Submitted:
17 January 2026
Posted:
19 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Plateaued Therapies and the Need for Mechanistic Innovation in Isoproterenol-Induced Myocardial Ischemia
1.2. Molecular Pathophysiology of ISO-Induced Myocardial Injury and Survival Pathways
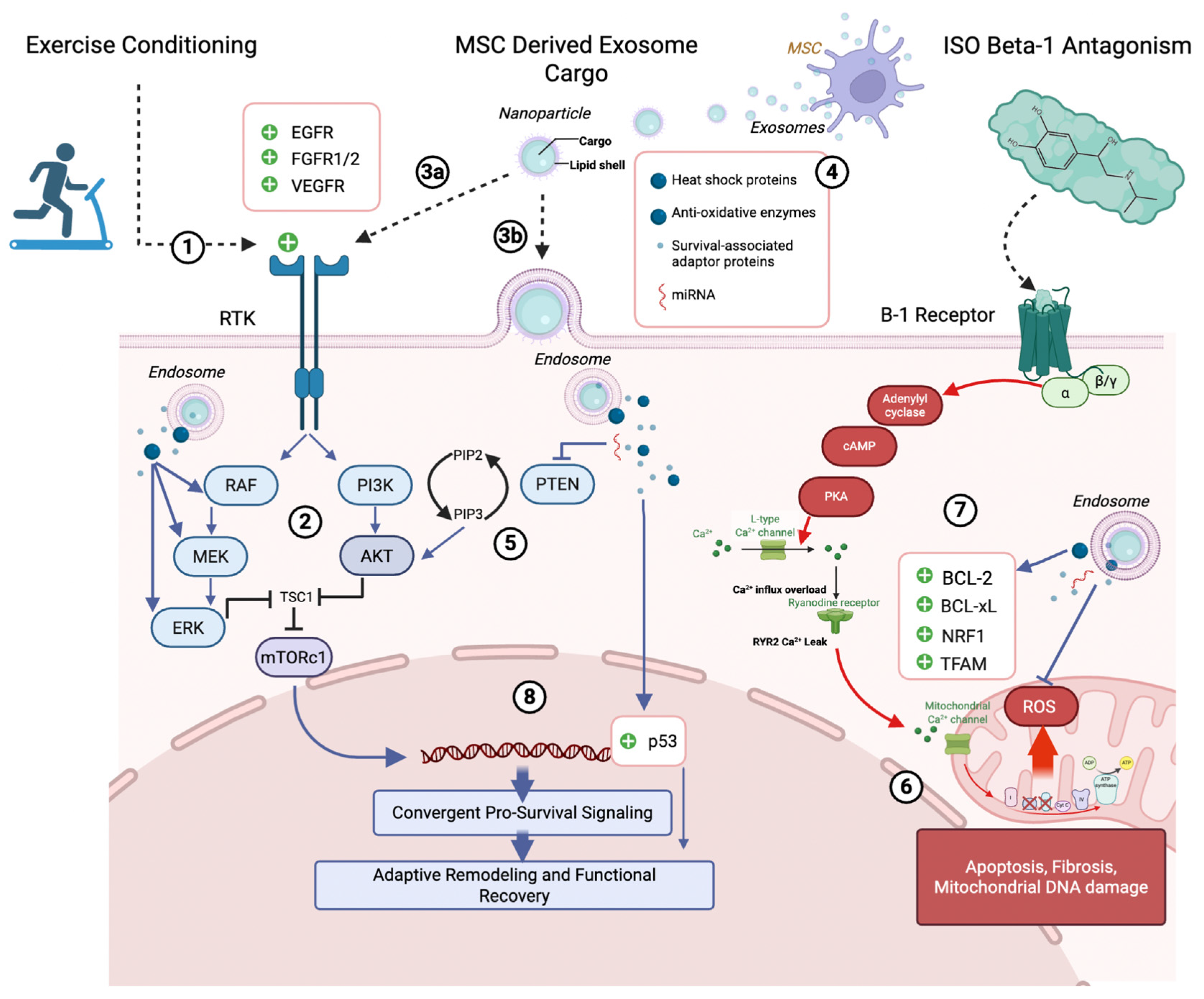
1.3. Exercise-Induced Intracellular Signaling in Cardioprotection Against ISO Injury
1.4. Cardioprotective Mechanisms of Mesenchymal Stem Cell–Derived Exosomes in Ischemic Myocardium
2. Methods
3. Stem Cell Source Considerations for Exosome-Based Therapy in Cardiac Ischemia
3.1. Pluripotent vs. Multipotent Stem Cells
3.2. Advantages and Limitations of Adipose versus Bone Marrow-Derived MSCs
4. Methods of Delivery
4.1. Intramycocardial Injection Approaches
4.2. Intrapericardial Catheter-Based Delivery
4.3. Intracoronary
4.4. Intravenous
5. Advancements
5.1. Stem-Cell Senescence
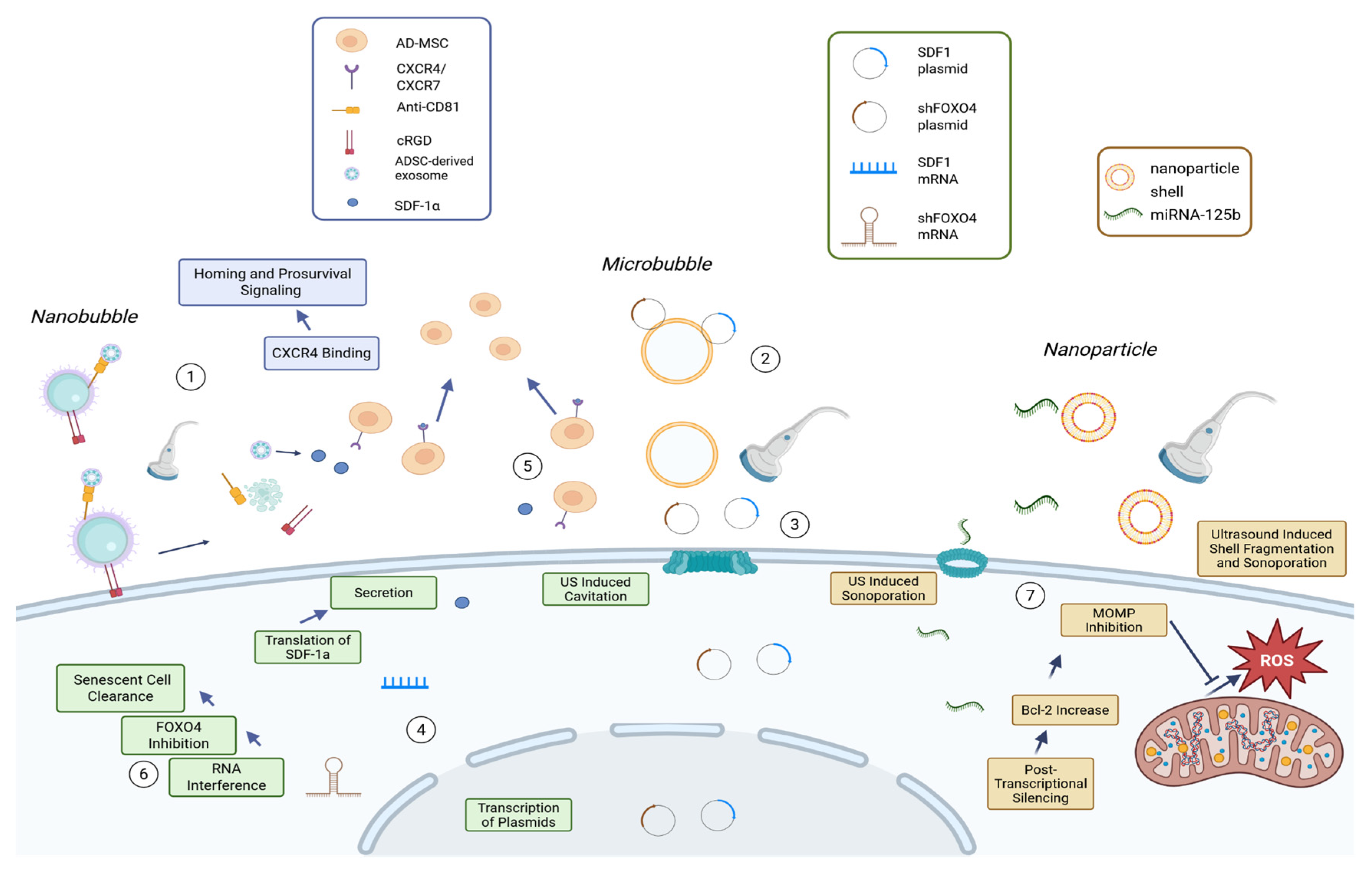
5.2. Bubble Technology
5.3. NanoParticles
| Intervention | Mechanism/ Procedure | Benefit | Drawback | Citation |
| Intramyocardial injection | Direct injection of stem cells into damaged myocardium | Direct targeted infusion of a large number of cells | Mechanical tissue damage; difficult infarct localization; higher arrhythmia incidence; suboptimal electromechanical coupling post-injection | Yuce, 2024 |
| Transendocardial intramyocardial injection | Percutaneous catheter-based, image-guided injections into endocardial surface targeting borderzone myocardium | Precise border zone access without open chest surgery; reported improvements in perfusion, EF, diastolic/LV function, and 6-minute walk metrics in preliminary work | Preliminary stage with inconsistent results | Yuce, 2024 |
| Epicardial intramyocardial injection | Surgical direct-visual injection into infarcted myocardium, typically adjunct during open-heart surgery | Direct visual access; described as not having coronary embolism risk | More invasive; leakage; dosing control challenges; inadequate donor-cell retention; studies limited to animals | Yuce, 2024 |
| Intrapericardial injection | Intrapericardial delivery of hydrogel compound containing MSCs; compared vs intramyocardial control; porcine feasibility/safety reported | ~10× higher viable cell retention vs intramyocardial in mice;increased exosome secretion and reduced myocardial apoptosis; no major adverse effects in mice; no abnormal cardiac events in pigs over 4 days | Follow-up in pigs only 4 days, so long-term effects not assessed | Li et al., 2022 |
