zhu, X.; Zhang, Q.; Su, L.; Yin, H.; Lu, T.; et al. STING signaling in inflammaging: a new target against musculoskeletal diseases. Front Immunol 2023, 14, 1227364. [Google Scholar] [CrossRef]
- Li, X.; Li, C.; Zhang, W.; Wang, Y.; Qian, P.; Huang, H. Inflammation and aging: signaling pathways and intervention therapies. Signal Transduction and Targeted Therapy 2023, 8, 239. [Google Scholar] [CrossRef]
- Shaw, A.C.; Joshi, S.; Greenwood, H.; Panda, A.; Lord, J.M. Aging of the innate immune system. Curr Opin Immunol 2010, 22, 507–513. [Google Scholar] [CrossRef]
- Caetano-Silva, M.E.; Shrestha, A.; Duff, A.F.; Kontic, D.; Brewster, P.C.; Kasperek, M.C.; Lin, C.H.; Wainwright, D.A.; Hernandez-Saavedra, D.; Woods, J.A.; et al. Aging amplifies a gut microbiota immunogenic signature linked to heightened inflammation. Aging Cell 2024, 23, e14190. [Google Scholar] [CrossRef]
- Strasser, B.; Ticinesi, A. Intestinal microbiome in normal ageing, frailty and cognition decline. Curr Opin Clin Nutr Metab Care 2023, 26, 8–16. [Google Scholar] [CrossRef]
- Müller, L.; Di Benedetto, S. Bridging the brain and gut: neuroimmune mechanisms of neuroinflammation and therapeutic insights. Front Cell Neurosci 2025, 19, 1590002. [Google Scholar] [CrossRef]
- Wen, N.N.; Sun, L.W.; Geng, Q.; Zheng, G.H. Gut microbiota changes associated with frailty in older adults: A systematic review of observational studies. World J Clin Cases 2024, 12, 6815–6825. [Google Scholar] [CrossRef]
- Xu, Y.; Mao, T.; Wang, Y.; Qi, X.; Zhao, W.; Chen, H.; Zhang, C.; Li, X. Effect of Gut Microbiota-Mediated Tryptophan Metabolism on Inflammaging in Frailty and Sarcopenia. J Gerontol A Biol Sci Med Sci 2024, 79. [Google Scholar] [CrossRef]
- Maurmann, R.M.; Schmitt, B.L.; Mosalmanzadeh, N.; Pence, B.D. Mitochondrial dysfunction at the cornerstone of inflammatory exacerbation in aged macrophages. Explor Immunol 2023, 3, 442–452. [Google Scholar] [CrossRef]
- Ginefra, P.; Hope, H.C.; Lorusso, G.; D’Amelio, P.; Vannini, N. The immunometabolic roots of aging. Current Opinion in Immunology 2024, 91, 102498. [Google Scholar] [CrossRef]
- Müller, L.; Di Benedetto, S.; Müller, V. The dual nature of neuroinflammation in networked brain. Front Immunol 2025, 16, 1659947. [Google Scholar] [CrossRef]
- Whittington, R.A.; Planel, E.; Terrando, N. Impaired Resolution of Inflammation in Alzheimer’s Disease: A Review. Front Immunol 2017, 8, 1464. [Google Scholar] [CrossRef] [PubMed]
- Conway, J.; A.D., N. Ageing of the gut microbiome: Potential influences on immune senescence and inflammageing. Ageing Res Rev 2021, 68, 101323. [Google Scholar] [CrossRef] [PubMed]
- Kalyanaraman, B.; Cheng, G.; Hardy, M. Gut microbiome, short-chain fatty acids, alpha-synuclein, neuroinflammation, and ROS/RNS: Relevance to Parkinson’s disease and therapeutic implications. Redox Biol 2024, 71, 103092. [Google Scholar] [CrossRef]
- Kohler, C.A.; Maes, M.; Slyepchenko, A.; Berk, M.; Solmi, M.; Lanctot, K.L.; Carvalho, A.F. The Gut-Brain Axis, Including the Microbiome, Leaky Gut and Bacterial Translocation: Mechanisms and Pathophysiological Role in Alzheimer’s Disease. Curr Pharm Des 2016, 22, 6152–6166. [Google Scholar] [CrossRef] [PubMed]