| Intracoronary infusion | Catheter-based delivery into infarct-related artery by inflating the catheter and infusing cells during inflation; used during catheterization | Human trials described with EF increases, reduced scarred myocardium, and lack of arrhythmia post-procedure | Long-term effects not established; procedural blinding was limited; >65 excluded; concern raised about potential bias in reporting/editorial handling; animal data suggest possible microvascular obstruction and myocardial injury | Heldman et al., 2008; Attar et al., 2023; Attar et al., 2025; Maxwell, 2025 |
| Embryoid body formation | Generate iPSC aggregates to drive spontaneous lineage commitment, then isolate MSC-like cells | Simple; cost-effective | Heterogeneity; scale-up challenges; limited microenvironment control | Barzegari et al., 2020; Dupuis and Oltra, 2021 |
| Specific differentiation | Predifferentiate iPSCs toward a lineage, then apply factors/conditions to yield iMSCs | Greater regenerative potential | More time-consuming; higher cost | Dias et al., 2023; Dupuis and Oltra, 2021 |
| Blood-based method | Culture iPSCs under blood-derived supplement conditions to promote iMSC phenotype | Low-cost; high proliferative potential | Potential immune reactivity if residual blood components/cell fragments persist | Dupuis and Oltra, 2021 |
| MSC switch method | Switch iPSC medium to MSC growth media; optional FACS to select subpopulations | Operationally straightforward; selection can improve consistency | Variability in signaling potency and paracrine profile; regulatory | Dupuis and Oltra, 2021; Dias et al., 2023 |
| Pathway inhibitor method | Use chemical pathway inhibitors to drive iPSC to iMSC differentiation | Can reduce heterogeneity via controlled signaling | Labor-intensive; scale-up/yield limitations | Dupuis and Oltra, 2021; Dias et al., 2023 |
| UTMD microbubble gene delivery | IV gene-loaded cationic microbubbles; ultrasound cavitation destroys bubbles to deliver genes | Enhances MSC homing; improved repair outcomes vs single-gene | Theoretical toxicity/embolism; transient gene expression | Jiang et al., 2025 |
| Targeted nanobubble–exosome delivery | IV nanobubble–antibody–exosome complex; LIPUS disrupts nanobubbles to drive exosome release/penetration | Improves myocardial retention/uptake vs controls; reduces non-cardiac sequestration | Preclinical window and assays limit safety claims; off-target biodistribution remains plausible | Wang et al., 2025[19] |
| Dual-membrane phase-change nanoparticles | IV phase-change nanoparticles with MSC + macrophage membranes; miRNA-125b surface-adsorbed | Anti-apoptotic and anti-fibrotic effect | Short miRNA activity; repeat dosing | Wang et al., 2025[20] |
6. Discussion
6.1. Integrative Model for Combining Exercise and MSC-Exosome Therapy in Isoproterenol-Induced Myocardial Ischemia
6.2. Current Clinical, Translational, and Preclinical Evidence for MSC-Based Cardiac Regeneration
6.3. Safety and Limitations
Author Contributions
Funding
References
- Attar A, Farjoud Kouhanjani M, Hessami K, et al. Effect of once versus twice intracoronary injection of allogeneic-derived mesenchymal stromal cells after acute myocardial infarction: BOOSTER-TAHA7 randomized clinical trial. Stem Cell Res Ther. 2023;14(1):264. Published 2023 Sep 23. [CrossRef]
- Attar A, Mirhosseini SA, Mathur A, et al. Prevention of acute myocardial infarction induced heart failure by intracoronary infusion of mesenchymal stem cells: phase 3 randomised clinical trial (PREVENT-TAHA8). BMJ. 2025;391:e083382. Published 2025 Oct 29. [CrossRef]
- Barzegari A, Gueguen V, Omidi Y, Ostadrahimi A, Nouri M, Pavon-Djavid G. The role of Hippo signaling pathway and mechanotransduction in tuning embryoid body formation and differentiation. J Cell Physiol. 2020;235(6):5072-5083. [CrossRef]
- Choudhery MS, Arif T, Mahmood R, et al. Induced Mesenchymal Stem Cells: An Emerging Source for Regenerative Medicine Applications. J Clin Med. 2025;14(6):2053. Published 2025 Mar 18. [CrossRef]
- Dias IX, Cordeiro A, Guimarães JAM, Silva KR. Potential and Limitations of Induced Pluripotent Stem Cells-Derived Mesenchymal Stem Cells in Musculoskeletal Disorders Treatment. Biomolecules. 2023;13(9):1342. Published 2023 Sep 4. [CrossRef]
- Dupuis V, Oltra E. Methods to produce induced pluripotent stem cell-derived mesenchymal stem cells: Mesenchymal stem cells from induced pluripotent stem cells. World J Stem Cells. 2021;13(8):1094-1111. [CrossRef]