- O’Riordan, K.J.; Moloney, G.M.; Keane, L.; Clarke, G.; Cryan, J.F. The gut microbiota-immune-brain axis: Therapeutic implications. Cell Rep Med 2025, 6, 101982. [Google Scholar] [CrossRef]
- Nirenjen, S.; Narayanan, J.; Tamilanban, T.; Subramaniyan, V.; Chitra, V.; Fuloria, N.K.; Wong, L.S.; Ramachawolran, G.; Sekar, M.; Gupta, G.; et al. Exploring the contribution of pro-inflammatory cytokines to impaired wound healing in diabetes. Front Immunol 2023, 14, 1216321. [Google Scholar] [CrossRef]
- Josephson, A.M.; Bradaschia-Correa, V.; Lee, S.; Leclerc, K.; Patel, K.S.; Muinos Lopez, E.; Litwa, H.P.; Neibart, S.S.; Kadiyala, M.; Wong, M.Z.; et al. Age-related inflammation triggers skeletal stem/progenitor cell dysfunction. Proc Natl Acad Sci U S A 2019, 116, 6995–7004. [Google Scholar] [CrossRef]
- Riparini, G.; Mackenzie, M.; Naz, F.; Brooks, S.; Jiang, K.; Deewan, A.; Dulek, B.; Islam, S.; Ko, K.D.; Tsai, W.L.; et al. Epigenetic dysregulation in aged muscle stem cells drives mesenchymal progenitor expansion via IL-6 and Spp1 signaling. Nature Aging 2025, 5, 2399–2416. [Google Scholar] [CrossRef]
- Mlynarska, E.; Kowalik, A.; Krajewska, A.; Krupinska, N.; Marcinkowska, W.; Motor, J.; Przybylak, A.; Tlustochowicz, K.; Rysz, J.; Franczyk, B. Inflammaging and Senescence-Driven Extracellular Matrix Remodeling in Age-Associated Cardiovascular Disease. Biomolecules 2025, 15. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro-Machado, E.; Gurgul-Convey, E.; Marzec, M.T. Immunometabolism in type 2 diabetes mellitus: tissue-specific interactions. Arch Med Sci 2023, 19, 895–911. [Google Scholar] [CrossRef]
- Meier, H.C.S.; Mitchell, C.; Karadimas, T.; Faul, J.D. Systemic inflammation and biological aging in the Health and Retirement Study. GeroScience 2023, 45, 3257–3265. [Google Scholar] [CrossRef] [PubMed]
- Spray, L.; Richardson, G.; Haendeler, J.; Altschmied, J.; Rumampouw, V.; Wallis, S.B.; Georgiopoulos, G.; White, S.; Unsworth, A.; Stellos, K.; et al. Cardiovascular inflammaging: Mechanisms, consequences, and therapeutic perspectives. Cell Reports Medicine 2025, 6, 102264. [Google Scholar] [CrossRef]
- Rohm, T.V.; Meier, D.T.; Olefsky, J.M.; Donath, M.Y. Inflammation in obesity, diabetes, and related disorders. Immunity 2022, 55, 31–55. [Google Scholar] [CrossRef] [PubMed]
- Abdelhamed, H.G.; Hassan, A.A.; Sakraan, A.A.; Al-Deeb, R.T.; Mousa, D.M.; Aboul Ezz, H.S.; Noor, N.A.; Khadrawy, Y.A.; Radwan, N.M. Brain interleukins and Alzheimer’s disease. Metab Brain Dis 2025, 40, 116. [Google Scholar] [CrossRef]
- Carr, L.; Mustafa, S.; Collins-Praino, L.E. The Hallmarks of Ageing in Microglia. Cell Mol Neurobiol 2025, 45, 45. [Google Scholar] [CrossRef]
- Antuna, E.; Cachan-Vega, C.; Bermejo-Millo, J.C.; Potes, Y.; Caballero, B.; Vega-Naredo, I.; Coto-Montes, A.; Garcia-Gonzalez, C. Inflammaging: Implications in Sarcopenia. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Damanti, S.; Senini, E.; De Lorenzo, R.; Merolla, A.; Santoro, S.; Festorazzi, C.; Messina, M.; Vitali, G.; Sciorati, C.; Manfredi, A.A.; et al. Molecular constraints of sarcopenia in the ageing muscle. Front Aging 2025, 6, 1588014. [Google Scholar] [CrossRef]