- Goto T, Nakamura Y, Ito Y, Miyagawa S. Regenerative medicine in cardiovascular disease. Regen Ther. 2024;26:859-866. Published 2024 Oct 5. [CrossRef]
- Heldman AW, Hare JM. Cell therapy for myocardial infarction: Special delivery. J Mol Cell Cardiol. 2008;44(3):473-476. [CrossRef]
- Ito, F., Kitani, T. Current Advances and Challenges in Stem Cell-Based Regenerative Therapy for Chronic Limb-Threatening Ischemia. Curr Treat Options Cardio Med 27, 4 (2025). [CrossRef]
- Izumi A, Yau TM, Fedak PWM, Fatehi Hassanabad A. Pericardial Delivery of Stem Cells: An Emerging Frontier in Myocardial Regeneration for Ischemic Heart Disease. Can J Cardiol. Published online October 14, 2025. [CrossRef]
- Jiang X, Chao L, Liu K, et al. Cationic microbubbles loading shFOXO4/SDF1 rejuvenate the aged heart and alleviate myocardial ischemia-reperfusion injury in the elderly. Free Radic Biol Med. 2025;237:210-227. [CrossRef]
- Khalili MR, Ahmadloo S, Mousavi SA, et al. Navigating mesenchymal stem cells doses and delivery routes in heart disease trials: A comprehensive overview. Regen Ther. 2025;29:117-127. Published 2025 Mar 13. [CrossRef]
- Li J, Hu S, Zhu D, et al. All Roads Lead to Rome (the Heart): Cell Retention and Outcomes From Various Delivery Routes of Cell Therapy Products to the Heart. J Am Heart Assoc. 2021;10(8):e020402. [CrossRef]
- Li J, Lv Y, Zhu D, et al. Intrapericardial hydrogel injection generates high cell retention and augments therapeutic effects of mesenchymal stem cells in myocardial infarction. Chem Eng J. 2022;427:131581. [CrossRef]
- EXPRESSION OF CONCERN: Prevention of acute myocardial infarction induced heart failure by intracoronary infusion of mesenchymal stem cells: phase 3 randomised clinical trial. BMJ. 2025;391:r2388. Published 2025 Nov 12. [CrossRef]
- Poliwoda S, Noor N, Downs E, et al. Stem cells: a comprehensive review of origins and emerging clinical roles in medical practice. Orthop Rev (Pavia). 2022;14(3):37498. Published 2022 Aug 25. [CrossRef]
- Stougiannou TM, Christodoulou KC, Dimarakis I, Mikroulis D, Karangelis D. To Repair a Broken Heart: Stem Cells in Ischemic Heart Disease. Curr Issues Mol Biol. 2024;46(3):2181-2208. Published 2024 Mar 8. [CrossRef]
- Tang XL, Wysoczynski M, Li Y, et al. Comparative Effects of Repeated Intravenous Infusions of Progenitor Cells in a Rat Model of Chronic Ischemic Cardiomyopathy. Stem Cell Rev Rep. Published online October 9, 2025. [CrossRef]