- Panezai, J.; Van Dyke, T.E. Resolution of inflammation: Intervention strategies and future applications. Toxicol Appl Pharmacol 2022, 449, 116089. [Google Scholar] [CrossRef] [PubMed]
- Perez-Hernandez, J.; Chiurchiu, V.; Perruche, S.; You, S. Regulation of T-Cell Immune Responses by Pro-Resolving Lipid Mediators. Front Immunol 2021, 12, 768133. [Google Scholar] [CrossRef] [PubMed]
- Peh, H.Y.; Chen, J. Pro-resolving lipid mediators and therapeutic innovations in resolution of inflammation. Pharmacol Ther 2025, 265, 108753. [Google Scholar] [CrossRef]
- Boucher, D.M.; Robichaud, S.; Lorant, V.; Leon, J.S.; Suliman, I.; Rasheed, A.; Susser, L.I.; Emerton, C.; Geoffrion, M.; De Jong, E.; et al. Age-Related Impairments in Immune Cell Efferocytosis and Autophagy Hinder Atherosclerosis Regression. Arterioscler Thromb Vasc Biol 2025, 45, 481–495. [Google Scholar] [CrossRef]
- Doran, A.C. Inflammation Resolution: Implications for Atherosclerosis. Circ Res 2022, 130, 130–148. [Google Scholar] [CrossRef] [PubMed]
- Nathan, C.; Ding, A. Nonresolving inflammation. Cell 2010, 140, 871–882. [Google Scholar] [CrossRef]
- Dalli, J.; Colas, R.A.; Serhan, C.N. Novel n-3 Immunoresolvents: Structures and Actions. Scientific Reports 2013, 3, 1940. [Google Scholar] [CrossRef]
- Marques, R.M.; Gonzalez-Nunez, M.; Walker, M.E.; Gomez, E.A.; Colas, R.A.; Montero-Melendez, T.; Perretti, M.; Dalli, J. Loss of 15-lipoxygenase disrupts Treg differentiation altering their pro-resolving functions. Cell Death & Differentiation 2021, 28, 3140–3160. [Google Scholar] [CrossRef]
- Yue, Z.; Nie, L.; Zhang, P.; Chen, Q.; Lv, Q.; Wang, Q. Tissue-resident macrophage inflammaging aggravates homeostasis dysregulation in age-related diseases. Cell Immunol 2021, 361, 104278. [Google Scholar] [CrossRef]
- Li, N.; Chen, Y.; Wang, Q.; Liu, X.; Han, C.; Qu, C.; Guan, X.; Zou, W.; Wang, X.; Li, A.; et al. Microenvironment-driven satellite cell regeneration and repair in aging-related sarcopenia: mechanisms and therapeutic frontiers. Stem Cell Res Ther 2025, 16, 545. [Google Scholar] [CrossRef]
- Kushioka, J.; Chow, S.K.; Toya, M.; Tsubosaka, M.; Shen, H.; Gao, Q.; Li, X.; Zhang, N.; Goodman, S.B. Bone regeneration in inflammation with aging and cell-based immunomodulatory therapy. Inflamm Regen 2023, 43, 29. [Google Scholar] [CrossRef]
- Rahman, F.A.; Angus, S.A.; Stokes, K.; Karpowicz, P.; Krause, M.P. Impaired ECM Remodeling and Macrophage Activity Define Necrosis and Regeneration Following Damage in Aged Skeletal Muscle. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Duong, L.; Radley, H.G.; Lee, B.; Dye, D.E.; Pixley, F.J.; Grounds, M.D.; Nelson, D.J.; Jackaman, C. Macrophage function in the elderly and impact on injury repair and cancer. Immunity & Ageing 2021, 18, 4. [Google Scholar] [CrossRef]