- Wang H, Jiang R, Zhong F, et al. Ultrasound-triggered targeted delivery of engineered ADSCs-derived exosomes with high SDF-1α levels to promote cardiac repair following myocardial infarction. Int J Pharm. 2025;681:125786. [CrossRef]
- Wang Z, Chen J, Wang J, et al. MSCs biomimetic ultrasonic phase change nanoparticles promote cardiac functional recovery after acute myocardial infarction. Biomaterials. 2025;313:122775. [CrossRef]
- Yuce K. The Application of Mesenchymal Stem Cells in Different Cardiovascular Disorders: Ways of Administration, and the Effectors. Stem Cell Rev Rep. 2024;20(7):1671-1691. [CrossRef]
- Zhang F, Hu G, Chen X, et al. Excessive branched-chain amino acid accumulation restricts mesenchymal stem cell-based therapy efficacy in myocardial infarction. Signal Transduct Target Ther. 2022;7(1):171. Published 2022 Jun 3. [CrossRef]
- Rittiphairoj T, Bulstra C, Ruampatana C, et al. The economic burden of ischaemic heart diseases on health systems: a systematic review. BMJ Glob Health. 2025;10(2):e015043. Published 2025 Feb 12. [CrossRef]
- World Health Organization. Cardiovascular diseases (CVDs) fact sheet (includes current global mortality estimates). Updated 2025.
- Hare JM, et al. Randomized Comparison of Allogeneic Versus Autologous Mesenchymal Stem Cells for Nonischemic Dilated Cardiomyopathy: POSEIDON-DCM Trial. J Am Coll Cardiol. 2017 Feb 7;69(5):526-537. Epub 2016 Nov 14. PMID: 27856208; PMCID: PMC5291766. [CrossRef]
- Heldman AW, et al. Transendocardial mesenchymal stem cells and mononuclear bone marrow cells for ischemic cardiomyopathy: the TAC-HFT randomized trial. JAMA. 2014 Jan 1;311(1):62-73. PMID: 24247587; PMCID: PMC4111133. [CrossRef]
- Bartunek J, et al; CHART Program. Cardiopoietic stem cell therapy in ischaemic heart failure: long-term clinical outcomes. ESC Heart Fail. 2020 Dec;7(6):3345-3354. Epub 2020 Oct 23. PMID: 33094909; PMCID: PMC7754898. [CrossRef]
- Raynaud CM, Yacoub MH. Clinical trials of bone marrow derived cells for ischemic heart failure. Time to move on? TIME, SWISS-AMI, CELLWAVE, POSEIDON and C-CURE. Glob Cardiol Sci Pract. 2013 Nov 1;2013(3):207-11. PMID: 24689022; PMCID: PMC3963753. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