- Singh, A.; Schurman, S.H.; Bektas, A.; Kaileh, M.; Roy, R.; Wilson, D.M., 3rd; Sen, R.; Ferrucci, L. Aging and Inflammation. Cold Spring Harb Perspect Med 2024, 14. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Pulido, S.; Yousefzadeh, M.J.; Mittelbrunn, M. Aging reshapes the adaptive immune system from healer to saboteur. Nat Aging 2025, 5, 1393–1403. [Google Scholar] [CrossRef]
- Salminen, A. Increased immunosuppression impairs tissue homeostasis with aging and age-related diseases. Journal of Molecular Medicine 2021, 99, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Tang, D. Immunosenescence promotes cancer development: from mechanisms to treatment strategies. Cell Communication and Signaling 2025, 23, 128. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.S.; Burton Sojo, G.; Sun, H.; Friedland, B.N.; McNamara, M.E.; Schmidt, M.O.; Wellstein, A. The Role of Aging and Senescence in Immune Checkpoint Inhibitor Response and Toxicity. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef]
- Ikeda, H.; Togashi, Y. Aging, cancer, and antitumor immunity. International Journal of Clinical Oncology 2022, 27, 316–322. [Google Scholar] [CrossRef]
- Lian, J.; Yue, Y.; Yu, W.; Zhang, Y. Immunosenescence: a key player in cancer development. Journal of Hematology & Oncology 2020, 13, 151. [Google Scholar] [CrossRef]
- Liu, Q.; Li, J.; Sun, X.; Lin, J.; Yu, Z.; Xiao, Y.; Li, D.; Sun, B.; Bao, H.; Liu, Y. Immunosenescence and cancer: molecular hallmarks, tumor microenvironment remodeling, and age-specific immunotherapy challenges. J Hematol Oncol 2025, 18, 81. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Qin, P.; Li, F.; Zhu, H.; You, H.; Zhang, Y.; Xu, B.; Li, T.; Zhang, F.; Han, L.; et al. The inter-link of ageing, cancer and immunity: findings from real-world retrospective study. Immunity & Ageing 2023, 20, 75. [Google Scholar] [CrossRef]
- Gao, D.; Kan, P.; He, Y.; Sun, S.; Tang, L.; Yang, F. Senescent immune cells in the tumor microenvironment: emerging insights into cancer immunotherapy resistance. Front Immunol 2025, 16, 1656733. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.; Cheng, J. Cellular Senescence and Immunosenescence in Melanoma: Insights From the Tumor Microenvironment. Cancer Med 2025, 14, e71223. [Google Scholar] [CrossRef] [PubMed]
- Norden, D.M.; Muccigrosso, M.M.; Godbout, J.P. Microglial priming and enhanced reactivity to secondary insult in aging, and traumatic CNS injury, and neurodegenerative disease. Neuropharmacology 2015, 96, 29–41. [Google Scholar] [CrossRef]
- Gao, C.; Jiang, J.; Tan, Y.; Chen, S. Microglia in neurodegenerative diseases: mechanism and potential therapeutic targets. Signal Transduction and Targeted Therapy 2023, 8, 359. [Google Scholar] [CrossRef]
- Alkhalifa, A.E.; Al-Ghraiybah, N.F.; Odum, J.; Shunnarah, J.G.; Austin, N.; Kaddoumi, A. Blood-Brain Barrier Breakdown in Alzheimer’s Disease: Mechanisms and Targeted Strategies. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Takata, F.; Nakagawa, S.; Matsumoto, J.; Dohgu, S. Blood-Brain Barrier Dysfunction Amplifies the Development of Neuroinflammation: Understanding of Cellular Events in Brain Microvascular Endothelial Cells for Prevention and Treatment of BBB Dysfunction. Front Cell Neurosci 2021, 15, 661838. [Google Scholar] [CrossRef]
- Müller, L.; Di Benedetto, S. Neuroimmune crosstalk in chronic neuroinflammation: microglial interactions and immune modulation. Front Cell Neurosci 2025, 19, 1575022. [Google Scholar] [CrossRef]
- Ajoolabady, A.; Pratico, D.; Vinciguerra, M.; Lip, G.Y.H.; Franceschi, C.; Ren, J. Inflammaging: mechanisms and role in the cardiac and vasculature. Trends Endocrinol Metab 2023, 34, 373–387. [Google Scholar] [CrossRef]
- Barcena, M.L.; Aslam, M.; Pozdniakova, S.; Norman, K.; Ladilov, Y. Cardiovascular Inflammaging: Mechanisms and Translational Aspects. Cells 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Snijckers, R.P.M.; Foks, A.C. Adaptive immunity and atherosclerosis: aging at its crossroads. Front Immunol 2024, 15, 1350471. [Google Scholar] [CrossRef]
- Acierno, C.; Frontuto, M.; De Stefano, G.F.; Erezanu, A.; Limone, A.; Morella, S.; Picaro, F.; Palazzo, D.; Gilio, M. From Steatosis to Immunosenescence: The Impact of Metabolic Dysfunction on Immune Aging in HIV and Non-HIV Populations. Biomedicines 2025, 13. [Google Scholar] [CrossRef] [PubMed]
- Mapuskar, K.A.; London, B.; Zacharias, Z.R.; Houtman, J.C.D.; Allen, B.G. Immunometabolism in the Aging Heart. J Am Heart Assoc 2025, 14, e039216. [Google Scholar] [CrossRef]
- Spray, L.; Richardson, G.; Haendeler, J.; Altschmied, J.; Rumampouw, V.; Wallis, S.B.; Georgiopoulos, G.; White, S.; Unsworth, A.; Stellos, K.; et al. Cardiovascular inflammaging: Mechanisms, consequences, and therapeutic perspectives. Cell Rep Med 2025, 6, 102264. [Google Scholar] [CrossRef]
- Laberge, R.M.; Sun, Y.; Orjalo, A.V.; Patil, C.K.; Freund, A.; Zhou, L.; Curran, S.C.; Davalos, A.R.; Wilson-Edell, K.A.; Liu, S.; et al. Author Correction: MTOR regulates the pro-tumorigenic senescence-associated secretory phenotype by promoting IL1A translation. Nat Cell Biol 2021, 23, 564–565. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.S.; Huang, Y.W.; Lee, T.H.; Chang, L.; Huang, Z.D.; Wu, T.Y.; Wang, T.J.; Ho, C.S. Rapamycin Alleviates Protein Aggregates, Reduces Neuroinflammation, and Rescues Demyelination in Globoid Cell Leukodystrophy. Cells 2023, 12. [Google Scholar] [CrossRef]
- Wang, F.; Song, Y.; Liu, P.; Ma, F.; Peng, Z.; Pang, Y.; Hu, H.; Zeng, L.; Luo, H.; Zhang, X. Rapamycin suppresses neuroinflammation and protects retinal ganglion cell loss after optic nerve crush. Int Immunopharmacol 2023, 119, 110171. [Google Scholar] [CrossRef]
- Hieber, C.; Grabbe, S.; Bros, M. Counteracting Immunosenescence-Which Therapeutic Strategies Are Promising? Biomolecules 2023, 13. [Google Scholar] [CrossRef]
- Kiss, T.; Nyul-Toth, A.; DelFavero, J.; Balasubramanian, P.; Tarantini, S.; Faakye, J.; Gulej, R.; Ahire, C.; Ungvari, A.; Yabluchanskiy, A.; et al. Spatial transcriptomic analysis reveals inflammatory foci defined by senescent cells in the white matter, hippocampi and cortical grey matter in the aged mouse brain. Geroscience 2022, 44, 661–681. [Google Scholar] [CrossRef] [PubMed]
- Kirkland, J.L.; Tchkonia, T. Senolytic drugs: from discovery to translation. J Intern Med 2020, 288, 518–536. [Google Scholar] [CrossRef] [PubMed]
- Hickson, L.J.; Langhi Prata, L.G.P.; Bobart, S.A.; Evans, T.K.; Giorgadze, N.; Hashmi, S.K.; Herrmann, S.M.; Jensen, M.D.; Jia, Q.; Jordan, K.L.; et al. Senolytics decrease senescent cells in humans: Preliminary report from a clinical trial of Dasatinib plus Quercetin in individuals with diabetic kidney disease. EBioMedicine 2019, 47, 446–456. [Google Scholar] [CrossRef]
- Burdusel, D.; Doeppner, T.R.; Surugiu, R.; Hermann, D.M.; Olaru, D.G.; Popa-Wagner, A. The Intersection of Epigenetics and Senolytics in Mechanisms of Aging and Therapeutic Approaches. Biomolecules 2024, 15. [Google Scholar] [CrossRef]
- De Maeyer, R.P.H.; van de Merwe, R.C.; Louie, R.; Bracken, O.V.; Devine, O.P.; Goldstein, D.R.; Uddin, M.; Akbar, A.N.; Gilroy, D.W. Blocking elevated p38 MAPK restores efferocytosis and inflammatory resolution in the elderly. Nat Immunol 2020, 21, 615–625. [Google Scholar] [CrossRef]
- Raza, A.; Crothers, J.W.; McGill, M.M.; Mawe, G.M.; Teuscher, C.; Krementsov, D.N. Anti-inflammatory roles of p38alpha MAPK in macrophages are context dependent and require IL-10. J Leukoc Biol 2017, 102, 1219–1227. [Google Scholar] [CrossRef] [PubMed]
- Dhapola, R.; Hota, S.S.; Sarma, P.; Bhattacharyya, A.; Medhi, B.; Reddy, D.H. Recent advances in molecular pathways and therapeutic implications targeting neuroinflammation for Alzheimer’s disease. Inflammopharmacology 2021, 29, 1669–1681. [Google Scholar] [CrossRef]
- Patel, Y.; Manturthi, S.; Tiwari, S.; Gahunia, E.; Courtemanche, A.; Gandelman, M.; Cote, M.; Gadde, S. Development of Pro-resolving and Pro-efferocytic Nanoparticles for Atherosclerosis Therapy. ACS Pharmacol Transl Sci 2024, 7, 3086–3095. [Google Scholar] [CrossRef]
- Wang, X.; Guo, D.; He, C.; Wang, X.; Wei, Y.; Zhang, F.; Wang, L.; Yang, Y. Clinical application of mesenchymal stem cells in immunosenescence: a qualitative review of their potential and challenges. Stem Cell Research & Therapy 2025, 16, 265. [Google Scholar] [CrossRef]
- Han, X.; Liao, R.; Li, X.; Zhang, C.; Huo, S.; Qin, L.; Xiong, Y.; He, T.; Xiao, G.; Zhang, T. Mesenchymal stem cells in treating human diseases: molecular mechanisms and clinical studies. Signal Transduct Target Ther 2025, 10, 262. [Google Scholar] [CrossRef]
- Song, N.; Scholtemeijer, M.; Shah, K. Mesenchymal Stem Cell Immunomodulation: Mechanisms and Therapeutic Potential. Trends Pharmacol Sci 2020, 41, 653–664. [Google Scholar] [CrossRef]
- Muller, L.; Tunger, A.; Wobus, M.; von Bonin, M.; Towers, R.; Bornhauser, M.; Dazzi, F.; Wehner, R.; Schmitz, M. Immunomodulatory Properties of Mesenchymal Stromal Cells: An Update. Front Cell Dev Biol 2021, 9, 637725. [Google Scholar] [CrossRef]
- Nguyen, T.Q.T.; Cho, K.A. Targeting immunosenescence and inflammaging: advancing longevity research. Experimental & Molecular Medicine 2025, 57, 1881–1892. [Google Scholar] [CrossRef]
- Strasser, B.; Wolters, M.; Weyh, C.; Kruger, K.; Ticinesi, A. The Effects of Lifestyle and Diet on Gut Microbiota Composition, Inflammation and Muscle Performance in Our Aging Society. Nutrients 2021, 13. [Google Scholar] [CrossRef]
- Martínez-Albert, E.; Lutz, N.D.; Hübener, R.; Dimitrov, S.; Lange, T.; Born, J.; Besedovsky, L. Sleep promotes T-cell migration towards CCL19 via growth hormone and prolactin signaling in humans. Brain, Behavior, and Immunity 2024, 118, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, A.C.; De Nys, L.; Brindle, R.C.; Drayson, M.T. Physical activity and sleep relate to antibody maintenance following naturalistic infection and/or vaccination in older adults. Brain, Behavior, & Immunity - Health 2023, 32, 100661. [Google Scholar] [CrossRef]
- Xie, L.; Kang, H.; Xu, Q.; Chen, M.J.; Liao, Y.; Thiyagarajan, M.; O’Donnell, J.; Christensen, D.J.; Nicholson, C.; Iliff, J.J.; et al. Sleep drives metabolite clearance from the adult brain. Science 2013, 342, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Zisapel, N. New perspectives on the role of melatonin in human sleep, circadian rhythms and their regulation. Br J Pharmacol 2018, 175, 3190–3199. [Google Scholar] [CrossRef] [PubMed]
- Bjorklund, G.; Shanaida, M.; Lysiuk, R.; Antonyak, H.; Klishch, I.; Shanaida, V.; Peana, M. Selenium: An Antioxidant with a Critical Role in Anti-Aging. Molecules 2022, 27. [Google Scholar] [CrossRef]
- Ecarnot, F.; Maggi, S. The impact of the Mediterranean diet on immune function in older adults. Aging Clin Exp Res 2024, 36, 117. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Gängler, S.; Wieczorek, M.; Belsky, D.W.; Ryan, J.; Kressig, R.W.; Stähelin, H.B.; Theiler, R.; Dawson-Hughes, B.; Rizzoli, R.; et al. Individual and additive effects of vitamin D, omega-3 and exercise on DNA methylation clocks of biological aging in older adults from the DO-HEALTH trial. Nature Aging 2025, 5, 376–385. [Google Scholar] [CrossRef]
- Chambers, E.S.; Vukmanovic-Stejic, M.; Turner, C.T.; Shih, B.B.; Trahair, H.; Pollara, G.; Tsaliki, E.; Rustin, M.; Freeman, T.C.; Mabbott, N.A.; et al. Vitamin D(3) replacement enhances antigen-specific immunity in older adults. Immunother Adv 2021, 1, ltaa008. [Google Scholar] [CrossRef] [PubMed]
- Ghaseminejad-Raeini, A.; Ghaderi, A.; Sharafi, A.; Nematollahi-Sani, B.; Moossavi, M.; Derakhshani, A.; Sarab, G.A. Immunomodulatory actions of vitamin D in various immune-related disorders: a comprehensive review. Front Immunol 2023, 14, 950465. [Google Scholar] [CrossRef] [PubMed]
- Rastgoo, S.; Pourvali, K.; Raeissadat, S.A.; Eslamian, G.; Zand, H. Co-administration of vitamin D and N-acetylcysteine to modulate immunosenescence in older adults with vitamin D deficiency: a randomized clinical trial. Front Immunol 2025, 16, 1570441. [Google Scholar] [CrossRef]
- Barry, A.R.; Dixon, D.L. Omega-3 fatty acids for the prevention of atherosclerotic cardiovascular disease. Pharmacotherapy 2021, 41, 1056–1065. [Google Scholar] [CrossRef]
- Djuricic, I.; Calder, P.C. Beneficial Outcomes of Omega-6 and Omega-3 Polyunsaturated Fatty Acids on Human Health: An Update for 2021. Nutrients 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Azzolino, D.; Bertoni, C.; De Cosmi, V.; Spolidoro, G.C.I.; Agostoni, C.; Lucchi, T.; Mazzocchi, A. Omega-3 polyunsatured fatty acids and physical performance across the lifespan: a narrative review. Front Nutr 2024, 11, 1414132. [Google Scholar] [CrossRef]
- Bodur, M.; Yilmaz, B.; Agagunduz, D.; Ozogul, Y. Immunomodulatory Effects of Omega-3 Fatty Acids: Mechanistic Insights and Health Implications. Mol Nutr Food Res 2025, 69, e202400752. [Google Scholar] [CrossRef]
- Kerlikowsky, F.; Kruger, K.; Hahn, A.; Schuchardt, J.P. Multimicronutrient and omega-3 fatty acid supplementation reduces low-grade inflammation in older participants: An exploratory study. Nutr Res 2025, 140, 46–58. [Google Scholar] [CrossRef]
- Pluta, R.; Ulamek-Koziol, M.; Januszewski, S.; Czuczwar, S.J. Gut microbiota and pro/prebiotics in Alzheimer’s disease. Aging (Albany NY) 2020, 12, 5539–5550. [Google Scholar] [CrossRef]
- Yu, L.W.; Agirman, G.; Hsiao, E.Y. The Gut Microbiome as a Regulator of the Neuroimmune Landscape. Annu Rev Immunol 2022, 40, 143–167. [Google Scholar] [CrossRef]
- Zhang, T.; Gao, G.; Kwok, L.Y.; Sun, Z. Gut microbiome-targeted therapies for Alzheimer’s disease. Gut Microbes 2023, 15, 2271613. [Google Scholar] [CrossRef]
- Dou, L.; Peng, Y.; Zhang, B.; Yang, H.; Zheng, K. Immune Remodeling during Aging and the Clinical Significance of Immunonutrition in Healthy Aging. Aging Dis 2024, 15, 1588–1601. [Google Scholar] [CrossRef]
- Silva, Y.P.; Bernardi, A.; Frozza, R.L. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front Endocrinol (Lausanne) 2020, 11, 25. [Google Scholar] [CrossRef]
- Nourazarain, A.; Vaziri, Y. Nutrigenomics meets multi-omics: integrating genetic, metabolic, and microbiome data for personalized nutrition strategies. Genes Nutr 2025, 20, 30. [Google Scholar] [CrossRef]
- Dolan, M.; Libby, K.A.; Ringel, A.E.; van Galen, P.; McAllister, S.S. Ageing, immune fitness and cancer. Nature Reviews Cancer 2025, 25, 848–872. [Google Scholar] [CrossRef]
- McGee, K.C.; Sullivan, J.; Hazeldine, J.; Schmunk, L.J.; Martin-Herranz, D.E.; Jackson, T.; Lord, J.M. A combination nutritional supplement reduces DNA methylation age only in older adults with a raised epigenetic age. Geroscience 2024, 46, 4333–4347. [Google Scholar] [CrossRef] [PubMed]
- Sun, E.D.; Nagvekar, R.; Pogson, A.N.; Brunet, A. Brain aging and rejuvenation at single-cell resolution. Neuron 2025, 113, 82–108. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Fan, D.; Yang, Y.; Gimple, R.C.; Zhou, S. Integrative multi-omics approaches to explore immune cell functions: Challenges and opportunities. iScience 2023, 26, 106359. [Google Scholar] [CrossRef] [PubMed]
- Riemann, L.; Gutierrez, R.; Odak, I.; Barros-Martins, J.; Roesner, L.M.; Leon Lara, X.; Falk, C.; Schulz, T.F.; Hansen, G.; Werfel, T.; et al. Integrative deep immune profiling of the elderly reveals systems-level signatures of aging, sex, smoking, and clinical traits. EBioMedicine 2025, 112, 105558. [Google Scholar] [CrossRef] [PubMed]
- Cisneros, B.; Garcia-Aguirre, I.; Unzueta, J.; Arrieta-Cruz, I.; Gonzalez-Morales, O.; Dominguez-Larrieta, J.M.; Tamez-Gonzalez, A.; Leyva-Gomez, G.; Magana, J.J. Immune system modulation in aging: Molecular mechanisms and therapeutic targets. Front Immunol 2022, 13, 1059173. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



